Abstract
Osteoarthritis (OA) is an evolving chronic joint disease with a huge global impact. Given the intricate nature of the etiopathogenesis and subsequent high heterogeneity in the clinical course of OA, it is crucial to discriminate between etiopathogenic endotypes and clinical phenotypes, especially in the early stages of the disease. In this sense, we propose that an OA phenotype should be properly assessed with a set of outcome measures including those specifically related to the main underlying pathophysiological mechanisms. Thus, each OA phenotype can be related to different and clinically meaningful outcomes. OA phenotyping would lead to an adequate patient stratification in well-designed clinical trials and the discovery of precise therapeutic approaches. A significant effort will be required in this field in light of inconclusive results of clinical trials of tissue-targeting agents for the treatment of OA.
Keywords
Introduction
Osteoarthritis (OA) is a very common and slowly progressive joint disease, which affects more than 300 million (15%) adults globally, particularly in aging populations. 1 OA represents a leading cause of pain and chronic disability with a profound burden on the quality of life of patients and on public health systems. As aging, obesity and poor lifestyle increase in the world population, the burden of musculoskeletal diseases, particularly OA, rises greatly and could be even bigger than expected. As a matter of fact, musculoskeletal diseases are now the second most common cause of disability-adjusted life years (DALYs), and DALYs for OA increased by 104.9% from 1990 to 2016 worldwide. 2 OA is also associated with increased rates of comorbidities, such as cardiovascular disease and diabetes mellitus, and an increased risk of cardiovascular disease-specific and all-cause mortality.3,4 In addition, the economic burden of OA is shown by high healthcare costs and high indirect costs. 5
Main etiopathogenic mechanisms in OA
Multiple pathophysiological mechanisms interacting in complex ways affect all joint structures, particularly articular cartilage, subchondral bone and synovial membrane at different time points throughout the long OA process. 6
The imbalance between mechanical loading and its absorption by the articular cartilage causes damage in joint tissues. The predominance of catabolic over anabolic events leads to a progressive loss of glycosaminoglycan content and collagen network with a subsequent significant reduction of tensile strength and compliance. Main etiopathogenic processes include the degradation of cartilage extracellular matrix, increased high subchondral bone turnover and synovial inflammation. Overloading may drive these alterations on normal joint tissues, while in other circumstances they can be secondary to the action of average mechanical loading upon joint tissues with an altered structure. These early changes can occur due to genetic alterations, sex hormone deficiency, aging and other major factors such as metabolic imbalance and low-grade chronic systemic inflammation in the biology of join tissues.
Various types of cells present in joint tissues under mechanical stress overexpress receptors of the innate immunity, namely toll-like receptors (TLRs). Tissue damage caused by overloading induce the release inside of the joint of a broad diversity of molecules, that is, hyaluronic acid, fibronectin fragments, small leucine-rich proteoglycans, collagen or cartilage oligomeric protein (COMP), which activate integrin receptors and the innate immune response, mainly the TLR2 and 4 mediated signalling pathways and complement system. 7 On the other side, proinflammatory adipokines such as leptin, adiponectin, resistin and others are the effector molecules of joint damage caused by the interaction between metabolism imbalance and the immune system in OA. 8 Not only does the association between OA and obesity occur by a direct overloading effect, but also by a chronic low-grade inflammation associated with obesity and metabolic syndrome. Furthermore, adipokines by their role in endothelial dysfunction and atherosclerosis may contribute to high mortality rates due to cardiovascular events in OA patients.
Therefore, mechanical stress, low-grade systemic inflammation and metabolic imbalance are main factors which play crucial roles on the onset and progression of OA. More importantly, these factors converge on the same etiopathogenic pathways whereby the chronic activation of innate immunity in chondrocytes, through TLR signalling, results in a robust activation of NF-κB, MAPK and PI3K dependent pathways. These signalling transduction cascades promote the production of pro-inflammatory cytokines, tissue-destructive enzymes and inflammasome components. The excess of glucose and/or lipids, the presence of crystals, as well as increased apoptosis also contribute to joint damage. 9
Establishing OA phenotypes
Since OA is an evolving disease, 6 the intricate nature of etiopathogenic events lead to a high heterogeneity in the clinical course and subsequent great difficulty on the development of an effective treatment for OA. In fact, although diverse candidate molecules for disease modifying OA drugs showed encouraging results in preclinical and early clinical studies, they fell short in achieving structural and clinical efficacy in phase III randomized clinical trials (RCTs). In this complex scenario, it is crucial to consider how to assess the efficacy of these drugs. What clinical symptoms or signs should be tracked to estimate the effects of candidate drugs? For instance, pain characteristics and severity over time can be chosen in knee OA. Notably, pain profile reflects the complexity of OA and its difficult assessment in RCTs. Indeed, the predominant joint tissue affected at a specific time expresses a particular type of pain in the early stages of OA, while in the advanced stages OA evolves towards a more uniform disease with persistent pain, and sometimes pain with neuropathic characteristics. Moreover, this picture becomes more complicated since silent periods have been observed during the OA process, which might explain the high rates of clinical improvement in some RCTs.
