Abstract
Background:
Previous experiences with non-medical switching of adalimumab (ADA) in patients with chronic inflammatory rheumatic diseases (CIRD) come mainly from phase III extension of randomised clinical trials and little from routine care.
Objectives:
To analyse treatment trajectories over 2 years in patients with CIRD conducting a non-medical switch from originator to biosimilar ADA.
Design:
A retrospective observational cohort study was conducted with data from a third-level rheumatology centre in Germany. CIRD patients on originator ADA who switched to ADA biosimilar from October 2018 onwards were identified and followed until September 2020.
Methods:
Patients’ characteristics were compared between the four a priori defined treatment trajectories ‘continued biosimilar ADA therapy’, ‘back-switch to originator ADA therapy’, ‘switch to another biological disease-modifying anti-rheumatic drug (bDMARD) therapy’ and ‘stopped bDMARD therapy/death/drop out’. Factors associated with continuing biosimilar ADA therapy were analysed using Cox proportional hazards regression analyses.
Results:
A total of 121 CIRD patients were included. Most patients (66.9%) continued therapy with biosimilar ADA over 2 years, with a treatment retention rate of 73.1%. Whereas 21 patients (17.4%) switched back to originator ADA, mainly due to adverse events, and 8 patients (6.6%) switched to a different bDMARD, mainly due to lack of effect. The estimated risk of withdrawal was lower for longer prior duration on originator ADA [hazard ratio (HR): 0.82; 95% CI: 0.69–0.97] and higher for higher C-reactive protein levels at baseline (HR: 1.18; 95% CI: 1.00–1.39). Male patients, older patients and those for whom originator ADA was their first bDMARD tended to have a lower risk of withdrawal.
Conclusion:
Our results indicated that three of four patients continue biosimilar ADA over 2 years with lower risks of withdrawal for male sex, older age, longer prior duration on originator ADA and originator ADA as first bDMARD.
Introduction
There is evidence that drug retention rates of adalimumab (ADA) in patients with chronic inflammatory rheumatic diseases (CIRD) are impaired by lack of effect/loss of efficacy and adverse events and that about 50% of users had discontinued ADA within 5 years. 1 With the introduction of ADA biosimilar disease-modifying anti-rheumatic drug (bsDMARD) starting in October 2018, non-medical switching from originator ADA to ADA bsDMARD is now increasingly part of daily practice in rheumatologic care. 2 Following the approval of the first bsDMARDs in the field of rheumatology by the European Commission in 2015, bsDMARDs have captured a steadily growing market. The approval of bsDMARDs is based on the totality of the evidence, demonstrating efficacy, safety and equivalence to the originator biologic DMARD (bDMARD).3–5 In all countries of the European Union, the annual therapy costs of the bsDMARD are considerably lower than those of the originator bDMARD. 2 The economic factors mean that, on the one hand, several bsDMARDs of an originator bDMARD are approved and, on the other hand, the health authorities pursue different strategies for the implementation of bsDMARDs. Both internationally and nationally, the proposals range from an unregulated approach (i.e. the decision-making authority lies solely on the side of the physician) 6 to rigid specifications of a mandatory switch from the originator bDMARD to the bsDMARD. 7
In Germany, the introduction of bsDMARDs is handled differently between the individual associations of panel doctors (Kassenärztliche Vereinigung – KV). The KV Westfalen-Lippe – which is the association within our federal state – specifies a bsDMARD quota of >90%, which in practice means that almost all patients have to start with or to be switched to a bsDMARD. Data collected in Denmark as part of a mandatory switch from etanercept (ETA) as the originator bDMARD to SB4 as the ETA bsDMARD show that the retention rate of the bsDMARD was higher than the retention rate of non-switchers but lower compared to a historical cohort with a treatment of originator bDMARD. 8 The authors conclude that patient factors and non-specific drug effects may have an impact on the retention rate.
