Abstract
Background:
The incidence of osteoporotic fracture increases with age, particularly in elderly populations with atrial fibrillation (AF). However, direct oral anticoagulants (DOACs) have less effect on osteoporotic fracture than vitamin K antagonists, it is unclear whether the risk of osteoporotic fracture is affected by different types and doses of DOACs in AF patients.
Methods:
This nationwide population-based cohort study included AF patients prescribed DOACs between 2011 and 2016 taken from the Taiwan National Health Insurance database. Adjusted hazard ratios (aHRs) for the risk of osteoporotic, hip, and spine fractures between DOAC users were compared using the Fine and Gray subdistribution hazard model to adjust for possible confounders.
Results:
A total of 56,795 patients who were prescribed DOACs were included in the present study. Among them, 24,597 patients received dabigatran, 26,968 received rivaroxaban, and 5230 received apixaban. After 2 years’ follow up, there was no significant difference in the incidence of osteoporotic, spine, or hip fracture among those receiving dabigatran, rivaroxaban, or apixaban. Subgroup analysis showed that patients taking dabigatran had a higher incidence of osteoporotic and hip fracture than those taking rivaroxaban and apixaban in cases with concomitant peripheral artery disease (PAD) or a history of hip fracture (p for interaction: 0.004 and 0.030, respectively). However, dabigatran users had a lower incidence of osteoporotic fracture and spine fracture in those receiving standard-dose DOACs compared with rivaroxaban and apixaban; whereas, they had a higher incidence of hip fractures when administered at low dose.
Conclusion:
AF patients with different DOACs did not have different risks of osteoporotic fracture overall. However, additional concomitant morbidities, such as PAD or a history of hip fracture, and standard/low doses might be associated with different risks for different DOACs. These findings should be taken into consideration in the clinic when the DOAC is being chosen.
Plain language summary
Anticoagulation therapy is an essential therapy in atrial fibrillation (AF) patients, but osteoporotic fracture is another important issue in these patients prescribed with anticoagulants. However, no study has been conducted to evaluate the impact of different DOACs on different types of osteoporotic fractures. In our findings, although different DOACs had no significantly different impact on osteoporotic fractures, dabigatran users had a slightly higher incidence of osteoporotic and hip fractures among different DOACs, particularly in those have simultaneously had peripheral artery disease, a history of hip fracture. In addition, when AF patients taking low-dose DOACs, dabigatran users also have higher incidence of hip fracture than those taking other DOACs.
Introduction
Atrial fibrillation (AF), the most common cardiac arrhythmia, was estimated to affect 33.5 million people worldwide in 2010, which was a notable increase in both incidence and prevalence compared with previous decades. 1 AF is an established risk factor for ischemic stroke, 2 and the risk of stroke has been estimated as between 1% and 20% per year.3,4 Therefore, oral anticoagulation with vitamin K antagonists (VKAs), was recommended in clinical practice in the early era 5 due to its documented efficacy for preventing ischemic stroke and prolonging life for patients with AF.6,7 Several direct oral anticoagulants (DOACs), including the direct thrombin inhibitor, dabigatran, and direct factor Xa inhibitors (rivaroxaban, apixaban, and edoxaban), have emerged as alternatives to VKAs for various indications. DOACs are being used more frequently because of their ease of administration and comparative efficacy compared with VKAs in reducing thromboembolism and major bleeding.8,9
The prevalence of osteoporosis and osteoporotic fracture both increase with age, particularly in elderly populations with AF. 10 In addition to being a key factor associated with thrombosis, vitamin K is also a cofactor in bone formation. 11 Osteocalcin, the principal non-collagenous protein in bone, is incorporated into bone via vitamin K-dependent gamma-carboxylation. VKAs decrease bone osteocalcin levels and usage leads to a decrease in bone hardness.12,13 Increased levels of undercarboxylated osteocalcin in the blood are associated with reduced bone mineral quality. 14 However, an association between the use of VKAs and decreased bone mineral density has not been found,15,16 suggesting that agents can affect bone structure without affecting bone density. Whether VKAs lead to an increase in bone fracture risk is controversial, as various studies have yielded different results.10,17,18 Furthermore, whether DOACs are better than VKAs, in terms of risk of osteoporotic fracture, is another dilemma. One Asian study found an increased risk of osteoporotic fractures in VKA users compared with dabigatran users. 19 In contrast, a recent systematic review found no increase in odds of fracture in VKA users versus controls or DOAC users. 20 Additionally, no difference in fracture risk was noted among different kinds of DOAC users. 21 However, these previous studies did not report the fracture risk of different anatomical locations, and they also did not consider the effect of DOAC dosage on fracture risk.
