Abstract
Chronic pain conditions affect at least 116 million US adults and more than one-third of adults worldwide. Nonsteroidal anti-inflammatory drugs (NSAIDs) are used extensively for the treatment of chronic pain due to their efficacy as anti-inflammatory and analgesic agents. Gastrointestinal toxicity is the most well known adverse effect of NSAID therapy and it may manifest as dyspepsia, ulcers, or bleeding. Current guidelines for the management of patients who require NSAIDs for chronic pain and inflammation recognize the potential toxicity associated with these drugs and the need for gastroprotection. DUEXIS® (ibuprofen 800 mg, famotidine 26.6 mg) is a proprietary combination, immediate release tablet containing 800 mg of ibuprofen and 26.6 mg of famotidine. The efficacy of DUEXIS® taken three times daily has been demonstrated in two large-scale controlled clinical trials (Registration Endoscopic Studies to Determine Ulcer Formation of HZT-501 Compared with Ibuprofen: Efficacy and Safety Studies (REDUCE) and REDUCE-2) which showed that this new formulation significantly reduced the risk of endoscopic upper gastrointestinal ulcers compared with ibuprofen alone (REDUCE-1, p < 0.0001, REDUCE-2, p <0.05). DUEXIS® was also superior to ibuprofen in decreasing the risk for gastric ulcers (REDUCE-1, p < 0.001, REDUCE-2, p < 0.05) as well as duodenal ulcers (REDUCE-1, p < 0.05, REDUCE-2, p < 0.05). Safety results from these two studies indicated that treatment-emergent adverse events occurred in 55% of patients treated with DUEXIS® versus 58.7% for ibuprofen, and serious adverse events were recorded for 3.2% of patients treated with DUEXIS® versus 3.3% of those on ibuprofen. Adverse events leading to discontinuation occurred in 6.7% of patients treated with DUEXIS® and 7.6% for ibuprofen. The combination of ibuprofen and famotidine in a single tablet has the potential to improve adherence to gastroprotective therapy in patients who require NSAID treatment and the use of a histamine type 2 receptor antagonist rather than a proton-pump inhibitor may decrease the risk for clinically significant drug interactions and adverse events (e.g. interaction with clopidogrel, fracture, pneumonia, Clostridium difficile infection).
Introduction
Chronic pain conditions affect at least 116 million US adults and more than one-third of adults worldwide [IOM, 2011; Tsang et al. 2008]. Nonsteroidal anti-inflammatory drugs (NSAIDs) are a mainstay of therapy for many of these individuals [Herndon et al. 2008]. Worldwide, over 73,000,000 prescriptions for NSAIDS are written yearly [Biederman, 2005]. Results compiled by the US Department of Health and Human Services indicate that NSAIDs were prescribed in 29% of all physician office and hospital outpatient visits in which drugs were prescribed in 2004–2005 [US DHHS, 2008].
While these drugs are effective, their use is associated with significant gastrointestinal (GI) toxicity in many patients, which may manifest as dyspepsia, ulcers, or bleeding. It has been estimated that endoscopically demonstrable ulcers occur in 15–30% of regular NSAID users and that the annual rate of upper GI (UGI) clinical events (complicated plus symptomatic uncomplicated ulcers) is approximately 2.5–4.5% [Laine, 2006]. Mortality and morbidity associated with NSAID GI toxicity is also substantial. It has been reported that 7000–10,000 NSAID users in the USA die each year as a result of ulcer perforations and bleeding [Lanza et al. 2009]. In addition, there are approximately 100,000 hospitalizations each year in the USA for NSAID-associated ulcer perforations or bleeding [Lanza et al. 2009].
Patient- and treatment-related risk factors for NSAID-associated GI adverse events (AEs) are well understood (Table 1) and guidelines for the prevention of NSAID-related ulcer complications have been published [Lanza et al. 2009]. However, despite these guidelines, which recommend gastroprotective therapy for at-risk patients taking NSAIDs, cotherapy is prescribed less than 50% of the time [Laine et al. 2009a].
