Abstract
Objective:
Synovitis, acne, pustulosis, hyperostosis, and osteitis (SAPHO) syndrome is a rare chronic inflammatory disorder and the underlying pathogenesis is unclear. In this study, 88 SAPHO patients and 118 healthy controls were recruited to investigate the role of serum-derived extracellular vesicles (SEVs) in SAPHO syndrome.
Methods:
Quantitative proteomics was applied for SEVs proteome identification, and ELISA and Western blotting was performed to verify the results of mass spectrum data. In vitro osteoclastogenesis and osteogenesis assay was used to confirm the effects of SEVs on bone metabolism.
Results:
Tandem mass tagging-based quantitative proteomic analysis of SAPHO SEVs revealed differential expressed proteins involved in bone metabolism. Of these, serum amyloid A-1 (SAA1) and C-reactive protein (CRP) were upregulated. Higher SAA1 levels in SAPHO patients were confirmed by ELISA. In addition, SAA1 levels were positively correlated with CRP, an inflammatory marker related to the condition of patients. In vitro celluler studies confirmed that SAPHO SEVs inhibited osteoclastogenesis in patients mainly in the active phase of the disease. Further analysis demonstrated that Nucleolin was upregulated in osteoclasts of active-phase patients under SAPHO SEVs stimulation.
Conclusion:
In this study, we identified SAA1 as an additional inflammation marker that can potentially assist the diagnosis of SAPHO syndrome, and speculated that Nucleolin is a key regulator of osteoclastogenesis in active-phase patients.
Introduction
Synovitis, acne, pustulosis, hyperostosis, and osteitis (SAPHO) syndrome is a rare chronic inflammatory disorder involving bone, joints, and skin that was first described in 19871 and occurs with an estimated prevalence in Caucasians of <1/10,000. 2 Bone and joint manifestations, including osteitis, synovitis and hyperostosis, are the hallmark of the SAPHO syndrome and affect a variety of regions of the body. 3 It occurs regardless of the presence of active dermatologic findings. 4 Bone and joint damage can induce pain and bone destruction, which cause spinal stiffness, and even vertebral collapse, thereby affecting the quality of life of patients with SAPHO syndrome.
According to the 2012 Seminars in Arthritis Rheumatism criteria, 4 SAPHO syndrome is classified mainly based on typical clinical manifestations (bone and cutaneous involvement), supplemented by imaging approaches to visualize hyperostosis of the characteristic sites (the anterior chest wall, axial skeleton), and combined with biopsy when necessary. However, no specific laboratory findings can be used to confirm the diagnosis of SAPHO syndrome. Commonly used inflammatory markers, such as C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR) are only applied to evaluate the disease status of SAPHO patients. 5 Thus, the diagnosis of SAPHO syndrome is still challenging.
The precise etiopathogenesis of SAPHO syndrome remains unclear. Although microbial infection and genetic mutations have been reported in a few cases of SAPHO syndrome,6–8 it is generally considered to be an autoinflammatory syndrome. 5 The osteoarticular manifestations in SAPHO patients are closely associated with immune dysregulation, mainly because the skeletal and immune systems share cells and cytokine effectors.9,10 Immune cells and cytokine effectors have powerful effects on bone metabolism and may mediated by extracellular vesicles (EVs).11–13 Previous studies have shown that EVs, mainly exosomes and shed microvesicles, are involved in bone metabolism in both normal physiological and disease states14,15 likewise in multiple inflammatory diseases. 16 In addition, EVs from monocytes, leukocyte and macrophage exert directly effects on osteoclastogenesis and osteogenesis,17–19 indicating that EVs function as mediators between the immune system and bone metabolism. As SAPHO syndrome is an inflammatory-mediated disease characterized mainly by systemic osteoarticular damage, we hypothesized that EVs play an important role in bone and joint damage in SAPHO syndrome.
In this study, we explored the role of serum-derived EVs (SEVs) in SAPHO syndrome, focusing mainly on identification of differential expressed proteins (DEPs) in SAPHO SEVs and the function of SEVs on bone metabolism.
Materials and methods
Patients and healthy controls
From October 2017 to August 2018, a total of 88 patients with SAPHO syndrome were recruited from the single-center dynamic cohort of SAPHO syndrome in Peking Union Medical College Hospital (PUMCH, Beijing, China). 20 All enrolled patients met the following inclusion criteria: (1) adults (⩾18 years old); (2) in accordance with the criteria for diagnosis of SAPHO reported by Nguyen et al.; 4 (3) anterior chest wall involvement. After enrolment, the blood samples were collected for basic laboratory tests as well as the measurement of inflammatory markers [CRP, ESR] and bone metabolism markers [β-isolated C-terminal peptide (β-CTX), Osteocalcin]. Patient-reported assessments, including Visual Analogue Score for global pain, Bath Ankylosing Spondylitis Disease Activity Index (BASDAI), and Ankylosing Spondylitis Disease Activity Score-CRP (ASDAS-CRP) were also conducted during the same disease episode (within a week) to evaluate the SAPHO syndrome disease activity. According to previous studies, SAPHO syndrome patients with BASDAI score ⩾4 and ASDAS score ⩾2.1, which indicated currently high disease activity, were defined at the active phase, otherwise patients were deemed to be in the resting phase of the disease. In addition, 118 healthy controls were also recruited from Physical Examination Center of PUMCH. The clinical information of SAPHO patients and healthy controls is shown in Supplemental Table 1.
Of the 88 SAPHO patients, 41 were used for SEVs mass spectrometry analysis and SEVs stimulation, 54 for ELISA, and 36 for peripheral blood mononuclear cells (PBMC) purification. For 118 healthy controls, 56 were used for SEVs MS analysis, 48 for SEVs stimulation, 84 for ELISA, 10 for PBMC purification, and one for human adipose tissue-derived mesenchymal stem cell (hADMSC) purification. The patients and healthy controls used in these experiments are partially overlapped. The enrolment criteria for all patients and healthy controls were the same, with no selection for each experiment. Detailed information is shown in Supplemental Table 1.