Therefore, we designed the present study using the Taiwan National Health Insurance database (NHIRD) to validate this issue. We assessed the incidence of osteoporotic fractures among AF patients who were prescribed different DOACs and compared the risk of osteoporotic fractures among them.
Materials and methods
Data source
This retrospective cohort study was conducted using the Taiwan NHIRD. The NHIRD is a national health and welfare administrative database from the Taiwan NHI program, which has been the universal health insurance system in Taiwan since 1995. The NHI data were collected by the National Health Informatics Project and was managed by the Health and Welfare Data Science Center. The NHI program covers approximately 99.8% of Taiwan’s 24 million inhabitants and details their complete outpatient visits, hospitalization records, prescription details, and disease status. It used the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) diagnostic and procedure codes before 2016 and the International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) codes from 2016 onwards. Previous studies have shown that high-quality information is provided in the NHIRD.22,23 To protect patient privacy and data security, all personal identifiers in the NHIRD are de-identified before access is given to researchers. The Ethics Institutional Review Board of Chang Gung Memorial Hospital approved the current study (IRB CGMH 201901426B0), and the requirement for informed consent was waived because of the anonymized data.
Patient population and exposure
The current study included all patients diagnosed with AF (atrial fibrillation or atrial flutter) between 2011 and 2016, who received DOAC treatment. AF was identified when patients were diagnosed at least once in inpatient services, or twice in outpatient services, with ICD-9-CM code 427.31 or ICD-10 codes I480–I482 and I4891. A previous study has confirmed the high accuracy of AF diagnosis in the NHIRD. 24 The index date was defined as the date of the patient’s first-ever prescription for DOAC between 2011 and 2016. To ensure we only included patients with AF, we excluded patients with a history of valvular heart disease who underwent surgery, to exclude possible off-label use of DOACs.
In addition, to ensure the comparability of outcomes between DOAC users, we also excluded patients younger than 50-years old at the index date and those with end-stage renal disease. Standard-dose and low-dose regimens of DOACs were defined according to several landmark trials of DOACs.25–27 Standard dose was considered to be dabigatran 150 mg twice daily (BID), rivaroxaban 20 or 15 mg/day, and apixaban 5 mg BID, while low dose was considered to be dabigatran 110 mg BID, rivaroxaban 10 mg/day, and apixaban 2.5 mg BID.
To ensure there was enough of a follow-up period, we excluded patients whose follow up was less than 3 months. To ensure that the osteoporotic fracture event was due to the medication, we excluded patients with osteoporotic fractures within 3 months after the index date. To minimize the potential confounding factors of osteoporosis and fracture, we also excluded patients who underwent hormone therapy. We also excluded patients who switched oral anticoagulants within 3 months after the index date and excluded those who were prescribed edoxaban, as only a few patients received edoxaban during the study period (Figure 1).

Flow chart for the inclusion of patients.
Covariates
The covariates were age, sex, monthly income, urbanization level, comorbidities, history of ischemic stroke, thrombo-embolism and fracture, Congestive heart failure,
Outcomes
The interested outcome was osteoporotic fracture after the index date and the different osteoporotic fracture locations, including hip fracture (ICD-9-CM code 820.x, ICD-10-CM codes S720.0x, S720.1x, S720.2x), and spine fracture (ICD-9- CM codes 733.13 and 805.x, ICD-10-CM codes S12.x, S22.x, S32.x, M800.x, M808.x, and M485.x) during follow up (Supplemental Table 1). According to the Taiwanese Osteoporosis Association guidelines, osteoporosis is diagnosed according to the T score derived from bone mineral density (osteoporosis T score ⩽−2.5) or according to fragility fractures diagnosed via clinical history. 30 All patients were followed up from the index date until the occurrence of a primary outcome, death, or the end of the database (31 December 2016) and the longest follow-up period was set as 2 years. The survival status was verified by the Taiwan Death Registry (TDR), including the mandatory registered date, causes, and location of death since 1971. The NHI data and TDR were directly linked by a unique and deidentified civil identification number.
