Abstract
Background:
Tanezumab is a nerve growth factor monoclonal antibody that may regulate pain in hip or knee osteoarthritis (OA). This meta-analysis was performed to evaluate the efficacy and safety of low and moderate doses of tanezumab in treating hip or knee OA.
Methods:
PubMed, EMBASE, the Cochrane Library, and Web of Science were comprehensively searched for clinical trials published before 1 May 2021. Patients were assessed via efficacy and safety outcomes.
Results:
Twelve randomized controlled trials including 6022 patients were identified. Both low and moderate doses of tanezumab significantly improved efficacy outcomes. However, only the point estimates (mean difference, MD) of moderate-dose tanezumab significantly exceeded the minimal clinically important differences (MCIDs). There were no significant differences in the incidence of treatment-related adverse events (AEs), withdrawals due to AEs, serious AEs, and total joint replacement between the tanezumab and placebo groups, whereas the incidence of AEs was higher in the tanezumab group (relative risk, RR = 1.10; 95% confidence interval, 95% CI = 1.04–1.17). The incidence of rapidly progressive OA was significantly higher in the combined low- and moderate-dose tanezumab groups than in the placebo group (RR = 5.01; 95% CI = 1.17–21.33). Furthermore, both low and moderate doses of tanezumab significantly increased the incidence of abnormal peripheral sensation (RR = 1.99, 95% CI = 1.21–3.28; RR = 2.64, 95% CI = 1.91–3.67, respectively). Compared with nonsteroidal anti-inflammatory drugs (NSAIDs) and opioids, tanezumab showed significantly improved efficacy outcomes (p < 0.05). However, the point estimates (MD) of tanezumab were not greater than the MCID. Pooled analysis showed no significant differences between tanezumab and NSAIDs and opioids in safety outcomes (p > 0.05).
Conclusion:
Tanezumab is efficacious in patients with hip or knee OA. Tanezumab is relatively well tolerated and safe but increases the incidence of AEs and reversible abnormal peripheral sensation. Additional studies on the occurrence of rapidly progressive OA are needed. A moderate dose of tanezumab may maximize the benefits for hip or knee OA.
Introduction
Osteoarthritis (OA) is a common disease worldwide. The pain and impairment in joint function caused by OA can lead to a decline in the quality of life of patients and even disability.1,2 The effective management of pain due to OA is a major concern for clinicians. Although the current recommendations suggest a combination of drug and nondrug treatments,3,4 many patients still suffer from inadequate pain relief. Moreover, currently used analgesics, including nonsteroidal anti-inflammatory drugs (NSAIDs) and opioids, are poorly tolerated by many patients, and there are concerns about the safety and efficacy of the long-term use of these drugs.5,6 Therefore, drugs that are more effective in relieving pain than are existing treatments and are well tolerated by patients urgently need to be developed.
With the emergence of in-depth research on pain mechanisms, new potential targets, especially the regulation of nociceptive and neurological pathways, have become the focus of recent drug development. Nerve growth factor (NGF) is a neurotrophic factor. After tissue injury or inflammation, NGF is released and binds to tropomyosin receptor kinase (Trk) A, which can lead to central sensitization, 7 induce the expression of peripheral and central pain-related substances, and make adjacent pain-sensing neurons sensitive to inflammation, thereby mediating pain.8 –10 NGF is also considered a pathological product of OA or an important pathogenic factor involved in the process of OA pain. The content of NGF in the synovial fluid of OA patients is higher than that in the synovial fluid of normal persons, 11 and NGF is also expressed in the synovial membrane of OA patients. 12 Furthermore, NGF expression increases significantly in traumatic and inflammatory sites. 13 The inhibition of NGF binding to its receptor and the downregulation of NGF expression can significantly reduce pain, 14 relieve OA symptoms, and improve limb function,15,16 thereby relieving OA pain.
Tanezumab is an IgG2 NGF monoclonal antibody that blocks NGF from interacting with its receptors, thereby blocking signals from nociceptive neurons responsible for pain.17,18 In 2010, tanezumab was tested in phase II human trials. The monoclonal antibodies against NGF currently used in clinical research include not only tanezumab but also fulranumab and fasinumab. However, there are few studies on fulranumab and fasinumab, and most of them are still in phase II trials, while tanezumab is the most studied monoclonal antibody to inhibit NGF. Studies on tanezumab in the treatment of hip or knee OA have been reported in meta-analyses, but these results were based on only statistical significant differences, and reporting only these results might be misleading in terms of clinical decision making. Thus, we carried out a specific analysis based on minimal clinically important difference (MCID) values. In addition, because there is a safety issue, the US Food and Drug Administration (FDA) has mandated that tanezumab and NSAIDs not be used in combination and called for more research on tanezumab at lower doses. Therefore, this study aimed to evaluate the efficacy and safety of lower doses of tanezumab monotherapy for OA of the hip or knee.
Methods
The systematic review was designed and reported based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) recommendations. 19
Search strategy and selection criteria
PubMed, EMBASE, the Cochrane Library, and Web of Science were comprehensively searched for English studies using the following keywords: ‘nerve growth factor antibody’, ‘NGF’, ‘anti-NGF’, ‘tanezumab’, ‘OA’, or ‘osteoarthritis’. Studies published from the inception of the database to 1 May 2021 were retrieved. The references of the articles included were also searched, and each article was manually cross-checked. Disagreements among the reviewers were resolved by consensus.
Study selection
With respect to the inclusion criteria, the two researchers independently screened the titles, abstracts, and full texts to determine which studies to include in this review. Any disagreements were discussed and resolved by a third reviewer.