The human adipose tissue used in the present study was obtained from donors who underwent liposuction at the Department of Plastic Surgery, PUMCH.
All patients and healthy controls provided written informed consent to participate and the study was approved by the Ethics Committee of PUMCH (No. ZS-994).
Isolation of EVs from human serum
EV isolation by ultracentrifugation was carried out as previously described. 21 Briefly, pooled serum was first centrifuged (10,000×g for 30 min at 4°C) to remove cell debris. The supernatant was filtered (0.22 μm pore size) and then re-ultracentrifuged (110,000×g for 90 min at 4°C) using a Beckman Optima L-100XP Ultracentrifuge. The supernatant was removed, and the pellet containing the SEVs was washed three times with 1 mL phosphate-buffered saline (PBS). The SEVs were then resuspended and dissolved in 8 M urea for tandem mass tag (TMT)-labeling. The protein concentration was determined using a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific).
For EV isolation using the Total Exosome Isolation Reagent (from serum) (Life Technologies, Carlsbad, CA, USA), pooled serum samples were diluted with an equal volume of PBS to decrease viscosity, followed by the addition of 0.2 volume of the Total Exosome Isolation Reagent. Samples were vortexed and incubated at 4°C for 30 min. Subsequently, samples were centrifuged (10,000×g for 30 min at room temperature) and the pellet containing the SEVs was resuspended in PBS. The SEVs were then lysed with RIPA buffer and the protein content was measured using BCA assay. SEVs isolated by reagent were used for Western blotting, nanoparticle tracking analysis (NTA), and SEVs stimulation in vitro osteoclastogenesis and osteogenesis assays.
TMT labeling
The detailed TMT labeling procedure is supplied in the supplemental methods. Healthy control SEVs were labeled with TMT-128 and SAPHO SEVs were labeled with TMT-131. For labeling of osteoclasts (OCs) in the response group, the OCs stimulated by healthy SEVs were labeled with TMT-129, and those stimulated by SAPHO SEVs were labeled with TMT-131. In the non-response group, the OCs stimulated by healthy SEVs were labeled with TMT-126 and those stimulated by SAPHO SEVs were labeled with TMT-130.
Bioinformatics analysis
The changes in protein expression in SAPHO SEVs were represented as the TMT-131/128 ratio (SAPHO/healthy control). According to the 95% prediction interval, log2(131/128) ratios ⩾1.24 was set as the threshold for upregulated proteins, while log2(131/128) ratios ⩽−2.17 were set as the threshold for downregulated proteins. The mass spectrometry proteomics data have been deposited in the ProteomeXchange Consortium via the PRIDE partner repository with the dataset identifier PXD012125.
The changes in protein expression in OCs stimulated by SAPHO SEVs were represented by the TMT-131/129 ratio in response group and 130/126 ratio in the non-response group. According to the 95% prediction interval, the log2(131/129) ratio ⩾0.20 was set as the threshold for upregulated proteins in the response group, while the ratio ⩽−0.22 was set as the threshold for downregulated proteins. Similarly, in the non-response group, the log2(130/126) ratio ⩾0.21 was set as the threshold for upregulated proteins, and the ratio ⩽−0.18 was set as the threshold for downregulated proteins. The mass spectrometry proteomics data have been deposited in the ProteomeXchange Consortium via the PRIDE partner repository with the dataset identifier PXD012218.
Gene ontology (GO) enrichment analyses were carried out using the DAVID online analysis tool (https://david.ncifcrf.gov/). Protein–protein interactions were illustrated using Cytoscape software (3.8) based on the STRING app.
Cell culture
The details of the methods used for purification of hADMSCs and PBMCs, the induction of osteogenesis from hADMSCs, and osteoclastogenesis from PBMCs are supplied in the supplemental methods.
Statistical analysis
The number of OCs was determined by counting Tartrate-Resistant Acid Phosphatase (TRAP) stain-positive cells with more than three nuclei. The area of OCs and mineralized point were measured by Image-J. SPSS 24.0 (IBM Corp., Armonk, NY), and GraphPad Prism (version 7.04; Nashville, TN, USA) software was used for data presentation and statistical analysis. Variables with normal distribution were expressed as mean ± standard deviation (SD), while variables with non-normal distribution were expressed as median and interquartile range, unless otherwise indicated. The χ 2 test was used for comparing the characteristics of patients (sex, BASDAI score, ASDAS score, ESR, CRP, β-CTX and Osteocalcin) in the response and non-response groups. The Mann–Whitney U-test analysis was used for non-normally distributed continuous variables (OC area and OC number). For normally distributed continuous variable (age) Student’s t-test was used. A value of p < 0.05 was considered to indicate statistical significance.
Details of reagents, NTA, high-performance liquid chromatography analysis, liquid chromatography tandem-mass spectrometry (LC-MS/MS) analysis, TMT labeling, protein identification, cell culture, bone resorption assay, TRAP staining, and Alizarin red staining are supplied in the supplemental methods.
Results
Project design
A flow chart of the study design is shown in Figure 1. The serum SEVs of 41 SAPHO patients and 56 healthy controls were isolated by ultracentrifugation or using appropriate reagents. The SEVs isolated by ultracentrifugation were used for TMT-based quantitative proteomics and those isolated by reagent were used for Western blotting, NTA and stimulation of osteogenesis and osteoclastogenesis. Expression of the EV markers CD63 and ALIX were verified by Western blotting. The particle diameters and concentration of EVs were evaluated by NTA.