Risk stratification for gastrointestinal toxicity in patients receiving nonsteroidal anti-inflammatory drugs (NSAIDs) (adapted from Lanza et al. [2009]).

The cost of managing serious AEs associated with NSAID gastrotoxicity is high, with estimated costs in the USA exceeding US$2 billion per year [Abdrabbo et al. 2004]. All of these findings support the view that there is a significant unmet need for an adjunctive therapy aimed at decreasing the GI toxicity of NSAIDs in patients who require these drugs for management of chronic pain. At present, there are four combination products aimed at decreasing the risk for NSAID-associated GI toxicity approved for use in the USA. These are the combinations of diclofenac and misoprostol [Bocanegra et al. 1998], naproxen and lansoprazole [Lai et al. 2005], naproxen and esomeprazole [Goldstein et al. 2010], and ibuprofen and famotidine [Laine et al. 2012]. Each of these combinations has been shown to have lower GI toxicity than the component NSAID alone. The combination of misoprostol with diclofenac is limited by high rates of abdominal pain, diarrhea, dyspepsia, nausea, and flatulence [Arthrotec prescribing information, 2010; Hawkey et al. 1998; Rostom et al. 2002] and concerns associated with combination treatments including a proton-pump inhibitor (PPI) are considered in detail below.
This paper describes the clinical efficacy and safety results obtained to date for DUEXIS® (ibuprofen 800 mg, famotidine 26.6 mg), a single tablet that contains the NSAID ibuprofen (800 mg) and the histamine type-2 receptor antagonist (H2RA) famotidine (26.6 mg).
Rationale for the development of DUEXIS®
DUEXIS® is a proprietary, single tablet formulation indicated for the relief of signs and symptoms of rheumatoid arthritis (RA) and osteoarthritis (OA) and to decrease the risk of developing UGI ulcers, which in the clinical trials was defined as a gastric or duodenal ulcer in patients who were taking ibuprofen for those indications. DUEXIS® is administered orally three times a day.
Ibuprofen is one of the most commonly prescribed NSAIDs in the USA [IMS Institute for Healthcare Informatics, 2011]. It has intermediate potency for the management of mild to moderate pain and inflammation; a relatively high safety margin compared with other NSAIDs; and is indicated for a wide range of chronic arthritic and nonarthritic conditions [Lee et al. 1976]. Relief of pain and inflammation with ibuprofen is known to be dose related [Ong et al. 2007; Schou et al. 1998] with an 800 mg three times a day regimen shown to be effective for relieving pain and inflammation in patients with OA [Day et al. 2000; Puopolo et al. 2007; Wiesenhutter et al. 2005; Moore et al. 2010] and RA [Neustadt, 1997; McLauglin, 1985]. In a 4-week, double-blind study in patients with RA, ibuprofen decreased the swollen joint count significantly at a dosage level of 2400 mg/day, but not at 1200 mg/day. The lower dosage of ibuprofen showed primarily analgesia effects [Godfrey and de la Cruz, 1975]. A large study with a total daily dose of 2400 mg of ibuprofen for acute pain from dental surgery indicated that onset of pain relief occurred within 30 min and lasted at least 6 h, which was superior to placebo and acetaminophen/codeine [Daniels et al. 2011].
