Abstract
Central endozepinergic signaling is implicated in glucose homeostasis. Ventromedial hypothalamic nucleus (VMN) metabolic monitoring governs glucose counter-regulation. VMN glucose-stimulatory nitric oxide (NO) and glucose-inhibitory γ-aminobutyric acid (GABA) neurons express the energy gauge 5’-AMP-activated protein kinase (AMPK). Current research addresses the premise that the astrocyte glio-peptide octadecaneuropeptide (ODN) imposes sex-dimorphic control of metabolic sensor activity and neurotransmitter signaling in these neurons. The ODN G-protein coupled-receptor antagonist cyclo(1−8)[DLeu5]OP (LV-1075) was administered intracerebroventricularly (icv) to euglycemic rats of each sex; additional groups were pretreated icv with the ODN isoactive surrogate ODN11−18 (OP) before insulin-induced hypoglycemia. Western blotting of laser-catapult-microdissected VMN NO and GABA neurons showed that hypoglycemia caused OP-reversible augmentation of phospho-, e.g., activated AMPK and nitric oxide synthase (nNOS) expression in rostral (female) or middle (male) VMN segments or ODN-dependent suppression of nNOS in male caudal VMN. OP prevented hypoglycemic down-regulation of glutamate decarboxylase profiles in female rat rostral VMN, without affecting AMPK activity. LV-1075 treatment of male, not female rats elevated plasma glucagon and corticosterone concentrations. Moreover, OP attenuated hypoglycemia-associated augmentation of these hormones in males only. Results identify, for each sex, regional VMN metabolic transmitter signals that are subject to endozepinergic regulation. Directional shifts and gain-or-loss of ODN control during eu- versus hypoglycemia infer that VMN neuron receptivity to or post-receptor processing of this stimulus may be modulated by energy state. In male, counter-regulatory hormone secretion may be governed principally by ODN-sensitive neural pathways, whereas this endocrine outflow may be controlled by parallel, redundant ODN-dependent and -independent mechanisms in female.
Keywords
Introduction
Brain astrocytes sustain nerve cell function by multiple mechanisms including regulation of microcirculatory blood flow, metabolic substrate fuel trafficking, and glio-transmitter release [Stobart & Anderson, 2013]. The 86-amino acid astrocyte glio-peptide diazepam-binding inhibitor (DBI; also referred to as acyl-CoA-binding protein, ACBP) is an endogenous benzodiazepine (BZ) receptor ligand (endozepine) that modulates anxiety and conflict behavior via negative allosteric modulation at the GABAA receptor-BZ-binding site (GABAAR-BZ) [Alho et al., 1985; Ferrero et al., 1986; Guidotti et al., 1983]. The DBI endoproteolytic cleavage product octadecaneuropeptide (ODN; DBI33−50) is a biologically-active peptide that regulates diverse brain functions [Tonon et al., 2020], including GABAAR-BZ-binding site-mediated anxiety-like and stress behaviors and ODN G-protein-coupled receptor (GPCR)-mediated control of food intake and body weight, and neuroprotective effects [Bahdoudi et al., 2018; de Mateos-Verchere et al., 2001; do Rego et al., 2007; Guidotti, 1991; Garcia de Mateos-Verchere et al., 1998; Ghouili et al., 2018; Guillebaud et al., 2020; Hamdi et al., 2012b, 2012a; Hamdi et al., 2015; Kaddour et al., 2013; Lebrun et al., 2020; Masmoudi-Kouki et al., 2018; Matsuda et al., 2007]. DBI/ACBP signaling is implicated in energy homeostasis and acts as an evident neurochemical component of the neural glucose-regulatory network as brain glucose availability regulates ODN release, which, in turn, controls systemic glucose levels [Alquier et al., 2021; Bouyakdan et al., 2019; Lanfray et al., 2013; Lebrun et al., 2020; Montégut et al., 2021; Pedro et al., 2019; Tonon et al., 2020].
The brain utilizes a disproportionate fraction of total body energy to maintain essential nerve cell functions, relying upon blood glucose as its primary energy source. Iatrogenic insulin-induced hypoglycemia (IIH) is an unremitting complication of requisite strict therapeutic management of insulin-dependent diabetes mellitus [Cryer, 2013; 2014]. Potential adverse consequences of insufficient glucose provision to the brain due to IIH include neurological impairment and injury or death of vulnerable CNS neurons. The brain responds to neuro-glucopenia by initiating autonomic, neuroendocrine, and behavioral functions that enhance body-wide glucose availability. The ventromedial hypothalamic nucleus (VMN) is important for assimilating nutrient, endocrine, and neurochemical metabolic cues that govern counter-regulation [Donovan & Watts, 2014; Watts & Donovan, 2010]. Specialized metabolic-sensory neurons in the VMN provide a continuous cellular energy readout by increasing (‘fuel-inhibited’) or reducing (‘fuel-excited’) synaptic firing when ambient energy substrate levels fall [Ashford et al., 1990; Oomura et al., 1969; Silver & Erecińska, 1998]. The medio-basal hypothalamus (MBH), which encompasses the VMN and other distinctive nuclei, provides cellular energy cues that control counter-regulatory endocrine and gluconeogenic functions [Borg et al., 1997; 2003]. Neurochemical effectors of ventromedial hypothalamic energy imbalance include the amino acid transmitter γ-aminobutyric acid (GABA), which attenuates hypoglycemic augmentation of glucagon and adrenomedullary epinephrine [Chan et al., 2006] as well as the gas nitric oxide (NO), a labile lipid-permeable free radical that stimulates counter-regulatory hormone release [Fioramonti et al., 2010; Routh et al., 2014]. GABA and NO signals originate within the VMN, as recent single-cell multiplex qPCR studies show that VMN neurons identified in situ as immunoreactive-positive for the marker protein glutamate decarboxylase65/67 (GAD) or neuronal nitric oxide synthase (nNOS) contain mRNA that encodes both GAD molecular weight variants [Ali et al., 2022] or nNOS [Briski, personal observation], respectively. VMN GABAergic and nitrergic neuron populations each express multiple estrogen receptor (ER) variants, e.g., ER-alpha, ER-beta, and G protein-coupled estrogen receptor-1 (GPER), as well as adrenergic receptor (AR) isoforms [Uddin et al., 2019]. Due to its limited diffusion range, NO is presumed to act within the VMN, but the identity(-ies) of local substrates is not known. Similarly, it is unclear if IIH-associated patterns of GABA transmission affect downstream neurons located within or outside the VMN.
The evolutionarily-conserved cellular energy gauge 5’-AMP-activated protein kinase (AMPK) is activated by phosphorylation in response to augmentation of the cellular AMP/ATP ratio [Hardie et al., 2016; Lin & Hardie, 2018]. In the hypothalamus, AMPK provides crucial input on brain cell ATP availability to neural pathways that regulate whole-body energy stability [López, 2018; Pimentel et al., 2013; Xue & Kahn, 2006]. MBH AMPK activity controls counter-regulatory reactivity to hypoglycemia [Han et al., 2005; McCrimmon et al., 2008]. VMN nitrergic and GABAergic nerve cell signaling may be directly enhanced or suppressed, respectively, by neuro-glucopenia as these site-specific cell populations express AMPK gene transcripts [Ali et al., 2022] and protein [Briski et al., 2020; Ibrahim et al., 2020]. The VMN is a likely source of AMPK glucoregulatory signaling as hypoglycemia augments AMPK phosphorylation in local NO and GABA neurons [Briski et al., 2020; Ibrahim et al., 2020].