Eligibility criteria
The inclusion criteria were as follows: (1) studies in which the participants were adult patients diagnosed with hip or knee OA; (2) studies in which the experimental group was treated with low or moderate doses of tanezumab monotherapy, and the control group was treated with a placebo or an active comparator; (3) studies in which at least the following outcome measures were included – Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) pain score (scale of 0–10), WOMAC physical function score (scale of 0–10), patient’s global assessment (PGA; 5-point Likert-type scale), adverse events (AEs), treatment-related AEs, serious adverse events (SAEs), withdrawals due to AEs, rapidly progressive OA (RPOA), and total joint replacement and abnormal peripheral sensation; and (4) studies that had a randomized controlled trial (RCT) design.
The exclusion criteria were as follows: (1) studies in which the experimental group was treated with tanezumab combined with other active treatments, and (2) studies with incomplete data.
Data extraction
The data were extracted independently by two researchers, and the variables included first author’s name, year of publication, phase of study, patient age, patient sex, sample size, Kellgren–Lawrence (K-L) grade, outcome measures, follow-up time, and type and dose of intervention. For studies by the same author, we selected the study with the longer follow-up time and complete data for the variables of interest. Disagreements were resolved through discussion, and if necessary, a third investigator made the final decision.
Risk of bias assessment
The quality of the methodology included in the study was assessed with the Cochrane Collaboration risk of bias assessment tool, 20 which evaluates the following domains: random sequence generation, allocation concealment, blinding of the participants and outcome assessors, incomplete outcome data, selective reporting, and other bias. Each component was considered to have a low, unclear, or high risk of bias.
Statistical analysis
We used Review Manager (version 5.3; Cochrane Collaboration, Oxford, UK) for statistical analysis of the data and Stata (version 14.0; StataCorp, College Station, Texas, USA) to assess publication bias. The mean baseline-to-end point changes and their standard deviations (SDs) were obtained. For the efficacy outcomes, including WOMAC pain, WOMAC physical function, and PGA scores, we calculated the mean differences (MDs) and 95% confidence intervals (CIs). For the safety outcomes, we calculated the relative risk (RR) values and 95% CIs. If only the standard error was reported, the SD was calculated based on the reported sample size and standard error. When the mean, standard error, or SD were not provided in an article, we extracted the values from charts or graphs, as needed. The Cochran Q test and I2 statistic were used to determine the magnitude of heterogeneity among studies. Heterogeneity levels of 0–25% were considered low, levels of 26–49% were considered moderate, and levels ⩾50% were considered high. If the level of heterogeneity was ⩾50%, a random-effects model was used; otherwise, a fixed-effects model was used. Publication bias of the outcome measures was assessed with Egger’s regression tests if the number of studies was >10. Subgroup analysis for tanezumab at different doses was performed; p < 0.05 indicated a significant difference. To assess whether the difference was clinically meaningful, the differences in the results reported by patients were directly compared with the MCIDs reported. According to previous reports, 21 the MCID of the WOMAC pain score (scale of 0–10) is 0.97, and the MCID of the WOMAC physical function score (scale of 0–10) is 0.93. However, to our knowledge, there are no well-established MCID criteria for PGA (5-point Likert-type scale). Therefore, only statistically significant differences are reported. When outcomes are statistically significant, and the point estimates (MD) are greater than the MCID, the result is considered to reflect a clinical effect.
Results
Selection of the included studies
Through the literature search, a total of 368 studies were retrieved. After the titles and abstracts were screened, 30 studies remained, and then, the full texts were read. According to the inclusion and exclusion criteria, 11 studies were finally selected. See Figure 1 for details.
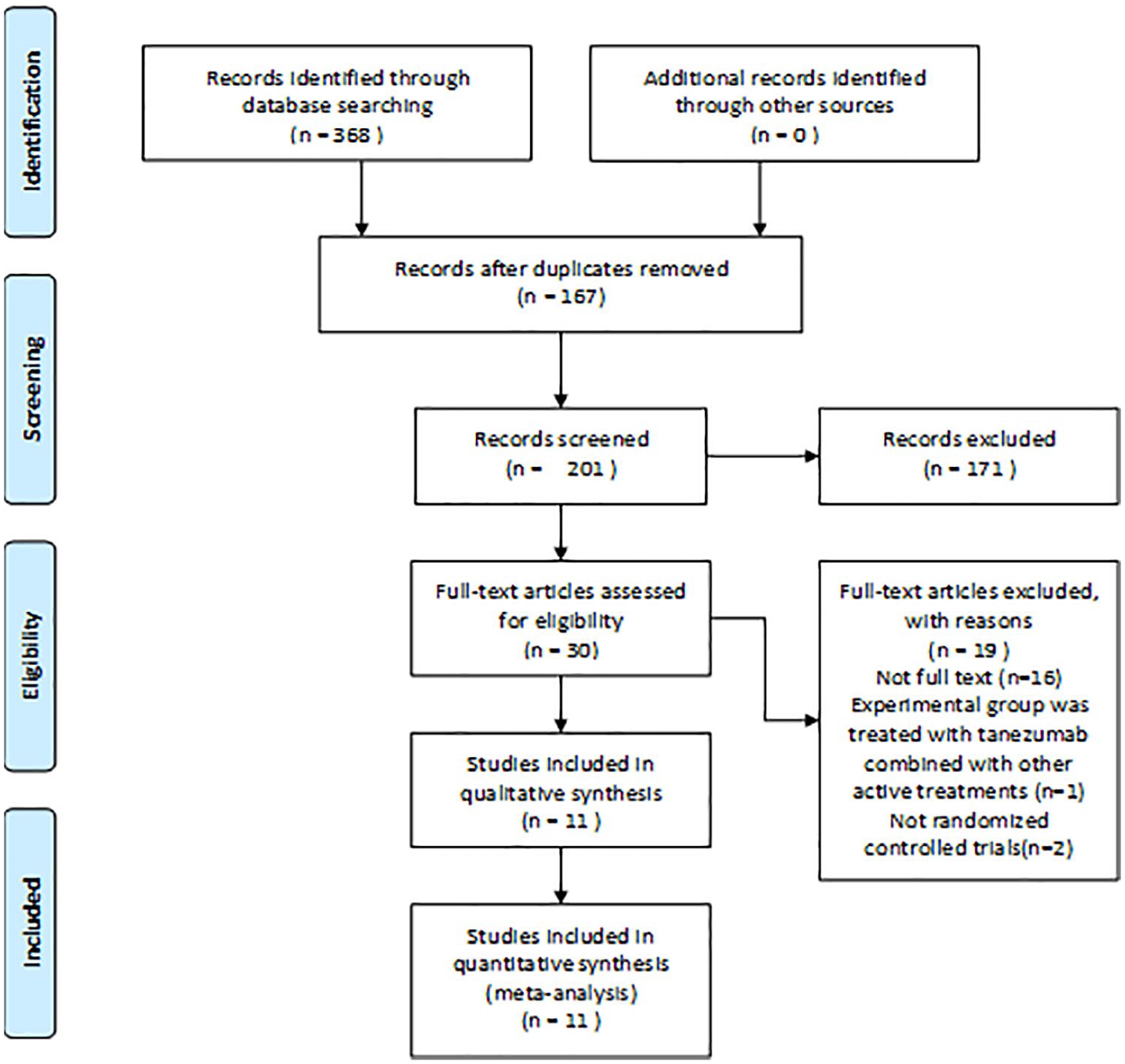
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) study selection flow diagram.
