Abstract
Aims:
Medical and non-medical switching strategies have been adopted in Europe in the last few years. We aimed to investigate persistence on treatment with a SB5 Adalimumab (SB5) biosimilar after switching from Adalimumab (ADA) originator among patients with inflammatory rheumatic musculoskeletal diseases (iRMD), identifying possible predictors of drug interruption and describing adverse events.
Method:
iRMD patients previously switched to SB5 after at least 6 months of ADA were enrolled. Data on concomitant medications, disease flares, and persistence on SB5 up to the last available follow up were collected retrospectively. Kaplan–Meier and Cox regression models were used.
Result:
A total of 172 patients (106 females, ADA duration 5.8 ± 3.8 years) were enrolled, including 34 rheumatoid arthritis, 59 psoriatic arthritis, and 61 axial spondyloarthritis patients. In a 10 ± 3 months follow up, 65 (37.8%) patients presented with adverse events, with 46 (26.7%) showing a clinically defined disease flare (no disease activity and patient reported outcomes assessment were available); 24 patients interrupted SB5 permanently (among them, 11 back-switched to ADA and 8 were prescribed a different biological therapy). Probability of persistence on SB5 was 94.7% at 6 months and 85.1% at 12 months. Baseline corticosteroid [hazard ratio (HR) 3.209, 95% confidence interval (CI) 1.193–8.635, p = 0.021] and therapy with nonsteroidal anti-inflammatory drugs (NSAIDs) (HR 2.876, 95% CI 1.229–6.727, p = 0.015), as well as the baseline corticosteroid dose (HR 1.200, 95%CI 1.026–1.403, p = 0.022) were predictors of drug interruption.
Conclusion:
Our data on persistence of treatment and adverse events are in line with previous reports. Further large cohort studies may confirm baseline corticosteroid and NSAIDs use as predictors of SB5 interruption, helping to identify patients at higher risk of failure after switching.
Introduction
Adalimumab (ADA) is a recombinant human immunoglobulin G1 (IgG1) specifically designed to bind and block tumor necrosis factor alpha (TNFi). 1 ADA, as well are other biologic disease modifying anti-rheumatic drugs (bDMARDs), is recommended for the treatment of inflammatory rheumatic musculoskeletal disorders (iRMD), i.e., rheumatoid arthritis (RA), psoriatic arthritis (PsA), axial spondyloarthritis (axSpA – both radiographic and non-radiographic), and juvenile idiopathic arthritis (JIA), as well as other non-primarily rheumatological conditions.2–5 A biosimilar drug is defined as a biological product highly similar to its originator molecule, without meaningful differences in terms of purity, quality, and efficacy. Given the growing request for advanced treatments for the management of iRMD, different ADA biosimilars have been developed in the last decade in order to reduce the economic burden on the public health care system.
The biosimilar SB5 (Imraldi®) was developed as an ADA biosimilar and received European Medicines Agency (EMA) marketing approval in August 2017, having an identical amino-acid sequence as well as similar physicochemical and in vitro functional properties to ADA. 6 Equivalence in pharmacokinetics features was demonstrated in a phase I study on 189 healthy subjects, 7 while its clinical efficacy was demonstrated in a phase III randomized clinical trial including RA patients at 24- and 52-week follow up.8,9 Switching from ADA to SB5 was demonstrated not to impact the efficacy and immunogenic profile of RA patients, 9 and short-term real-life data confirmed efficacy and results in inflammatory bowel diseases (IBD), plaque psoriasis, PsA patients switched to SB5, as well as in mixed cohorts of iRMD.10–12
The aim of the present study was to report our real-life experience with switching from ADA to SB5 over a follow-up period up to 18 months in a cohort of patients attending third-level rheumatology centers in Tuscany, Italy. In details, our primary objectives were the measurement of persistence on treatment with SB5 and the identification of predictors of SB5 therapy interruption. As a secondary objective, we wanted to identify the types of adverse events (AEs) reported and their prevalence.
Materials and methods
Adult patients affected by RA, PsA, AS, JIA, and other rheumatologic diseases treated with ADA for at least 6 months, who had undergone medical or non-medical switching from ADA to SB5 while in clinically stable condition, were enrolled in the study. The procedure of switching had been performed mainly for non-medical reasons, as previously reported. 13 Each patient had been informed during a clinical consultation from the treating physician regarding both medical and non-medical aspects of the practice, presenting both data from the literature and previous personal experience with biosimilar drugs. Exclusion criteria were age <18 years at the time of switching, treatment with ADA lasting for less than 6 months, and lack of follow-up visits after switching. All enrolled patients signed informed consent for data collection and analysis and the study received local Institutional Review Board (IRB) approval (CAEVC 15659).
