Abstract
Background:
Recent surveys in chronic inflammatory rheumatic diseases (CIRD) showed a high degree of vaccine hesitancy. Current knowledge about patients’ attitudes toward vaccination against SARS-CoV-2 is limited.
Objectives:
To assess the willingness of CIRD patients to be vaccinated against SARS-CoV-2 and to identify the influencing factors compared with non-CIRD patients.
Methods:
In this cross-sectional study, two cohorts of consecutive patients with and without CIRD were recruited in parallel when presenting to our tertiary hospital and asked to answer questions of a structured interview to assess vaccination willingness to SARS-CoV-2 their experience with SARS-CoV-2 and their personal history of infections and vaccinations. Vaccination willingness was assessed using a numerical rating scale (0: fully disagree; 10: fully agree). Arbitrarily defined cut-offs were used to define definite (score ⩾7) and probable willingness (score of 5 or 6) to be vaccinated. Factors associated with willingness were assessed using Kendall’s tau-b correlation measure and linear regression analysis.
Results:
A total of 514 CIRD and 100 non-CIRD patients, mean age of 54.7 ± 12.8 and 55.6 ± 9.8 years, respectively, were included. Definite and probable willingness to be vaccinated against SARS-CoV-2 was declared by 79.6% and 90.7% versus 76.0% and 85.0% of CIRD and non-CIRD patients, respectively. Only 60% of CIRD patients believed that the vaccines against SARS-CoV-2 were safe, and 42% indicated to be afraid of side effects. Vaccination willingness was significantly correlated with being in a risk group for COVID-19 (tau-b = −0.149), hypertension (tau-b = 0.14), and information about disease prevention (tau-b = 0.19), while a history of infections or immunosuppressive therapy was not. Vaccination willingness was significantly associated with higher education (b = 0.65) and age (b = 0.06).
Conclusion:
This survey highlights several predictors of relevance for the vaccination willingness of patients with CIRD and controls including appropriate information about its relevance. The good news, however, is that the vast majority of CIRD patients indicated their willingness to be vaccinated. However, there was some uncertainty regarding the safety and efficacy of the vaccines. Since the major influencing factors were education and information about SARS-CoV-2 Vaccine and COVID-19 Disease, patient education should be improved soon.
Introduction
The current pandemic, announced by the World Health Organization (WHO) on 11 March 2020, caused by the novel beta severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and its mutations 1 has, according to the WHO, meanwhile killed several million people worldwide. Whether patients with chronic inflammatory rheumatic diseases (CIRD) are at higher risk for a severe course of COVID-19 infections than the general population was unclear, but the known risk factors male sex, age, and pulmonary and cardiovascular disease are also relevant for patients with CIRD, such as rheumatoid arthritis (RA), axial spondyloarthritis (axSpA), psoriasis arthritis (PsA), and connective tissue diseases (CTD). 2 An increased risk of infections associated with both, the disease itself and immunosuppressive medication, has been demonstrated.3,4 Unfavorable outcomes of COVID-19 have been shown to be associated with medication, especially rituximab, but also JAK inhibitors and higher doses of corticosteroids.5,6
Vaccinations should be offered to all patients with CIRD, especially to those on or before immunosuppressive therapy. 7 However, the vaccination rate against pneumococcal infections and influenza recently tested in our center even before the pandemic started had a rather disappointing result: less than 50% of CIRD patients were vaccinated against pneumococci and influenza. 8 The German government has arranged national shutdowns and other regulatory activities including the vaccination campaign starting in January 2021. Vaccination against the SARS-CoV-2 is strongly recommended worldwide as a major disease control measure.
Furthermore, a great hesitancy to be vaccinated has been reported from other countries in the general population9–13 and CIRD patients.14,15 On this background, we started this survey prior to the start of vaccination of CIRD patients in Germany and assessed the vaccination willingness against SARS-CoV-2 of these patients, compared the results with non-CIRD patients, and investigated different factors of influence on vaccination compliance.