Famotidine is known to prevent NSAID-induced UGI ulceration by reducing gastric acid secretion [Taha et al. 1996; Smith et al. 1985; FDA, 1986a, 1986b]. It has a longer duration of action than other H2RAs and when administered three times a day is able to maintain a more consistent gastric pH level [Tidmarsh and Rodriguez, 2009] making it an appropriate addition to a three times a day regimen of a NSAID to bring about optimal gastroprotection. Selection of the 80 mg total daily dose of famotidine used in DUEXIS® is based on results demonstrating superiority of this dose over 40 mg/day for ulcer prevention. Taha and colleagues assessed the efficacy of two doses of famotidine (20 mg and 40 mg, each given orally twice daily) versus placebo in preventing peptic ulcers in 285 patients without peptic ulcers who were receiving long-term NSAID therapy for RA (82%) or OA (18%). At the end of 24 weeks, the cumulative incidence of gastric ulcers was 20% in the placebo group, 13% in the patients receiving 20 mg of famotidine twice daily (p = 0.24 versus placebo), and 8% in the group receiving 40 mg of famotidine twice daily (p = 0.03 versus placebo). The proportion of patients in whom duodenal ulcers developed was significantly lower with both doses of famotidine than with placebo (13% in the placebo group, 4% in the low-dose famotidine group, p = 0.04, and 2% in the high-dose famotidine group, p = 0.01). There were no significant differences between the safety and tolerability of 40 and 80 mg/day famotidine in this study [Taha et al. 1996]. Results from a 48-week study of 40 mg famotidine administered twice daily for 48 weeks for the prevention of recurrence of aspirin-related peptic ulcers or erosions indicated that the only notable adverse event was dyspepsia (12.2% of patients) [Ng et al. 2010].
Pharmacology
Ibuprofen
NSAIDs are believed to exert their anti- inflammatory and analgesic effects by inhibiting the enzyme cyclooxygenase (COX), and thus inhibiting prostaglandin synthesis. There are at least two variants of cyclooxygenase (COX-1 and COX-2). Ibuprofen inhibits both COX-1 and COX- 2 [Vane, 1996; Steinmeyer, 2000]. Multiple studies have demonstrated the comparable therapeutic benefits of ibuprofen with selective COX-2 inhibitors (coxibs) and other NSAIDs in controlling inflammation and pain [Rainsford, 2009].
Famotidine
Famotidine competitively inhibits histamine H2 receptors, thus reducing basal, nocturnal, and stimulated gastric acid secretion. Pepsin secretion is reduced resulting in decreased peptic activity [Langtry et al. 1989]. Famotidine, through inhibition of H2 receptors present on parietal cells in the stomach, prevents NSAID-induced UGI ulceration by reducing gastric acid secretion [Smith et al. 1985; Takeda et al. 1992]. Clinical trials have demonstrated that famotidine is effective for the prevention of NSAID-associated ulcers [Taha et al. 1996]. Famotidine has a longer duration of action than other H2RAs [Schunack, 1989; Smith et al. 1985], and unlike cimetidine and ranitidine, famotidine does not impact the activity of alcohol dehydrogenase, the inhibition of which can result in increased serum alcohol concentrations when alcohol is ingested with these compounds [Howden and Tytgat, 1996]. Studies with PPIs have also shown no impact on the activity of alcohol dehydrogenase [Blume et al. 2006].
In addition, studies with famotidine have shown no significant interference with the disposition of compounds metabolized by the hepatic microsomal enzymes (e.g. cytochrome P450 system); therefore, no drug interactions of clinical importance with famotidine have been identified [Pepcid SmPC, MSD, 2011].
It is important to note that the European League Against Rheumatism (EULAR) recommendations for the management of early arthritis have indicated that there is category I-a evidence (i.e. evidence from meta-analyses of randomized, controlled trials) that H2RAs can significantly reduce NSAID-associated GI complications, such as the incidence of GI bleeding [Combe et al. 2007].
Clinical trial results have repeatedly demonstrated the efficacy of famotidine for the prevention of endoscopic ulcers in patients receiving NSAIDs. An early small-scale study evaluated the mucosal protection provided by famotidine in healthy volunteers receiving naproxen 500 mg twice daily with either famotidine 20 mg twice daily or placebo. The results showed a statistically significant reduction in naproxen-induced mucosal damage with famotidine 20 mg twice daily [Aabakken et al. 1990]. A subsequent study confirmed the efficacy and safety of famotidine in patients with arthritis receiving long-term NSAID therapy [Taha et al. 1996]. Results from this 24-week double-blind, parallel-group, randomized comparison of placebo with low-dose famotidine (20 mg twice daily) or high-dose famotidine (40 mg twice daily) as prophylaxis against endoscopically detected gastric or duodenal ulceration showed that the 24-week cumulative incidence of gastric ulcers was 20% in the placebo group, 13% in the low-dose famotidine group, and 8% in the high-dose famotidine group. Additionally, the proportion of patients who developed duodenal ulcers was significantly lower with both doses of famotidine versus placebo (13% placebo, 4% low-dose famotidine, 2% high-dose famotidine) [Taha et al. 1996].