Neuroanatomical mapping studies document widespread endozepine expression among interconnected hypothalamic metabolic loci, including the VMN [Alho et al., 1985; Malagon et al., 1993; Tonon et al., 1990]. Endozepines are known to control behavioral responses to brain glucose dyshomeostasis. Hyperphagia initiated by central glucose anti-metabolite administration is attenuated by the ODN-GPCR agonist, the ODN C-terminal octapeptide (OP) fragment (H-Arg-Pro-Gly-Leu-Leu-Asp-Leu-Lys-OH), whereas intracerebroventricular (icv) glucose-induced hypophagia is blunted by the ODN-GPCR antagonist cyclo1−8[DLeu5]OP (LV-1075) [Lanfray et al., 2013]. The present study used a well-characterized experimental model for insulin (INS)-induced hypoglycemia (IIH), replicating route and formulation of insulin administered in the clinical setting [Parajape & Briski, 2005], to investigate the hypothesis that ODN imposes sex-dimorphic control of VMN nitrergic and/or GABAergic nerve cell sensor activation and metabolic transmitter marker protein [e.g., neuronal nitric oxide synthase (nNOS), glutamate decarboxylase65/67 (GAD)] expression alongside plasma counter-regulatory hormone profiles during this metabolic stress. IIH causes sex-specific changes in VMN tissue nNOS and GAD protein expression [Ali et al., 2019; Mahmood et al., 2018; Napit et al., 2019]. In accordance with current U.S. National Institutes of Health policy emphasis on consideration of sex as a critical biological variable, the current experimental design incorporated adult rats of each sex to investigate the corollary notion that ODN regulation of these parameters may vary in magnitude and/or direction in one or both sexes between conditions of systemic glucostasis versus glucoprivation. Glucose stimulates endozepine release; evidence that the ODN GPCR antagonist LV-1075 elevates circulating glucose levels [Lanfray et al., 2013] and that central ODN action increases whole-body glucose utilization [Bouyakdan et al., 2019] infers that ODN signaling may act as a feedback mechanism for tonic suppression of glycemia. Research here employed pharmacological tools to address the premise that euglycemia-associated patterns of ODN release impose an inhibitory tone on VMN glucose-stimulatory neurotransmission and counter-regulatory hormone secretion, whereas IIH-mediated diminution of ODN may augment those functions. The current experimental design evaluated effects of 1) icv LV-1075 administration to euglycemic rats of each sex or 2) icv ODN GPCR agonist OP pretreatment of male and female rats prior to IIH on neurochemical marker expression and AMPK activity in VMN nerve cell types implicated in neural regulation of glucose homeostasis and on plasma glucose and counter-regulatory hormone profiles. Recent analyses of VMN micropunch-dissected tissue samples obtained at different rostro-caudal levels provide novel evidence for regional variability in hypoglycemic patterns of total and phosphorylated AMPK protein expression in this critical glucose-regulatory brain structure in male, but not female rats [Uddin et al., 2020]. Herein, high-neuroanatomical resolution laser-catapult microdissection technology was used in conjunction with Western blotting to investigate patterns of target protein expression in pure subpopulations of nitrergic and GABAergic neurons collected at different levels over the length of the VMN.
Materials and Methods
Experimental Design
Description of Primary Antisera Used.
Results
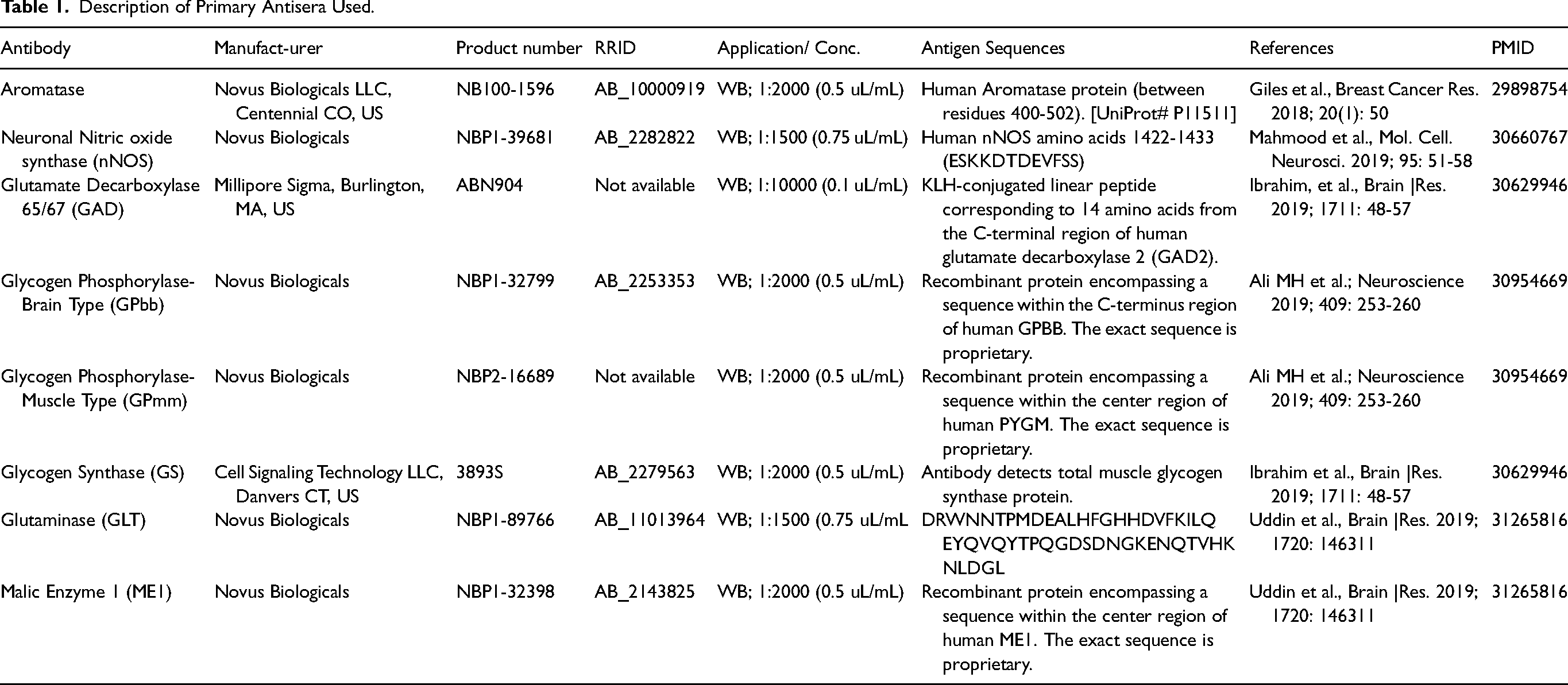
Central endozepinergic signaling is implicated in systemic glucose control, yet potential cell targets within the VMN, a key integrative element of the brain glucose-regulatory network, are not known. Current research utilized single-cell microdissection techniques to determine if, and where within the VMN ODN may regulate metabolic sensor activity and neurotransmitter signaling in characterized glucose-stimulatory or -inhibitory nerve cell populations. Figure 1 depicts combinatory immunocytochemical labeling and laser-catapult microdissection of nitrergic and GABAergic neurons from the VMN, a prominent bilateral structural feature of the MBH. The MBH is depicted in panel A-1 as a black oval within the diencephalon (brown square). Enlargement of the diencephalon in the rat brain coronal brain section in panel A-II shows the orientation of the VMN (oval area denoted by brown dotted line) within the MBH. The methylene blue-stained tissue section in panel A-III displays the clear demarcation of the VMN (brown dotted line) from surrounding structures, including the arcuate hypothalamic nucleus (ARH). Individual nerve cells in VMH tissue sections were identified by GAD (Panel B-I) or nNOS (Panel C-I) -ir prior to laser-catapult harvesting; representative immunolabeled neurons are indicated by blue arrows. Panels B-II – B-IV and C-II – C-IV portray sequential actions, e.g., sequential positioning of a continuous laser cut (shown as a green line) around individual neurons, followed by laser beam ejection of those neurons whereby single-cell microdissection is achieved in the absence of destruction of surrounding tissue and minimal inclusion of adjacent tissue.
Laser-Catapult microdissection of Immunolabeled Ventromedial Hypothalamic Nucleus (VMN) γ-Aminobutyric Acid (GABA) or Nitric Oxide (NO) Neurons. The VMN and other critical neuroendocrine structures reside in the mediobasal hypothalamus (MBH), which is depicted by the solid blue midline oval in Panel A-I. The VMN is a bilateral component of the MBH, as indicated by blue ovals in Panel A-II, and is neuroanatomically distinct from adjacent structures, including the hypothalamic arcuate nucleus (ARH) (Panel A-III). Panel B-1 shows the widespread distribution of glutamate decarboxylase65/67 (GAD)-immunoreactive (ir)-positive neuron perikarya (blue arrows) in the VMN. Panels B-II through B-IV portray a representative GAD-ir-positive neuron (blue arrow) after immunostaining and encirclement by green laser track, and residual tissue vacancy after laser pulse-mediated ejection of that nerve cell, leaving minimal destruction of adjacent neural tissue. Panel B-V illustrates an uncropped immunoblot containing a single detected antigen band of anticipated molecular weight after Western blotting of lysates of laser-harvested VMN GAD-ir-positive neurons. A typical pattern of distribution of VMN neuronal nitric oxide synthase (nNOS)-ir-positive neurons is presented in Panel C-I. Panels C-II through C-IV illustrate a representative immunostained nitrergic nerve cell body (blue arrow), its location within a laser track, and intact tissue remaining after harvesting of that cell. Panel C-V shows an uncropped immunoblot featuring a single immunodetectable protein band from VMN nNOS-ir cell lysates identified by the primary antibody utilized for immunostaining in situ.