Characteristics of the included studies
A total of 12 RCTs, including 6022 patients, were included in the review. One of the studies 22 involved two RCTs. According to the K-L grade, the degree of hip or knee OA ranged from moderate to severe. Two studies23,24 were phase II trials, and nine studies22,25 –32 were phase III trials. The characteristics of the included clinical trials are summarized in Table 1. The doses of tanezumab in phase II trials were 10, 25, 50, 100, and 200 µg/kg, while in phase III trials, the doses of tanezumab were 2.5, 5, and 10 mg. However, the dose–response analysis and pharmacokinetic/pharmacodynamic study of tanezumab showed that sex and the dose regimen of adjusting an individual’s body weight negligibly reduced the variability in systemic exposure compared with that predicted with a fixed-dose regimen. 33 Therefore, the data from the phase II and phase III trials were combined in this study. At present, according to the FDA regulations, high doses of tanezumab cannot be used for experimental research; thus, we studied only low and moderate doses of tanezumab. The combination of the 10 and 25 µg/kg doses in the phase II trials and the 2.5 mg doses in the phase III trials was studied and considered the low-dose group; the 50 µg/kg doses in the phase II trials and 5 mg doses in the phase III trials were combined and defined as the moderate-dose group. In all subsequent analyses, we conducted a subgroup analysis based on low and moderate doses.
Characteristics of the included studies.

AEs, adverse events; K-L grade, Kellgren–Lawrence grade for classifying OA severity, ranging from 0 (no OA) to IV (severe OA); PGA, patient’s global assessment; RPOA, rapidly progressive osteoarthritis; SAEs, serious adverse events; TJR, total joint replacement; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.
There were 10 placebo-controlled studies and 4 active-controlled studies. The active-controlled agents included oxycodone, celecoxib, and naproxen. However, in each of the active-controlled RCTs, low doses of tanezumab were not reported, so we analyzed only moderate doses of tanezumab.
Risk of bias assessment
The quality of all of the studies was relatively high. Although all studies were randomized controlled studies, five studies22,26,28,29,32 did not report the specific randomization methods used. Two studies22,26 did not clearly indicate whether the participants and outcome assessors were blinded, so the risk of bias for this domain was judged as unclear. Allocation concealment was implemented in all studies. The complete results for the risk of bias assessment are presented in Supplemental Appendix Figures A1 and A2. Egger’s test suggested that there was no significant publication bias for the WOMAC pain score (p = 0.627), WOMAC physical score (p = 0.589), AEs (p = 0.435), SAEs (p = 0.666), or abnormal peripheral sensation (p = 0.672).
Placebo-controlled trials
Efficacy
WOMAC pain score: A total of 11 RCTs (4305 patients) reported WOMAC pain scores. The results of the pooled analysis showed that tanezumab significantly improved WOMAC pain scores compared with placebo (MD = −0.98, 95% CI = −1.15 to −0.81, p < 0.00001, I2 = 0%). In the subgroup analysis, 7 studies were included in the low-dose group, and 11 studies were included in the moderate-dose group. The results of the pooled analysis for the low-dose group showed that tanezumab significantly relieved patients’ pain compared with placebo (MD = −0.90, 95% CI = −1.19 to −0.61, p < 0.00001, I2 = 35%). Similarly, moderate doses of tanezumab also significantly relieved pain in patients compared with placebo (MD = −1.02, 95% CI = −1.24 to −0.81, p < 0.00001, I2 = 0%). There were no significant differences between the two treatment groups (combined low-dose group versus combined moderate-dose group; p = 0.50, I2 = 0%; Figure 2). In comparison with the MCID (0.97), the point estimates (MD) of the low-dose tanezumab group were not greater than the MCID, but the point estimates (MD) of the moderate-dose tanezumab group significantly exceeded the MCID.

Forest plot of WOMAC pain scores detailing mean differences and 95% CIs comparing tanezumab with placebo.