Pharmacokinetics of DUEXIS®
Ibuprofen and famotidine are rapidly absorbed after administration of a single dose of DUEXIS®. Maximum plasma concentration (Cmax) values for ibuprofen are 67 ± 18 ng/ml and are reached 1.5 ± 0.6 h after oral administration of DUEXIS®. Cmax values for famotidine are 163 ± 53 ng/ml and are reached 2.1 ± 0.8 h after oral administration of DUEXIS®. Both ibuprofen and famotidine are rapidly eliminated. Ibuprofen is eliminated from the systemic circulation with elimination half-life (t1/2) values of 2.3 ± 0.4 h following administration of a single dose of DUEXIS®. Famotidine is eliminated from the systemic circulation with t1/2 values of 3.2 ± 0.8 h following administration of a single dose of DUEXIS® [Tidmarsh and Rodriguez, 2009]. Oral administration of DUEXIS® to healthy subjects is bioequivalent to concurrent oral administration of equivalent doses of commercially available ibuprofen and famotidine. The pharmacokinetics of famotidine is altered in subjects with renal impairment and renal impairment slightly alters the pharmacokinetics of ibuprofen [Tidmarsh and Rodriguez, 2009].
Clinical efficacy of DUEXIS®
Data characterizing the efficacy of DUEXIS® were generated from two pivotal phase III, multicenter, randomized, double-blind, parallel group studies (REDUCE-1 and REDUCE-2) for the reduction of the risk of development of ibuprofen-associated, UGI ulcers in patients who require use of ibuprofen for the treatment of chronic pain [Weinblatt et al. 2009; Laine et al. 2012]. Patients enrolled in REDUCE-1 and REDUCE-2 were expected to need daily administration of an NSAID for at least 6 months for conditions such as OA, RA, chronic low back pain, chronic regional pain syndrome, and chronic soft tissue pain. The patients enrolled in the two studies had the primary risk factor of taking a high-dose NSAID increasing their UGI risk 2.5 fold versus a low-dose NSAID [Garcia-Rodriguez and Tolosa, 2007; Laine et al. 2010]. Patients were randomly assigned in a 2:1 ratio to DUEXIS® or matched comparator, ibuprofen 800 mg tablets three times a day. Concomitant low-dose aspirin, up to 325 mg daily and anticoagulant therapies were permitted. Protocol required endoscopy to be performed at baseline, 8, 16 and 24 weeks.
REDUCE-1 and REDUCE-2 included 812 and 570 patients, respectively, in their primary analysis populations. The majority of patients were women (68.2% of patients in REDUCE-1 and 67.7% of the patients in REDUCE-2). The mean age of patients in REDUCE-1 and REDUCE-2 were 55.7 and 55.4 years, respectively; 82% of patients in both trials were under 65 years of age. Overall, the primary populations in REDUCE-1 and REDUCE-2 were 77.2% and 82.6% white; and 19.7% and 13.5% black or African American.
Efficacy results from both studies showed that DUEXIS® was significantly superior to ibuprofen alone in decreasing the risk for UGI ulcers (REDUCE-1, p < 0.0001; REDUCE-2, p < 0.002). DUEXIS® was significantly superior to ibuprofen in decreasing the risk for gastric ulcers (REDUCE-1, p < 0.001; REDUCE-2, p < 0.05) (Figure 1) (DUEXIS® prescribing information, 2011). Pooled analysis of the trials showed a significant decrease in UGI ulcers with a number needed to treat (NNT) of 11, as well as significant decreases in both ulcer components: gastric ulcers and duodenal ulcers [Laine et al. 2012]. Additional crude rate sensitivity analysis of the results from REDUCE-1 and REDUCE-2 are reported in the DUEXIS® prescribing information, which mirror those reported by Laine and colleagues (Tables 2 and 3). Factors associated with significantly increased risk for the occurrence of UGI ulcers included taking ibuprofen without famotidine, history of a prior ulcer, and age at least 65 years (all p < 0.05) [Laine et al. 2009b]. The relative risks and their 95% confidence intervals for UGI ulcers of the treatment effect of DUEXIS® versus ibuprofen for each subgroup population were derived with only the patients in that subgroup population included in the proportional hazards regression model. Treatment was the only factor included in the model. The forest plot of these overall results as well as those for each subgroup is shown in Figure 2 [Weinblatt et al. 2009; Laine et al. 2012].