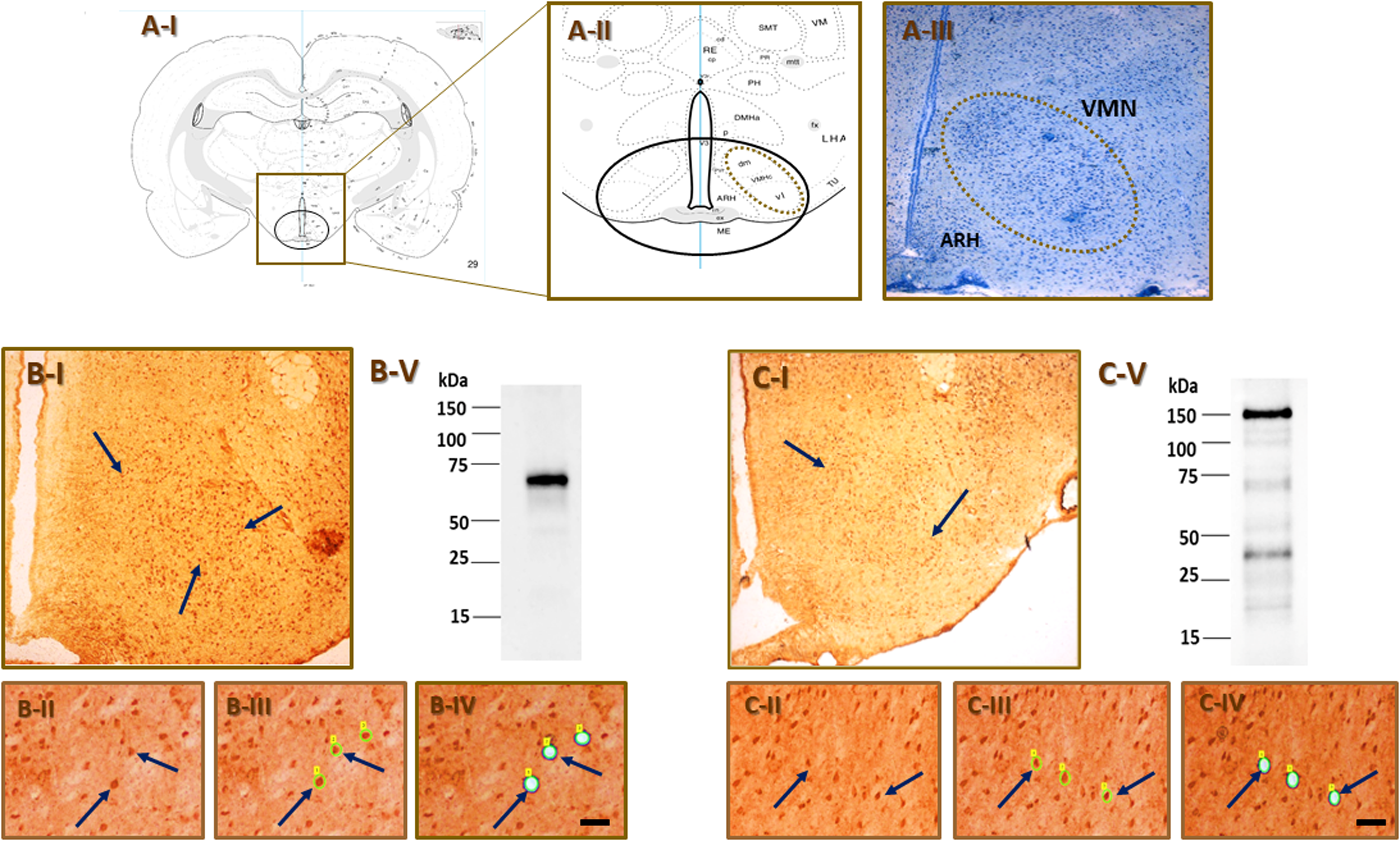
Present studies used a pharmacological approach to address the premise that ODN action on its GPCR imposes tonic control of VMN NO marker and/or activated AMPK protein profiles in nitrergic neurons located in distinct rostro-caudal levels of the euglycemic male and female rat VMN. Figure 2 depicts effects of icv administration of the ODN-GPCR antagonist LV-1075 on nNOS (Row A), AMPK (Row B), and pAMPK (Row C) protein expression and mean pAMPK/AMPK ratios (Row D) in nNOS-ir neurons collected from rostral [left-hand column], middle [middle column], or caudal [right-hand column] regions of the male and female VMN. Outcomes of statistical analyses are presented in Supplementary Table 1. Data show that endogenous ODNergic tonus blockade reduced or increased nNOS protein levels in nitrergic neurons taken from male and female rat rostral VMN, respectively (Panel A.1). Neurons located in this segment also exhibited increased total AMPK protein content in the female (Panel B.1), and diminished or amplified pAMPK expression in male or female, respectively, in response to LV-1075 treatment (Panel C.1). Middle VMN NO neurons exhibited an increase in nNOS content (Panel A.2), alongside decreased AMPK (Panel B.2) and increased pAMPK levels (Panel C.2) in LV-1075-injected male, but not female rats. In the caudal VMN, nNOS protein profiles were refractory to ODN-GPCR antagonism in both sexes (Panel A.3), yet this treatment elevated AMPK (Figure B.3) or decreased pAMPK (Panel C.3) expression in nitrergic neurons obtained from female versus male rats, respectively. Male rats responded to LV-1075 treatment with bi-directional, segment-specific changes in mean nitrergic nerve cell pAMPK/AMPK ratios, as those values were increased in the middle VMN (Panel D.2), yet diminished in rostral (Panel D.1) and caudal (Panel D.3) regions. Yet, in the female, only caudal VMN nitrergic neurons showed a change, e.g., reduction in mean pAMPK/AMPK expression in response to ODN-GPCR blockade.
Effects of the octadecaneuropeptide GPCR antagonist Cyclo1−8[DLeu5]OP (LV-1075) on ventromedial hypothalamic nucleus (VMN) nitric oxide (NO)_neuron 5’-AMP-activated protein kinase (AMPK) activity and transmitter marker protein expression in male versus female rats. Groups of male and female rats were euthanized one hour after intracerebroventricular (icv) administration of vehicle (V; 0.9% NaCl; groups 1 and 2) or LV-1075 (20µg/7 µL NaCl [Lanfray et al., 2013] for brain dissection. Neurons immunocytochemically labeled for nitric oxide synthase (nNOS) were laser-catapult-microdissected from 10 micron-thick fresh frozen sections cut from successive rostro-caudal segments of the VMN. In each treatment group, aliquots of NO nerve cell lysates from individual subjects were combined to create separate triplicate (n = 3) sample pools for Western blot analysis of individual target proteins. Graphs show mean normalized nNOS (Row A), AMPK (Row B), and phospho-AMPK (pAMPK; Row C) protein optical density (O.D.) measures ± S.E.M. for nitrergic neurons collected from the rostral (left-hand column), middle (middle column), or caudal (right-hand column) VMN of male (white bars) and female (gray bars) rats treated by icv injection of V (solid bars; n = 3 male nerve cell lysate pools, n = 3 female nerve cell lysate pools) or LV-1075 (20 µg/rat; cross-hatched bars; n = 3 male nerve cell lysate pools, n = 3 female nerve cell lysate pools). Effects of LV-1075 treatment on the mean ratio of pAMKP/AMPK protein expression in rostral, middle, and caudal VMN nitrergic neurons are shown in Row D. Circles depict individual independent data points. Data were analyzed by two-way analysis of variance and Student-Newman-Keuls post-hoc test using GraphPad Prism (Volume 8) software; outcomes are presented in Supplementary Table 1. *p < 0.05, **p < 0.01, ***p < 0.001.