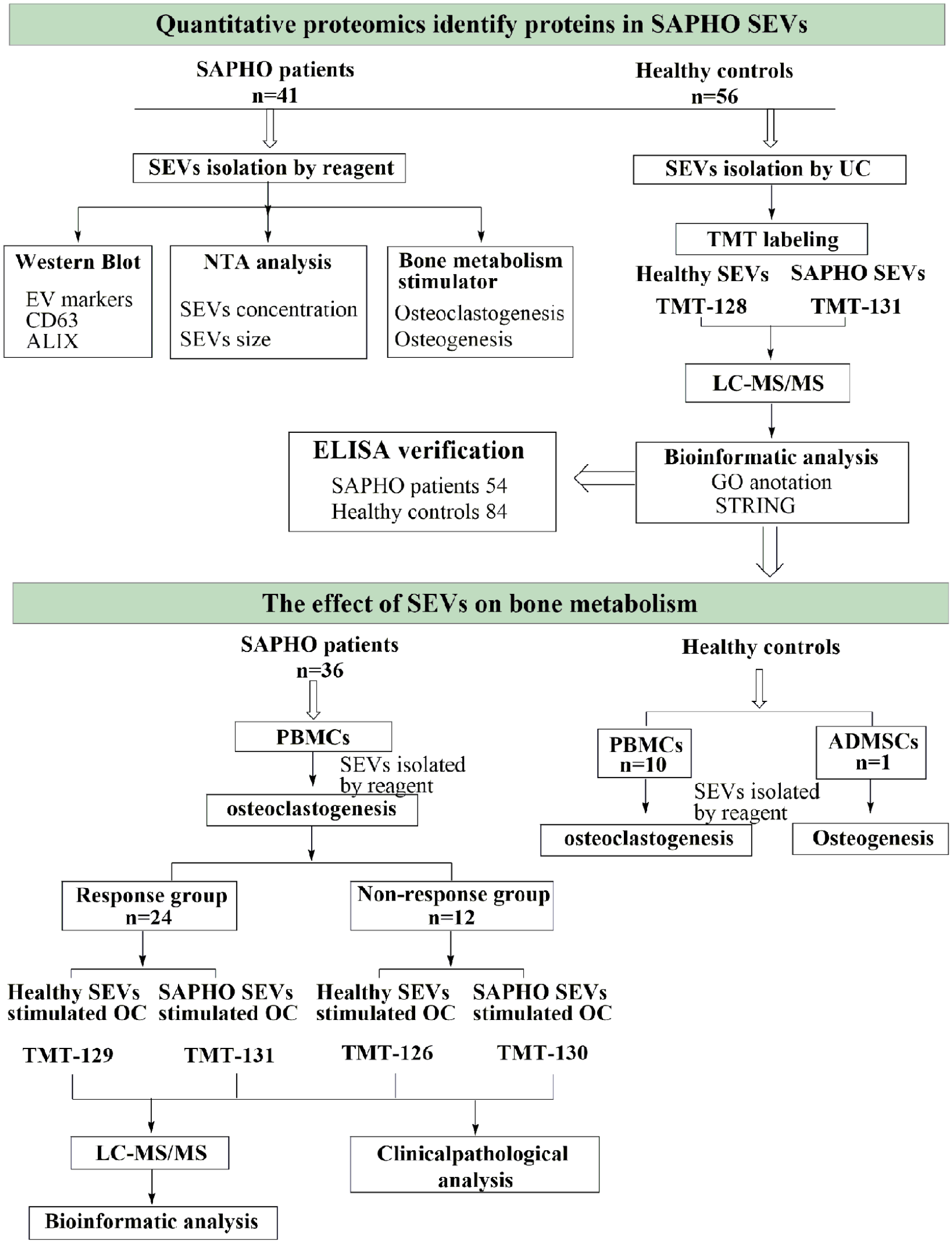
The workflow of the project. SEVs, serum-derived extracellular vesicles; EV, extracellular vesicles; NTA, nanoparticle tracking analysis; UC, ultracentrifugation; TMT, tandem mass tag; LC-MS/MS, liquid chromatography tandem-mass spectrometry; GO, gene ontology; STRING, protein-protein interaction networks analysis by STRING (version 11.0); PBMCs, peripheral blood mononuclear cells; ADMSCs, adipose tissue-derived mesenchymal stem cells; OC, osteoclast.
For verification of the mass spectrometry results showing higher SAA1 level in SAPHO SEVs, serum samples from 54 SAPHO patients and 84 healthy controls were analyzed by ELISA.
The function of SEVs in bone metabolism was further investigated in another 36 SAPHO patients and 11 healthy controls. The PBMCs isolated from 36 SAPHO patients and 10 healthy controls were induced to form OCs. The hADMSCs from one healthy control were induced to form osteoblasts (OBs). The effect of SEVs on osteoclastogenesis and osteogenesis were evaluated by TRAP staining and Alizarin red staining, respectively.
While exploring the effect of SEVs on osteoclastogenesis, we found that OC differentiation was inhibited by SAPHO SEVs in 24 SAPHO patients; these patients were classified as the response group. In contrast, no changes under SAPHO and healthy SEV stimulation were observed in the other 12 SAPHO patients; these patients were classified as the non-response group. Statistical analysis and quantitative proteomics were further applied to explain the clinical and molecular mechanisms of the response to SEVs in SAPHO patients.
Verification of SEVs
The SEVs isolated using reagents were used for Western blotting and NTA analysis. Western blot analysis showed that EV markers CD63 and ALIX were both enriched in the SEV pellet fraction, but not in whole serum and the supernatant fraction (Figure 2a). NTA showed that mean particle diameter of SEVs in the healthy controls and SAPHO patients was 176 nm and 152 nm, respectively (Figure 2b), with no significant difference between these two groups (Figure 2c). In addition, there was no significant differences in particle concentration of SEVs between healthy controls and SAPHO patients (Figure 2d).

Serum-derived extracellular vesicle (SEV) purification and identification. (a) Purified SEVs were verified by Western blot detection of EV markers ALIX and CD63. Equal amounts of proteins from each sample were loaded. The Coomassie Blue staining of the total proteins separated by SDS-PAGE is shown. (b) Nanoparticle tracking analysis (NTA) analysis of SEVs of SAPHO patients and healthy controls. (c) The particle size of two types of SEVs. (d) The particle concentration of two types of SEVs. (e) The overlapped proteins between identified SEVs proteins and the ExoCarta database. (f–h) Gene ontology annotation of SEVs proteins identified by mass spectrometry based on biological process (f), cellular component (g), and molecular function (h).