Demographic data (diagnosis, age, gender, disease duration), previous and concomitant iRMD treatments [bDMARDs, conventional synthetic disease modifying anti rheumatic drugs (csDMARDs), corticosteroids (CCS), non-steroidal anti-inflammatory drugs (NSAIDs)] were collected. Clinimetric disease activity and patient-reported outcomes were also collected from patients’ files.
AEs, defined as undesirable experiences associated with the use of a medical product in a patient exposed to it, were also recorded if reported during the study period. We categorized them into local and systemic reactions to the drug, the latter including also clinically defined articular, cutaneous, gastrointestinal, and ocular disease flares. Data collection covered the time from the date of switching from ADA originator to SB5 (baseline) to latest available follow up, defined as the most recent date while on SB5 therapy or the date of SB5 discontinuation.
Mean and standard deviation (SD) are reported for each continuous variable, while absolute frequencies and percentages are presented for categorical variables. We evaluated the change from baseline to follow up of categorical variables using the McNemar test, while the change in continuous variables with Student’s t test for paired samples.
Persistence on treatment, defined as continuation of SB5 therapy at the time of the last follow up, was represented graphically using Kaplan–Meier curves. The probability of treatment persistence at 6 and 12 months and the respective 95% confidence interval (CI) were calculated using Kaplan–Meier estimation.
Potential treatment interruption predictive factors, including type of iRMD, first line versus subsequent bDMARD therapy lines, monotherapy versus csDMARD combination therapy, gender, age, disease duration, ADA therapy duration, local and systemic AEs, baseline use of NSAIDs, baseline use and dosage of steroids were analyzed. Log-rank test and Kaplan–Meier curves were used to test the association between survival and baseline factors, while Cox univariate regression model was performed to calculate the hazard ratio (HR) and its 95% CI. We also analyzed the interaction between type of iRMD and concomitant medications, still in predicting SB5 treatment interruption, using a multiple Cox model with interaction term.
Results
A total of 172 patients (106 females, 61.6%, mean age 53.3 ± 15.5 years, mean disease duration 13.2 ± 7.6 years) attending the Rheumatology outpatient clinics of the Florence and Siena University Hospitals were eligible for our study.
The study population was: PsA (n = 59, 34.3%), axSpA (n = 61, 35.5%), RA (n = 34, 19.8%), JIA (n = 11, 6.4%), and 7 (4.1%) patients with other diagnosis (5 Behçet disease and 2 idiopathic uveitis). The patients had received ADA for an average duration of 5.8 ± 3.8 years prior to switching to SB5 and it represented the first, second, and third line of bDMARD therapy in 108 (62.8%), 48 (27.9%), and 16 (9.3%) patients, respectively.
As presented in Table 1, 79 (45.9%) patients received combination treatment with csDMARDs, mostly methotrexate (n = 40, 23.9%), followed by sulfasalazine (n = 25, 14.5%); one out of four patients was also receiving cyclic/chronic NSAIDs or CCS co-treatment.
Concomitant medications profile at baseline and last available follow up.