Data on ADA bsDMARD efficacy and safety in routine care have been published to a limited extent in recent years, but data on treatment pathways over the long term are lacking. Documentation of such treatment switches is essential in clinical practice, as clinical trials for switching from the originator bDMARD or other mode of action to bsDMARDs are not part of the procedure provided by the European Medical Agency. For the originator ADA (Humira®), four ADA bsDMARDs are approved in Germany. ADA is approved for the treatment of rheumatoid arthritis (RA), axial spondyloarthritis (axSpA), plaque psoriasis in children and adolescents, hidradenitis suppurativa (acne inversa), Crohn’s disease (also in children and adolescents), juvenile idiopathic arthritis, uveitis (also in children and adolescents), psoriasis, ulcerative colitis and psoriatic arthritis (PsA) in Germany.
The Rheumazentrum Ruhrgebiet treats patients with CIRD with originator bDMARDs and several bsDMARDs. In the course of treatment, the effectiveness and safety of the treatment, including documentation of patients and disease characteristics, comorbidities, disease activity and physical function, are regularly assessed. In the long-term care of patients with CIRD, drug therapy may be changed for a variety of reasons: adverse effects, lack of effect, contraindications and patient refusal are the most common reasons.9–11 In a bsDMARD therapy setting, there may be an additional situation that patients report a lack of effect under bsDMARD, although they had well-suppressed disease activity under the originator bDMARD. Subjective complaints were the main reason for biosimilar discontinuation. 12
The aim of this study was to investigate treatment trajectories over 2 years in patients with CIRD who conducted a non-medical switch from originator ADA to ADA bsDMARD according to the local guideline specifying a bsDMARD quota of >90%.
Patients and methods
Study design
This was a retrospective observational cohort study conducted with data from patients attending a third-level rheumatology centre in Germany between October 2018 and September 2020. Patients with CIRD on ADA originator who switched to ADA bsDMARD were included. Clinical information was retrieved from every visit (in general, each patient had one visit each quarter) and included b/bsDMARD treatment, reason for treatment withdrawal, C-reactive protein (CRP; mg/L), assessment of disease activity and physical function. Disease activity was assessed by Disease Activity Score of 28 joints (DAS28) 13 for RA and PsA patients and by Ankylosing Spondylitis Disease Activity Score (ASDAS)14,15 and Bath Ankylosing Spondylitis Disease Activity Index (0–10) 16 for axSpA patients. Disease remission was defined as DAS28 < 2.6 17 for RA and PsA and as ASDAS < 1.3 for axSpA.18,19 Physical function was assessed by Health Assessment Questionnaire (0–3) 20 for RA and PsA and Bath Ankylosing Spondylitis Functional Index (0–10) 21 for axSpA.
At baseline (i.e. date of switch), additional information was retrieved, including DMARD treatment history, prior treatment duration of originator ADA, concomitant treatment with conventional synthetic DMARDs, glucocorticoids or non-steroidal anti-inflammatory drugs (NSAIDs) and comorbidities, assessed by Charlson Comorbidity Index.
Outcome
Patients were followed for up to 2 years. The primary outcome was the frequency of the four predefined treatment trajectories ‘continued ADA bsDMARD therapy’, ‘back-switch to originator ADA therapy’, ‘switch to other bDMARD therapy’ and ‘stopped bDMARD therapy/death/drop out’. Treatment retention of ADA bsDMARD was the secondary outcome and was defined as the number of days on ADA bsDMARD until withdrawal, death, lost to follow-up or end of study period, whichever came first. Further secondary outcomes were factors associated with continuing ADA bsDMARD therapy.
In addition, the following outcomes were assessed: (a) frequency and reasons for ADA bsDMARD withdrawal and (b) changes in disease activity between baseline and post-switch, stratified by treatment trajectory.
Statistical analysis
Descriptive analyses were performed using medians and 25th–75th percentiles (interquartile range – IQR) for quantitative variables and absolute frequencies and proportions (%) for qualitative ones.
The Kaplan–Meier method, a non-parametric approach, was used to estimate the probability of continuing ADA bsDMARD therapy over the 2-year study period. Patients whose treatment regime was not switched back to originator ADA nor a different bDMARD were censored either at study completion or premature study discontinuation. The cumulative incidence for the competing events of switching therapy either back to originator ADA or to a different bDMARD was calculated using a non-parametric estimation.