The SEVs purified by ultracentrifugation were used for TMT-based quantitative proteomics. A total of 1620 proteins were identified both in healthy controls and SAPHO patients, of which 803 trusted proteins were identified (score ⩾10, unique peptide ⩾2). The Uniprot Accession numbers of trusted proteins were then mapped to 768 Entrez Gene IDs. Based on the Entrez Gene ID, 692 of 768 proteins (90%) overlapped with the ExoCarta database (Figure 2e, http://www.exocarta.org, release date: 29 July 2015).
GO analysis showed that the total proteins identified in SEVs were mainly enriched in immune regulation-related process, such as complement activation, Fcγ receptor signaling pathway, and proteolysis (Figure 2f), and located in the extracellular matrix, extracellular region, extracellular space, and exosome (Figure 2g). The molecular functions of these proteins were antigen binding, heparin binding, and cadherin binding involved in cell–cell adhesion (Figure 2h). These data indicated that the purification of EVs from serum was successful.
Characteristics of DEPs in SAPHO SEVs
Based on the criteria list in the Materials and Methods section, 49 DEPs were filtered in SAPHO SEVs with 10 proteins upregulated and 39 downregulated (Supplemental Table S2). GO analysis showed that the downregulated proteins were enriched in the following biological processes: platelet degranulation, signal peptide processing, peptidyl-glutamic acid carboxylation, and extracellular matrix organization (Figure 3a). Apart from the extracellular space, extracellular region, and extracellular exosome, the downregulated proteins also located in EVs generating cell organelles, such as endoplasmic reticulum lumen, platelet alpha granule lumen, and Golgi lumen (Figure 3a). In addition, the enriched molecular functions were bone metabolism related, such as endopeptidase inhibitor activity, calcium ion binding, collagen binding, and heparin binding (Figure 3a). The upregulated proteins were not significantly enriched in any GO items, but nonetheless, CRP and SAA1, two acute-phase proteins, are highly related to immune response and inflammation. The protein–protein interaction analysis based on STRING database showed close interconnection between the DEPs (Figure 3b).

Biological analysis of DEPs in SAPHO SEVs. (a) Gene ontology annotation of the DEPs identified by mass spectrometry in SAPHO SEVs based on biological process, cellular component, and molecular function. (b) The protein–protein interaction network of DEPs in SAPHO SEVs, constructed using the STRING online tool. Red represents upregulation and blue represents downregulation. The proteins involved in bone metabolism are shown in yellow text. (c) Serum SAA1 levels in healthy controls and SAPHO patients detected by ELISA. (d) SEVs SAA1 levels of healthy controls and SAPHO patients detected by ELISA. In (c) and (d), the patients were further separated into resting and active-phase groups and the SAA1 levels was compared. (e, f) ROC curve analysis of the SAA1 in serum (e) and SEVs (f) for SAPHO diagnosis. (g, h) Pearson correlation analysis of CRP levels with serum (g) and SEVs SAA1 levels (g). Data represent the mean ± standard deviation. **p < 0.01, ****p < 0.0001, two-tailed unpaired t-test. SAA1, Serum amyloid A-1; SEVs, serum-derived extracellular vesicles; AUC, area under curve; CRP, C-reaction protein.
SAA1 is highly expressed in SAPHO patients
CRP is a laboratory inflammatory indicator associated with high levels of disease activity in SAPHO patients. 19 However, the relationship between SAA1 and SAPHO remains unclear. To investigate this relationship, we analyzed the levels of SAA1 in 54 SAPHO patients and 84 healthy controls by ELISA. Compared with the healthy controls, the SAA1 level of SAPHO patients was elevated both in serum and SEVs (Figure 3c and 3d). However, there was no significant difference between the resting and active phases of the disease. Furthermore, receiver operating characteristic (ROC) curve analysis showed that SAA1 could be used to distinguish SAPHO patients from healthy controls with moderate accuracy, with an area under the curve (AUC) of 0.62 for serum SAA1 and AUC 0.71 for SEVs SAA1 (Figure 3e and 3f). The best cut-off value for serum SAA1 was identified as 1539.47 ng/ml, with a specificity of 0.88 and a sensitivity of 0.35. For SEVs, the best cut-off value was identified as 15.12 ng/mg, with a specificity of 0.68 and a sensitivity of 0.65. Pearson correlation analysis showed that SAA1 was positively correlated with serum CRP, both in serum (r = 0.48, p = 0.0002, Figure 3g) and SEVs (r = 0.47, p = 0.0003, Figure 3h). These results implicated that SAA1, as an additional inflammation marker, may potentially assist SAPHO diagnosis.
SAPHO SEVs inhibit osteoclastogenesis in SAPHO patients in the active phase of the disease
Of the 49 DEPs, 18 proteins (37%) were identified as bone metabolism regulators, which were labeled with yellow in STRING network (Figure 3d). The details of protein function in bone metabolism are shown in Table 1. This result indicated that SEVs play roles in the abnormal bone metabolism associated with SAPHO.
Bone metabolism related DEPs.

To investigate the influence of SEVs on osteoclastogenesis in SAPHO patients, PBMCs were purified and OC differentiation was induced. In 24 (2/3) SAPHO patients, osteoclastogenesis was significantly inhibited by SAPHO SEVs. TRAP staining revealed significantly fewer and smaller OCs under SAPHO SEVs stimulation than that under healthy SEVs stimulation (Figure 4a). In the other 12 cases, however, there were no significant differences between OCs stimulated by SEVs derived from SAPHO patients and healthy controls (Figure 4b). Detailed statistical results of OC area and number were shown in Figure 4c. The patients with inhibited OCs by SAPHO SEVs were classified as the response group, and those with no changes in OCs were classified as the non-response group. Representative TRAP staining images of OCs of all 36 SAPHO patients are shown in Supplemental Figure S1.