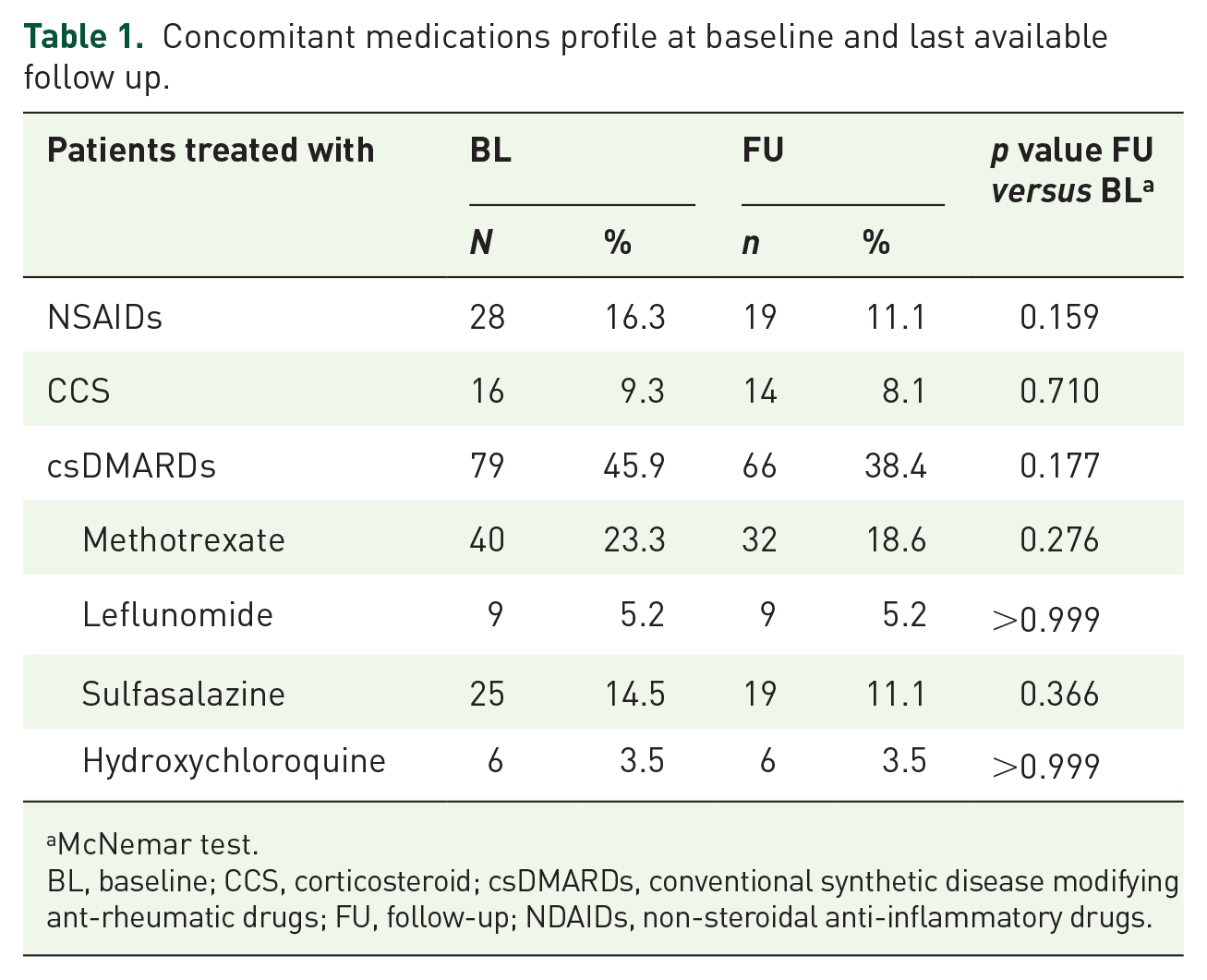
McNemar test.
BL, baseline; CCS, corticosteroid; csDMARDs, conventional synthetic disease modifying ant-rheumatic drugs; FU, follow-up; NDAIDs, non-steroidal anti-inflammatory drugs.
CCS and NSAIDs, as well as csDMARDs, were used with a variable trend among the different diseases, as expected (see Supplemental Table S1). In particular, CCS and methotrexate were used more frequently in RA and PsA patients, while NSAIDs and sulfasalazine were used in PsA and axSpA. In a 10 ± 3 months average observation from baseline to follow up, we observed an overall numerical reduction of CCS, NSAIDs, and csDMARDs users, although no statistically significant difference was detected (Table 1). In addition, the mean CCS dosage did not change significantly from baseline to the end of observation (from 2.93 ± 3.21 to 3.45 ± 4.65 mg/day, p = 0.704).
Given the heterogeneity of the diseases and the consistent amount of missing data in both disease activity and patient-reported outcomes assessment, these data were not used for analysis and, therefore, no efficacy assessment was performed.
Treatment persistence
At the 6 months timepoint, 8/150 patients interrupted SB5, 13/86 additional patients interrupted the bDMARD at 12 months, while 3 interruptions were recorded after the first year of treatment.
The probabilities of persistence on SB5 treatment in our cohort at 6 and 12 months follow up were 94.7% (95% CI 90.1–97.2) and 85.1% (95% CI 77.9–90.2), respectively (Figure 1).

Survival curve of persistence on SB5 treatment up to 12 months.