Based on very relevant molecular physiopathological mechanisms of the disease, and by differentiating aetiologies from risk factors, our group suggested to classify primary OA several years ago. 10 Indeed, we proposed three OA subsets depending on the main underlying pathophysiological pathways: type I or genetically determined; type II, oestrogen hormone-dependent; and type III age-related. As a result of the interaction between these mechanisms and extra-articular risk factors in joint tissues, we further suggest four interchangeable clinical profiles –biomechanical, inflammatory, metabolic and osteoporotic may occur in OA patients during the early stages of the disease 11 (Figure 1).
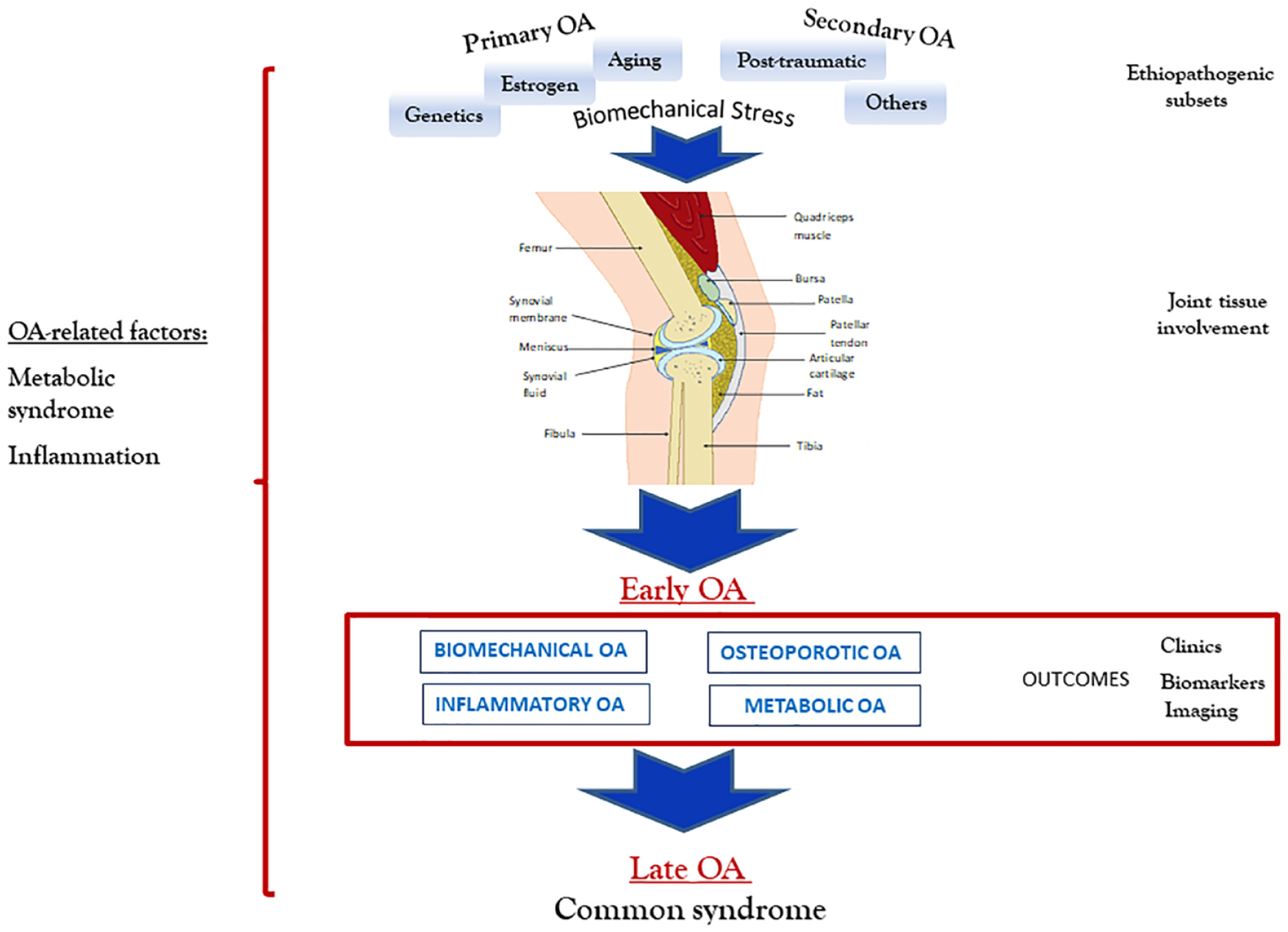
Pathogenic progression of osteoarthritis (OA). Biomechanical stress induced by main biological processes is the basis of primary OA events in different joint tissues, modulated by extra-articular risk factors. Subsequent interchangeable clinical settings may occur along early stages of OA, whereas a common syndrome occurs during late stages of the disease.