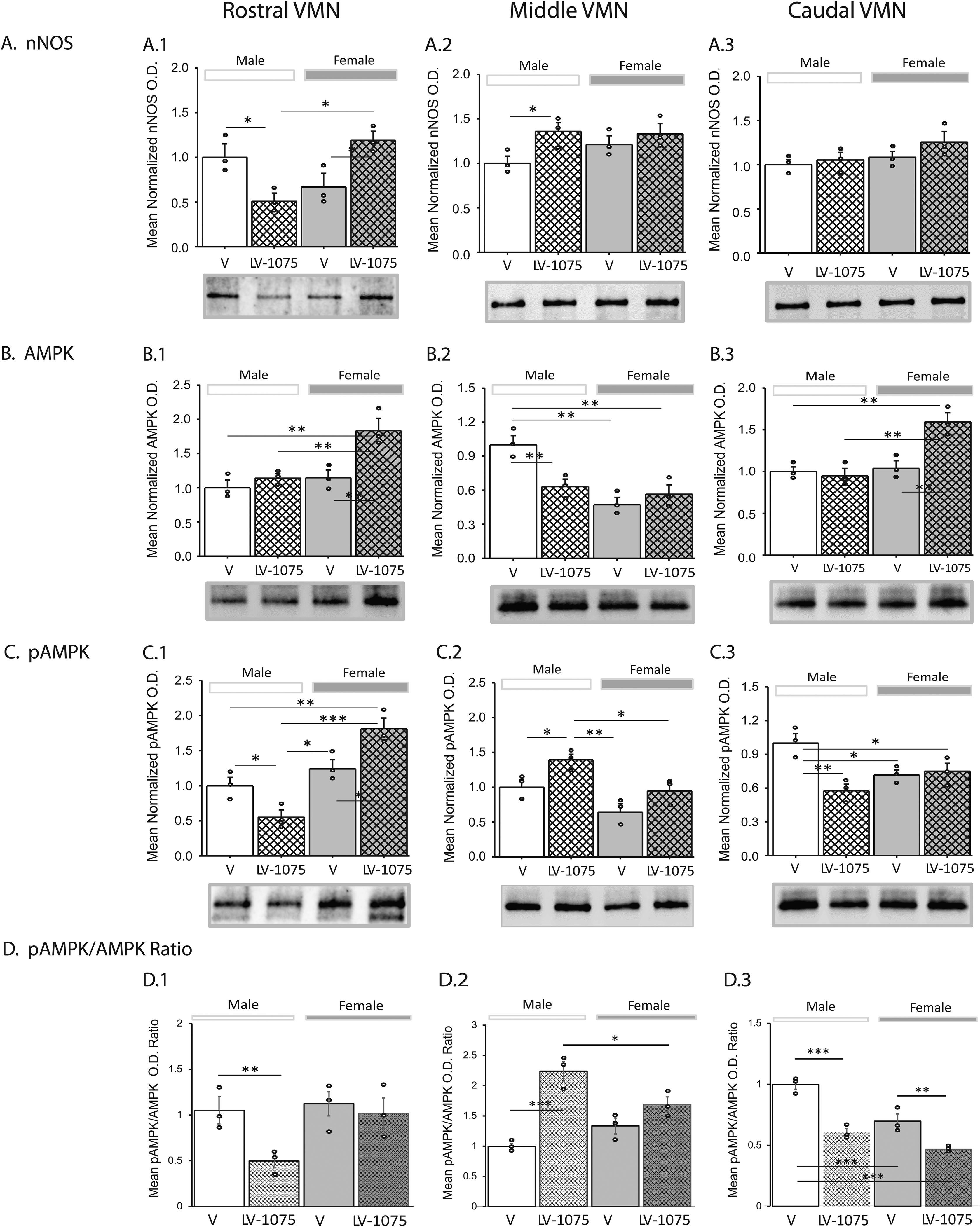
Current work investigated whether ODN regulates expression of GAD, a protein marker for the glucose-inhibitory neurochemical GABA, and/or GABAergic nerve cell AMPK activity in the VMN of either sex during glucose homeostasis. Effects of LV-1075 on rostral [left-hand column], middle [middle column], and caudal [right-hand column] VMN GABAergic neuron target protein expression and mean pAMPK/AMPK ratios in male and female rats are illustrated in Figure 3. Outcomes of statistical analyses are presented in Supplementary Table 1. Endogenous ODNergic blockade reduced total GAD content (Row A) in GABA nerve cells collected from rostral (Panel A.1) and caudal regions (Panel A.3) of the female VMN, but did not alter this protein profile in any segment of the male VMN. Rostral VMN GABAergic neurons exhibited decreased or increased AMPK levels (Panel B.1) in LV-1075-treated male and female rats, respectively. In other VMN segments, endogenous ODN signaling inhibition caused similar changes in GABA nerve cell AMPK expression in each sex, e.g., reduction in middle VMN (Panel B.2) versus augmentation in caudal VMN (Panel B.3). Male rats injected with LV-1075 icv exhibited either suppression (rostral) or augmentation (middle) of GABA nerve cell pAMPK expression, depending upon rostro-caudal VMN segment (Row C). Drug-treated females were also characterized by region-specific pAMPK protein responses to LV-1075, as this profile was correspondingly increased or decreased in middle (Panel C.2) versus caudal (Panel C.3) VMN. Mean VMN GABA neuron pAMPK/AMPK ratios were altered along the rostro-caudal length of the female VMN, e.g., down-regulated in rostral (Panel D.1) and caudal (Panel D.3) VMN, but elevated in the middle segment (Panel D.2) after LV-1075 administration. However, in the male, ODN-GPCR antagonism had no effect on this ratio in VMN GABAergic nerve cells.
VMN γ-Aminobutyric acid (GABA) neuron AMPK activation and glutamate Decarboxylase65/67 (GAD) protein expression in Icv LV-1075 - injected male and female rats. Groups of male and female rats were euthanized one hour after intracerebroventricular (icv) administration of vehicle (V; 0.9% NaCl; groups 1 and 2) or LV-1075 (20µg/7 µL NaCl [Lanfray et al., 2013] for brain dissection. Neurons immunocytochemically labeled for GAD were laser-catapult-microdissected from 10 micron-thick fresh frozen sections cut from successive rostro-caudal segments of the VMN. In each treatment group, aliquots of GABA nerve cell lysates from individual subjects were combined to create separate triplicate (n = 3) sample pools for Western blot analysis of individual target proteins. Data show mean normalized GAD (Row A), AMPK (Row B), and pAMPK (Row C) protein O.D. measures ± S.E.M. for GABAergic neurons taken from the rostral (left-hand column), middle (middle column), or caudal (right-hand column) VMN after icv LV-1075 administration. Treatment groups are depicted as follows: V- (solid white bars; n = 3 lysate pools) versus LV-1075 (cross-hatched white bars; n = 3 lysate pools)-injected male rats; V- (solid gray bars; n = 3 lysate pools) versus LV-1075- (cross-hatched gray bars, n = 3 lysate pools) injected female rats. Effects of LV-1075 treatment on the mean ratio of pAMKP/AMPK protein expression in rostral, middle, and caudal VMN GABAergic neurons are shown in Row D. Circles depict individual independent data points. Data were analyzed by two-way analysis of variance and Student-Newman-Keuls post-hoc test using GraphPad Prism (Volume 8) software; outcomes are presented in Supplementary Table 1. *p < 0.05, **p < 0.01, ***p < 0.001.
Research outcomes described below were obtained using a validated whole-animal model for experimental IIH to address the question that ODN may impose segment-specific control of hypoglycemic patterns of VMN nitrergic nerve cell transmitter marker and activated AMPK protein expression in each sex. Effects of sc INS injection on VMN nitrergic neuron nNOS (Row A), AMPK (Row B), pAMPK (Row C) protein expression and mean pAMPK/AMPK ratios (Row D) were examined in male and female rats after icv pretreatment with the ODN isoactive fragment OP or V (Figure 4). Outcomes of statistical analyses are presented in Supplementary Table 2. In each sex, VMN NO neurons exhibited segment-specific adjustments in nNOS expression during hypoglycemia, as this protein was up-regulated in rostral (Panel A.1) and middle (Panel A.2) VMN in males and rostral in female VMN, but down-regulated in the caudal VMN (Panel A.3) in male and female rats. OP administration normalized nNOS expression in nitrergic neurons collected from middle and caudal VMN segments in INS-injected males, and the rostral VMN in INS-treated females. In male rats, hypoglycemia increased NO nerve cell AMPK protein content in the rostral (Panel B.1) and middle (Panel B.2) VMN, but decreased this profile in the caudal VMN (Panel B.3). In this sex, OP pretreatment prevented hypoglycemic patterns of AMPK expression in middle VMN nitrergic neurons only. Female rats exhibited hypoglycemic down- or up-regulation of nitrergic neuron AMPK in rostral and middle VMN segments, respectively; these responses were not averted by OP. Hypoglycemia-associated augmentation of NO nerve cell pAMPK profiles occurred throughout the male VMN (Panels C1-C3); in each region, this stimulatory response was totally reversed by OP pretreatment. On the other hand, female rats exhibited OP-reversible (rostral VMN; Panel C1) or -exacerbated effects (caudal VMN; Panel C3) of hypoglycemia on nitrergic neuron pAMPK expression. As shown in Panels D.1-D.3, hypoglycemic male rats showed no change in rostral and middle VMN NO nerve cell mean pAMPK/AMPK expression, yet OP-reversible augmentation of this ratio occurred in the caudal VMN. In the hypoglycemic female, mean pAMPK/AMPK ratios were altered in rostral (OP-reversible) and middle (OP-non-reversible) VMN nitrergic nerve cells.