Overall, 24 patients discontinued SB5 treatment (see Figure 2): disease flares determined SB5 interruption in 18 cases, with other AEs (no disease flares) in the remaining 6 patients. After SB5, 11 were back-switched to ADA, 8 were swapped to another bDMARDs or small molecule (1 Etanercept, 4 Golimumab, 1 Apremilast, 1 Infliximab, and 1 Secukinumab), 3 were managed with NSAIDs/CCS cycles, and 2 with csDMARD monotherapy. At last available follow- up, disease control was regained in 16/24 cases, including all patients back-switched to ADA (Table 2).
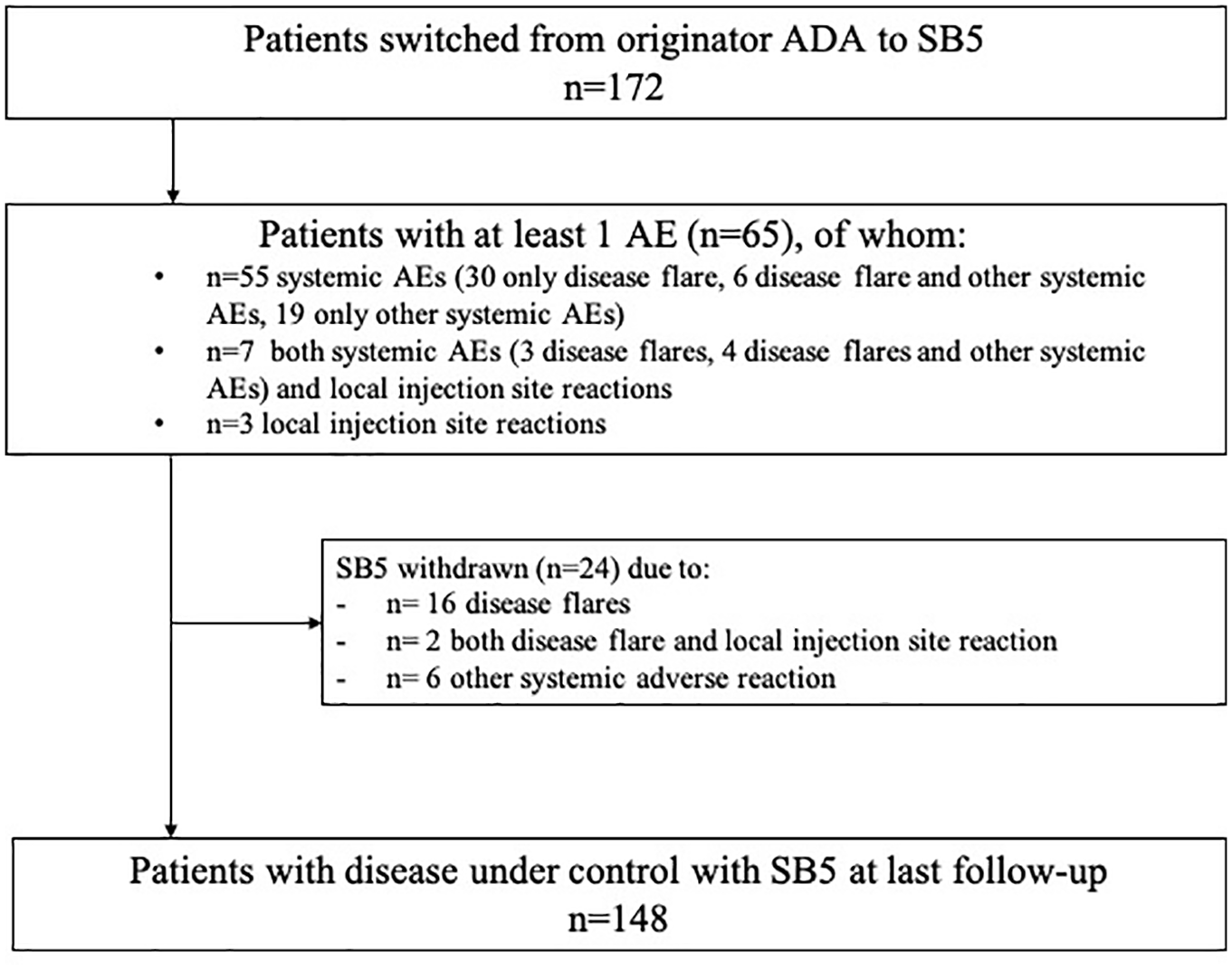
Description chart of population, AEs, and reason for SB5 interruption.