A significant effort has been also devoted in phenotyping OA for several other research groups. A total of 79 knee OA phenotypes were reported in the included studies of a recent systematic review. From such a variety of proposed phenotypes, which makes no sense at all, the authors identified six main clinical phenotypes: chronic pain, inflammatory, metabolic syndrome, bone and cartilage metabolism, mechanical overload and minimal joint disease. 12 Their phenotype allocation was shown to be successful for 84% of cases with an overlap of 20%. 13 Another systematic review identified that some characteristics such as pain sensitization, psychological distress, radiographic severity, body mass index (BMI), muscle strength, inflammation and comorbidities are associated with different clinical phenotypes in knee OA patients, whereas sex, obesity, other metabolic abnormalities, the pattern of cartilage damage and inflammation may determine distinct structural knee OA phenotypes. 14
Molecular biomarkers are expected to become useful tools to differentiate between subgroups of patients whose disease is triggered by specific main etiopathogenic mechanisms, proposed as ‘molecular endotypes’, and manifested by their corresponding clinical phenotypes. 15 Metabolomic studies in synovial fluid of human knees identified high inflammation in early and late OA, oxidative stress in late OA, or structural deterioration in early and late OA. 16 Recently, a machine learning approach in a big dataset determined key variables that differentiate progression versus non-progression phenotypes in knee OA patients at 48 months. Baseline variables contributing to progression included bone marrow lesions, osteophytes, medial meniscal extrusion, and urine C-terminal crosslinked telopeptide type II collagen (uCTX-II), whereas the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) pain, lateral meniscal extrusion and serum N-terminal pro-peptide of collagen IIA, were strongly associated with non-progression. 17 Hence, the use of molecular biomarkers together with imaging, pain and function assessments may greatly help to identify distinct OA phenotypes, with the following progress in the design of more effective, stratified and individualized therapeutic strategies in OA.
At this point, it is crucial to distinguish phenotypes of outcomes. An OA phenotype is determined by either a single or combination of disease attributes that characterize a subgroup of patients sharing distinct underlying pathophysiological mechanisms. In turn, the outcome is defined as an event or measure in the study participants that is used to assess the effectiveness and/or safety of an intervention studied in clinical trials. Establishing different phenotypes by using the same outcomes would lead to erroneous interpretation having no practical utility. In this sense, we believe that each OA phenotype should be related to clinically meaningful outcomes, which will be different between OA phenotypes.
Outcomes and endpoints according to knee OA phenotypes
The identification of distinct profiles among OA patients will necessarily involve the use of a core set of outcome measures including clinical variables, biological and imaging markers related to specific pathophysiological mechanisms involved in each clinical phenotype.