Statistical analysis
Patient characteristics at baseline among the study groups (dabigatran, rivaroxaban, and apixaban) were compared using one-way analysis of variance for continuous variables and the chi-squared test for categorical variables. The risk of all-cause mortality among groups was compared using Cox’s proportional hazard model. The cumulative incidence of osteoporotic fracture, hip fracture, and spine fracture among the study groups was compared using the Fine and Gray subdistribution hazard model, which considers all-cause mortality during follow up as a competing risk. Subgroup analysis by the different levels of selected baseline characteristics [including age, sex, AF duration, hypertension, diabetes mellitus, dyslipidemia, peripheral artery disease (PAD), heart failure, chronic obstructive pulmonary disease, chronic kidney disease, epilepsy or seizure, malignancy, history of hip fracture, history of non-hip fracture, osteoporosis, old ischemic stroke anti-osteoporosis drugs, antipsychotics, and antidepressants] was conducted on the following outcomes: osteoporotic fracture, hip fracture, spine fracture and composite effect, during the 2-year follow up.
The risk of outcomes among the three distinct DOAC treatment groups was compared and stratified by the dose (standard dose versus low dose). Finally, the risk of outcomes between the standard-dose and the low-dose groups were compared for all patients with DOACs. The above survival analyses were adjusted for all of the covariates listed in Table 1, except the follow-up month was replaced with the index date. A two-sided p value < 0.05 was considered statistically significant, and no adjustment was made for multiple testing (multiplicity) in the present study. Given the fact that drug adherence and prior VKA use might be crucial to the outcomes, additional sensitivity analyses were performed. First, drug censoring was considered in which the patient data were censored at the date of drug switch from the original type of DOAC to another DOAC or VKA during the entire follow up. Second, prior VKA use was additionally adjusted in the survival model. Third, both drug censoring and prior VKA use were considered. All statistical analyses were performed using SAS version 9.4 (SAS Institute, Cary, NC, USA). The direct-adjusted cumulative incidence function under the Fine and Gray subdistribution method was generated using an SAS macro ‘%CIFCOX’.