Features of patients back-switched to ADA originator and their clinical outcome.

ADA, adalimumab; AE, adverse event; axSpA, axial spondylarthritis; PsA, psoriatic arthritis; RA, rheumatoid arthritis; SB5, ADA biosimilar; SSZ, sulfasalazine.
Predictors of SB5 interruption
Among tested factors, baseline therapy with NSAIDs (HR 2.876, 95% CI 1.229–6.727, p = 0.015) and CCS (HR 3.209, 95% CI 1.193–8.635, p = 0.021), together with baseline CCS dose (HR 1.200, 95% CI 1.026–1.403, p = 0.022), were seen as significant predictive factors of SB5 treatment interruption (Figures 3 and 4). None of the other variables was associated with SB5 interruption.

Survival curve of persistence on SB5 treatment up to 12 months, comparing patients treated, or not, with NSAIDs at baseline.
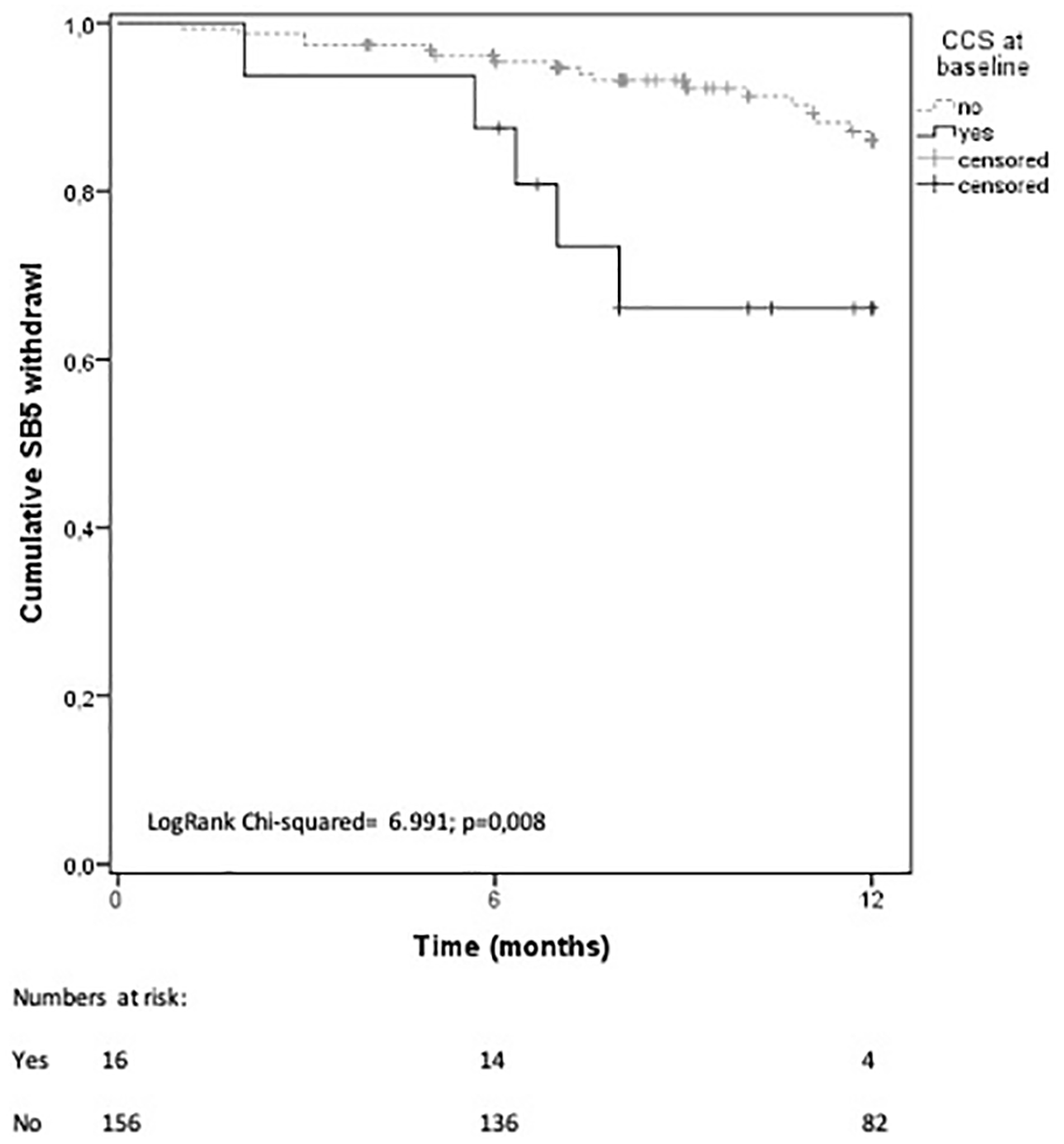
Survival curve of persistence on SB5 treatment up to 12 months, comparing patients treated, or not, with CCS at baseline.
