Abstract
The objective of this study is to investigate the effect of long noncoding RNA (lncRNA) XIST on postoperative pain and inflammation of plantar incision pain (PIP) in rats and its underlying mechanisms.
PIP rat models were established by plantar incision. Rats in the sham group were subjected to povidone-iodine scrubbing, and no incision was made. To explore the role of XIST/miR-340-5p/RAB1A in postoperative pain and inflammation, PIP rats were separately or simultaneously injected with lentivirus containing sh-NC, sh-XIST, mimic NC, miR-340-5p mimic, inhibitor NC, miR-340-5p inhibitor, pcDNA3.1, or pcDNA3.1-RAB1A through an intrathecal catheter. The paw withdrawal threshold (PWT) and paw withdrawal latency (PWL) values of rats in each group were assessed to evaluate the pain behavior. RT-qPCR and Western blot were utilized to determine the levels of XIST, miR-340-5p, RAB1A, and NF-κB pathway-related proteins (p-IκBα, IκBα, p-p65, and p65). The concentrations of inflammatory cytokines (TNF-α, IL-1β, and IL-6) in rat spinal dorsal horn tissues were inspected by ELISA. H and E staining was applied to observe the pathological changes of neurons in the spinal dorsal horn, TUNEL staining to detect neuronal apoptosis, and immunohistochemistry to measure RAB1A level.
Plantar incision surgery caused decreased PWT and PWL values, enhanced levels of XIST, RAB1A, and inflammatory cytokines, along with an increased proportion of apoptotic neurons. The pain sensitivity and inflammation of rats were motivated after plantar incision surgery. Intrathecal injection of sh-XIST or miR-340-5p mimic ameliorated the pain and inflammation of PIP rats, while silencing of miR-340-5p or overexpression of RAB1A partly reversed the effect of sh-XIST on PIP rats. XIST targeted miR-340-5p and miR-340-5p negatively regulated RAB1A. The XIST/miR-340-5p/RAB1A axis activated the NF-κB signaling pathway.
LncRNA XIST aggravates inflammatory response and postoperative pain of PIP rats by activating the NF-κB pathway via the miR-340-5p/RAB1A axis.
Introduction
Surgery is a first-line therapy choice for many diseases and can lead to numerous complications (Xu et al., 2020). Postoperative pain is a severe acute pain that occurs in patients undergoing a surgery, and there are no adequate analgesics to control the pain (Xu et al., 2019). Although analgesic technologies, such as advanced and multimodal analgesia, have been improving, 39% to 50% of surgical patients still experience moderate to severe acute postoperative pain, which severely affects their recovery (Brennan, 2011). Therefore, investigating the underlying mechanisms of incisional pain and its mediators would facilitate the discovery of novel therapies. Inflammation resulting from surgical injury may contribute to the sensitization of peripheral nociceptors, which may further give rise to hyperalgesia and allodynia (Chen et al., 2018). Furthermore, postoperative pain is proven to be exacerbated by proinflammatory cytokines such as IL-1β, IL-6, and TNF-α, and these factors are implicated in the hyperalgesia and pain mechanism (Kim et al., 2018). Therefore, further elucidation of the interaction among inflammation, postoperative pain, and plantar incision pain (PIP) is warranted to reveal novel therapeutic targets.
Long noncoding RNA (lncRNA), a class of oligonucleotides longer than 200 nucleotides, bears no protein-coding potentials (Ma et al., 2020; Wang et al., 2020). Increasing evidence indicated that lncRNAs exert crucial functions in gene regulation and biological process, such as inflammation. For example, neuropathic pain and neuroinflammation can be aggravated by lncRNA MALAT1 by regulating the miR-129-5p/HMGB1 axis (Ma et al., 2020). X inactivate-specific transcript (XIST), located on the long (q) arm of the X chromosome, contributes to neuropathic pain progression in rats (Sun et al., 2018; Wei et al., 2018). More important, XIST is validated to be relative to the inflammatory response of bovine mammary epithelial cells by linking the NF-κB/NLRP3 inflammasome pathway (Ma et al., 2019). However, the role of XIST in postoperative pain remains poorly elucidated. MicroRNA (miRNA) is a small noncoding RNA of approximately 22 nucleotides that serves as a posttranscriptional regulatory factor of its messenger RNA (mRNA) target (Catalanotto et al., 2016; Xu et al., 2018). Besides, miRNAs are reported to involve in various neuropathological alterations (Peng et al., 2017). A previous study clarified that miR-340-5p relieves chronic constriction injury (CCI)-induced neuropathic pain by targeting RAP1A in rat models (Gao et al., 2019). Additionally, excess administration of miR-340-5p attenuates the spinal cord injury-induced neuroinflammation of rats (Qian et al., 2020). It is well known that lncRNAs can function as competitive endogenous RNAs (ceRNAs) to modulate disease development by sponging miRNAs and targeting mRNAs (Zhao et al., 2020). Herein, we attempted to identify lncRNA-miRNA-mRNA pair, which might interact with each other, thus affecting postoperative pain. In our current study, we identified miR-340-5p as a potential target gene of XIST and an upstream factor of RAB1A. RAB1A is a member of the RAB family (Zhang et al., 2019) and has been previously reported to promote neuroinflammation through activation of the NF-κB pathway (Song et al., 2020). The miR-340/RAB27B axis is actively involved in the occurrence of nonsmall cell lung cancer (Takahashi et al., 2020). In consideration of the potential relationship, we arranged this study to explore the interaction among XIST/miR-340-5p/RAB1A, postoperative pain, and inflammatory response in a rat model following plantar incision.