Methods
The reporting of this study conforms to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statement for cross-sectional studies. 16 In the preparation phase of this study, we developed a detailed structured questionnaire to answer all study questions (see Supplementary file 1).
After approval by the ethical committee, inpatients and outpatients of our tertiary rheumatology hospital were consecutively parallelly recruited. A total of 781 CIRD and 155 non-CIRD patients were asked to participate in the study between February and April 2021 (see Supplementary file 2). Finally, 514 CIRD and 100 non-CIRD (degenerative spinal cord disease, osteoarthritis, and fibromyalgia) patients agreed to participate in the study. To describe the recruited population, sociodemographic data, comorbidities, disease characteristics, medication, and knowledge about their therapy were documented. The structured questionnaire included dichotomous answer options (yes/no) regarding prior vaccination experience (pneumococcal infections, influenza, and travel vaccination) and a numerical rating scale (NRS) ranging from 0 (fully disagree) to 10 (fully agree) to assess the attitude toward and vaccination willingness against SARS-CoV-2. Guided by various European League Against Rheumatism (EULAR) recommendations in which the NRS scale is often used to measure the level of agreement 17 and the Guidelines on Measuring Subjective Well-being, 18 a threshold of ⩾7 was arbitrarily set for a definite positive answer and a score of 5 or 6 for probable willingness to be vaccinated.
Other questions were related to the history of previous infections in general but also to the experience with the diagnosis of a SARS-CoV-2 infection or death due to COVID-19 among the household or close circle. To assess patients’ susceptibility to infections, we used the questions of the SPUR (Serious, Persistent, Unusual or Recurrent) questionnaire which are also used to screen for immunodeficiency, 19 including secondary forms in patients with CIRD treated with rituximab. 20 The only question that was taken as evidence of a history of severe infections included sepsis, meningitis, osteomyelitis, and chronic complicated sinusitis. We also asked about a history of pneumonia, a history of recurrent infections, and infections with unusual pathogens (Table 1).
Patient demographics, disease characteristics, and infection history.
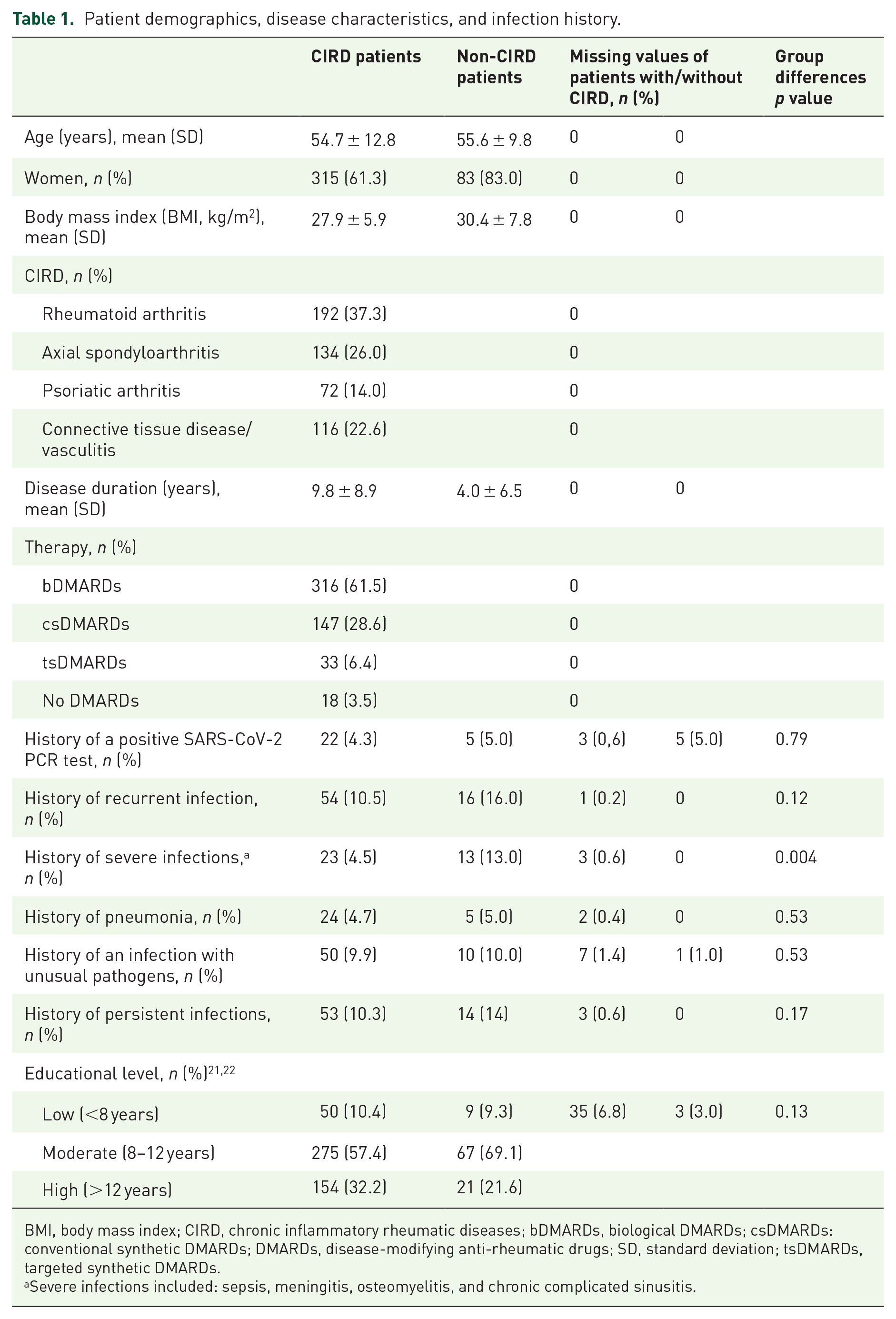