Overall incidence rate of patients who developed at least one gastric or upper gastrointestinal ulcer in REDUCE-1 and REDUCE-2 (adapted from DUEXIS® Prescribing Information [2011]). DUEXIS®, 800 mg of ibuprofen, 26.6 mg of famotidine.
Overall incidence rate of patients who developed at least one gastric or upper gastrointestinal (UGI) ulcer – REDUCE-1 [DUEXIS® prescribing information, 2011].

Cochran-Mantel-Haenszel test.
Classifying patients who terminated early as not having an ulcer.
Classifying patients who terminated early due to an adverse event, were lost to follow up, discontinued due to the discretion of the sponsor or the investigator, or did not have an endoscopy performed within 14 days of their last dose of study drug as having an ulcer.
DUEXIS®, 800 mg of ibuprofen, 26.6 mg of famotidine.
Overall incidence rates of patients who developed at least one upper gastrointestinal (UGI) or gastric ulcer – REDUCE 2 [DUEXIS® prescribing information, 2011].

Cochran-Mantel-Haenszel test.
Classifying patients who terminated early as not having an ulcer.
Classifying patients who terminated early due to an adverse event, were lost to follow up, discontinued due to the discretion of the sponsor or the investigator, or did not have an endoscopy performed within 14 days of their last dose of study drug as having an ulcer.
DUEXIS®, 800 mg of ibuprofen, 26.6 mg of famotidine.

Forest plots of the relative risk (95% confidence interval) of upper gastrointestinal ulcers for HZT-501 versus ibuprofen in subgroup analyses [Laine et al. 2012]. HZT-501, DUEXIS® (ibuprofen 800 mg, famotidine 26.6 mg); IBU, ibuprofen.
Safety of DUEXIS®
The safety profile for DUEXIS® is based on results from the two previously described phase III trials that included a total of 1533 patients. Treatment emergent adverse events (TEAEs) were noted for 55% of patients treated with DUEXIS® versus 58.7% for ibuprofen; and serious AEs were recorded for 3.2% of patients treated with DUEXIS® and 3.3% of those on ibuprofen.
AEs leading to discontinuation occurred in 6.7% of patients treated with DUEXIS® and 7.6% for ibuprofen. Only two AEs were reported in either study at a rate of at least 5%: dyspepsia (4.7% for DUEXIS® versus 8.0% for ibuprofen) and nausea (5.8% versus 4.7%). The proportion of patients with one or more predefined symptoms consistent with dyspepsia was 12.3% for DUEXIS® and 12.3% for ibuprofen. GI complications were reported for three patients given DUEXIS® and none who received ibuprofen. These were GI bleeding episodes in which hemoglobin dropped 1.6, 2.1, and 3.1 g/dl without clinical evidence of overt GI bleeding, transfusions, or hospitalizations [Laine et al. 2012]. A summary of all AEs reported for at least 2% of patients treated with DUEXIS® in REDUCE-1 and REDUCE-2 is provided in Table 4 [DUEXIS® prescribing information, 2011]. There was one death reported in REDUCE-1 in the ibuprofen group and it was attributed to acetaminophen toxicity [Laine et al. 2012]. Additional longer-term safety data for DUEXIS® are available from 179 patients who were entered into a 28-week follow-up study after completion of either REDUCE-1 or REDUCE-2. During this follow-up evaluation, DUEXIS® was administered to 132 patients (52 weeks of treatment was completed by 112) and 47 were treated with ibuprofen (38 completed 52 treatment weeks). The incidence of treatment-related AEs was comparable between the two groups for the 52-week period; and there were no statistically significant differences in discontinuation rates or safety parameters [Goldstein et al. 2011].