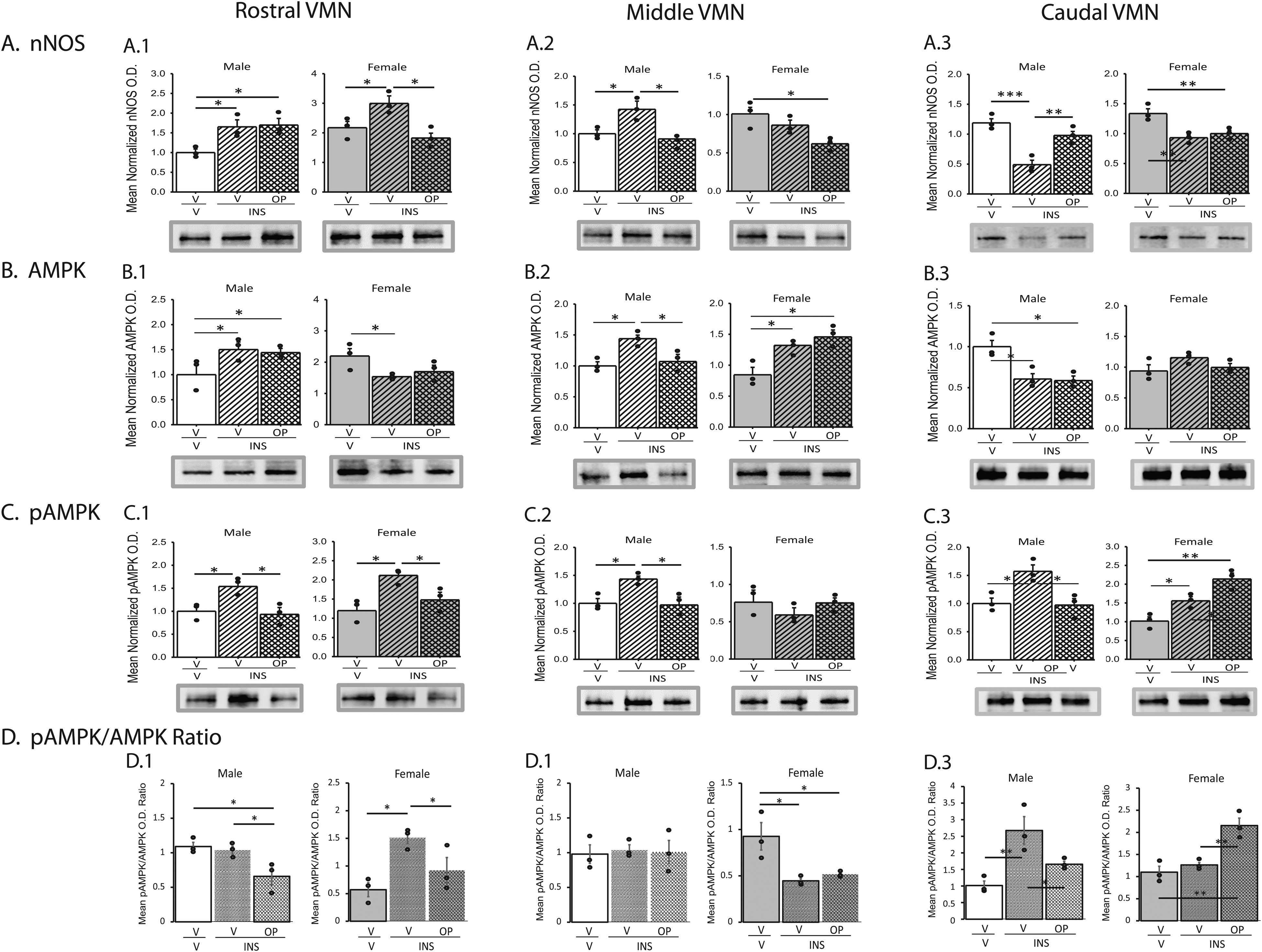
Effects of hypoglycemia on VMN nitrergic neuron AMPK activation and nNOS protein expression in male versus female rats: impact of octadecaneuropeptide GPCR agonist octapeptide (OP) pretreatment. Male and female rats were pretreated by icv administration of vehicle (V) or OP (2.0 ug/rat) before subcutaneous (sc) injection of vehicle (V) or insulin (INS; 10.0 U neutral protamine Hagedorn insulin/100 g bw). Animals were euthanized one hour after sc injections for brain dissection and processing of tissue sections cut through the VMN for nNOS immunolabeling. VMN NO neurons were collected from rostral (left-hand column), middle (middle column), and caudal (right-hand column) VMN segments after injections. Data were analyzed by one-way analysis of variance and Student-Newman-Keuls post-hoc test using GraphPad Prism (Volume 8) software. Graphs show mean normalized nitrergic neuron nNOS (Row A), AMPK (Row B), and pAMPK (Row C) protein O.D. measures ± S.E.M. for groups of male or female rats treated as follows: V icv /Vsc (males: solid white bars, n = 3 lysate pools/females: solid gray bars, n = 3 lysate pools); V icv /INSsc (males: diagonal-striped white bars, n = 3 lysate pools/females: diagonal-striped gray bars, n = 3 lysate pools); OP icv /INSsc (males: cross-hatched white bars, n = 3 lysate pools/females: cross-hatched gray bars, n = 3 lysate pools). Effects of IIH, with or without OP pretreatment on mean nitrergic nerve cell pAMKP/AMPK expression ratios are presented in D. Circles depict individual independent data points. Data for each sex were analyzed by one-way analysis of variance and Student-Newman-Keuls post-hoc test using GraphPad Prism (Volume 8) software; outcomes are presented in Supplementary Table 2. *p < 0.05, **p < 0.01, ***p < 0.001.
An additional objective was to investigate whether VMN GABAergic nerve cell metabolic sensing and transmitter signaling may be controlled by ODN in specific rostro-caudal segments of the male and female VMN during hypoglycemia. Figure 5 depicts the effects of OP pretreatment on VMN GABA nerve cell target protein expression in male and female rats. Outcomes of statistical analyses are presented in Supplementary Table 2. As shown in Panels A.1–A.3, hypoglycemia repressed GABA neuron GAD protein expression in rostral (both sexes), middle (females only), and caudal (males only) segments of the VMN. These inhibitory responses were not prevented by OP. In hypoglycemic male rats, GABAergic AMPK profiles were increased (rostral VMN, Panel B.1) or decreased (middle VMN, Panel B.2; caudal VMN, Panel B.3) compared to controls; OP normalized AMPK expression in rostral VMN GABA neurons only. Female rats exhibited OP-reversible hypoglycemic augmentation of AMPK content in caudal VMN GABAergic cells (Panel B.3). GABA neuron pAMPK expression was up-regulated in the female middle (Panel C.2) and caudal (Panel C.3) VMN; in middle VMN this protein profile was partially restored by OP pretreatment while pAMPK content was decreased below control levels in the caudal region. Hypoglycemic stimulation of caudal VMN GABA cell pAMPK protein in male rats was refractory to OP. In male rats, GABAergic nerve cell mean pAMPK/AMPK expression ratios were unaffected by IIH in rostral and middle segments of the VMN, but up-regulated in both OP- or V-pretreated INS-injected animals (Panel D.3). IIH enhanced pAMPK/AMPK ratio values in this transmitter nerve cell type in middle VMN, a response that was blunted by OP pretreatment.
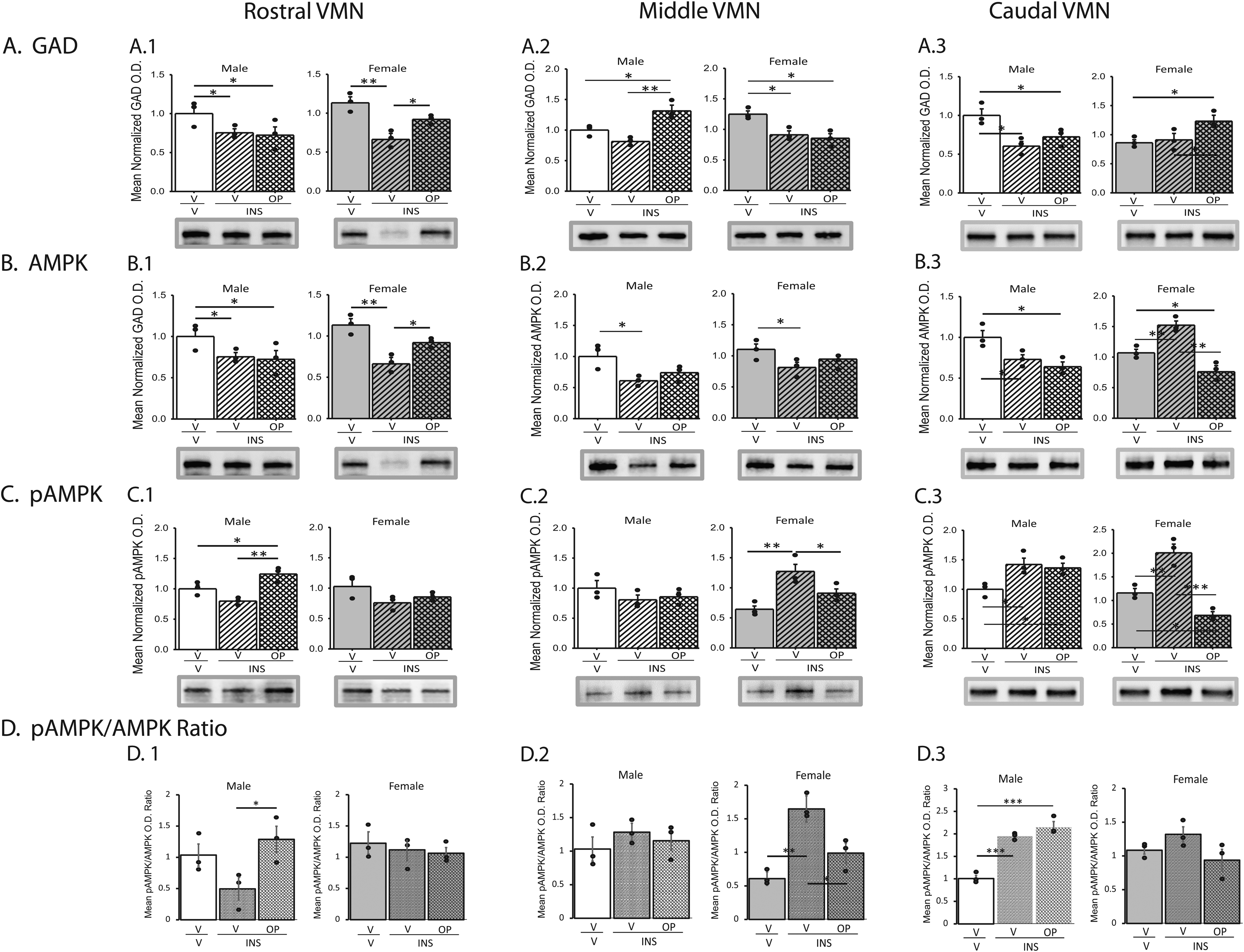
Impact of OP on hypoglycemic patterns of VMN GABAergic AMPK activation and GAD protein expression in male and female rats. Male and female rats were pretreated by icv administration of vehicle (V) or OP (2.0 ug/rat) before subcutaneous (sc) injection of vehicle (V) or insulin (INS; 10.0 U neutral protamine Hagedorn insulin/100 g bw). Animals were euthanized one hour after sc injections for brain dissection and processing of tissue sections cut through the VMN for GAD immunolabeling. VMN GABAergic neurons were collected from rostral (left-hand column), middle (middle column), and caudal (right-hand column) VMN segments after injections. Graphs show mean normalized GABA nerve cell GAD (Row A), AMPK (Row B), and pAMPK (Row C) protein O.D. measures ± S.E.M. for the following treatment groups: V icv /Vsc (males: solid white bars, n = 3 lysate pools/females: solid gray bars, n = 3 lysate pools); V icv /INSsc (males: diagonal-striped white bars, n = 3 lysate pools/females: diagonal-striped gray bars, n = 3 lysate pools); OP icv /INSsc (males: cross-hatched white bars, n = 3 lysate pools; females/cross-hatched gray bars, n = 3 lysate pools). Effects of IIH, with or without OP pretreatment on mean GABAergic neuron pAMKP/AMPK expression ratios are presented in D. Circles depict individual independent data points. Data for each sex were analyzed by one-way analysis of variance and Student-Newman-Keuls post-hoc test using GraphPad Prism (Volume 8) software; outcomes are presented in Supplementary Table 2. *p < 0.05, **p < 0.01, ***p < 0.001.