BMI, body mass index; CIRD, chronic inflammatory rheumatic diseases; bDMARDs, biological DMARDs; csDMARDs: conventional synthetic DMARDs; DMARDs, disease-modifying anti-rheumatic drugs; SD, standard deviation; tsDMARDs, targeted synthetic DMARDs.
Severe infections included: sepsis, meningitis, osteomyelitis, and chronic complicated sinusitis.
In addition, participants were asked about their confidence in SARS-CoV-2 vaccination and the fear of side effects because of it, their attitudes toward vaccination in general, and their compliance with hygiene rules to protect against SARS-CoV-2. One question asked about the identification with a risk group.
The level of education was assessed using a simplified version of the International Standard Classification of Education (ISCED).21,22
For comparison, a separate cohort of patients without CIRD, but diagnosed with degenerative spine conditions, osteoarthritis, or fibromyalgia was recruited for the study.
The locally appointed ethics committee of the Ruhr-University Bochum (Nr: 20-7143), Germany, has specifically approved this study and the research protocol. Written informed consent has been obtained from all subjects (or their legally authorized representative).
Statistical analyses
For the sample size calculation of this study, we used the data of our own recent study in which the prevalence of vaccination against pneumococci and influenza among CIRD patients was around 50%. 8 Thus, using a confidence interval of 95% and ±5% margin of error, we calculated that the required sample size of our study must be in the order of 502 patients with CIRD. For the control group, there was no sample size calculation. These patients were consecutively included when reporting to our hospital in the same time period.
For the description of the cohorts, established methods were used. Continuous variables were calculated as mean and standard deviation (SD) and categorical variables as percentages. The Shapiro–Wilk test was used to assess the normality of distribution of numeric variables. Non-parametric tests for independent-samples (Mann–Whitney U test for two groups or Kruskal–Wallis H test for three or more groups) and Fischer’s exact test were employed to check for differences in percentage between groups. To determine the correlation between different variables such as comorbidity and vaccination willingness of participants, Kendall’s tau-b correlation was used. Linear regression analyses were performed to find factors influencing the willingness to be vaccinated in relation to demographic data (gender, age, body mass index, education level) of CIRD patients.