Baseline characteristics among atrial fibrillation patients with different types of direct oral anticoagulants.
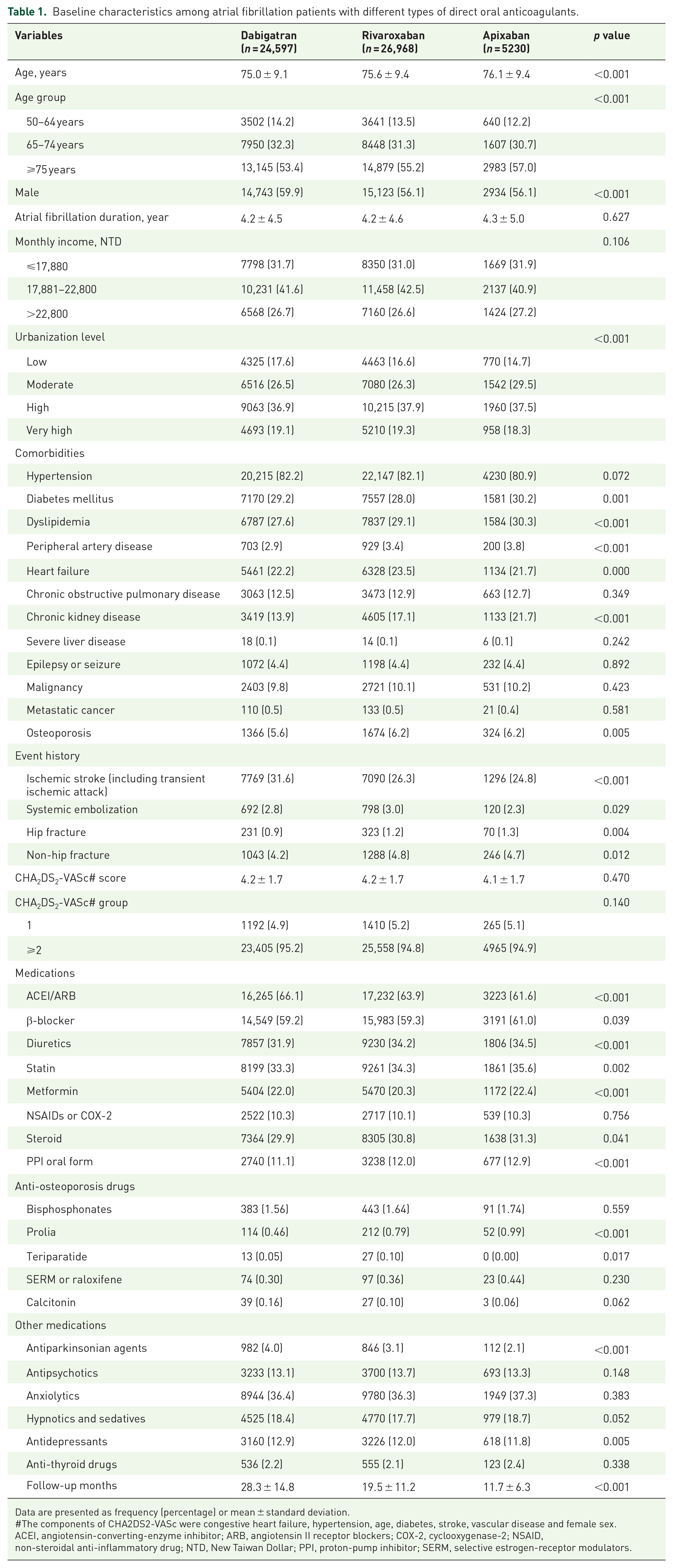
Data are presented as frequency (percentage) or mean ± standard deviation.
The components of CHA2DS2-VASc were congestive heart failure, hypertension, age, diabetes, stroke, vascular disease and female sex.
ACEI, angiotensin-converting-enzyme inhibitor; ARB, angiotensin II receptor blockers; COX-2, cyclooxygenase-2; NSAID, non-steroidal anti-inflammatory drug; NTD, New Taiwan Dollar; PPI, proton-pump inhibitor; SERM, selective estrogen-receptor modulators.
Results
Patient characteristics
A total of 75,083 AF patients received DOACs between 2011 and 2016. After applying the exclusion criteria, 56,795 patients remained for inclusion within the study. These patients were divided into the dabigatran (24,597 patients), rivaroxaban (26,968 patients), and apixaban (5230 patients) study groups (Figure 1). Baseline characteristics are listed in Table 1. The mean age was 75–76-years old for all groups, and the age distribution was similar among the three groups. The mean duration between the time of AF diagnosis and the index date for DOAC prescription was around 4.2 years. Hypertension was the main comorbidity across all three groups. The frequency of dabigatran prescription was highest in patients with a history of stroke, but lowest in patients with chronic kidney disease across all three groups. The CHA2DS2-VASc score was no different among the three groups (CHA2DS2-VASc: dabigatran versus rivaroxaban versus apixaban: 4.2 ± 1.7 versus 4.2 ± 1.7 versus 4.1 ± 1.7, respectively). As for fracture events, the prevalence of different types of fracture and osteoporosis was similar in all three groups. The use of other medications, including angiotensin-converting enzyme inhibitors/angiotensin receptor blockers (ACEi/ARB), statins, and hypoglycemic drugs, was similar among the three groups. As for anti-osteoporosis drugs and other drugs potentially affecting fracture, the frequency of use was similar among the three groups (Table 1).