WOMAC physical score: A total of 11 RCTs (4305 patients) reported WOMAC physical scores. The results of the pooled analysis showed that tanezumab significantly improved WOMAC physical scores compared with placebo (MD = −1.04, 95% CI = −1.20 to −0.87, p < 0.00001, I2 = 0%). In the subgroup analysis, 7 studies were included in the low-dose group, and 11 studies were included in the moderate-dose group. The pooled estimate of the WOMAC physical score demonstrated significant differences in favor of low doses of tanezumab (MD = −0.93, 95% CI = −1.21 to −0.65, p < 0.00001, I2 = 29%). Similarly, the moderate-dose group had significantly better WOMAC physical scores than the placebo group (MD = –1.09, 95% CI = –1.30 to −0.89, p < 0.00001, I2 = 0%). There were no significant differences between the two treatment groups (p = 0.35, I2 = 0%; Figure 3). In comparison with the MCID (0.93), the point estimates (MD) of the low-dose tanezumab group were not greater than the MCID, but the point estimates (MD) of the moderate-dose tanezumab group significantly exceeded the MCID.
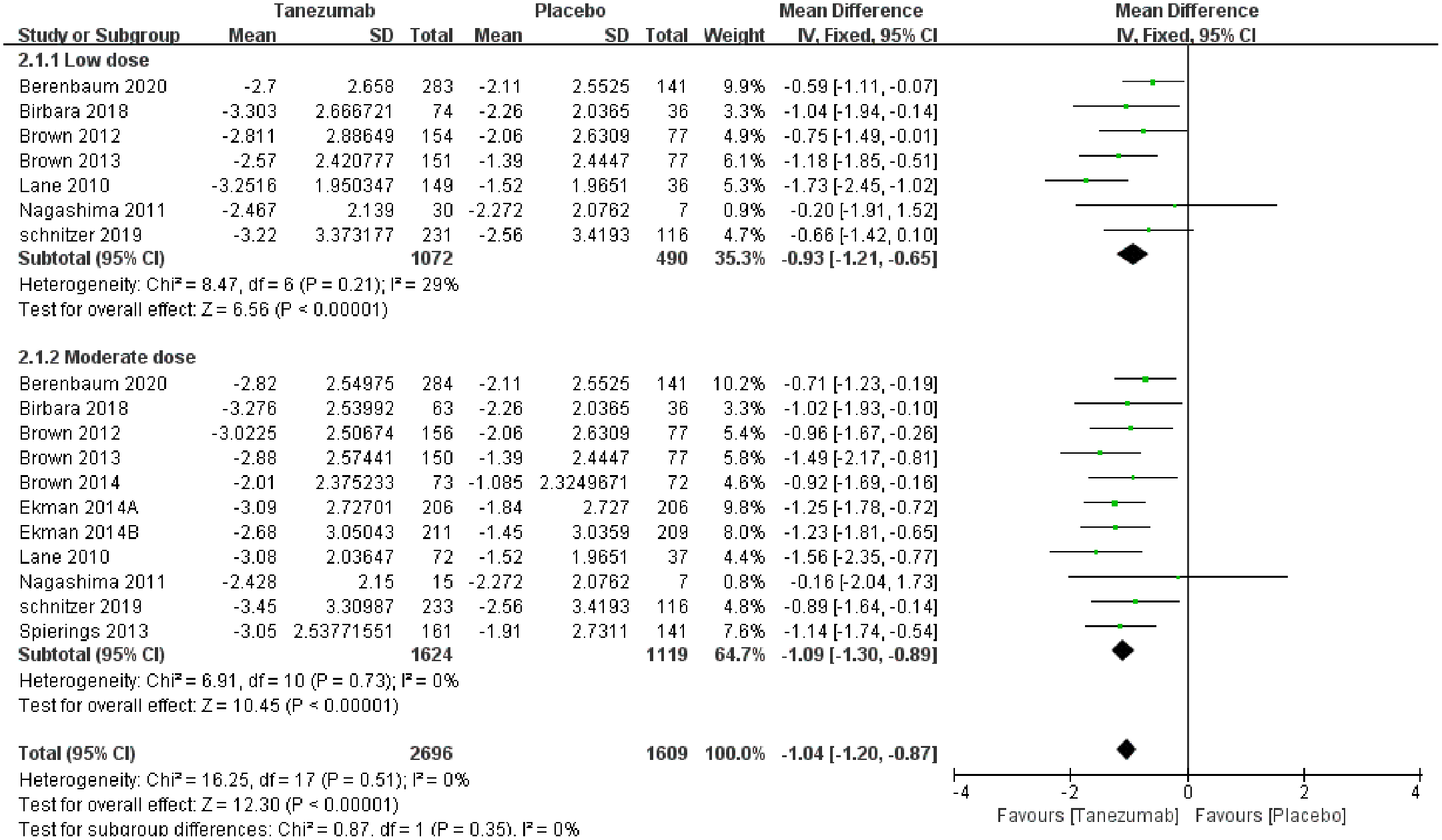
Forest plot of WOMAC physical scores detailing mean differences and 95% CIs comparing tanezumab with placebo.
PGA: A total of nine RCTs (3951 patients) reported PGA results. The results of the pooled analysis showed that tanezumab significantly improved PGA compared with the placebo (MD = −0.29, 95% CI = −0.35 to −0.23, p < 0.00001, I2 = 0%). In the subgroup analysis, five studies were included in the low-dose group, and nine studies were included in the moderate-dose group. Pooled analysis showed that low doses of tanezumab resulted in greater improvements in PGA than the placebo (MD = –0.24, 95% CI = −0.34 to −0.13, p < 0.00001, I2 = 0%; Figure 4). Similarly, the moderate-dose group had significantly better PGA results than the placebo group (MD = −0.31, 95% CI = −0.39 to −0.24, p < 0.00001, I2 = 0%). There were no significant differences between the two treatment groups (p = 0.24, I2 = 27%).

Forest plot of patient’s global assessment detailing mean differences and 95% CIs comparing tanezumab with placebo.