Factors associated with continuing ADA bsDMARD therapy were analysed using univariable and multivariable Cox proportional hazards regression analyses and included the a priori defined baseline variables such as age, gender, CRP, disease remission, number of previous bDMARDs and prior treatment duration of originator ADA. Results are presented as hazard ratios (HRs) with 95% confidence intervals (CIs). The proportional hazards assumption was assessed.
Data analyses were performed with R Statistical Software (v4.1.2; R Core Team 2021). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 22
Results
A total of 121 CIRD patients on treatment with originator ADA were switched to ADA bsDMARD after an average treatment duration of originator ADA of 41 months. Originator ADA was the first bDMARD for 61%. Most patients had axSpA (N = 74), followed by RA (N = 24), PsA (N = 17) and other [N = 6, including juvenile idiopathic arthritis and synovitis-acne-pustulosis-hyperostosis-osteitis (SAPHO) syndrome]. The median disease duration was 5 years and one-third of patients were in remission at baseline (i.e. date of non-medical switch). Baseline characteristics stratified by disease are shown in Table 1.
Clinical characteristics of patients at the time of switching from originator to biosimilar ADA stratified by indication.

Numbers are stated as median (IQR) or n (%).
Remission defined as DAS28 < 2.6 (RA, PsA), ASDAS < 1.3 (axSpA).
ADA, adalimumab; ASDAS, Ankylosing Spondylitis Disease Activity Score; axSpA, axial spondyloarthritis; BASDAI, Bath Ankylosing Spondylitis Disease Activity Index (0–10); BASFI, Bath Ankylosing Spondylitis Functional Index (functional status, 0–10); b/csDMARD, biological/conventional synthetic disease-modifying anti-rheumatic drug; CRP, C-reactive protein; DAS28, Disease Activity Score of 28 joints; HAQ, Health Assessment Questionnaire (functional status, 0–3); IQR, interquartile range; NSAID, non-steroidal anti-inflammatory drug; PsA, psoriatic arthritis; RA, rheumatoid arthritis.
Treatment trajectories
Most patients, 81 (66.9%), continued therapy with ADA bsDMARD, including two patients with treatment breaks in between. A total of 21 patients (17.4%) switched back to originator ADA and 8 patients (6.6%) switched to a different bDMARD, including 6 patients who switched to another tumour necrosis factor inhibitor and 2 who switched to a different mode of action. The remaining 11 patients (9.1%) either stopped treatment or died.
Patients who continued ADA bsDMARD were more frequently male than those who switched therapy back to originator ADA (60% versus 43%) and those who switched to a different bDMARD (60% versus 25%). Moreover, they were older with a longer disease duration than those who switched therapy back to originator ADA or a different bDMARD (Table 2). The prior duration of originator ADA treatment was increased in patients who continued ADA bsDMARD therapy (48 months) compared to those who switched therapy back to originator ADA (32 months) and those who switched to a different bDMARD (9 months). Concomitant treatment with NSAIDs, conventional synthetic disease-modifying anti-rheumatic drugs (csDMARDs) and glucocorticoids at baseline was somewhat increased in patients who continued ADA bsDMARD therapy, while patients who switched therapy had more previous bDMARD therapies (Table 2).
Clinical characteristics of patients at the time of switching from originator to biosimilar ADA, stratified by treatment trajectory.

Numbers are stated as median (IQR) or n (%).
Remission defined as DAS28 < 2.6 (RA, PsA), ASDAS < 1.3 (axSpA).
ADA, adalimumab; ASDAS, Ankylosing Spondylitis Disease Activity Score; axSpA, axial spondyloarthritis; b/csDMARD, biological/conventional synthetic disease-modifying anti-rheumatic drug; CRP, C-reactive protein; DAS28, Disease Activity Score of 28 joints; IQR, interquartile range; NSAID, non-steroidal anti-inflammatory drug; PsA, psoriatic arthritis; RA, rheumatoid arthritis.