Different types of osteoporotic fractures among those receiving dabigatran, rivaroxaban, and apixaban
In the 2-year observation period, the incidence of osteoporotic fracture was 6–10% in AF patients receiving DOAC therapy (Table 2). The results showed no significant difference in the incidence of osteoporotic fracture among those receiving dabigatran, rivaroxaban or apixaban (dabigatran versus rivaroxaban versus apixaban: 9.82% versus 8.44% versus 6.06%, respectively) [Figure 2(a)]. There were also no significant differences in the incidence of spine fractures among the three groups (dabigatran versus rivaroxaban versus apixaban: 3.79% versus 3.43% versus 2.58%, respectively) [Figure 2(b)]. However, there was relative high incidence of hip fractures in AF patients taking dabigatran compared with those taking apixaban or rivaroxaban. (dabigatran versus rivaroxaban versus apixaban: 1.69% versus 1.36% versus 0.90%, respectively), particularly when those taking dabigatran and those taking rivaroxaban were compared [adjusted hazard ratio (aHR) = 1.31, 1.24 and 1.15; and 95% confidence intervals (CIs) = 0.94–1.82, 1.01–1.53 and 0.98–1.35 for 6, 12 and 24 months’ follow up, respectively] [Table 2 and Figure 2(c)]. The additional sensitivity analyses showed that the long-term effect of DOACs did not change, after controlling drug censoring (Supplemental Table 2), prior VKA use (Supplemental Table 3), or both (Supplemental Table 4).
The incidences of different types of fractures in atrial fibrillation patients prescribed with different types of direct oral anticoagulants. * .

Data not specified are presented as frequency (percentage).
All covariates listed in Table 1 were adjusted, except the follow-up months.
aHR, adjusted hazard ratio; CI, confidence interval.
The incidences of different types of fractures in atrial fibrillation patients prescribed with a standard dose/low dose of different types of direct oral anticoagulants * (the sum of number of standard and low dose in each group: dabigatran n = 24,597; rivaroxaban n = 26,968; apixaban n = 5230).