Biomechanical OA
The most important biomechanical contributors to the development and the progression of OA are overweight, joint malalignment, loss of meniscal function and ligament injury in the biomechanical phenotype. Thus, surrogates for mechanical stress such as weight and fat-free mass were strongly related to knee OA. 18 Indeed, weight gain led to an increased progression of knee OA structural features, synovitis, patellofemoral bone marrow lesions (BMLs) and cartilage defects in overweight and obese women over 2.5 years. 19 In contrast, more than 5% weight loss was associated with a slower increase in global cartilage T2 and deep layer cartilage T2 scores compared with stable weight after a follow up of 96 months. 20 Furthermore, weight loss was also associated with improvements in the quality (increased proteoglycan content) and quantity (reduced thickness loss) on medial knee articular cartilage over 1 year. 21
Radiographic joint space width (JSW) or narrowing (JSN) is frequently used for OA diagnosis and monitoring OA progression, and is still the only approved end point by regulatory bodies in clinical trials. However, the use of radiographic JSN exhibits some conspicuous limitations. It identifies patients with advanced OA who are more likely to be non-responsive to therapeutic interventions. It does not show correlation with the severity of clinical symptoms, being a poorly responsive endpoint that requires long and large clinical trials (2–4 years duration with about 1000 subjects) to determine therapy efficacy. Thus, radiographic JSN may impede drug development in OA.22,23
On the other hand, only magnetic resonance imaging (MRI) currently provides a good assessment of all joint structures.23,24 MRI has displayed lesions in the tibiofemoral joint of middle aged and elderly people with radiographs not showing any OA feature, regardless of pain. 25 Quantitative measurements of cartilage volume and thickness have been used as outcomes in intervention studies. Indeed, some systematic reviews have demonstrated that MRI biomarkers of OA hold concurrent and predictive validity, with good responsiveness and reliability.26,27 Data from a recent study with over 4 years of follow-up endorsed these previous results. 28 Hence, the OARSI–FDA Working Group considers MRI as a suitable imaging tool to assess cartilage morphology in clinical trials assessing the development and progression of OA. 29 In addition, MRI-assessed structural pathology has been also related with symptomatic knee OA. A 10.7 years follow-up study demonstrated that cartilage defects, BMLs and effusion-synovitis were associated with worse pain trajectories in older populations and knee OA patients. 30 On the other hand, greater baseline pain, and fluctuating and persistent pain at the knee over 1 year predicted an increase of cartilage volume loss, incidence and progression of radiographic knee OA at 4 years follow-up. 31
Furthermore, a detailed analysis with cartilage segmentation in knee plates has reported subregional changes in different cohorts.32–34 Notably, cartilage thickening predominantly at the external subregion of medial femoral condyle was seen in individuals with pre-radiographic and radiographic knee OA (Kellgren/Lawrence stage 2).
35
This finding may be consistent with cartilage swelling/hypertrophy described as a sign of early OA in in vivo study.
36
In turn, compositional MRI detects early biochemical alterations in joint tissues before morphologic changes can be seen in conventional MRI.37,38 As a matter of fact, local cartilage regions of interest had higher T2-values compared with the surrounding cartilage 4 years prior to lesion onset.
39
Thus, MRI has become a valuable imaging modality for a better understanding of the natural history of OA and for the development of new therapies. Moreover, emerging hybrid imaging techniques including positron emission tomography
The assessment of early OA changes in joint structures using dynamic imaging techniques also looks like an attractive approach to examine the outcome of joint loading. In this regard, the degree of ultrasound (US)-estimated medial meniscal subluxation correlated with radiographic medial tibiofemoral JSN, being remarkably higher in weight-bearing than in non-weight-bearing positions.40,41 Likewise, MRI studies showed that medial meniscal subluxation is significantly associated with cartilage volume loss, 42 and both meniscal protrusion and meniscal root tears are very prevalent in patients with accelerated knee OA. 43 In turn, malalignment has been strongly related with knee OA progression at 26- months follow-up. 44 Frontal plane alignment was more strongly associated with tibiofemoral than patellofemoral OA worsening over 7 years. 45 Nevertheless, patella alta correlated with the worsening of MRI-determined patellofemoral OA over 24 months. 46 Malalignment and meniscal damage together may increase the risk of cartilage loss even more, likely due to the high dynamic load on local articular cartilage. 47