Incidence of adverse events reported for at least 2% of patients treated with DUEXIS® or ibuprofen in REDUCE-1 and REDUCE-2 [DUEXIS® prescribing information, 2011].

DUEXIS®, 800 mg of ibuprofen, 26.6 mg of famotidine.
Potential advantages of DUEXIS®
Adherence
While gastroprotection is important for many patients taking NSAIDs for chronic pain, both physician prescribing and patient adherence to prescribed gastroprotective therapy are poor.
Despite guidelines recommending gastroprotective therapy for patients taking NSAIDs [Wilcox et al. 2006], less than 50% of NSAID users are prescribed cotherapy even with educational reminders [Laine et al. 2009a]. When cotherapy is prescribed, it is uncommon for patients to take protective gastric acid suppressive therapy when using NSAIDs due to problems adhering to complicated dosing schedules and multiple medicines within their regimen [Laine et al. 2009a;
Pan et al. 2008; Abraham et al. 2005; Goldstein et al. 2006; Sturkenboom et al. 2003]. It is well documented that most patients who experience serious GI complications from NSAID treatment do not experience antecedent symptoms [Wolfe et al. 1999; Singh et al. 1996], and this may also contribute to poor adherence to gastroprotective therapy. It is important to note that poor adherence to GI-protective therapy in patients taking NSAIDs is associated with increased risk for UGI events. A nested case-control study that analyzed information from 618,684 NSAID users from the General Practice Research Database in the UK and the HealthSearch/CSD Longitudinal Patient Database in Italy indicated that subjects who were less than 80% adherent to gastroprotective therapy had a more than two-fold increased risk for all UGI events and a nearly twofold increased risk for GI bleeding alone compared with patients who were at least 80% adherent to gastroprotective treatment [van Soest et al. 2011].
A single tablet that combines an NSAID with a gastroprotective agent may simplify prescribing and improve physician adherence to treatment guidelines and it may also improve patient adherence to therapy. Previous studies have shown that adherence to prescribed therapy is improved when one pill rather than two is required [Bangalore et al. 2007]. Thus, the availability of a single tablet that decreases the risk for UGI ulcers has the potential to improve adherence versus administration of separate agents for the treatment of the signs and symptoms of OA and RA and improve outcomes for patients with chronic pain.
Combination of a proton-pump inhibitor with a nonsteroidal anti-inflammatory drug
The combination of ibuprofen with famotidine is not the only option available for gastroprotection in patients taking a conventional NSAID. A combination of enteric-coated naproxen and esomeprazole has also been shown to provide significant gastroprotection in patients requiring chronic NSAID treatment [Goldstein et al. 2010]. A comparison of the efficacy and safety results from REDUCE-1 and REDUCE-2 and the two studies that compared enteric-coated naproxen plus esomeprazole with enteric-coated naproxen alone is provided in Table 5. While the results in Table 5 indicate acceptable safety profiles for both medications, there are a number of important safety considerations that differentiate famotidine from PPIs.
Comparison of results for DUEXIS® and esomeprazole plus EC naproxen (VIMOVO) in phase III clinical trials [Laine et al. 2012; Goldstein et al. 2010; DUEXIS® Prescribing Information, 2011; VIMOVO prescribing information, 2012].