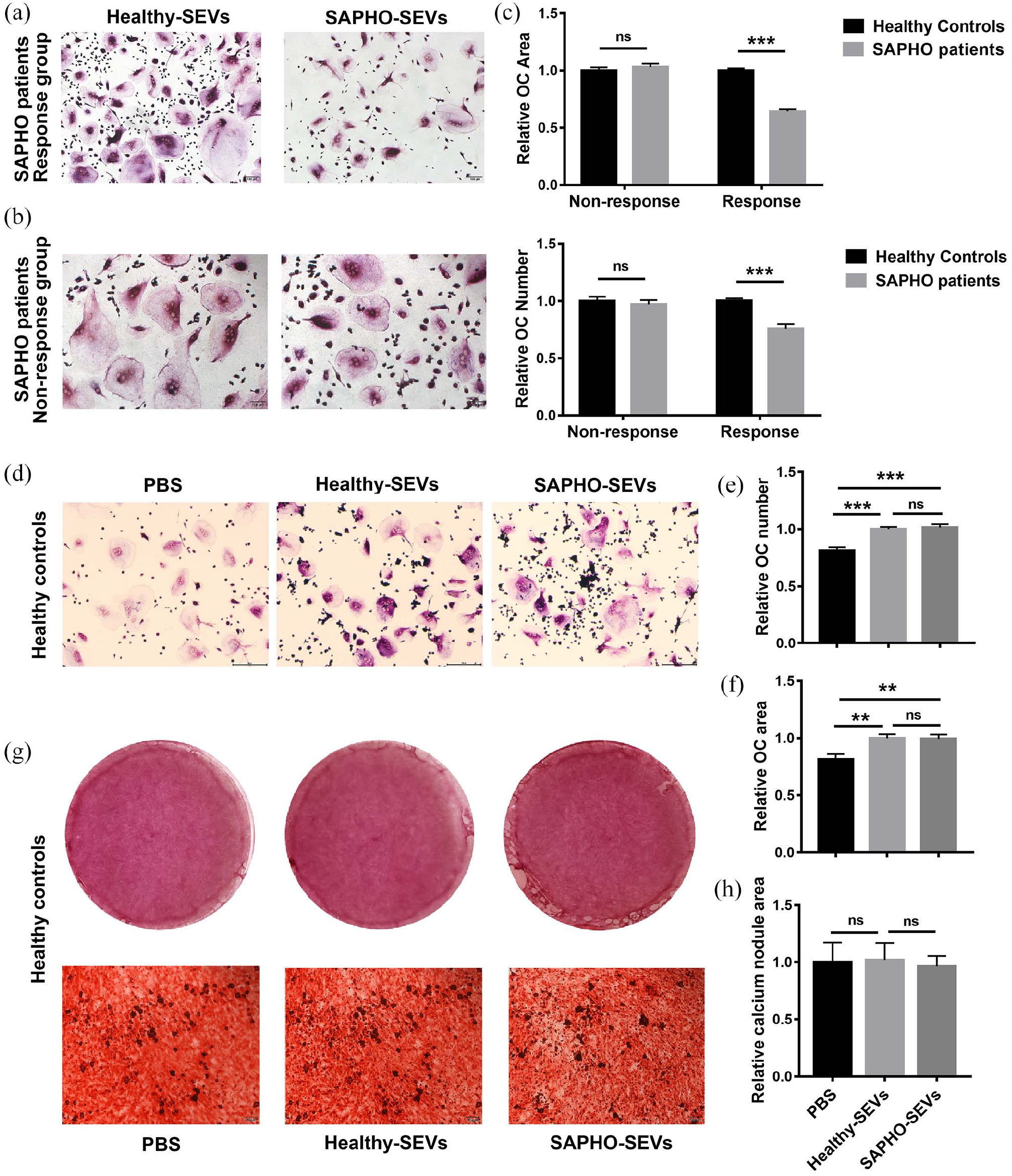
SAPHO SEVs inhibit osteoclastogenesis SAPHO patients in the active phase of the disease. (a, b) Representative TRAP staining images of OCs in patients in the response group (a) and the non-response group (b) under SEVs stimulation. The TRAP staining images of each patient are shown in Supplemental Figure S1. (c) OC number and area calculation and comparison. The average number or area of OCs in each patient stimulated by healthy SEVs was normalized to 1. (d) Representative TRAP staining images of OCs in healthy controls under SEVs or PBS stimulation. (e, f) OC number (e) and area (f) calculation and comparison. The average number or area of OCs in each healthy control stimulated by healthy SEVs was normalized to 1. (g) Alizarin red staining images of calcium node in osteoblasts differentiated from human ADMSCs under SEVs or PBS stimulation. (h) Calcium node area calculation and comparison of osteoblasts differentiated from ADMSCs under SEVs or PBS stimulation. Data represent the mean ± standard deviation. ***p < 0.001, two-tailed unpaired t-test. SEVs, serum-derived extracellular vesicles; OC, osteoclast.
To investigate the potential correlation of clinical factors and patients’ responses, the clinical features of SAPHO patients in the response and non-response group were compared and assessed (Table 2). Chi-square test showed that the patients’ response was associated with BASDAI score (|rs| = 0.378, p = 0.023), ASDAS score (|rs| = 0.359, p = 0.031) and CRP level (|rs| = 0.347, p = 0.037). According to both the BASDAI score (8/8) or ASDAS score (13/15), it can be seen that almost all the active-phase patients were within the response group. However, for the resting-phase patients, there was no significant difference of patients’ number in the response and the non-response groups, with approximately even distribution. This indicates that SAPHO patients in the active phase are more susceptible to the effects of SAPHO SEVs on osteoclastogenesis than those in the resting phase. In addition, the patients’ response was not correlated with demographic characteristics (such as sex and age) and bone metabolism markers (such as Osteocalcin and β-CTX).
Relationship between patient response and clinical features.

Statistically significant (p < 0.05). OC, osteoclast; ESR, erythrocyte sedimentation rate CRP, C-reaction protein; β-CTX, β-isolated C-terminal peptide. The bold indicates significant difference.
SAPHO SEVs have no influence on osteoclastogenesis and osteogenesis in healthy people
The function of SEVs on bone metabolism in healthy individuals was evaluated in 11 healthy controls. TRAP staining showed that both healthy and SAPHO SEVs promote the OC differentiation, but no significant difference was observed between the effects of the two types of SEVs (Figure 4d–f). Representative TRAP staining images of 10 healthy controls are shown in Supplemental Figure S2.