Adverse events
A total of 65 patients (37.8%) experienced at least one AE, including 1 (0.6%) serious AE (severe infection requiring hospitalization). In this cohort, 10 (5.8%) patients complained about local injection site reactions, 62 (33.7%) about at least one systemic AE, with 7 (4.1%) reporting both.
In detail, 19/62 patients experienced infectious complications, 10/62 complained about constitutional AEs, 3/62 experienced alterations of their laboratory serological profile (liver function tests increase), and 3/62 of other non-serious events; 46 patients reported a loss of SB5 treatment efficacy, resulting in 42 articular, 7 cutaneous, 5 gastrointestinal, and 3 ocular disease flares, in a variable combination. Among the cases with clinically defined disease flares, 17 patients (37%) were treated with temporary NSAIDs/CCS cycles, 6 (13%) with reduction of SB5 dosing interval, 3 (6.6%) were managed with a wait and see approach and 7 (15.2%) were given csDMARD, either in terms of adding the drug or increasing the dosage. All these changes allowed the return to a satisfactory disease control in 28/33 cases. For the remaining 13 cases, SB5 was interrupted at the time of disease flare (see Figure 2).
Discussion
Our data show that ADA/SB5 switched iRMD patients maintain a satisfactory level of persistence in treatment, with loss of efficacy as the most frequent cause for drug discontinuation. Moreover, concomitant NSAIDs or CCS baseline treatment were identified as predictive factors of treatment discontinuation.
SB5 retention rate and safety profile have been tested previously in several studies. In the phase III clinical trial conducted by Weinblatt et al., 8 SB5 retention rate was observed to be 93.6% after 28 weeks follow up, similar to those who had not switched (96.1%) and comparable with what we found out in our cohort study. Similarly, the safety profile was also comparable: 37.6% versus 33.1% patients with at least one AE in switched and non-switched patients, respectively. This is in line with 37.8% cases with AEs seen in our population.
Other real-life short-duration cohort studies showed similar results: in a 10-week follow up evaluation including 93 IBD patients switched from ADA to SB5, no meaningful difference in clinical outcome and safety profile was found when comparing switched patients with the originator cohort. 10 Furthermore, Di Cesare et al. reported no significant safety issues in plaque psoriasis without associated arthritis and PsA patients, 11 even if a direct comparison with our results cannot be made due to the very small numbers of patients in the SB5 switch cohort and the short follow-up period. In addition, they observed discontinuation of SB5 therapy in 1/12 PsA and in 2/20 plaque psoriasis patients after 12 weeks follow up. In our cohort, discontinuation rates were similar in the short-term 6-month assessment, with 5.3% discontinuations, despite an increase in the longer follow up, which still remained satisfactory in terms of treatment persistence. Moreover, besides SB5, our results were comparable with those found in literature for other ADA biosimilars such as ZRC-3197, ABP501, and BI 695501.14–18
In addition to other evidence on SB5, our data are also supported by evidence available for the persistence on treatment with ADA derived from national cohorts. In particular, Iannone et al. showed data on the survival of ADA treatment used as a first line bDMARD in a large multi-center RA population. 19 At 3 years, ADA retention rate was 40.6%, with a relative 15–20% drug interruption per year. Similar numbers were also confirmed by Favalli et al., 20 showing 10-year treatment retention with ADA in 23.5% of a mixed population of RA, PsA, and axSPA. Although these populations were different in nature, representing mainly bDMARD-naive patients, we may indirectly consider these numbers as in line with our population, showing 85.1% probability of SB5 continuation after 1 year from switching, therefore supporting the concept of a continuum started with ADA treatment initiation.