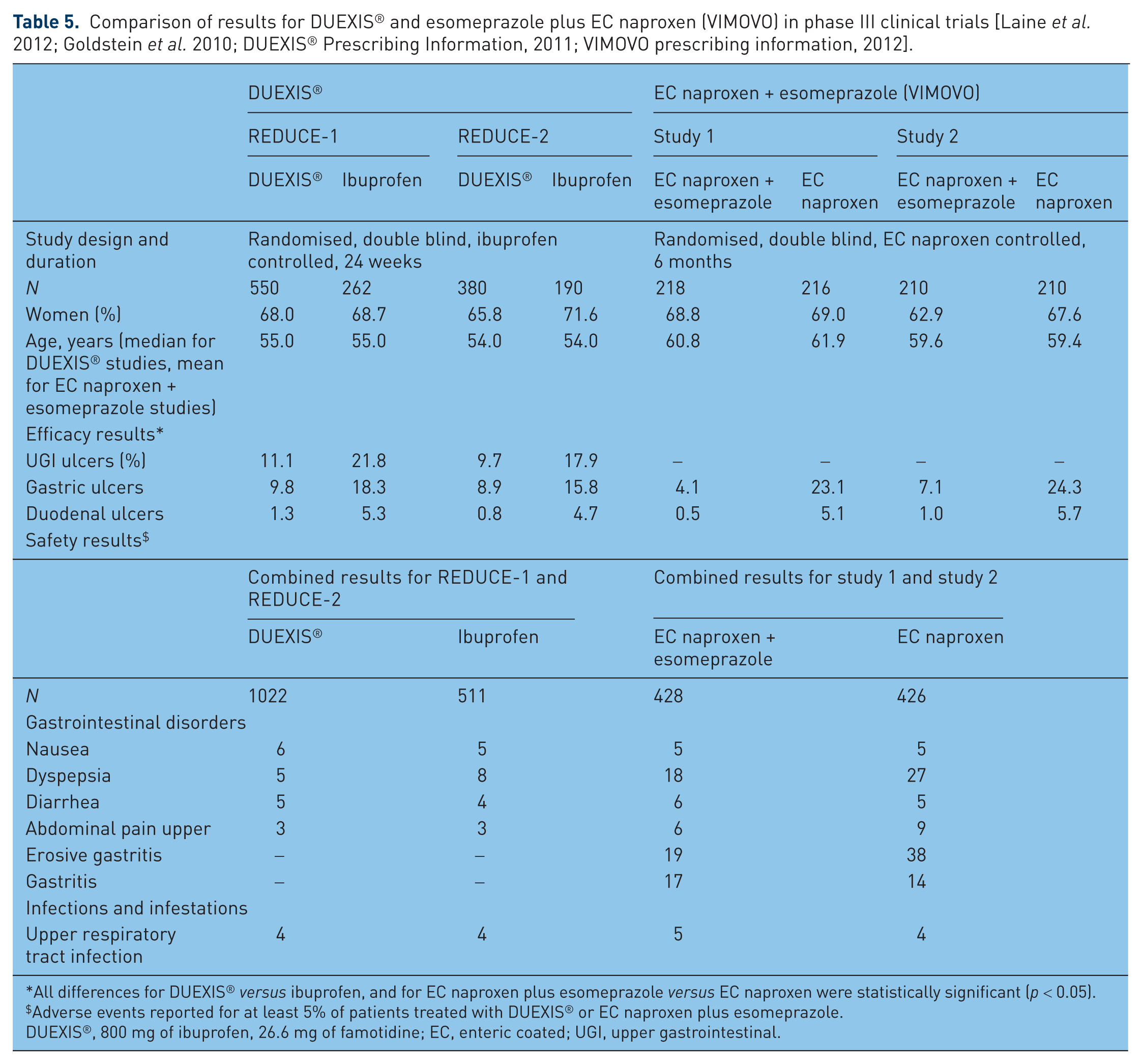
All differences for DUEXIS® versus ibuprofen, and for EC naproxen plus esomeprazole versus EC naproxen were statistically significant (p < 0.05).
Adverse events reported for at least 5% of patients treated with DUEXIS® or EC naproxen plus esomeprazole.
DUEXIS®, 800 mg of ibuprofen, 26.6 mg of famotidine; EC, enteric coated; UGI, upper gastrointestinal.