Data not specified are presented as frequency (percentage).
All covariates listed in Table 1 were adjusted, except the follow-up months.
aHR, adjusted hazard ratio; CI, confidence interval; DOAC, direct oral anticoagulant.
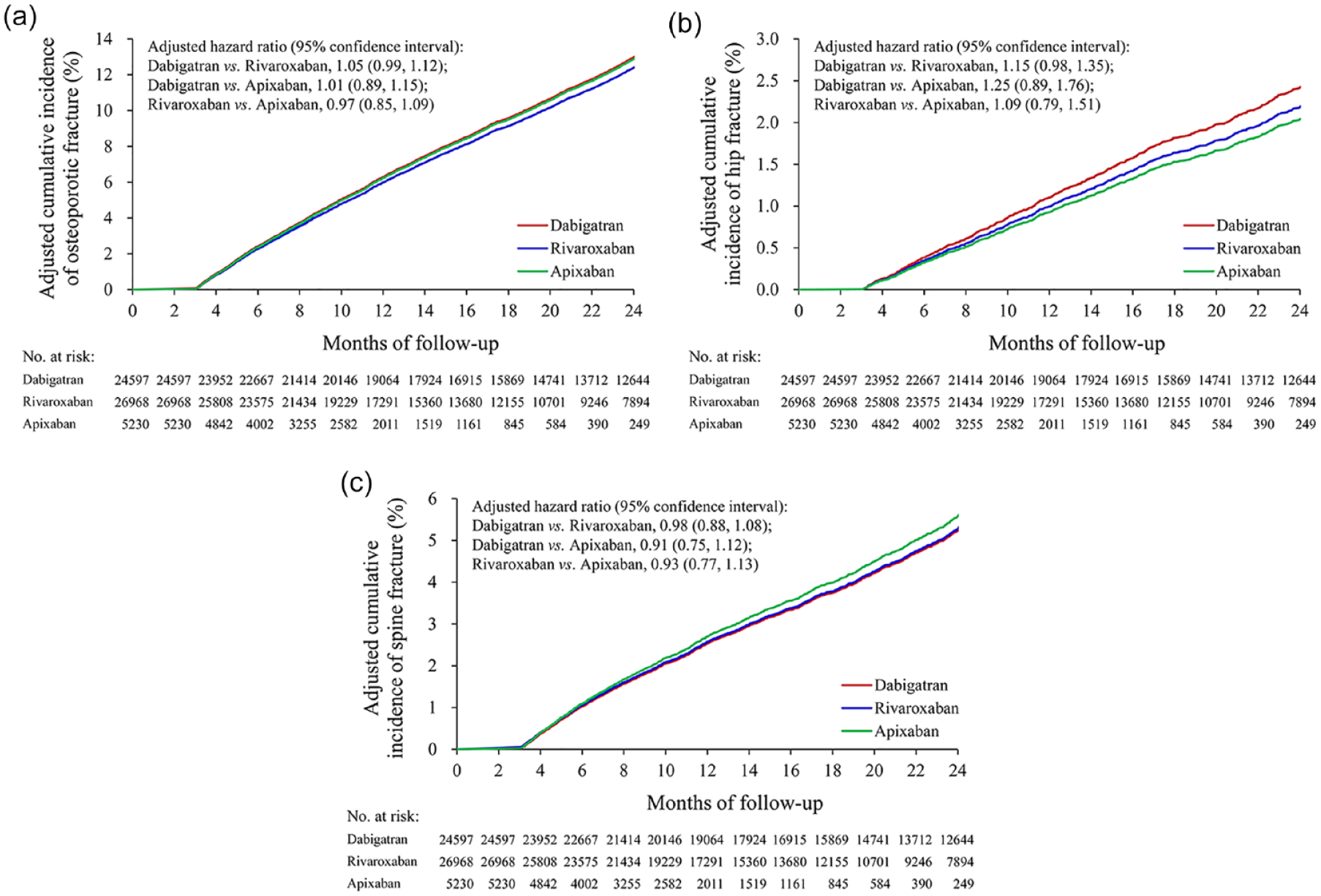
Direct-adjusted cumulative incidence of osteoporotic fractures, hip fractures, and spine fractures in patients receiving dabigatran, rivaroxaban and apixaban treatment.
Subgroup analysis by baseline characteristics
The results of the subgroup analysis demonstrated that most baseline characteristics did not significantly modify the association between type of DOAC and osteoporotic fracture (Figure 3). However, the results revealed that PAD and a history of hip fracture might play a role in the association between type of DOAC and the incidence of osteoporotic fracture (p for interaction = 0.004 and 0.030, respectively). AF patients taking dabigatran had a higher incidence of osteoporotic fracture compared with those taking rivaroxaban or apixaban in patients with concomitant PAD or a history of hip fracture (AF patients with PAD: dabigatran versus rivaroxaban versus apixaban: 14.8% versus 7.6% versus 3.0%, respectively; AF patients with a history of hip fracture: dabigatran versus rivaroxaban versus apixaban: 35.1% versus 26.3% versus 11.4%, respectively). Because there was a trend for a higher incidence of hip fracture in AF patients receiving dabigatran, subgroup analyses were also performed. The results were similar to that of osteoporotic fracture, and there was a significantly higher incidence of hip fracture in AF patients taking dabigatran compared with those taking rivaroxaban or apixaban when they had PAD or a history of hip fracture (Figure 4). The full results of the subgroup analyses on the three outcomes are listed in Supplemental Tables 5–7. Meanwhile, prior VKA use did not significantly modify the association between type of DOACs and osteoporotic fracture (Supplemental Table 8).

Subgroup analysis for osteoporotic fractures at 2 years’ follow up.

Subgroup analysis for hip fracture at 2 years’ follow up.