Biological markers aiming to detect very early OA changes in joint issues are being intensely studied. Both baseline urinary C-terminal telopeptide of type II collagen (uCTX-II) and urinary alpha isomerized version of collagen type I (uCTXIα) levels significantly predicted pain and structural worsening of knee OA over 48 months. 48 High baseline serum cartilage oligomeric matrix protein (sCOMP) levels were associated with incident radiographic knee OA in two studies, one with an average 6.3 years follow-up 49 and another with 10 years follow-up. 50 In addition, a large scale meta-analysis described that uCTX-II levels are significantly associated with the risk of prevalence of hand, hip and knee OA progression and incidence of knee OA. 51 A systematic review found strong evidence for uCTX-II level as a prognostic marker for knee OA progression and sCOMP level as a prognostic marker for incidence of knee and hip OA. 52 These findings were confirmed by a longitudinal study that showed significant associations between uCTX-II and sCOMP levels and knee or hip OA progression at 5 years follow-up. 53 However, a recent metanalysis only revealed a moderate performance of sCOMP and uCTX-II for diagnosing knee or hip OA, although it concluded that sCOMP may predict OA progression. 54 So far, none of the candidate biomarkers have shown to be sufficiently discriminative for diagnosis or prediction of prognosis in OA and have yet to be fully validated for acting as a surrogate outcome in OA. 55
Muscle strength also plays a relevant role in load distribution across a joint surface. Loss of quadricep strength may reduce its shock-absorbing potential on the knee causing large dynamic loads on articular cartilage, and subsequent progressive cartilage degeneration. 56 Indeed, quadricep weakness increased the risk of worsening lateral patellofemoral cartilage damage at 7 years follow-up in women with or at risk of knee OA. 57 In this regard, muscle torque may be used as a surrogate outcome for muscle capacity on protecting joints. The amount of physical activity, measured by a pedometer, was deleteriously related to knee OA progression at 2–5 years follow up. 58 Likewise, high and very low Physical Activity Scale for the Elderly (PASE) scores were associated with high progression of cartilage T2 measurements in asymptomatic middle-aged individuals over a period of 4 years. 59 Furthermore, increasing levels of physical activity associated with high risk of knee OA have been observed in overweight and obese men after 96 months of follow-up. 60 Long-term pain and function after treatment, known as the carry-over effect of an interventional agent, should be assessed. 61 Remarkably, knee pain when using stairs has been described as the first symptom to appear in early OA, followed by pain when walking and standing. 62 Furthermore, subjects who developed accelerated knee OA over 48 months apprised greater pain when walking and straightening the leg, and had difficulty in lying down compared with those who developed common knee OA. 63 Subjects with the greatest functional impairment – WOMAC physical function scores between 40 and 68 – had an increased risk of undergoing total knee replacement over 30 months in a large cohort study of persons with or at high risk of symptomatic knee OA. 64 In addition to X-rays, MRI, US, and potentially biological markers, these other outcome measurements may be useful options for conducting well-designed clinical trials in patients with biomechanical OA.
Osteoporotic OA
Greater prevalence of OA in women than men and the dramatic rise in OA prevalence among postmenopausal women,65–69 which are associated with the presence of oestrogen receptors in joint tissues,70–74 hint that OA is significantly related to sexual hormone status, particularly to oestrogen levels. 75 Generalized involvement of joints with predominant node formation and signs of inflammation in interphalangeal joints of the hands have been described in postmenopausal women since the earliest studies of OA.76,77 A subset of middle-aged women who develop erosions associated with transient inflammation in the interphalangeal joints are characteristic of erosive OA. 78 Lower levels of serum E2 were reported in postmenopausal women who developed radiographically defined knee OA. 79 However, oestrogen replacement therapy has shown mixed results in OA, probably because of methodological flaws in the performed studies. Hence, oestrogen deficiency plays a conspicuous role in a distinctive OA, which develops in women during the early years following menopause.
In patients with this phenotype, increased remodelling and impaired structure of subchondral bone may play key roles in the development and progression of OA, responding to bone active drugs. 80 These events can be assessed by imaging techniques such as digital X-ray, computerized tomography and MRI used in research studies, but technically difficult to handle at daily clinical practice. MRI studies have identified BMLs that are imaging features of OA with a characteristic signal pattern in subchondral bone. BMLs associated with OA correspond to fibrosis, necrosis, oedema and bleeding of fatty marrow as well as trabecular alterations on histopathology. 81 BMLs, as well as cartilage defects and effusion-synovitis, were associated with worse pain trajectories over 10.7 years in older populations, particularly in those with radiographic knee OA. 30 Notably, the size of BMLs significantly correlates with the progression of articular cartilage loss, the incidence of knee arthroplasty and pain fluctuations in patients with knee OA.82–84 Likewise, osteophytes were independently associated with knee OA structural progression and the incidence of total knee replacement. 82 Furthermore, in accord with the presence of osteophytes in MRI, OA can be classified in either hypertrophic or atrophic (osteoporotic) OA phenotypes. 23 Therefore, BML volume and osteophytes arise as attractive potential outcomes for studying the development and progression of subchondral bone damage in OA and its response to therapeutic interventions.