Limitations associated with use of a proton-pump inhibitor for gastroprotection in patients taking nonsteroidal anti-inflammatory drugs
Clopidogrel interaction
While controversial, there is evidence that coadministration of a PPI interferes with formation of the active metabolite of the commonly used antiplatelet agent clopidogrel, decreasing inhibition of platelet aggregation, and potentially increasing the risk for ischemic events [O'Donoghue et al. 2009; Kwok and Loke, 2010]. While the increased risk with concomitant administration of PPIs with clopidogrel has not reached statistical significance in all studies, the Food and Drug Administration (FDA) issued a public health warning on the possible interaction between clopidogrel and PPIs, predominantly omeprazole, stating ‘the decision to add a PPI to clopidogrel therapy should be weighed against the apparent increased risk of adverse cardiovascular events when the combination is used’ [FDA, 2009]. Available results suggest no significant interaction between famotidine (administered at a total dosage of 40 mg/day) and clopidogrel [Taha et al. 2009]. This is consistent with the lack of effect of famotidine on cytochrome P450 isozymes [Humphries and Merritt, 1999; Humphries, 1987].
Increased risk with proton-pump inhibitors for fracture
Treatment with a PPI has consistently been associated with an increased risk for fractures; however, this has not been the case for H2RAs [Eom et al. 2011a, 2011b; Kwok et al. 2011; Yu et al. 2011]. Long-term use of PPIs significantly increased the risk for any fracture [adjusted odds ratio (OR) 1.30, 95% confidence interval (CI) 1.15–1.48] and hip fracture (adjusted OR 1.34, 95% CI 1.09–1.66), but long-term H2RA use was not significantly associated with fracture risk [Eom et al. 2011a].
It is also important to note that results from one study have shown that PPI-associated increased fracture risk may occur with relatively short-term administration of these drugs. A population-based case-control study that included 1241 cases with newly diagnosed hip fracture and controls that were pair matched to cases by age and sex showed that receiving more than 28 defined daily dose (DDDs) of PPIs was associated with an increased risk for hip fracture in multivariate analyses (adjustments for matching variables and medication use) (at 29–70 DDDs, OR 1.67, 95% CI 1.11–2.51 and at >70 DDDs, OR 2.51, 95% CI 1.77–3.55). There was also a significant trend toward increased hip fracture risk with increasing cumulative DDDs of PPIs (p < 0.0001) [Chiu et al. 2010]. While the mechanisms underlying increased fracture risk in patients taking PPIs are not fully understood [Gray et al. 2010], these data led the FDA to issue a warning to consumers and healthcare professionals in May 2010 about a possible increased risk of fractures of the hip, wrist, and spine with high doses or long-term use of PPIs [FDA, 2010] and requested changes to product labeling to describe this increased risk. In considering this potentially important difference between H2RAs and PPIs, it is important to note that these results do not necessarily reflect potential risks for famotidine administered at a total dose of 80 mg/day. As noted above, this dose has been used in only a very small number of studies.
Pneumonia and Clostridum difficile colitis
Other associated safety issues reported with PPIs include an increased risk of pneumonia and Clostridium difficile diarrhea. Comparatively, the increased risk of pneumonia or C. difficile diarrhea is lower in patients receiving H2RAs [Rodríguez et al. 2009; Dalton et al. 2009; Jayatilaka et al. 2007]. Here again, these results may not reflect risk for these adverse events that may be associated with famotidine delivered at a dose of 80 mg/day.
Conclusions
NSAIDs, including ibuprofen, are effective for decreasing pain and inflammation in patients with a wide range of acute and chronic conditions. However, these agents are associated with the potential to develop UGI ulcers and consequent serious and life-threatening bleeding and perforation. Therapy with DUEXIS® significantly decreases the risk of UGI ulcers associated with ibuprofen without altering ibuprofen’s pharmacokinetic profile. Use of a single tablet that contains both ibuprofen and famotidine may provide advantages over other approaches to lowering risk for NSAID-associated UGI ulcers and may also improve patient adherence to therapy. DUEXIS® helps fulfill an important need for many patients with chronic pain who require treatment with NSAIDs who are also at risk for NSAID-associated GI ulcers.
Footnotes
Acknowledgements
Editorial support has been provided by Robert Rhoades, PhD and Valorie Thompson.