Pharmacological manipulation of central ODN GPCR signaling is reported to affect circulating glucose levels in the male rat [Lanfray et al., 2013], but it is unclear if icv LV-1075 or OP effects on VMN glucose-regulatory nerve cell target proteins coincide with alterations in counter-regulatory hormone secretion in eu- or hypoglycemic rats, respectively. Effects of LV-1075 administration on male and female rat plasma glucose (Panel A1), the counter-regulatory hormones corticosterone (Panel A2) and glucagon (Panel A3), and free fatty acid (FFA) concentrations are shown in Figure 6. Outcomes of statistical analyses are presented in Supplementary Table 3. Circulating glucose levels (Panel A1) were refractory to LV-1075 in each sex. Male, but not female rats exhibited increased plasma corticosterone (Panel A2) and glucagon (Panel A3) levels in response to LV-1075. This treatment decreased FFA levels in each sex (Figure A.4). Data presented in Row B depict effects of OP pretreatment on INS-induced hypoglycemic glucose profiles (Panel B1), and hypoglycemia-associated plasma corticosterone (Panel B2), glucagon (Panel B3), and FFA (Panel B4) concentrations. Current data show that OP pretreatment exacerbated diminished glycemic profiles in INS-injected male rats, similar to prior studies in that sex [Lanfray et al., 2013] but not females. Hypoglycemia elevated corticosterone and glucagon secretion in each sex; these responses were abolished by OP administration in male, but not female rats. Circulating FFA levels were decreased in hypoglycemic rats of each sex; here, OP pretreatment prevented this inhibitory response in males, but not females. These data show that ODN regulation of counter-regulatory hormone secretion in the rat is sex-specific. Outcomes infer that ODN GPRC signaling is a critical determinant of eu- and hypoglycemic patterns of corticosterone and glucagon release in male, but not female rats. Results also suggest that metabolic status governs ODN control of circulating FFA levels in the female, but not male.
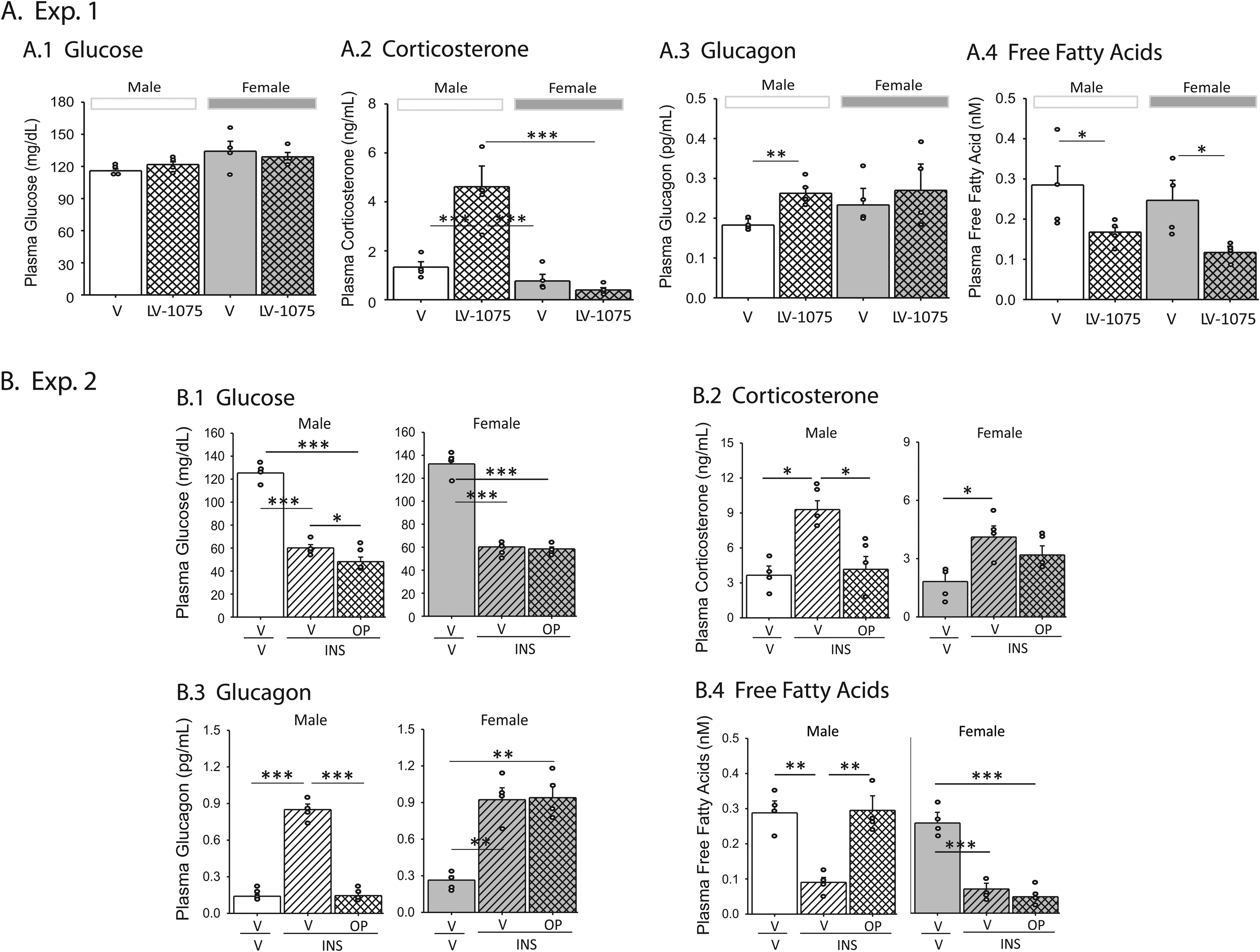
Pharmacological analysis of ODN regulation of plasma glucose, counter-regulatory hormone, and free fatty acid (FFA) levels in male and female rats. Graphs in Row A depict effects of icv V or LV-1075 administration (Experiment 1) on circulating glucose (Figure A1), corticosterone (Figure A2), glucagon (Figure A3), and FFA (Figure A4) concentrations in euglycemic male and female rats. Samples were collected 1 hour post-injection. Data were analyzed by two-way analysis of variance and Student-Newman-Keuls post-hoc test using GraphPad Prism (Volume 8) software. Data show mean values ± S.E.M. for groups of male or female rats injected with V (males: solid white bars, n = 5)/females: solid gray bars, n = 5) or LV-1075 (males: white cross-hatched bars, n = 5/females: gray cross-hatched bars, n = 5). Graphs in Row B illustrate effects of OP pretreatment on plasma glucose (Figure B1), corticosterone (Figure B2), glucagon (Figure B3) and FFA (Figure B4) levels in the following treatment groups in Experiment 2: V icv /Vsc (males: solid white bars, n = 5/females: solid gray bars, n = 5), V icv /INSsc (males: diagonal-striped white bars, n = 5/females: diagonal-striped gray bars, n = 5), and OP icv /INSsc (males: cross-hatched white bars, n = 5; females/cross-hatched gray bars, n = 5) treatment groups. Samples were obtained one hour after V or INS injection. Data were analyzed by two-way analysis of variance and Student-Newman-Keuls post-hoc test using GraphPad Prism (Volume 8) software; outcomes are presented in Supplementary Table 3. Circles depict individual independent data points. *p < 0.05, **p < 0.01, ***p < 0.001.