Subgroup analysis of different types of osteoporotic fractures among those with standard-/low-dose dabigatran, rivaroxaban or apixaban
We further divided AF patients into low- and standard-dose users and analyzed the outcomes among different types of DOACs. In standard-dose DOACs users, AF patients taking dabigatran had the lowest incidence of different types of fractures among the three groups (Figure 5). AF patients taking dabigatran had a lower incidence of spine fracture than those taking rivaroxaban or apixaban at the 6-month, 1-year, and 2-year follow up. Dabigatran users also had a lower risk of osteoporotic fracture compared with rivaroxaban and apixaban users at the 2-year follow up (upper part of Table 3). However, in low-dose users, AF patients taking dabigatran had the highest incidence of osteoporotic and hip fracture among the three groups (Figure 6). Meanwhile, AF patients taking dabigatran had a higher risk of hip fracture compared with those taking apixaban at the 6-month and 1-year follow up, but not at the 2-year follow up (lower part of Table 3). In addition, when the DOAC types were pooled together, the results showed that there was no significant difference between the standard-dose and low-dose groups (Supplemental Table 9).

Direct-adjusted cumulative incidence of osteoporotic fractures, hip fractures, and spine fractures in patients receiving standard dose of dabigatran, rivaroxaban, and apixaban treatment.

Direct-adjusted cumulative incidence of osteoporotic fractures, hip fractures, and spine fractures in patients receiving low dose of dabigatran, rivaroxaban, and apixaban treatment.
Discussion
The nationwide study of patients with AF revealed no significant difference for the risk of osteoporotic fracture at 2 years’ follow up between patients taking different DOACs. However, dabigatran users had a slightly higher risk of hip fracture compared with rivaroxaban and apixaban users at 6-months and 1-year follow up. In AF patients with PAD or a history of hip fracture, and those taking low-dose DOACs, dabigatran users had an even higher risk of developing any one of the composite outcomes, compared with rivaroxaban users.
A previous experimental study showed that dabigatran could inhibit osteoblast and osteoclast differentiation activity, 31 subsequently impairing bone quality, and increasing the risk of osteoporotic fracture. Although our results showed no significant difference in the incidence rate of osteoporotic fracture at 2 years’ follow up, dabigatran seems to be associated with a higher risk of hip fracture when compared with rivaroxaban and apixaban at 6 months and 1 year. Meanwhile, in some subgroups of AF patients, such as those with PAD or a history of hip fracture, dabigatran should not be the recommended anticoagulant when the risks and benefits are considered.
The advantages of DOACs over VKAs in the risk of developing osteoporosis or osteoporotic fractures when treating AF have been established in previous studies.19,32–34 When compared with VKA users, DOAC users had a lower risk of newly diagnosed osteoporosis, 32 any fracture, or major osteoporotic fracture.19,33,34 Apixaban users also had a lower risk of hip fracture when compared with VKA users. 34 Furthermore, Lau et al. 21 compared the fracture risk between different types of DOACs, and found no significant differences for the rate of osteoporotic fracture from a head-to-head comparison at 2 years. However, they did not perform subgroup analyses for fracture location and other subgroups. Findings from the present nationwide study with 2-year follow up showed that dabigatran users had a higher risk of hip fracture in specific patient populations, such as those with PAD and a history of hip fractures.
Since osteoporosis and AF share similar risk factors, prevention of osteoporosis and subsequent fracture is essential when choosing the type of DOAC for AF, especially in certain circumstances. PAD has been associated with a higher risk of falling and subsequent fractures.35–37 PAD was also associated with higher hip-bone-loss rates and an increased risk of non-spine fractures in men.35,36 Furthermore, a large nationwide cohort study showed that PAD was associated with a higher risk of all types of fracture, including hip and non-hip fractures. 37 In the current study, 3% of AF patients had PAD, concomitantly. According to the subgroup analysis, PAD increased the risk of osteoporotic fractures, including hip fractures, in AF patients taking dabigatran, but not in patients taking other types of DOACs. Therefore, dabigatran should not be the first choice DOAC in patients with PAD, especially in patients with an established risk of osteoporosis.