Safety
AEs: A total of 11 RCTs (4372 patients) reported AEs. The results of the pooled analysis showed that the incidence of AEs was significantly higher in the tanezumab group than in the placebo group (RR = 1.10, 95% CI = 1.04–1.17, p = 0.0007, I2 = 1%). In the subgroup analysis, 7 studies were included in the low-dose group, and 11 studies were included in the moderate-dose group. The pooled results for the low-dose group showed that the incidence of AEs was significantly higher in the tanezumab group than in the placebo group (RR = 1.10, 95% CI = 1.01–1.21, p = 0.03, I2 = 38%). Similarly, moderate doses of tanezumab also led to a higher risk of AEs than placebo (RR = 1.10, 95% CI = 1.03–1.19, p = 0.008, I2 = 0%; Table 2). The RR values of the two treatment groups (low-dose group versus moderate-dose group) were the same, without a significant difference (p = 0.99, I2 = 0%).
The safety outcomes of placebo-controlled trials.

CI, confidence interval; OA, osteoarthritis; RR, relative risk.
Treatment-related AEs: A total of five RCTs (2204 patients) reported treatment-related AEs. The results of the pooled analysis showed no significant difference in the incidence of treatment-related AEs between the tanezumab and placebo groups (RR = 1.15, 95% CI = 0.92–1.43, p = 0.22, I2 = 26%). In the subgroup analysis, four studies were included in the low-dose group, and five studies were included in the moderate-dose group. Pooled analysis showed no significant difference between the low-dose tanezumab and placebo groups in terms of treatment-related AEs (RR = 1.17, 95% CI = 0.83–1.63, p = 0.37, I2 = 46%). Similarly, there were no differences between the moderate-dose group and placebo group in treatment-related AEs (RR = 1.13, 95% CI = 0.85–1.52; p = 0.40, I2 = 24%; Table 2). The RR values of the two treatment groups (low-dose group versus moderate-dose group) were similar, without a significant difference (p = 0.90, I2 = 0%).
Withdrawals due to AEs: A total of nine RCTs (3612 patients) reported withdrawals due to AEs. The results of the pooled analysis showed that there was no significant difference in the incidence of withdrawals due to AEs between the tanezumab and placebo groups (RR = 1.19, 95% CI = 0.74–1.91, p = 0.47, I2 = 0%). In the subgroup analysis, five studies were included in the low-dose group, and nine studies were included in the moderate-dose group. The combined results for the low-dose group showed no significant difference in the incidence of withdrawals due to AEs between the tanezumab and placebo groups (RR = 1.20, 95% CI = 0.42–3.37, p = 0.74, I2 = 0%). Similarly, there was no significant difference in the incidence of withdrawals due to AEs between the moderate-dose tanezumab and placebo groups (RR = 1.19, 95% CI = 0.70–2.02, p = 0.52, I2 = 0%; Table 2). The RR values of the two treatment groups were similar, without a significant difference (p = 0.99, I2 = 0%).
SAEs: A total of 11 RCTs (4372 patients) reported SAEs. The results of the pooled analysis showed no significant difference in the incidence of SAEs between the tanezumab and placebo groups (RR = 1.21, 95% CI = 0.87–1.66, p = 0.25, I2 = 0%). In the subgroup analysis, 7 studies were included in the low-dose group, and 11 studies were included in the moderate-dose group. Pooled analysis showed no significant difference between the low-dose tanezumab and placebo groups in SAEs (RR = 1.30, 95% CI = 0.77–2.18, p = 0.33, I2 = 0%). Similarly, the combined results for the moderate-dose group showed no significant difference in the incidence of SAEs between the tanezumab and placebo groups (RR = 1.15, 95% CI = 0.87–1.66, p = 0.50, I2 = 16%; Table 2). The RR values were similar for the two treatment groups (p = 0.72, I2 = 0%).
RPOA: A total of two RCTs (1545 patients) reported RPOA results. The results of the pooled analysis showed that the incidence of RPOA was significantly higher in the tanezumab group than in the placebo group (RR = 5.01, 95% CI = 1.17–21.33, p = 0.03, I2 = 0%). In the subgroup analysis, two studies were included in the low-dose group, and two studies were included in the moderate-dose group. The pooled estimate of RPOA demonstrated that there were no significant differences between the low-dose tanezumab and placebo groups (RR = 5.02, 95% CI = 0.65–39.02, p = 0.12, I2 = 0%). Similarly, there were no significant differences between the moderate-dose group and placebo group in treatment-related AEs (RR = 4.99, 95% CI = 0.64–38.78, p = 0.12, I2 = 0%). The RR values of the two treatment groups were similar (p = 1.00, I2 = 0%; see Figure 5 and Table 2).