Treatment retention
Information about the likelihood of continuing ADA bsDMARD therapy over the 2-year study period is given in Figure 1(a). Whereas the probability of switching therapy back to originator ADA or to a different bDMARD is illustrated in Figure 1(b). The 2-year ADA bsDMARD treatment retention rate was 73.1% (Kaplan–Meier estimation).

Probability of continuing ADA bsDMARD therapy (a) and probability of switching therapy back to originator ADA or to a different bDMARD (b) in patients with CIRD over 2 years (N = 121).
Results from univariable Cox regression analyses showed that the estimated risk of withdrawal was lower for longer prior duration on originator ADA (in years; HR: 0.82; 95% CI: 0.69–0.97; Table 3). Moreover, male patients (HR: 0.47; 95% CI: 0.22–1.00), older patients (in 10 years; HR: 0.85; 95% CI: 0.65–1.11), patients in DAS28/ASDAS remission at baseline (HR: 0.34; 95% CI: 0.10–1.18) and those for whom originator ADA was their first bDMARD (HR: 0.58; 95% CI: 0.28–1.20) tended to have a lower risk of withdrawal. Whereas a higher risk of withdrawal was observed for higher CRP levels at baseline (HR: 1.18; 95% CI: 1.00–1.39). Similar results were obtained from two multivariable Cox regression analyses, which included the a priori defined variables such as age, sex, first bDMARD originator ADA, prior duration on originator ADA and either CRP at baseline (Figure 2) or DAS28/ASDAS remission at baseline (Figure 3). In the former model, a higher risk of withdrawal was observed for higher CRP levels at baseline (HR: 1.23; 95% CI: 1.01–1.49) and a tendency for a lower risk of withdrawal for patients in DAS28/ASDAS remission at baseline (HR: 0.28; 95% CI: 0.079–1.0) was observed in the latter model.
Factors associated with ADA bsDMARD withdrawal: Results from univariable Cox regression analyses.

Bold values denote statistical significance at the p < 0.05 level.