Discussion
VMN nitrergic and GABAergic neurons express the high-sensitivity energy gauge AMPK, and are implicated in neural control of counter-regulatory hormone secretion. Herein, pharmacological tools were used in conjunction with combinatory immunochemistry/laser-catapult microdissection/Western blot techniques to address the premise that the astrocyte transmitter ODN imposes sex-specific control of VMN NO and/or GABA neuron energy sensor and metabolic transmitter signaling during eu- and/or hypoglycemia. Results show that OP attenuates hypoglycemic augmentation of nNOS and pAMPK profiles in rostral (female) and middle (male) VMN nitrergic neurons; endogenous ODN input suppresses baseline expression of these proteins in those same sites. On the other hand, down-regulated NO signaling in the hypoglycemic male caudal VMN may be opposed by ODN. GABAergic transmission is evidently liable to ODN control in the female VMN, specifically the rostral segment, but not in this structure in the male. Further effort is needed to identify, in each sex, metabolic cues that may govern nitrergic and GABAergic cell responses to this gliopeptide. Endozepinergic signaling evidently affects principal neural pathways controlling counter-regulatory hormone release in the male, whereas ODN-dependent and -independent mechanisms may operate in parallel to regulate this outflow in the female.
ODN regulation of VMN NO neurotransmission will possibly occur in a rostro-caudal region– specific manner in each sex. Administration of the ODN-GPCR antagonist LV-1075 to male rats either inhibited, elevated, or did not alter nNOS protein expression in rostral, middle, or caudal levels of the VMN, respectively; thus, endogenous ODN may correspondingly amplify, diminish, or have no impact on basal NO signaling, in a segment-specific manner. On the other hand, LV-1075 had a singular stimulatory effect on nNOS profiles in females, which was observed in the rostral VMN. Notably, in males, there is an evident loss- or gain-of-ODN influence on VMN nitrergic signaling, as the ODN-GPCR agonist OP either did not alter (rostral VMN) or suppressed (caudal VMN) hypoglycemic patterns of nNOS expression. It should be noted that measurable changes in total cellular nNOS protein levels due to pharmacological treatments administered here do not constitute definitive evidence for simultaneous adjustments in enzyme activity. Confirmation of neuron population-specific modifications in nNOS activity state will necessitate application of analytical tools of sufficient sensitivity for quantification of enzyme function in pure cell samples. These findings bolster the need to investigate whether metabolic state-specific cues modulate NO neuron receptivity to and/or post-receptor processing of ODN stimulation at specific levels of the VMN. The current project did not assess if or how hypoglycemia may affect endogenous VMN ODN release in either sex, as analytical tools for quantification of this polypeptide in small-volume brain tissue samples corresponding to rostro-caudal VMN segments collected here are not available. Therefore, it remains to be determined if differential action of ODN during eu- versus hypoglycemia reflect, in part, adjusted glio-peptide transmitter signal strength in male or female. It is acknowledged that conclusions drawn from observations here of OP effects on target proteins are subject to future verification that ODN secretion is not, in fact, suppressed to physiologically-negligible levels during hypoglycemia.
Under conditions of systemic glucose balance, ODN evidently regulates total AMPK protein expression in rostro-caudal segment-defined subsets of VMN nitrergic neurons in each sex, as this protein was down-regulated in NO nerve cells in the male middle VMN, yet increased in the female rostral and caudal VMN. In the male, LV-1075 caused divergent changes in AMPK and pAMPK protein profiles in each VMN region, reflected by diminution (rostral and caudal VMN) or augmentation (middle VMN) of mean nitrergic neuron pAMPK/AMPK ratios. In that sex, ODN-GPCR signaling is thus likely capable of stimulating or suppressing AMPK enzyme specific activity in a VMN segment-specific manner. In the female, NO nerve cell pAMPK/AMPK ratios were refractory to LV-1075, except in the caudal VMN where the mean value was decreased. It is of interest to point out that ODN may control AMPK sensor activity in this VMN transmitter cell type by regulating AMPK and/or pAMPK expression versus AMPK levels alone in male or female, respectively. In the male rostral and middle VMN, ODN stimulation or suppression of NO signaling may reflect, in part, augmentation versus inhibition, respectively, of AMPK activity. On the other hand, up-regulation of nNOS protein in female rostral VMNnitrergic neurons in the absence of change in pAMPK/AMPK ratio suggests that ODN may inhibit glucose-stimulatory NO transmission in that segment by AMPK-independent mechanisms. In caudal VMN of each sex, LV-1075 had dissimilar effects on NO nerve cell nNOS protein expression (no change) and pAMPK/AMPK ratio (decreased), suggesting that in this segment, positive ODN effects on sensor activation may be offset by concurrent inhibitory inputs, culminating in no net change in nitrergic transmission.
The neuroanatomical cell sampling strategy employed here provides unique evidence for regional heterogeneity of VMN NO nerve cell reactivity to hypoglycemia. For example, INS-injected male rats exhibited increased or decreased nNOS protein expression in nitrergic neurons harvested from rostral and middle versus caudal VMN segments. On the other hand, NO nerve cells from hypoglycemic females contained augmented nNOS protein only in the rostral VMN, as this protein profile was either unaffected or decreased relative to other VMN segments. Results additionally document region-specific VMN nitrergic neuron sensitivity to ODN input during hypoglycemia in each sex. Whereas IIH up-regulated pAMPK protein expression in NO neurons along the rostro-caudal length of the male VMN, only the caudal segment population showed a change in mean pAMPK/AMPK expression ratio, suggesting that nitrergic signaling in that region alone may be linked to AMPK activity. In the female, evidence that nNOS and pAMPK/AMPK ratio responses to IIH coincide only in the rostral VMN implies that sensor-controlled NO transmission may occur in that location alone. Current data show that the ODN-GPCR agonist OP reversed hypoglycemic patterns of nNOS protein expression in nitrergic neurons residing in select segments of the male and female VMN. Evidence that OP concurrently attenuated IIH-associated changes in mean pAMPK/AMPK ratios in the female rostral VMN and male caudal VMN, by action on pAMPK protein expression, infers that AMPK activity state may be critical for ODN-GPCR regulation of local nitrergic transmission during hypoglycemia. Interestingly, OP elevated nNOS protein levels, but decreased the pAMPK/AMPK ratio in NO cells in the hypoglycemic male caudal VMN, results that infer that ODN may paradoxically stimulate NO signaling in this segment by reducing AMPK specific activity. Present data document a hypoglycemia-associated directional shift in ODN control of NO nerve cell AMPK activity state in specific regions of the male and female VMN as, for example, in the former sex, this stimulus exerts a positive effect on the mean pAMPK/AMPK ratio in the rostral and caudal VMN, but instead suppresses sensor activation in sites during hypoglycemia.
Present outcomes provide unique proof that VMN GABAergic signaling is subject to ODN control in female, but not male rats. In response to LV-1075 administration, euglycemic females exhibited down-regulated GABAergic nerve cell GAD protein in rostral and caudal VMN segments; GAD-immunolabeled neurons taken from these same locations in the male exhibited no effect of this treatment on GAD profiles. In euglycemic females, AMPK activity state may be critical for ODN-GPCR stimulation of rostral and caudal GAD expression as LV-1075 significantly decreased mean pAMPK/AMPK ratio values in those sites. Notably, ODN-GPCR antagonism reduced this ratio by increasing total AMPK levels, without altering pAMPK protein expression. GABAergic neurons collected from male rats showed no effect of LV-1075 on this index of enzyme specific activity. Hypoglycemia inhibited GAD protein expression in GABA neurons micro-dissected from the rostral VMN of each sex, as well as GAD-immunopositive nerve cells obtained from middle or caudal segments of female and male VMN, respectively. In females, OP administration prior to INS injection reversed hypoglycemic suppression of GABAergic nerve cell GAD expression in the rostral VMN, but had no effect on this protein in any segment of the hypoglycemic male rat VMN. OP regulation of rostral VMN GABA transmission in hypoglycemic females evidently does not involve AMPK as this treatment did not affect the mean pAMPK/AMPK ratio in GAD-expressing neurons residing in that site.