To evaluate the effect of SEVs on osteogenesis in healthy individuals, in vitro osteogenesis assay was performed. Human ADMSCs from healthy controls were incubated with β-GP, L-AA, and dexamethasone to induce OB formation in the presence or absence of SEVs. After 24 days, calcium nodules appeared and were stained with Alizarin Red S. The result showed that there were no significant differences in the calcium nodes formed under stimulation with PBS, SEVs derived from SAPHO patients, or SEVs derived from healthy controls (Figure 4(g) and 4h). These results suggested that SAPHO SEVs did not disturb the osteoclastogenesis and osteogenesis processes in healthy individuals.
Antigen presentation-associated proteins are downregulated in OCs of patients in response group under SAPHO SEVs stimulation
To clarify the molecular mechanisms of the response of SAPHO patients to SEVs, we analyzed the proteomic differences between OCs from SAPHO patients in the response and non-response groups (Figure 1). The identified total and DEPs are shown in Supplemental Table 3. The ratios of TMT-131/129 and TMT-130/126 were determined as a representation of the changes induced by SAPHO SEVs in response group and non-response group, respectively. The DEPs in the response group and the non-responsive group were screened out based on the 95% prediction interval. Protein expression heatmaps showed dramatic differences between the DEPs in the response and non-response group (Figure 5a). The overlaps of the upregulated and downregulated proteins between the two groups were less than 18% and 34%, respectively (Figure 5b and 5c). The protein–protein interaction network of DEPs in the response group showed that the downregulated proteins were enriched in the antigen processing and presentation process, including MHC class I protein (HLA-A, HLA-C, LILRB4, TAPBP), MHC class II proteins (HLA-DRA, HLA-DRB1, HLA-DRB5, HLA-DPA1, CD74), and the Fcγ receptor family proteins (FCGR1A and FCGR3A) (Figure 5d).
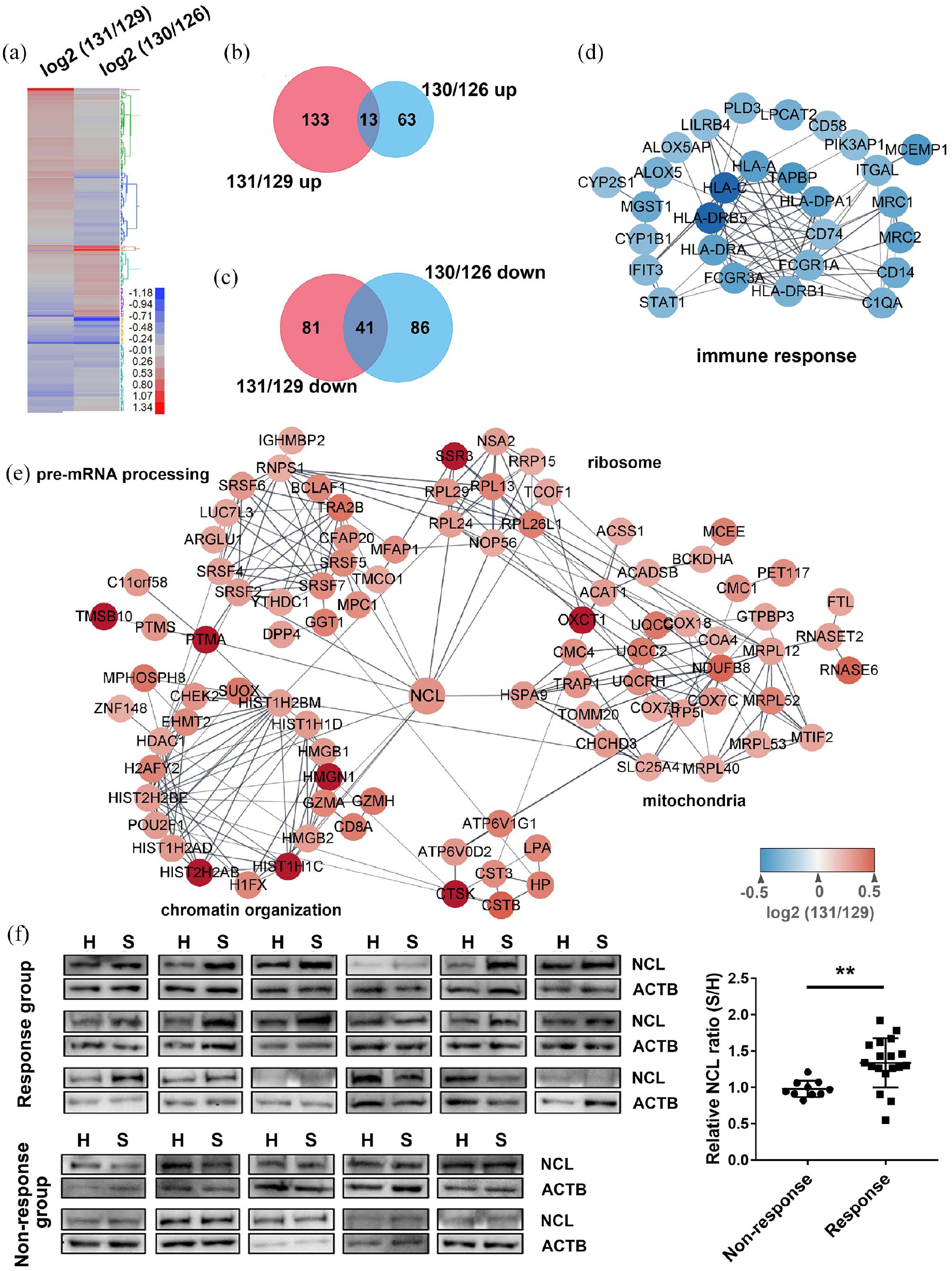
Bioinformatic analysis of DEPs in OCs in the response group patients. (a) Heatmap of DEPs in OCs from the response (131/129) and non-response (130/126) group under SAPHO SEVs stimulation. (b, c) Venn diagram of upregulated proteins (b) and downregulated proteins (c) in the response (131/129) and non-response (130/126) group. (d, e) Protein–protein interaction network of downregulated proteins (d) and upregulated proteins (e) in OCs of response group patients under SAPHO SEVs stimulation. (f) Western Blot verification of NCL upregulation in OCs of the response group patients under SAPHO SEVs stimulation. No changes in NCL expression were induced by SAPHO SEVs in the non-response group. Grayscale analysis results are displayed in dot plots. Data represent the mean ± standard deviation. **p < 0.01, two-tailed unpaired t-test. H, healthy; S, SAPHO; NCL, nucleolin; ACTB, beta actin.