In addition, subchondral trabecular bone texture has emerged as a promising imaging biomarker for knee OA. Changes at 12–18 months of MRI-assessed subchondral bone texture predicted radiographic knee OA progression at 36 months. 85 Even radiographic subchondral trabecular bone texture predicted risk of radiographic and/or pain progression in individuals with knee OA at baseline and over 12 and 24 months. 86 In contrast, the use of bone formation and resorption markers to assess subchondral bone turnover in OA has not been well established. So far, biomarkers such as type I collagen-degradation epitopes and osteocalcin have shown limited diagnostic potential.48,87
The simultaneous use of dual X-ray absorptiometry (DXA), to assess subchondral bone mineral density (BMD) and bone scintigraphy to detect patients on high subchondral bone remodelling, arises as a reasonable approach in daily practice. 88 In fact, a recent study showed that periarticular bone assessments including baseline and most rates of change in medial:lateral BMD and MRI-assessed trabecular morphometry in proximal tibia is associated with radiographic knee OA structural progression over 12–18 months. 89 Likewise, baseline subchondral BMD positively predicted cartilage defect development at the medial tibial site. 90 In turn, late-phase bone scintigraphy demonstrated that agent retention in the tibiofemoral compartment is associated with severity of knee symptoms. 91 Furthermore, a (99m) Tc-DPD-SPECT/CT showed that elevated subchondral uptake is directly associated with the grade of cartilage lesions and with the Whole-Organ Magnetic Resonance Imaging Score (WORMS) sum in patients with knee OA. 92 Scintigraphy would allow to assess the de novo rate of subchondral bone remodelling, thus acting as an early surrogate marker, whereas DXA would provide an index of accumulative effect therefore acting in some extent as a final outcome for subchondral bone status. Moreover, a study using a radioisotope with PET showed an increase of bone metabolism in the proximal femur of patients with symptomatic hip OA, suggesting that this method may detect early OA changes. 93 However, several methodological issues in the study of OA subchondral bone should be addressed before these assessments may become suitable outcome measures for clinical trials in osteoporotic OA.
Metabolic OA
Metabolic syndrome components, high levels of proinflammatory adipokines, diabetes mellitus and cardiovascular events may notably contribute to the development and progression of joint damage in patients with metabolic OA. In this regard, metabolic syndrome was associated with hand OA, after adjustment for weight, whereas high abdominal circumference, hypertension, high fat consumption and self-reported diabetes mellitus were associated with early cartilage degradation measured with T2 relaxation times in knees of middle-aged subjects. 94 Likewise, a recent study demonstrated that metabolic syndrome and low high-density lipoprotein (HDL) is related with medial tibial cartilage volume loss and increase of BML size during a mean follow-up period of 10.7 years. 95 Both baseline leptin levels and change in leptin levels correlated with longitudinal cartilage thinning. 96 Furthermore, baseline leptin levels were associated with the presence of osteophytes, synovitis and effusion, cartilage defects, BMLs and meniscal tears assessed by MRI 10 years later in middle-aged women. 97 These findings suggest that leptin may play a relevant role in the maintenance of cartilage homeostasis, and thus hold great potential to become a valuable outcome measurement in metabolic OA. Furthermore, recent studies describe that serum leptin levels partially mediate the associations between osteoarthritis and adiposity, as assessed with BMI and percentage total body fat, 98 as well as between knee OA and elevated BMI alone. 99 Indeed, BMI has been shown to exert a major causal effect on the risk of OA, particularly at weight-bearing joints.100,101 Fatty acid levels have also been related to cartilage loss and synovitis. Indeed, a negative association was found between total omega-3 fatty acids or docosahexaenoic acid-specific omega-3 and patellofemoral cartilage loss. In contrast, omega-6 fatty acids and arachidonic acid were positively associated with synovitis. 102
Diabetes mellitus has been described as a risk factor for OA progression and poor arthroplasty outcomes. 103 Meta-analyses confirmed an association between diabetes mellitus and the development or presence of radiographic and symptomatic OA,104,105 although a recent systematic review concluded that little evidence supports this relationship. 106 A longitudinal population-based study showed that diabetes mellitus predicts the development of severe OA undergoing hip or knee arthroplasty, and moreover, the probability of arthroplasty increased with the duration of diabetes mellitus. 107 Accordingly, high haemoglobin A1c levels increased linearly with the risk of complications in total joint arthroplasty. 108 Interestingly, medication-treated diabetes was not associated with the incidence of knee OA but independently reduced knee OA progression. 109 High systolic blood pressure and pulse pressure were associated with increased incidence of radiographic knee OA, while treatment with ⩾3 antihypertensive medications reduced it. 110 Moreover, the presence of hypertension and diabetes mellitus was associated with bone loss at subchondral plate in knee OA. 111
Cardiovascular disease, particularly angina and congestive heart failure, was initially associated with prevalent OA in a population-based cross-sectional study. 112 In a subsequent longitudinal analysis, OA was an independent predictor of cardiovascular disease, and, furthermore, the risk of cardiovascular disease was even higher among individuals with OA who underwent total joint replacements. 113 On the other side, individuals with high cardiovascular risk were more likely to have OA-related arthroplasty up to a 30-year follow-up. 114 In turn, the use of statins has been associated with a lower prevalence of generalized OA and with a more than 50% reduction in knee OA progression.115,116 Statin use was also associated with reduced risk of radiographic knee JSN progression in patients with nodal OA followed up annually over 8 years. 117 The validation of these factors as outcomes would dramatically improve the detection of early stages of the disease, and thus modify its progression through appropriate therapeutic interventions in patients with metabolic OA.