A history of hip fractures is a significant risk factor for secondary hip fractures.38,39 Having a hip fracture may suggest unrecognized and untreated osteoporosis. In the elderly, a history of hip fracture might cause gait abnormality and has been associated with severe fall injuries, 40 and in turn, second hip fractures. Therefore, secondary prevention for hip fracture must incorporate fall prevention. 41 The current study identified that dabigatran potentiated the risk of hip fractures in AF patients, especially when they had a history of hip fracture. Therefore, DOACs other than dabigatran should be selected to treat AF, especially in those patients with a history of hip fracture.
In a previous Danish study, AF patients with more comorbidities, those receiving dabigatran treatment, and those aged older than 65 years were more likely to have a reduced DOAC dose. 42 An elevated risk of stroke, acute myocardial infarction, and major bleeding events were also associated with a reduced dosage of DOACs.42,43 In the current study, we found that the dose of DOACs might affect osteoporotic fracture risk. Compared with standard-dose DOAC users, low-dose DOAC users had a higher risk of fracture, especially spine and osteoporotic fractures. Since evidence that DOACs affect bone structure and strength remains lacking, an explanation for the current findings may be the characteristics of low-dose DOACs users. As mentioned previously, reduced doses of DOACs were associated with those patients who had more comorbidities and an older age, both of which are risk factors for falling and subsequent fractures. However, the percentage difference between the regular and low dose was different among the DOACs. Additionally, the heterogeneity between standard-dose and low-dose strategies might be associated with comorbidities and the degree of fragile status. Therefore, further study is warranted to confirm this dose–response issue.
To the best of our knowledge, this is the first population-based study to compare the risk of osteoporotic fractures at different locations following the administration of different kinds of DOACs in patients with non-valvular AF. A main strength of the current study was its largescale, nationwide design, using the Taiwan NHIRD, which has been recognized as providing high-quality data for large drug surveillance studies.22,23,44 However, there were still several limitations to the current research. First, misclassification bias is inevitable, since the diagnostic accuracy of osteoporosis and osteoporotic fractures is uncertain. We could only use the administrative diagnosis codes to identify outcomes in this retrospective database study rather than directly evaluating patients. Second, uncontrolled confounders were a potential limitation, since we did not have information on the clinical characteristics related to osteoporosis, including smoking history, bone mineral density, and serum calcium and vitamin D. Third, the details of drug adherence were not reported in this study, although the outcomes did not change after controlling the drug censoring in the sensitivity analysis. However, the mean of proportion of days covered (PDC)/medication possession ratio was 81% in 12 months from several observation studies, 45 and even the PDC was >90% in 3 years, from a Japan observation study. 46 Therefore, we supposed that the impact of drug discontinuation might be little in our largescale population. Fourth, the present study was conducted in an Asian population in a single country, which makes generalization of the findings to people of other countries or races uncertain. Further studies are needed to examine the external validity of the findings.
Conclusions
Osteoporotic fracture is a key issue in AF patients, but a dilemma existed in AF patients who needed VKA for stroke prevention. Although DOACs were proven to yield better results than VKA in osteoporosis and osteoporotic fractures, rare studies mentioned outcome differences among the different DOACs. Our national cohort study showed that different DOACs did not have significantly different risks of osteoporotic fracture overall. However, additional concomitant morbidities, such as PAD or a history of hip fracture, and standard/low dose might be associated with different risks when taking different DOACs. Therefore, our findings should be taken into consideration in the clinic when DOACs are chosen.
Supplemental Material
sj-pdf-1-tab-10.1177_1759720X211011374 – Supplemental material for Direct oral anticoagulants and the risk of osteoporotic fractures in patients with non-valvular atrial fibrillation
Supplemental material, sj-pdf-1-tab-10.1177_1759720X211011374 for Direct oral anticoagulants and the risk of osteoporotic fractures in patients with non-valvular atrial fibrillation by Liang-Tseng Kuo, Su-Ju Lin, Victor Chien-Chia Wu, Jung-Jung Chang, Pao-Hsien Chu and Yu-Sheng Lin in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
The authors would like to thank Alfred Hsing-Fen Lin and Zoe Ya-Jhu Syu for their assistance with the statistical analysis.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
This work was supported by grants from Chang Gung Memorial Hospital, Taiwan (grant CFRPG6K0011).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