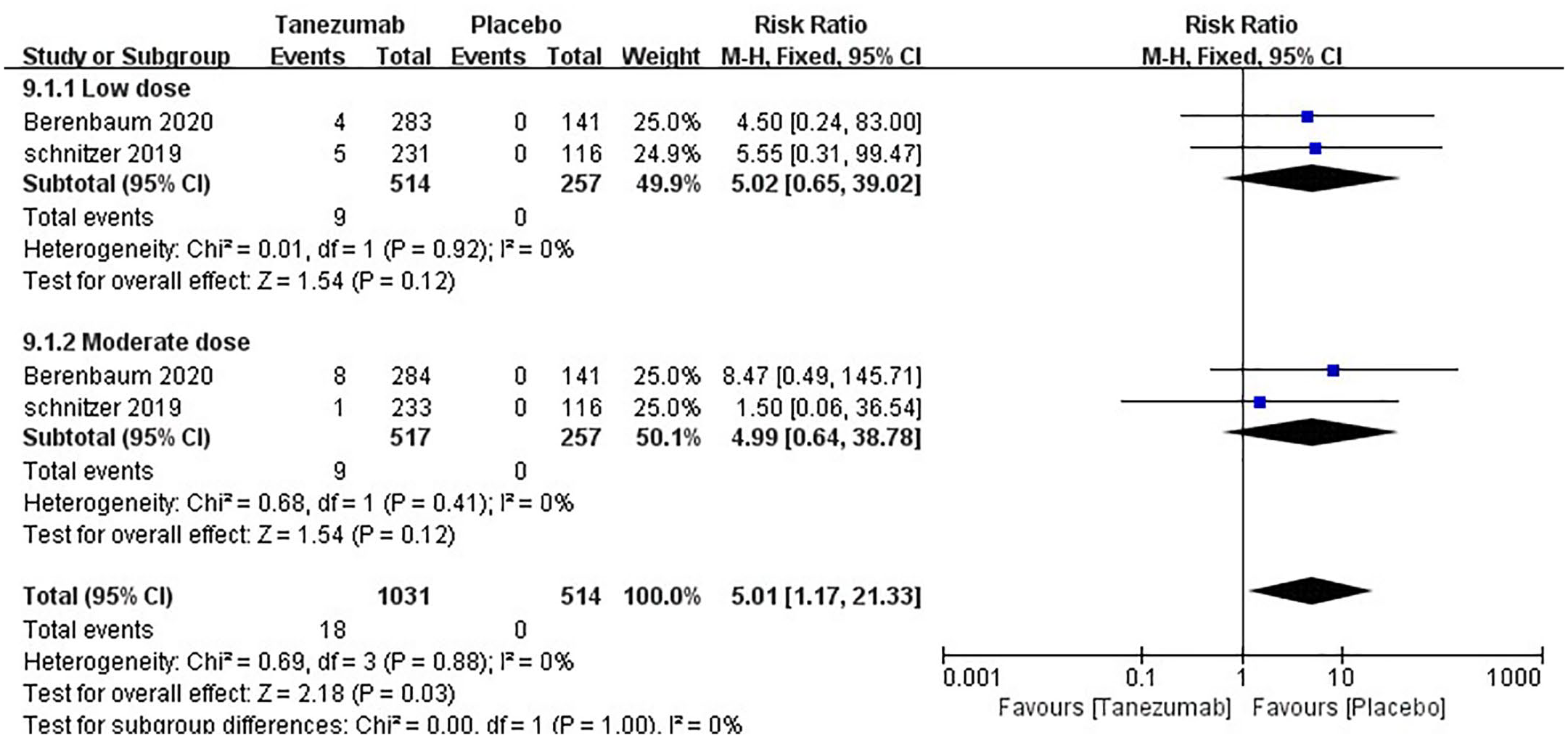
Forest plot of rapidly progressive OA detailing relative risk and 95% CIs comparing tanezumab with placebo.
Total joint replacement: A total of four RCTs (2525 patients) reported total joint replacement results. The results of the pooled analysis showed that there was no significant difference in the incidence of total joint replacement between the tanezumab and placebo groups (RR = 1.33, 95% CI = 0.87–2.04, p = 0.19, I2 = 0%). In the subgroup analysis, four studies were included in the low-dose group, and four studies were included in the moderate-dose group. The pooled analysis results showed no significant differences in the incidence of total joint replacement between the low-dose tanezumab and placebo groups (RR = 1.18, 95% CI = 0.64–2.16, p = 0.60, I2 = 0%). Similarly, there was no significant difference in the incidence of total joint replacement between the moderate-dose tanezumab and placebo groups (RR = 1.49, 95% CI = 0.82–2.72, p = 0.19, I2 = 21%; Table 2). There was no significant difference between the two treatment groups (p = 0.59, I2 = 0%).
Abnormal peripheral sensation: A total of 11 RCTs (4372 patients) reported abnormal peripheral sensation results. The results of the pooled analysis showed that the incidence of abnormal peripheral sensation was significantly higher in the tanezumab group than in the placebo group (RR = 2.42, 95% CI = 1.84–3.18, p < 0.00001, I2 = 16%). In the subgroup analysis, 7 studies were included in the low-dose group, and 11 studies were included in the moderate-dose group. The pooled results for the low-dose group showed that the incidence of abnormal peripheral sensation was significantly higher in the tanezumab group than in the placebo group (RR = 2.00, 95% CI = 1.21–3.29, p = 0.006, I2 = 0%). Similarly, moderate doses of tanezumab also led to a higher risk of abnormal peripheral sensation than placebo (RR = 2.64, 95% CI = 1.90–3.66, p < 0.00001, I2 = 44%; Table 2). There was no significant difference between the two treatment groups (p = 0.35, I2 = 0%).
Active-controlled trials
Efficacy
WOMAC pain score: A total of four RCTs (2203 patients) reported WOMAC pain scores. The results of the pooled analysis showed that moderate doses of tanezumab relieved pain significantly more than the active comparator did (MD = –0.62, 95% CI = –0.85 to −0.39, p < 0.00001, I2 = 0%; Supplemental Appendix Figure A3). However, the point estimates (MD) of the moderate-dose tanezumab group were not greater than the MCID (0.97).
WOMAC physical score: A total of four RCTs (2219 patients) reported WOMAC physical scores. Pooled analysis showed that moderate doses of tanezumab resulted in greater improvements in the WOMAC physical score than did the active comparator (MD = −0.70, 95% CI = −0.92 to −0.47, p < 0.00001, I2 = 0%; Supplemental Appendix Figure A4). However, the point estimates (MD) of the moderate-dose tanezumab group were not greater than the MCID (0.93).
PGA: A total of four RCTs (2218 patients) reported PGA results. The pooled estimate of PGA demonstrated significant differences in favor of moderate doses of tanezumab (MD = –0.20, 95% CI = −0.36 to −0.05, p = 0.008, I2 = 70%; Supplemental Appendix Figure A5).