ADA, adalimumab; ASDAS, Ankylosing Spondylitis Disease Activity Score; b/bsDMARD, biologic/biosimilar disease-modifying anti-rheumatic drug; CI, confidence interval; CRP, C-reactive protein; DAS28, Disease Activity Score of 28 joints; HR, hazard ratio.

Forest plot showing factors associated with 2-year treatment withdrawal after switching from originator to biosimilar ADA: Results of multivariable Cox regression analysis (N = 119).
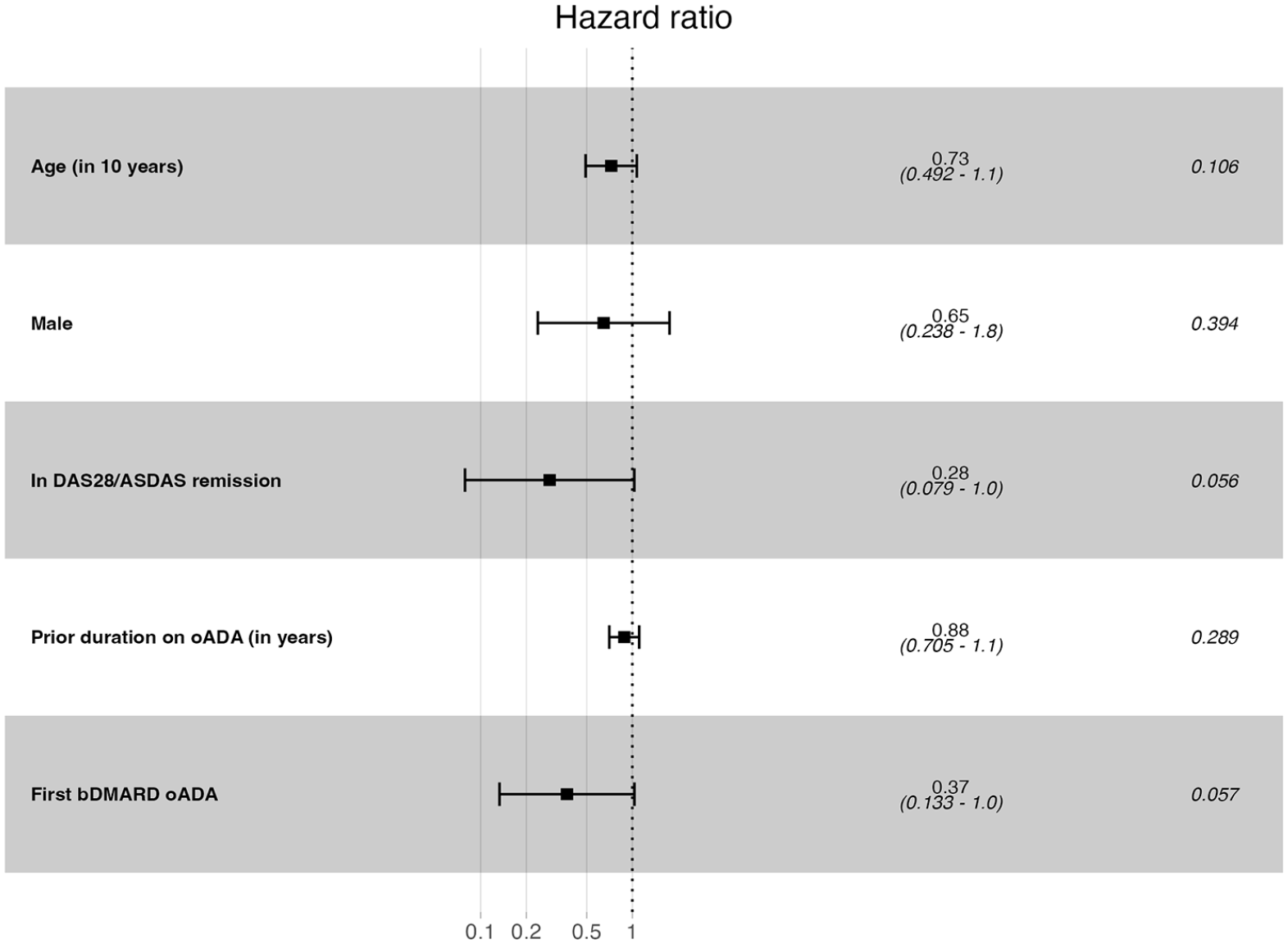
Forest plot showing factors associated with 2-year treatment withdrawal after switching from originator to biosimilar ADA: Results of multivariable Cox regression analysis (N = 78).
Reasons for withdrawal
During a 2-year follow-up, 40 patients (33.1%) withdrew from treatment with ADA bsDMARD, mainly due to lack of effect (35%), adverse events (35%) and loss to follow-up (22%; Table 4). Patients who switched back to originator ADA had a treatment duration on ADA bsDMARD of a median of 3 months (IQR: 1–9 months) and those who switched to a different bDMARD therapy of a median of 7.5 months (IQR: 5.6–12.1 months). Back-switch was mainly due to adverse events (55%), while the switch to a different bDMARD was mainly due to lack of effect (75%).
Main reasons for withdrawal during 2-year follow-up.

Numbers are stated as n (%).
Disease activity at baseline and post-switch
The median ASDAS in patients with axSpA who continued ADA bsDMARD therapy decreased from 1.90 at baseline (IQR: 1.10–2.70) to 1.45 (IQR: 1.08–2.20) at the study end. Whereas patients with axSpA who withdrew from ADA bsDMARD therapy had a higher median ASDAS at baseline of 2.80 (IQR: 2.25–3.30), which decreased to 2.75 (IQR: 2.42–3.20) at the date of withdrawal (Supplemental Table 1). A similar result was observed for patients with RA or PsA. Those who continued ADA bsDMARD therapy had a lower DAS28 at baseline (median: 2.49; IQR: 2.15–3.77) than those who withdrew from ADA bsDMARD therapy (median: 3.50; IQR: 2.15–4.00; Supplemental Table 2).