Our study identified baseline NSAIDs or CCS co-treatments as predictive factors for bDMARDs discontinuation in SB5 switched patients. Glintborg et al. previously tested predictive factors for biosimilar SB4 etanercept treatment discontinuation 21 : in this cohort, RA diagnosis, treatment with originator Etanercept for less than 1 year and absence of disease remission at switch timepoint were the only significant predictors, without significant risk for NSAIDs and CCS baseline concomitant treatment. Despite the lack of data on disease activity and patient reported outcome, our current data may indirectly support the hypothesis of non-biosimilar related features, such as demographics, disease status and concomitant medications, being involved in the interruption of the biosimilar medication during follow up. In this light, we may consider that the baseline use of NSAIDs or CCS could represent a surrogate for sub-optimal disease control, which could have possibly led the patient to a disease flare or ADA interruption in a similar time. In this view, our data may confirm the results of Glintborg et al. in terms of disease non-remission at the time of switch as a predictor of biosimilar interruption. 21 Given the lack of clinimetric assessments allowing the quantification from either the patient or the physician perspective, we could not support these data with more robust analysis. Despite this, there is a clear signal of the need for alternative approaches to patients in non-complete disease control, who would possibly not benefit from either continuing the originator drug or switching to the biosimilar compound.
As previously presented also for other compounds, back-switching to the originator bDMARD is possible and frequently allows a return to the previous status of disease control. 13 In this light, we might not exclude a possible impact of the nocebo effect. Although the registered loss or lack of therapeutic effect were based on the physician clinical judgment and evaluation, this could have been influenced by the increase in patient-reported pain, morning stiffness, or joint tenderness. Again, the lack of disease activity and patient-reported outcome in our cohort could has not allowed us to clearly identify patient-reported versus clinical/clinimetric measurable disease flares.
To the best of our knowledge, our study describes the largest real-life cohort of patients switched to SB5, including all iRMD for which SB5 has been licensed and with a longer follow-up observation in comparison with the literature. In addition, we identified predictors of treatment interruption. On the other hand, some limitations should be addressed: first, the lack of physician activity indexes and patient-reported outcomes has not allowed us to present efficacy data to support our indirect evidence of sub-optimal disease control at baseline as a predictive factor of SB5 interruption during follow up. Moreover, the observational retrospective nature of the study might have impaired the collection of data in terms of ongoing medications and AEs, as well as determined the non-eligibility of patients without baseline/follow-up data. The lack of a non-switched group as a direct comparator was indeed another limitation of our analysis; unfortunately, this was not possible as it was not part of the study protocol approved and due to non-accessibility of medical charts of historical patients treated with ADA. Finally, no specific definition for disease control using clinimetric scale was used, leaving the attribution of clinical status to the treating physician. This absence of standard inclusion criteria might have biased patient selection and our result. Despite this, our manuscript is representative of a real-life scenario, in which medical and non-medical switching were performed simultaneously and confirmed successful also for other compounds. 13
In conclusion, our study confirms the high value of SB5 persistence on treatment at 6 and 12 months after switching from ADA, with a safety profile in line with the current literature. Our results need to be confirmed in larger real-life studies and initiatives, in particular regarding the potential increased risk of treatment interruption in patients who were treated at baseline with NSAIDs and CCS, and the need of awareness of higher risk of failure in patients in sub-optimal disease control at the time of switching. 22
Supplemental Material
sj-docx-1-tab-10.1177_1759720X211033679 – Supplemental material for Switching from originator adalimumab to biosimilar SB5 in a rheumatology cohort: persistence on treatment, predictors of drug interruption and safety analysis
Supplemental material, sj-docx-1-tab-10.1177_1759720X211033679 for Switching from originator adalimumab to biosimilar SB5 in a rheumatology cohort: persistence on treatment, predictors of drug interruption and safety analysis by Cosimo Bruni, Stefano Gentileschi, Giovanni Pacini, Marco Bardelli, Lorenzo Tofani, Francesca Bartoli, Caterina Baldi, Laura Cometi, Ginevra Fiori, Francesca Nacci, Luca Cantarini, Serena Guiducci, Alberto Moggi-Pignone, Bruno Frediani and Marco Matucci-Cerinic in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Author contributions
All authors contributed to the study conception, design, and interpretation of results. Material preparation, data collection, and analysis were performed by CB, SG, GP, and LT. The first draft of the manuscript was written by GP and CB, all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Conflict of interest statement
MMC reports speaker’s and advisory board honoraria from Biogen Italia.
All other authors declare no conflict of interest with the scientific content of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