Nucleolin is a core protein involved in osteoclastogenesis in response group patients under SAPHO SEVs stimulation
The proteins that were upregulated in the OCs from the patients in the response group following stimulation by SAPHO SEVs were enriched in cellular processes including chromatin organization, pre-mRNA processing, mRNA pre-treatment, ribosome organization and mitochondria. These proteins were linked by Nucleolin (NCL) (Figure 5e). Western blot analysis verified higher NCL level in response group OCs stimulated by SAPHO SEVs. In contrast, there was no difference of NCL level between the OCs stimulated by SAPHO SEVs and healthy SEVs in the non-response group (Figure 5f).
Discussion
SAA1 as an inflammation marker may potentially assist SAPHO syndrome diagnosis
SAPHO syndrome is a chronic inflammatory disease involving bone, joints, and skin, with unclear etiology and pathogenesis. The diagnosis of SAPHO syndrome is based mainly on the clinical manifestations and radiological or magnetic resonance imaging. Due to the variety of clinical manifestations and sharing features with other established disease categories, the accurate diagnosis of SAPHO syndrome remains challenging. At present, there are no specific diagnostic laboratory tests of SAPHO. Although laboratory tests of non-specific inflammatory markers such as CRP and ESR are commonly used to evaluate the condition of patients, no consistent correlation with SAPHO has been identified.20,42–44
Higher circulating concentrations of the acute-phase protein SAA1 are associated with inflammatory conditions,45,46 such as rheumatoid arthritis (RA) and ankylosing spondylitis (AS).47–49 In the current study, using 54 SAPHO patients and 84 healthy controls, we found that SAA1 levels were increased in SAPHO patients, and were correlated with CRP levels. ROC curves analysis indicated that SAA1 could distinguish SAPHO patients from healthy controls with moderate accuracy. In addition, to evaluate whether SAA1 could also distinguish SAPHO from other inflammation disease, 22 RA and 29 AS patients were recruited and their SEVs’ SAA1 level was also detected. We found that SAA1 level was increased in SAPHO, RA and AS patients compared with healthy controls. However, no significant difference was seen among SAPHO, RA and AS patients, although the level of SEVs’ SAA1 in SAPHO is slightly higher than that in RA and AS patients, especially in AS patients (Supplemental Figure S3). Due to the limitation of the number of patients and the type of disease, a definite conclusion could not be drawn. Thus, to determine whether SAA1 could be an additional inflammation marker assist SAPHO diagnosis, a larger sample size and more disease types are required.
Wekell et al. 50 reported that SAA1 appears to be a more sensitive inflammatory marker than CRP when monitoring the improvement (the resting) and relapses (the active) phase in SAPHO syndrome. However, our small-scale study showed that there were no significant differences in SAA1 levels between SAPHO patients in the active and resting phase of the disease, which is not consistent with the finding of Wekell et al. 50 It can be speculated that this discrepancy is due to differences in the focus of these two studies, with our study comparing SAA1 levels between different patients in the resting and active phases, whereas Wekell and colleagues investigated the dynamic changes of SAA1 in the same patients during disease progression. This difference may also be related to the wide difference in sample sizes in these two studies, with only two patients recruited into the study conducted by Wekell et al. Thus, whether SAA1 could be used as a monitoring marker for disease activity still needs to be studied.
SAPHO SEVs inhibit osteoclastogenesis in patients in the active phase of the disease
SAPHO syndrome is a stable entity, with a good long-term prognosis.42,43 The osteoarticular manifestations in patients with SAPHO syndrome show little progression over more than 5 years. A few patients achieve disease-free status after a limited course lasting 3–6 months. And most patients have a chronic course characterized by fluctuating intermittent periods of exacerbation and short-term improvement. During the active phase (exacerbation period), osteolysis occurs in the sternoclavicular joint, causing pain. After a period of time, the osteolysis disappears and the pain is relieved for unknown reasons, and the patient enters the resting phase (improvement period). This relapsing–remitting course and good prognosis suggests that the patients may have self-relief ability. At present, the mechanism regulating the transition from active to resting phase is still unclear. Our results show that SAPHO SEVs specifically inhibit osteogenesis in patients in the active phase of the disease, suggesting that some unknown factors in SEVs may induce the transition from the active to the resting phase.
NCL plays roles in osteoclastogenesis in active phase: Patients under SEVs stimulation
NCL, which is the major nucleolar protein in eukaryotic cells, is involved in chromatin decondensation and ribosome synthesis and maturation.51–53 Our proteomic data analysis showed that NCL is a core protein connecting proteins in pre-RNA processing, ribosome, and chromatin organization-related proteins, which is consistent with previous reports.52,54 Proteins in the mitochondria, a key cellular organelle in OC differentiation,55,56 were also linked with NCL by HSPA9. Furthermore, in our previous study, we observed high levels of NCL protein in patients with osteopenia and osteoporosis, indicating NCL promotes osteoclastogenesis. 57 These results implicate NCL as a novel regulator of OC differentiation, possibly through regulation of chromatin organization, pre-mRNA processing, and mitochondria. However, further studies are required to clarify the exact role of NCL in OC differentiation.