Safety
Withdrawals due to AEs: A total of four RCTs (2233 patients) reported withdrawals due to AEs. Pooled analysis showed no significant difference between moderate doses of tanezumab and the active comparator in terms of withdrawals due to AEs (RR = 0.54, 95% CI = 0.20–1.40, p = 0.20, I2 = 83%; Supplemental Appendix Figure A6).
SAEs: A total of four RCTs (2233 patients) reported SAE results. The results of the pooled analysis showed no significant differences in the incidence of SAEs between the moderate-dose group and active-comparator group (RR = 0.95, 95% CI = 0.67–1.34, p = 0.76, I2 = 3%; Supplemental Appendix Figure A7).
Discussion
The efficacy of tanezumab in treating OA has been assessed in some systematic reviews. Schnitzer and Marks 34 reported that the anti-NGF antibody was effective in treating hip or knee OA, and found that high doses (10 mg, and 100 or 200 µg/kg) of tanezumab showed greater efficacy than a placebo but were accompanied by a higher incidence of adverse reactions. Chen et al. 35 conducted a meta-analysis of studies on tanezumab in the treatment of hip or knee OA, and found that low and high doses of tanezumab had similar effects in relieving pain and improving function. However, the use of tanezumab significantly increased the incidence of withdrawals due to AEs. Similarly, Kan et al. 36 also found in their meta-analysis that the use of tanezumab could significantly relieve pain and improved function but increased the incidence of withdrawals due to AEs. However, when analyzing the incidence of AEs, the authors mixed different doses (low to high) of tanezumab and mixed studies using tanezumab monotherapy and tanezumab + other active interventions. We conducted a subgroup analysis of only low- and moderate-dose tanezumab monotherapy and did not find that tanezumab increased the incidence of withdrawals due to AEs. Therefore, high-dose tanezumab and tanezumab + other active interventions are likely to be responsible for the increased incidence of withdrawals due to AEs. The FDA also found these issues; as a result, the FDA suggests limiting the use of these drugs. Therefore, the focus of this study was to evaluate the efficacy and safety of low- and moderate-dose tanezumab monotherapy in the treatment of hip or knee OA, as well as to define the optimal dose to maximize benefits and minimize risks.
The results of this meta-analysis showed that low and moderate doses of tanezumab significantly relieved pain and improved function, and there were no significant differences in the incidence of treatment-related AEs, withdrawals due to AEs, serious AEs, and total joint replacement between the tanezumab and placebo groups; however, tanezumab increased the risk of AEs and reversible peripheral sensory abnormalities. To more accurately study the clinical efficacy of tanezumab in the treatment of OA, we also introduced the MCID value for detailed evaluations. The clinical results based on the MCID values showed that the point estimates (MD) of moderate-dose tanezumab versus a placebo exceeded the MCID of the WOMAC pain and WOMAC physical function scores. However, the point estimates (MD) of low-dose tanezumab versus placebo were not greater than the MCID values. Thus, the clinical effect of moderate-dose tanezumab is more significant than that of low-dose tanezumab. However, the incidence of adverse reactions was similar between the two groups with different doses, and the incidence of adverse reactions did not increase with increasing dose.
There were four RCTs comparing the efficacy of moderate-dose tanezumab therapy and active-comparator therapy (two RCTs used naproxen, one RCT used oxycodone, and one RCT used naproxen or celecoxib). The final results showed that the therapeutic effects of tanezumab were significantly larger than those of the active comparator, but the MDs were smaller than those of tanezumab versus the placebo, and the point estimates (MD) were not greater than the MCID values for WOMAC pain and WOMAC physical function scores. In terms of safety, there were no significant differences between tanezumab and the active comparator.
The remarkable efficacy of tanezumab in the treatment of hip or knee OA is very promising. For moderate-to-severe OA of the hip or knee for whom standard analgesics were not effective or could not be taken, the onset of efficacy of subcutaneous tanezumab was within the first week, and efficacy was maintained through the 24-week treatment period. 37 However, because of safety risks and an inadequate plan to manage them, the European Medicines Agency and the FDA adopted a negative opinion for the tanezumab marketing authorization application. Therefore, its safety is still the current focus of attention. We evaluated the safety of low and moderate doses of tanezumab in the treatment of hip or knee OA from several aspects. Overall, the incidence rates of treatment-related AEs, withdrawals due to AEs, and SAEs with low and moderate doses of tanezumab were low, and there were no significant differences between the tanezumab groups and placebo groups or active-comparator groups. Although the incidence of AEs was higher in the tanezumab group than in the placebo group, the RR values were relatively small, and the RR values of tanezumab at low doses were similar to those at moderate doses. Moreover, in the assessment of individual adverse reactions, there was no significant difference in the incidence of total joint replacement between tanezumab and the placebo.