The VMN is a critical component of the brain sex-dimorphic neural network that is imprinted by estradiol during development [Simerly, 2002]. In the adult, ERs are highly expressed in the ventrolateral subdivision VMN (VMNvl) [Yang et al., 2013], which is involved in sex-specific control of several behavioral and physiological functions, including sexual and maternal behaviors, physical activity, and metabolic homeostasis [Correa et al., 2015; Hashikawa et al., 2017; Martínez de Morentin et al., 2014; Musatov et al., 2007; Yang et al., 2013]. Neurons that exhibit diminished or enhanced synaptic firing during glucoprivation have been identified in the VMNvl [Cotero & Routh, 2009; Santiago et al., 2006]. There is a need for further research to investigate, within rostro-caudal VMN segments documented here to contain ODN-sensitive GABA- and/or nitrergic neurons, whether ODN regulatory effects on either cell population vary among ventrolateral, central, and dorsomedial subdivisions of this structure, and if so, involve ER-dependent mechanisms. It is noted that stabilization of plasma estradiol levels at metestrus-like levels, accomplished here by sc capsule implantation in OVX female rats, does not replicate the dynamic estrous cycle pattern of estradiol secretion that occurs in ovary-intact adult animals. Thus, it should be considered that VMN glucose-regulatory neuron responses to the pharmacological agents used here may differ in the presence of unvarying estradiol concentrations versus exposure to fluctuating endogenous estradiol release.
Project outcomes document sex-dimorphic ODN control of glucose counter-regulatory hormone secretion. Results show that plasma glucagon and corticosterone levels were elevated in LV-1075 - treated male, but not female rats relative to V controls. These data infer that ODN signaling imposes a negative brake on release of these hormones in one, but not both sexes, during glucostasis. Similarly, OP pretreatment abolished hypoglycemia-associated augmentation of both hormone profiles in male rats only, signifying that ODN signaling during this metabolic stress may curb these critical counter-active responses in that sex. Additional effort is justified to address the question of whether OP-sensitive VMN NO neurons are involved in this down-regulating treatment effect. While current results document parallel effects of pharmacological treatments on VMN neurotransmitter marker protein expression and counter-regulatory hormone profiles in the male, it should be kept in mind that further research is needed to establish whether those neurochemicals mediate ODN regulatory effects on hormone endpoints. Despite LV-1075 and OP regulation of VMN gluco-regulatory nerve cell signaling in the female, neither treatment affected plasma glucagon or corticosterone levels in this sex. Thus, current results support the view that in the female, redundant, parallel ODN-dependent and -independent pathways may control release of these hormones during either glucose sufficiency or deficiency. LV-1075 treatment suppressed circulating FFA levels in both sexes, indicative of a positive influence on systemic provision of these substrate fuels. The ability of OP to reverse hypoglycemia-associated suppression of FFA concentrations in male, but not female rats infers that metabolic state may determine whether ODN-sensitive or -insensitive circuitries control FFA availability in the latter sex. Male rats showed no change in plasma glucose levels one hour after administration of the ODN GPCR antagonist LV-1075 and slight exacerbation of hypoglycemia in response to the ODN agonist OP, despite documented contrary effects of these treatments on counter-regulatory hormone levels at the same post-injection interval. Since circulating glucose profiles were not measured between drug administration and sacrifice, the possibility that one or both drugs may have altered blood glucose over some point in time prior to + 1 h cannot be discounted. Indeed, 2013] report that the duration of icv LV-1075 – induced hyperglycemia is less than 60 min. It is plausible that after central LV-1075 or OP delivery, plasma glucose profiles may exhibit a dynamic flux owing to modulation of icv drug-associated patterns of counter-regulatory hormone release and/or induction of counter-active hepatic gluconeogenic or glycogenolytic functions by glucose and energy sensor readouts. Current data may thus provide a snapshot of a temporal phase during which plasma glucose levels are normalized or nearly normalized after LV-1075 or OP treatment, respectively, as an adaptive reaction to central actions of these drugs. In any case, measurement of glycemic profiles at only a single post-treatment time point remains a limitation of this study.
Endozepines are widely expressed over the interconnected network of hypothalamic loci that govern systemic metabolic balance [Alho et al., 1985; Malagon et al., 1993; Tonon et al., 1990]. The current strategy of icv drug administration does not preclude the possibility that, in one or both sexes, observed drug treatment effects on VMN nerve cell target protein expression or plasma hormone profiles may reflect, in part, adjustments in ODN GPCR signaling to cell substrates present within as well as outside the VMN. It will be necessary for future research to ascertain, for each sex (1) the neuroanatomical distribution of hypothalamic DBI-secreting astrocytes that regulate glucose homeostasis; (2) the neurochemical phenotype(s) of afferent projections to the VMN that may convey this regulatory stimulus to local glucose-regulatory NO and GABA neurons; (3) to characterize the metabolic cues that govern astrocyte DBI release under conditions of glucose sufficiency versus deficiency. The presumption that VMN neurotransmitter activity governing hypoglycemic hyperglucagonemia and hypercorticosteronemia is controlled by ODN GPCR signaling within that nucleus will require further work involving stereotactic delivery of pharmacological or genetic reagents directly to the VMN.
In summary, present outcomes document the neuroanatomical distribution of ODN-sensitive nitrergic and GABAergic neurons within the rat VMN. Results provide novel proof of segment-specific endozepinergic regulation of basal and hypoglycemic patterns of NO transmission in each sex, and suggest that mechanisms of this control may involve AMPK activation state. Data also show that VMN GABAergic transmission is liable to ODN control in the female only. Gain- or loss-of-effect of ODN control of gluco-regulatory marker protein expression infers that metabolic state-specific cues that may govern nitrergic and GABAergic nerve cell reactivity to this gliopeptide. Endozepinergic signaling evidently affects neural pathways controlling counter-regulatory hormone release in the male, whereas ODN-dependent and -independent mechanisms may operate in parallel to regulate this outflow in the female.
Supplemental Material
sj-docx-1-asn-10.1177_17590914231167230 - Supplemental material for Sex-Dimorphic Octadecaneuropeptide (ODN) Regulation of Ventromedial Hypothalamic Nucleus Glucoregulatory Neuron Function and Counterregulatory Hormone Secretion
Supplemental material, sj-docx-1-asn-10.1177_17590914231167230 for Sex-Dimorphic Octadecaneuropeptide (ODN) Regulation of Ventromedial Hypothalamic Nucleus Glucoregulatory Neuron Function and Counterregulatory Hormone Secretion by Karen P. Briski, Prabhat R. Napit, Abdulrahman Alhamyani, Jérôme Leprince and A.S.M. Hasan Mahmood in ASN Neuro
Supplemental Material
sj-docx-2-asn-10.1177_17590914231167230 - Supplemental material for Sex-Dimorphic Octadecaneuropeptide (ODN) Regulation of Ventromedial Hypothalamic Nucleus Glucoregulatory Neuron Function and Counterregulatory Hormone Secretion
Supplemental material, sj-docx-2-asn-10.1177_17590914231167230 for Sex-Dimorphic Octadecaneuropeptide (ODN) Regulation of Ventromedial Hypothalamic Nucleus Glucoregulatory Neuron Function and Counterregulatory Hormone Secretion by Karen P. Briski, Prabhat R. Napit, Abdulrahman Alhamyani, Jérôme Leprince and A.S.M. Hasan Mahmood in ASN Neuro
Supplemental Material
sj-docx-3-asn-10.1177_17590914231167230 - Supplemental material for Sex-Dimorphic Octadecaneuropeptide (ODN) Regulation of Ventromedial Hypothalamic Nucleus Glucoregulatory Neuron Function and Counterregulatory Hormone Secretion
Supplemental material, sj-docx-3-asn-10.1177_17590914231167230 for Sex-Dimorphic Octadecaneuropeptide (ODN) Regulation of Ventromedial Hypothalamic Nucleus Glucoregulatory Neuron Function and Counterregulatory Hormone Secretion by Karen P. Briski, Prabhat R. Napit, Abdulrahman Alhamyani, Jérôme Leprince and A.S.M. Hasan Mahmood in ASN Neuro
Footnotes
Abbreviations
Acknowledgements
The authors wish to express their sincere thanks and appreciation to Dr. Jean-Denis Troadec, Aix-Marseille University, UMR 7291 CNRS, Cognitive Neurosciences Laboratory, Marseilles, France, for his helpful comments and suggestions on manuscript revisions.
Author's Note
Abdulrahman Alhamyani is also affiliated at Pharmaceuticals Chemistry Department, Faculty of Clinical Pharmacy, Al Baha University, Saudi Arabia.
Availability of Data and Material
All data generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code Availability
Not applicable.
Consent for Publication
(include appropriate statements)
Consent to Participate
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
All animal experimental was carried out in compliance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals, 8th Edition. Sex of animals used is included, along with discussion of sex impacts on study outcomes.
Funding
National Institutes of Health DK-109382
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