Results
A total of 781 CIRD and 155 non-CIRD were initially approached to participate in the study (see Supplementary file 2.) In the end, 514 CIRD patients and 100 non-CIRD (degenerative spinal cord disease, osteoarthritis, or fibromyalgia) were included between February and April 2021, all in the hospital and all during the same time period. The detailed demographics are shown in Table 1. The mean age was similar, but the percentage of women was higher in the non-CIRD group. The educational level differed slightly between groups, however, without statistical significance. The disease duration was longer for CIRD compared with non-CIRD patients (Table 1).
Only 63.7% of CIRD patients thought that their medication was ‘immunosuppressive’. Of interest, recurrent and severe infections were reported more often by non-CIRD patients (16.0% and 13.0%) versus CIRD patients (10.5% and 4.5%), respectively. For the ‘history of severe infection’, the difference was even significant.
The frequency of a positive polymerase chain reaction (PCR) test for SARS-CoV-2 was comparable in the groups: 4.3% in CIRD versus 5.0% in non-CIRD patients. About 20% of patients in both groups reported to have experienced COVID-19-associated deaths in their acquaintance. Furthermore, a larger percentage of CIRD patients felt that they were at increased risk for COVID-19: 60.4% versus 48.5% non-CIRD (p = 0.026).
Compliance with hygiene rules was similar in the groups with over 90% reporting strict adherence.
Slightly more CIRD (79.6%) than non-CIRD patients (76.0%) were definitely willing to be vaccinated against SARS-CoV-2 (score ⩾7), but this was not statistically significant (p = 0.43). The detailed response rates of CIRD patients are shown in Figure 1. If the cut-off was changed to ⩾5, more than 90% of CIRD patients had a rather positive attitude to vaccination against SARS-CoV-2. In both groups, about 60% of the patients mentioned that they would rather get vaccinated immediately. The confidence in vaccination safety was relatively low in CIRD patients, with 58.9% with a cut-off score ⩾7 and about 80% with the cut-off ⩾5. (Figure 1). The level of confidence was even lower in the non-CIRD group (51.0%) and it reached 73.0% when adding the patients who marked an answer ⩾5. Furthermore, many CIRD patients (41.6%) reported to be afraid of side effects of the vaccination, and this proportion was even higher (62.3%) when using the cut-off ⩾5. Almost half of non-CIRD patients were concerned about side effects (48.0%), and this was also higher (67%) when values above 5 were used.

Percentual depiction of vaccination willingness, fear of vaccinations side effects, and confidence in vaccination safety among CIRD patients.
A generally negative attitude toward vaccination (NRS ⩾7) was more often found in non-CIRD than in CIRD patients: 6 % versus 1.58% (p = 0.016). When specifically asked whether the vaccination against SARS-CoV-2 is useless (NRS ⩾7), 1.9% of CIRD and 3% of non-CIRD found this statement correct (p = 0.45). Moreover, when comparing the differences in vaccination willingness based on the number of comorbidities, CIRD patients with a status of ‘no comorbidities’ had a lower vaccine acceptancy (p = 0.002).
The willingness to be vaccinated against SARS-CoV-2 showed a weak positive correlation with the level of information on influenza and pneumococcal infections as well as a history of travel vaccination (Table 2) in the CIRD group. This was not the case in the other group. Neither the number nor the type of current immunomodulatory therapy correlated with vaccination willingness. CIRD patients who thought that they are at risk of a COVID-19 infection were more likely to accept a SARS-CoV-2 vaccination. In addition, neither the history of previous infections in general nor the death due to SARS-CoV-2 among the household or close circle was correlated with the decision to get vaccinated against SARS-CoV-2 in CIRD and non-CIRD patients, respectively.
Correlation analyses of vaccination willingness and possible predictors.

CIRD, chronic inflammatory rheumatic diseases; DMARDs; DMARDs, disease-modifying anti-rheumatic drugs; PCR, polymerase chain reaction.
Correlation significant at the 0.05 level (two-tailed), if not stated otherwise.
Correlation significant at the 0.01 level (two-tailed).
Correlation significant at the 0.05 level (one-tailed).