SEVs negatively regulate immune responses in active-phase SAPHO patients
EVs are involved in numerous physiological processes, and vesicles from both immune and non-immune cells have important roles in immune regulation. 58 Immunized blood-borne EVs have been shown to reduce an active immune response to a specific antigen. 59 It has been hypothesized that SEVs suppress body responses to peripheral self-antigens and to commonly encountered foreign antigens to inhibit chronic inflammation and autoimmunity. 58
SAPHO is generally considered as an autoinflammatory syndrome, with elevated pro-inflammatory cytokines (IL-1, IL-8, IL-17, and IL-18) and Th17 cells,5,60 implying that patients are under a highly inflammatory condition. Indeed, SAPHO patients are mostly suffering from long-term chronic inflammation. Antigen-presenting cells express higher levels of MHC class II proteins under inflammatory condition, thereby enhancing self-antigen presentation, 61 suggesting that SAPHO patients are likely to have autoimmune reactions. However, current reports suggest that there is no significant increase in self-antigen content in SAPHO patients. 62 OCs are derived from monocytes/macrophages and inherit the characteristics of monocytes as antigen-presenting cells, including the capacity to engulf pathogens and present antigens to T cells via MHC family proteins.63,64 In this study, SAPHO SEVs reduced the OC expression of proteins in MHC and FCGR families, which play important roles in the antigen presentation process. It is reasonable to speculate that SEVs weaken the self-antigen presentation ability, thereby ameliorating the responses to self-antigens in SAPHO patients. In addition, a previous study showed that loss of MHC class II proteins does not alter osteoclastogenesis and osteogenesis; 65 therefore, we speculate that the SAPHO SEVs regulate the antigen presentation process and osteoclastogenesis in parallel, rather than being a causal relationship. The effects of SEVs on antigen presentation may also promote the transition of SAPHO patients from the active to the resting phase of the disease. Further investigation of the role of SAPHO SEVs in antigen presentation may provide a greater understanding of the pathogenesis of SAPHO syndrome and potential therapeutic strategies.
Limitations
Since SAPHO is a rare disease, the sample size of patients included in this study is limited. SAA1 is considered as a non-specific inflammatory marker, which is elevated in a variety of inflammatory diseases, like RA and AS. A diagnostic marker specific for SAPHO should not only be able to distinguish SAPHO from healthy status, but also from other inflammation-related diseases that have clinical manifestations similar to SAPHO, including RA and AS. Therefore, SEVs’ SAA1 levels were also detected in patients with RA and AS. We obtained some preliminary results. However, due to the limitation of the number of patients and the type of disease, a definite conclusion could not be drawn. Thus, further studies with larger sample size and more disease types are required to determine whether SAA1 as an inflammation marker could assist the diagnosis of SAPHO syndrome.
Conclusion
This study speculates that SAA1, as an additional inflammation marker, may assist SAPHO syndrome diagnosis. This study also shows that the SAPHO SEVs inhibit osteoclastogenesis in active-phase patients, and demonstrates the role of NCL as a core regulator in this process. However, further studies are required to evaluate the diagnostic performance of SAA1 and fully elucidate the specific mechanism by which SEVs affect osteoclastogenesis.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X211006966 – Supplemental material for Serum-derived extracellular vesicles inhibit osteoclastogenesis in active-phase patients with SAPHO syndrome
Supplemental material, sj-docx-1-tab-10.1177_1759720X211006966 for Serum-derived extracellular vesicles inhibit osteoclastogenesis in active-phase patients with SAPHO syndrome by Yanpan Gao, Yanyu Chen, Lun Wang, Chen Li and Wei Ge in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X211006966 – Supplemental material for Serum-derived extracellular vesicles inhibit osteoclastogenesis in active-phase patients with SAPHO syndrome
Supplemental material, sj-docx-2-tab-10.1177_1759720X211006966 for Serum-derived extracellular vesicles inhibit osteoclastogenesis in active-phase patients with SAPHO syndrome by Yanpan Gao, Yanyu Chen, Lun Wang, Chen Li and Wei Ge in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-xlsx-3-tab-10.1177_1759720X211006966 – Supplemental material for Serum-derived extracellular vesicles inhibit osteoclastogenesis in active-phase patients with SAPHO syndrome
Supplemental material, sj-xlsx-3-tab-10.1177_1759720X211006966 for Serum-derived extracellular vesicles inhibit osteoclastogenesis in active-phase patients with SAPHO syndrome by Yanpan Gao, Yanyu Chen, Lun Wang, Chen Li and Wei Ge in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-xlsx-4-tab-10.1177_1759720X211006966 – Supplemental material for Serum-derived extracellular vesicles inhibit osteoclastogenesis in active-phase patients with SAPHO syndrome
Supplemental material, sj-xlsx-4-tab-10.1177_1759720X211006966 for Serum-derived extracellular vesicles inhibit osteoclastogenesis in active-phase patients with SAPHO syndrome by Yanpan Gao, Yanyu Chen, Lun Wang, Chen Li and Wei Ge in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-xlsx-5-tab-10.1177_1759720X211006966 – Supplemental material for Serum-derived extracellular vesicles inhibit osteoclastogenesis in active-phase patients with SAPHO syndrome
Supplemental material, sj-xlsx-5-tab-10.1177_1759720X211006966 for Serum-derived extracellular vesicles inhibit osteoclastogenesis in active-phase patients with SAPHO syndrome by Yanpan Gao, Yanyu Chen, Lun Wang, Chen Li and Wei Ge in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Conflict of interest statement
The authors declare that there is no conflict of interest.
Ethics approval
The experiments were conducted with full compliance with local, national, ethical, and regulatory principles and local licensing regulations. This study was approved by the Ethics Committee of PUMCH (No. ZS-994).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Yanpan Gao was supported by the National Natural Science Foundation of China (81902258), the Beijing Natural Science Foundation (7192127) and the Non-profit Central Research Institute Fund of Chinese Academy of Medical Sciences (2019-RC-HL-006). Wei Ge was supported by the National Natural Science Foundation of China (81971023). Chen Li was supported by the CAMS Innovation Fund for Medical Sciences (CIFMS) (2017-I2M-3-001). Yanyu Chen was supported by the Graduate innovation fund of Peking Union Medical College (2019-1001-04).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