Two studies reported RPOA, and there was no significant difference between tanezumab and the placebo in the subgroup analysis of low or moderate doses. But when the doses were combined, the use of tanezumab significantly increased the incidence of RPOA. At present, the pathological mechanism of RPOA caused by NGF inhibitors is still unclear. Some scholars have reported that NGF can promote tissue repair, and the increased expression of NGF in chondrocytes of osteoarthritic articular cartilage may play an important role in preventing harmful changes by regulating the proliferation and differentiation of chondrocytes. Therefore, NGF inhibitors may adversely affect bone remodeling, resulting in an increased risk of RPOA.38,39 Patients with subchondral insufficiency fractures before treatment are at a greater risk of developing RPOA. 40 In addition, NSAIDs may also inhibit subchondral microfracture repair, which may explain why the simultaneous use of NSAIDs may increase the risk of AEs associated with anti-NGF therapy in patients with joint failure. 41 However, it is worth noting that the two RCTs25,30 that reported the incidence of RPOA excluded predetermined imaging evidence of specific bone or joint safety (such as RPOA, atrophic or malnourished OA, subchondral fracture, spontaneous knee joint necrosis, osteonecrosis or pathological fracture, and patients with long-term use of NSAIDs). However, the final results of the meta-analysis showed that there was still an increased RPOA risk. Another explanation is that the analgesic effect of anti-NGF drugs is obvious, which may increase the load of the degenerative part of the involved joint, thus promoting the occurrence of RPOA. However, joint failure also occurs in non-weight-bearing joints, including the shoulders, and in joints other than the index joints evaluated in clinical studies. 8 Some scholars also believe that in most cases, the destructive arthropathy that leads to rapidly progressive joint injury may be a neuropathic joint disease since NGF may play a role in the regulation of neurosensory neurons. 42 Although the available data cannot completely rule out this possibility, there is a lack of evidence of severe neuropathy and loss of protective sensation in weight-bearing joints.40,42 As joint-related adverse reactions include clinical entities with different radiological characteristics, there may be a variety of mechanisms contributing to the development of RPOA. 43 Therefore, more research is needed to explore the causes of RPOA.
In terms of abnormal peripheral sensation, our results showed that compared with a placebo, both low and moderate doses of tanezumab could significantly increase the incidence of abnormal peripheral sensation (including abnormal sensation, hypoesthesia, and hypersensitivity). However, the severity of this condition is usually rated as mild or moderate, and most of these cases are short-lived and do not have any permanent sequelae. Most commonly, this sensory disorder disappears within 1 to 2 weeks or 1 month after the first dose.23,28,44,45,46 In general, most patients with a final neurological diagnosis of new or worsening peripheral neuropathy are diagnosed with some form of mononeuropathy (mainly carpal tunnel syndrome) or radiculopathy based on clinically significant signs or diagnostic tests. Very few patients are diagnosed with multiple neuropathies.28,29,32,44,45,47 These results do not reflect the expected pattern for neurotoxic compounds, which typically cause prolonged durations of multiple neuropathies in affected patients. The association of tanezumab with mononeuropathy symptoms suggests that these manifestations may be the result of the inhibitory effect of NGF.31,48 An RCT that evaluated the safety of tanezumab in the treatment of OA showed that when tanezumab was used to treat chronic pain in individuals without known peripheral neuropathies, it was not associated with structural neurotoxic effects on large motor or sensory nerves, autonomic nerves, or small sensory nerve fibers in the skin. 27 Therefore, neurosensory symptoms induced by anti-NGF therapy may be caused by reversible functional changes or homeostasis in peripheral nerve activity. A short-term follow-up study of tanezumab in the treatment of two types of peripheral neuropathic pain also showed that patients’ neurological condition did not deteriorate with treatment. 49 In addition, no significant adverse reactions of tanezumab were found in other assessments, such as cardiovascular, liver, renal function, gastrointestinal, and other types of laboratory tests.22,31 Therefore, tanezumab is generally safe. Cost-effectiveness analysis showed that the pain relief provided by anti-NGF therapy was significant enough that even rapid progressive OA, which was present in 10% of patients, did not offset the overall improvement in quality-adjusted life years achieved 50 and that the cost of anti-NGF therapy might be as low as $400 per dose. 50 Of course, it should be noted that these analyses are based entirely on models that used arbitrary values of the costs related to pain and are intended to inform policy makers rather than clinicians, as individuals have significantly different perspectives on risks and benefits. 51
At present, the existing research on tanezumab in the treatment of hip or knee OA is relatively extensive; thus, the total sample size studied is relatively large, and the final results are precise. Moreover, the homogeneity of the whole study is relatively good. However, some shortcomings of this article are still noteworthy. First, our study is based on only English articles and does not include non-English studies, so the results may be affected by publication bias. However, Egger’s test results indicate that there is no risk of publication bias. For the risk assessment of RPOA, only 2 studies were included, and more studies still need to be conducted in the future. Finally, there is a known potential risk, and all of the studies included were sponsored by pharmaceutical companies.
Conclusion
Based on the current research evidence, lower doses of tanezumab monotherapy can significantly improve pain, function, and PGA in patients with hip or knee OA. Tanezumab is relatively well tolerated and safe, but increases the incidence of AEs and reversible abnormal peripheral sensation. The occurrence of RPOA is still worthy of attention, and more studies are needed to explore this aspect. In terms of dose selection, moderate doses of tanezumab have a more pronounced clinical effect than low doses of tanezumab, and the safety and tolerance were similar between the two doses, with no significant differences. Therefore, a moderate dose of tanezumab may maximize the benefits for hip or knee OA.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X211067639 – Supplemental material for Based on minimal clinically important difference values, a moderate dose of tanezumab may be a better option for treating hip or knee osteoarthritis: a meta-analysis of randomized controlled trials
Supplemental material, sj-docx-1-tab-10.1177_1759720X211067639 for Based on minimal clinically important difference values, a moderate dose of tanezumab may be a better option for treating hip or knee osteoarthritis: a meta-analysis of randomized controlled trials by Di Zhao, Ming-hui Luo, Jian-ke Pan, Ling-feng Zeng, Gui-hong Liang, Yan-hong Han, Jun Liu and Wei-yi Yang in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (No. 81873314, No. 81473698, No. 82004386, No. 82004383), the Science and Technology Research Project of Guangdong Provincial Hospital of Chinese Medicine (No. YN2019ML08, No. YN2015MS15), Science and Technology Program of Guangzhou (No. 202102010273), the Project of Guangdong Provincial Department of Finance (No. [2014]157, No. [2018]8), and the Science and Technology Planning Project of Guangdong Province (No. 2020A1414050050).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