Kendall’s tau-b correlation coefficient.
Multivariable linear regression analysis revealed that vaccination willingness was associated with education (b = 0.65, p = 0.001) and age (b = 0.06, p < 0.001); for detailed information, see Table 3. In contrast, there was no influence of established risk factors for severe outcomes of a COVID-19 infection except for hypertension (Table 2).
Linear regression of vaccination willingness and CIRD patient demographics.
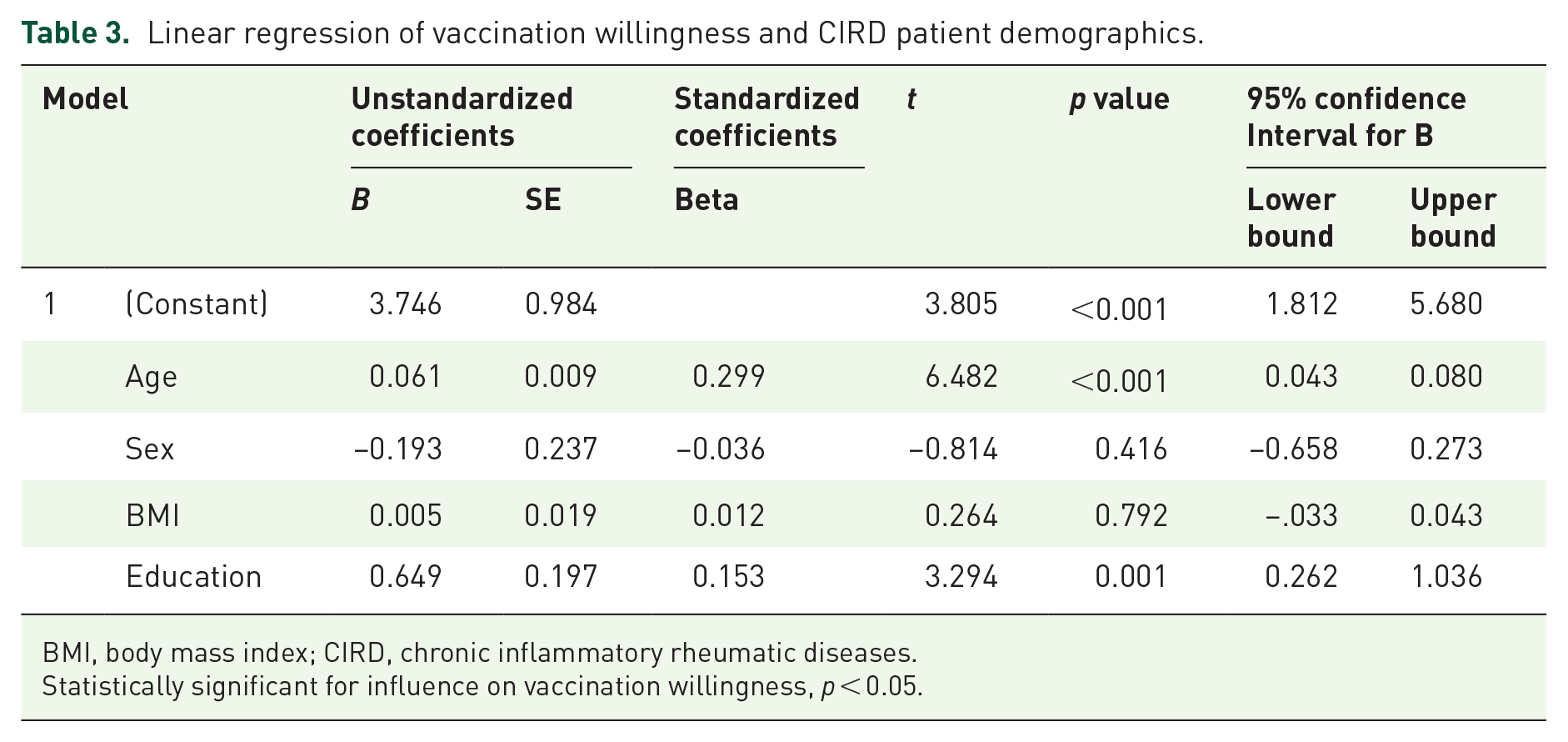
BMI, body mass index; CIRD, chronic inflammatory rheumatic diseases.