Discussion
In this retrospective observational cohort study, we investigated treatment trajectories over 2 years in patients with CIRD, who switched from originator ADA to ADA bsDMARD. Most patients continued therapy with ADA bsDMARD with a treatment retention rate of 73.1%, while 21 patients (17.4%) switched back to originator ADA and 8 patients (6.6%) switched to a different bDMARD. We found lower estimated risks of withdrawal for male patients, older patients, those with longer prior duration on originator ADA and for whom originator ADA was their first bDMARD, while a higher estimated risk of withdrawal was observed for higher CRP levels at baseline. The main reasons for withdrawal were adverse events and lack of effect, the former was more frequent in back-switches, and the latter in switches to a different bDMARD.
Previous experiences with non-medical switching of ADA in patients with CIRD come mainly from phase III extension of randomised clinical trials5,23–26 and little from routine care. In a recent study of CIRD patients in routine care, who were mandated to switch from ADA originator to ADA bsDMARD, 89.5% of patients maintained treatment 1 year after switching, while only 4% of patients switched back to originator ADA. 7 In this study, the main reasons for withdrawal were adverse events and lack of effect, similar to our results. In another study, over 93% of patients with stable disease maintained treatment 1 year after switching, while only 6.6% of patients switched back to originator ADA. 27 The retention rate reported in both previous studies was higher in comparison to our study, which may be explained by the proportion of patients who were in remission at baseline. While only 33% were in remission in our study, 61% were in Nabi et al. 7 and van Adrichem et al. 27 included only patients in remission or with low disease activity.
While the few studies with a non-medical switch setting of ADA in patients with CIRD in routine care focussed on comparative analyses of retention rates,7,28 this is, to the best of our knowledge, the first study focussing on factors associated with withdrawal. Our results corroborate those from other real-life studies examining treatment retention in CIRD patients, although results are not completely comparable through to differences in study design. Previous studies that examined RA, PsA or axSpA reported that men have better treatment retention of bDMARDs compared to women6,29 and a better treatment retention has also been demonstrated in patients who had previously been treated with fewer bDMARDs.6,30–33 Although these results were not obtained in a switch setting, they are consistent with our findings. Furthermore, a recent study of patients with CIRD who had undergone medical or non-medical switching from ADA originator to ADA bsDMARD found an increased risk of treatment withdrawal in patients who were treated at baseline with NSAIDs or glucocorticoids. 34 Considering baseline use of NSAIDs or glucocorticoids as a surrogate for sub-optimal disease control, those results may be regarded as indirect evidence of higher risks of ADA bsDMARD withdrawal for patients with sub-optimal disease control at baseline, 34 which is in line with our findings.
A recent study of patients with CIRD switching from ETA originator to ETA bsDMARD found that patients with longer previous duration on originator ETA had lower estimated risks of ETA bsDMARD withdrawal, as did RA patients in remission or with low disease activity, 35 both results are comparable to our findings.
Strengths of the current study include the follow-up time of 2 years and the study design in which all patients were switched to a bsDMARD in accordance with the local guideline. Whereas the small sample size and the large number of missing values on disease activity (ASDAS/DAS28) to otherwise highly complete data represent limitations. To avoid overfitting, we restricted the number of variables in the multivariable Cox regression analyses to 5.
Conclusion
In conclusion, in this observational study of patients, who conducted a non-medical switch from originator to biosimilar ADA according to the local guideline, patients’ treatment trajectories over 2 years were analysed. Our results indicated that three of four patients continued biosimilar ADA with lower estimated risks of withdrawal for male patients, older patients, those with longer prior duration on originator ADA and for whom originator ADA was their first bDMARD. A back-switch to originator ADA (17.4%) was mainly due to adverse events and a switch to another bDMARD (6.6%) due to lack of effect, with higher estimated risks of withdrawal for higher CRP levels.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X231197087 – Supplemental material for Treatment with adalimumab in patients with chronic inflammatory rheumatic diseases: a study of treatment trajectories on a patient level in routine care
Supplemental material, sj-docx-1-tab-10.1177_1759720X231197087 for Treatment with adalimumab in patients with chronic inflammatory rheumatic diseases: a study of treatment trajectories on a patient level in routine care by Imke Redeker, Stefan Moustakis, Styliani Tsiami, Xenofon Baraliakos, Ioana Andreica, Bjoern Buehring, Jürgen Braun and Uta Kiltz in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