Inflammatory OA
Severe local synovitis is developed by a subgroup of patients, in contrast of the chronic systemic low-grade inflammation seen in all subtypes of OA. Evaluation of synovial inflammation can be properly carried out by MRI and US. In fact, contrast-enhanced MRI offers a good assessment of the extent and degree of synovitis, whereas the presence of signal changes in Hoffa fat pad or joint effusion assessed on non-contrast-enhanced MRI can be considered as indirect markers of synovitis. MRI-detected synovial inflammation positively correlates with pain and radiographic progression in knee OA.30,118–121 Moreover, synovial tissue volume lessens following steroid therapy and rebounds in those whose pain relapses in knee OA. 122 Histologic analysis of synovial biopsy specimens, as well as both contrast and non-contrast MRI studies of knee joints, have shown that mild-to-moderate synovial inflammation is associated with severe radiographic OA. 123 Likewise, a further study demonstrated that contrast enhanced MRI-detected synovitis is strongly associated with radiographic severity and MRI-assessed widespread cartilage damage in knee OA. 124
Synovitis has been also associated with incident radiographic knee OA, specifically with a total synovitis score of 3 or higher on a 0–9 scale. 125 Furthermore, effusion-synovitis and Hoffa-synovitis have been observed to strongly predict by 1–2 years the development of incident radiographic knee OA.126,127 In this sense, a very recent study reported that effusion/synovitis preceded the development of accelerated knee OA by 2 years, boosting the chance for synovitis to become a prognostic outcome. 128
Synovial hypertrophy, increased vascularity and synovial fluid can be also commonly detected with US in joints affected by OA. 129 Signs of inflammation determined by US are more frequently seen in erosive OA hands than in nonerosive OA hands. 130 Both US-assessed suprapatellar effusion and medial compartment synovitis were directly related with knee pain in motion, sitting and rest in patients with radiographic knee OA.131,132 Notably, US-detected effusion, as well as, severity of both radiographic damage and pain, were predictors of subsequent joint replacement over a 3-year follow-up. 133 Thus, synovitis detected by US as an outcome measure also seems attractive for multicentre trials assessing OA in patients with inflammatory phenotypes. In turn, superolateral Hoffa’s fat pad (SHFP) oedema was associated with simultaneous cartilage damage, BMLs and osteophytes in lateral patella, and importantly predicted longitudinal patellar cartilage loss over 24 months. 134 Hence, MRI-examined SHFP oedema may become an outcome measure for patellar OA.