Statistically significant for influence on vaccination willingness, p < 0.05.
Discussion
This study shows that the majority of CIRD patients were willing to be vaccinated against SARS-CoV-2, but at least 20% still need to be convinced about its importance. This number could be increased by systematically providing convincing medical information. The vaccination willingness of CIRD patients was slightly better than in the non-inflammatory group. How does this compare to population-based data? The Robert-Koch-Institute (RKI), the German authority for the survey of diseases reporting to the Ministry of Health, published results of the COVIMO study. 23 During the survey period (end of April 2021), 1005 adults were interviewed about vaccination against SARS-CoV-2. The vaccination willingness of the population was also generally positive with a proportion of those who ‘definitely do not want to be vaccinated’ of only 4.6% versus 72.6 % of respondents who stated that they ‘definitely want’ to be vaccinated; 24 these results are rather similar to ours.
Although the willingness of CIRD patients was high and the majority thought that the vaccines were safe, many patients were afraid of side effects, and this was before the national discussion on the safety of viral vector vaccines officially started in April 2021 with a dear doctor letter (when our recruitment period ended).
The non-CIRD group had a shorter disease duration in our study. This can only be by chance since patients were consecutively recruited and within the same time period. Nevertheless, disease duration was not associated with vaccination readiness in our analysis, which is concordant with other studies. 25
Interestingly, the non-CIRD patients reported a higher frequency of severe infections than the CIRD group, while other questions related to infections (persistent, recurrent, or with unusual pathogens) showed a similar frequency in both groups. However, there are two major methodological problems that need to be mentioned in this regard: (1) recall bias because infections may have occurred a long time ago and (2) ascertainment because patients’ reports were not validated by searching hospital or physician’s records.
Whether CIRD patients, knowing that they have an increased risk of infection due to immunosuppression, have better protected themselves against infections than other patients is unknown. Furthermore, the total number of subjects with severe infections was rather small. Thus, we are reluctant to generalize the results or draw major conclusions. However, it seems that this topic may be worth to be studied in more detail in the future.
The education level of CIRD patients was slightly higher than in non-CIRD, but the correlation to vaccination willingness was strong. This was different for age which showed a weaker correlation. Other correlations were less surprising: patients who were well informed about influenza and pneumococcal infections and those who had undergone vaccination related to traveling were more often willing to be vaccinated against SARS-CoV-2. The same was for the patients who stated themselves as being at high risk.
More surprising were the results related to comorbidities, since only arterial hypertension came out as an influencing factor, while diabetes, chronic kidney and lung diseases, the diagnosis of cancer, and osteoporosis did not seem to have an influence on vaccination willingness in both patient groups.
Interestingly, neither death experience due to SARS-CoV-2 among the household or close circle nor a history of severe infections played a significant role in the decision of getting vaccinated against SARS-CoV-2; these results are concordant with other studies. 26
Our study has some limitations. Although we did have a sample size calculation for the CIRD patients, we did not have that for the control group which was just recruited in parallel. However, since the percentage of patients willing to be vaccinated was almost exactly as in a much larger German study, 27 we do not think that this challenges the credibility of our data. Furthermore, we have assessed the vaccination willingness using the questionnaire developed by us which has not been validated. However, we are not aware of a validated instrument for patients with CIRD in this regard. In addition, we have set arbitrary cut-offs for definite and possible vaccination willingness. This decision was based on international guidelines and recommendations.16,17 Employing an NRS had the advantage to use an instrument that was familiar and easy to use for both patients and physicians.
Furthermore, the control group of the non-CIRD patients was not ‘ideal’ in not having the same proportion of females. In addition, the group of non-CIRD patients included many patients with fibromyalgia syndrome. As it is known that this disease is often associated with anxiety, depression, and negativism,28,29 we cannot exclude confounding by indication in the non-CIRD group. However, depression does also frequently occur in CIRD patients such as those with axSpA, as recently reported. 30 Furthermore, we have recently shown that, early in the pandemic, anxiety was not a major driver for the decision to reduce immunosuppressive therapy. 31
Importantly, we did not differentiate the willingness to get vaccinated based on the administered vaccine, one reason being that AstraZeneca Vaccine was granted marketing authorization by the European Commission on 29 January 2021, exactly when starting the recruitment. Indeed, in two recent studies from Italy,32,33 a low willingness to be vaccinated against SARS-CoV-2 has been reported, mainly due to skepticism toward AstraZeneca vaccine. In the meantime, the situation has somewhat changed, and the efficacy of the mRNA BNT162b2 vaccine has been well documented on the individual level 34 but also on the society level in terms of herd immunity, 35 which, in light of only minor side effects of the vaccine, 36 results in a magnificent benefit/risk ratio.
Both, the knowledge about the effectiveness of vaccination and the side effects, were still limited in patients with CIRD and unclear at the time of the study start, and CIRD patients were usually not included in the trials aiming for approval of the vaccines. However, in the meantime, the situation is much clearer, and there are no concerns related to the vaccination against SARS-CoV-2 based on several studies.37–41 Indeed, there are no more side effects after vaccination against SARS-CoV-2 in CIRD patients compared with the general population. Moreover, flares after vaccination were rare. 42 The EULAR recommendations for vaccination provide a solid basis for vaccination strategies in patients with CIRD. 43
In summary, our survey highlights several predictors that are relevant for vaccination willingness including appropriate information about its relevance. The good news, however, is that the vast majority of CIRD patients indicated their willingness to be vaccinated. This was very recently confirmed in a large Canadian study which showed a high COVID-19 vaccine uptake among individuals with immune-mediated inflammatory diseases. 44
However, although 96.5% of CIRD patients were on immunosuppressive therapy, only 60.5% were aware of it and this is alarming, and it can be changed by appropriate education. Thus, information programs about vaccinations in general and COVID-19 in particular are necessary since a well-perceived own risk for COVID-19 may well increase vaccine acceptancy. The fact that experienced death due to SARS-CoV-2 among the household or close circle and known risk factors for a severe COVID-19 did not show an interference with vaccine acceptancy is concerning and underlines the need for a personalized assessment and personalized patient education.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X221093760 – Supplemental material for SARS-CoV-2 vaccination willingness and predictors in patients with chronic inflammatory rheumatic diseases (CIRD) and without CIRD
Supplemental material, sj-docx-1-tab-10.1177_1759720X221093760 for SARS-CoV-2 vaccination willingness and predictors in patients with chronic inflammatory rheumatic diseases (CIRD) and without CIRD by Iulia Roman, Ioana Andreica, Xenofon Baraliakos, Imke Redeker, Uta Kiltz and Jürgen Braun in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X221093760 – Supplemental material for SARS-CoV-2 vaccination willingness and predictors in patients with chronic inflammatory rheumatic diseases (CIRD) and without CIRD
Supplemental material, sj-docx-2-tab-10.1177_1759720X221093760 for SARS-CoV-2 vaccination willingness and predictors in patients with chronic inflammatory rheumatic diseases (CIRD) and without CIRD by Iulia Roman, Ioana Andreica, Xenofon Baraliakos, Imke Redeker, Uta Kiltz and Jürgen Braun in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
We wish to thank all persons who participated in the current study. Furthermore, we thank Dr Styliani Tsiami, Tanja Kobylinski, and the clinical departments for assisting with recruitment of participants.
Compliance with ethical guidelines
The study was performed in accordance with the Helsinki Declaration of 1964, and its later amendments. All subjects provided informed consent to participate in the study. The locally appointed ethics committee of the Ruhr-University Bochum (Nr: 20-7143), Germany, has specifically approved this study and the research protocol.
Author contribution(s)
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We acknowledge support by the Open Access Publication Funds of the Ruhr-Universität Bochum.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding authors on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