Biomarkers that reflect systemic and synovial inflammation are being studied in OA. A meta-analysis found a significant association between serum levels of high sensitivity C-reactive protein (hs-CRP) and pain or discapacity in OA patients. 135 Moreover, increased serum hs-CRP and IL-6 were associated with an increase in BML scores in patients with knee OA over 2 years.136,137 High plasma PGE2 and 15-HETE levels identified patients with symptomatic knee OA, and elevated levels of peripheral blood leukocyte IL-1β, TNFα and COX-2 gene expression determined a high risk of radiographic progression over 24 months in these patients. 138 Similarly, plasma lipopolysaccharide binding protein and serum TLR4 were also associated with knee OA progression over 16–18 months. 139 Synovial fluid (SF) biomarkers drive slightly higher expectations to reflect the local underlying molecular changes in knee OA. Indeed, SF IL-1β and IL-18 levels have been associated with OA severity and progression. 140 In the same way, high levels of inflammatory macrophages, CD163 and CD14, neutrophils, elastase and related markers (VEGF, MMP-3, TIMP-1, sICAM-1, sVCAM-1, and MCP-1) in SF and plasma were associated with an inflammatory subset of patients with knee OA that showed structural progression and severe pain.141,142 Nevertheless, as for other potential biomarkers, the role of inflammatory biomarkers for predicting structural damage in OA so far remains unclear, being necessary for more large scale, high-quality and homogenous studies. Hence, an appropriate assessment of synovitis features by MRI or US, particularly contrast-enhanced MRI, holds the potential for the development of useful outcomes and could be used in clinical trials of new drugs that target synovitis in OA patients with inflammatory phenotype. Finally, clinical trials in patients with inflammatory OA should also assess long-term pain and function after treatment (carry-over effect) of an interventional agent. 61
Composite indexes
Although brief unidimensional measures like the visual analogue scale or the Likert scale are widely used for established OA, more robust multidimensional outcome measures may be needed in early OA to address the diverse clinical manifestations originate from the evolving OA process. 143 Therefore, simplified composite indexes that include information of the changes in various domains seems suitable to evaluate the response to therapeutic agents in clinical trials, as proposed in the OMERACT-OARSI set of responder criteria. Pain, physical function, quality of life, patient’s global assessment of the target joint and adverse effects, including death, have been established as mandatory core domains to be assessed in all hip and/or knee OA clinical trials. 144 Composite indexes should have great validity, simple and consistent medical language, and a statistical analysis without multiple tests. However, the format of such indexes entails a high level of symptoms at entry for the patient to fulfil the set of criteria. The inclusion of appropriate risk factors or imaging features as valid outcomes into composite indexes may help to identify specific subsets of individuals, such as those who develop certain OA phenotypes or accelerated knee OA.145,146
Conclusion
OA is an evolving chronic joint disease with a great global impact. The intricate nature of etiopathogenic events affecting all joint tissues lead to a high heterogeneity of the clinical course of OA. In this complex scenario, it becomes crucial to discriminate distinct etiopathogenic endotypes and clinical phenotypes, especially in the early stages of the disease. We propose establishing an OA phenotype that should comprise the usage of a set of distinctive outcome measures including those related to main pathophysiological mechanisms (Table 1). However, setting up OA phenotypes is highly challenging precisely because the complex etiopathogenesis and varied clinical manifestations throughout an evolving process can lead to the identification of multiple subsets of patients with no specific outcome measures and lacking clinical meaning. In this regard, the current proposal may contribute to better phenotyping of OA patients; therefore, it would lead to well-designed clinical trials and the discovery of precise therapeutic approaches for each OA phenotype. Hence, a significant effort will be required in this field, given the discordant results of clinical trials using tissue-targeting agents for the treatment of OA.

ADA-criteria DM, Type 2 diabetes diagnostic criteria by the American Diabetes Association; BMD, bone mineral density; BMI, bone mass index; CT, computed tomography; CV, cardiovascular disease; DHA, docosahexaenoic acid; DXA, dual X-ray absorptiometry; HbA1c, hemoglobin A1c; HFP, Hoffa’s fat pad; hs-CRP, high sensitivity C-reactive protein; ICOAP, Intermittent and Constant Osteoarthritis Pain Questionnaire; JSN, joint space narrowing; JSW, joint space width; KOOS, Knee Injury and Osteoarthritis Outcome Score; KOOS-p, pain subscale of KOOS; KOOS-f, function in daily living and function in sport and recreation subscales of KOOS; MRI, magnetic resonance imaging; OA, osteoarthritis; p,sf CD163, CD164, plasma and synovial fluid CD163 and CD164 levels; PET, positron emission tomography; PUFA, Polyunsaturated fatty acids; s,u CTXI, serum and urinary C-terminal telopeptide of type I collagen; SB, subchondral bone; sCOMP, serum cartilage oligomeric matrix protein; shsCRP, serum high-sensitivity C-reactive protein; sOsteocalcin, serum osteocalcin; SPECT, single-photon emission computed tomography; sTLR4, serum toll-like receptor 4; uCTXII, urinary C-terminal telopeptide of type II collagen; US, ultrasound; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index; WOMAC-f, function subscale of WOMAC; WOMAC-p, pain subscale of WOMAC.
Footnotes
Conflict of interest statement
GH-B received grants from Novartis, Sandoz, Pfizer, Amgen, Mylan and Servier. GH-B and RL have a national patent on use of 6-shogaol for the treatment of osteoporosis and a national patent on use of osteostatin for the treatment of osteoarthritis. JAR-B and LAM-T report no conflict.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Instituto de Salud Carlos III, grants PI16/00065, PI16/00991, PI18/00261. This work is co-funded by Fondo Europeo de Desarrollo Regional (FEDER).