The aim of this study was to examine the regulatory relationship between XIST, miR-340-5p, and RAB1A in order to improve the current understanding of the mechanisms underlying postoperative pain. The present study demonstrated that XIST may intensify postoperative pain in PIP rats by activating the NF-κB pathway via the miR-340-5p/RAB1A axis. These findings provided a novel experimental basis for targeted therapy for incisional pain from the aspect of lncRNA-miRNA-mRNA interaction.
Materials and Methods
Ethical Statement
The design of the experiments of this study was approved by the Ethical Committee of Third Xiangya Hospital, Central South University (No: 2020sydw0630). All experiments in this study were conducted based on the Ethical Guidelines for Animal Experiments of Third Xiangya Hospital, Central South University.
Experimental Animals
Healthy Sprague Dawley (SD) rats were provided by the Shanghai laboratory animal center of the Chinese Academy of Sciences (Shanghai, China), with a total of 50 male rats weighing 193.2 ± 14.6 g. Rats were housed in a laboratory with humidity of 55 ± 5%, and room temperature of 24 ± 1°C using a 12 h light-dark cycle (light: 6 am ∼ 6 pm; dark: 6 pm ∼ 6 am) to adapt to the environment for at least one week. Then, they were randomly assigned into ten groups with five rats in each group: Sham, PIP (plantar incision pain), PIP + sh-NC, PIP + sh-XIST, PIP + mimic NC, PIP + miR-340-5p mimic, PIP + sh-XIST + inhibitor NC, PIP + sh-XIST + miR-340-5p inhibitor, PIP + sh-XIST + pcDNA3.1, and PIP + sh-XIST + pcDNA3.1-RAB1A groups.
Planter Incision
An animal model of postoperative pain was generated by plantar incision as stated in a previous study (Xing et al., 2017). Briefly, rats were anesthetized with 100 mg/kg ketamine and 12 mg/kg xylazine, and the plantar surface of the left hind paw was scrubbed thrice with 10% povidone-iodine. A 1 cm incision was made in the plantar aspect of the left hind paw beginning 0.5 cm from the proximal edge of the heel and extending toward the toes, and skin, fascia, and muscle were incised. The plantaris muscle was isolated, elevated slightly, incised longitudinally, and then put back to its original position. The exposed incision site of the plantaris muscle was desiccated and scrubbed with sterile gauze until hemorrhage was stopped. Then, the skin was opposed with suture using 2-0 nylon. The animals were allowed to return to their home cages after recovering from anesthesia. The incisions were examined daily, and rats with wound infection or dehiscence were excluded from the study. Rats in the sham group were subjected to povidone-iodine scrubbing and no incision was made.
Intrathecal Catheter Implantation and Intrathecal Injection
Animals under ketamine (100 mg/kg) and xylazine (12 mg/kg) anesthesia were implanted with an intrathecal catheter using the method as Yaksh and Rudy described (Yaksh & Rudy, 1976). Briefly, about a 2 cm long midline incision was made in the skin on the midline between the lower margins of both ears (lower margin of occipital prominences), and the subcutaneous tissues were separated to expose the atlanto-occipital membrane. A 1 ∼ 2 mm incision was made on the membrane, and a polyethylene-10 intrathecal catheter (BD, Franklin Lake, NJ) was then placed caudally at approximately 3.3 cm to the T4 spinal cord. The correct intrathecal localization of the catheter was confirmed by the backflow of spinal fluid. The incision was subsequently sutured, with the catheter finally opening cephalic and firmly fixed. The catheter outside of the skin was retained about 3 cm, and the catheter orifice was closed by the application of heat. All rats were fed in single cages after catheterization. To further confirm whether the intrathecal catheter was located in the subarachnoid space of rats, they were monitored for any neurologic deficit (nerve injury or paralysis) on the 7th day after catheterization, and any showing signs of a neurologic deficit were removed from the study.
The intrathecal injection was given once a day, three days before the plantar incision. Rats were separately or simultaneously subjected to ten microliters lentivirus containing sh-NC, sh-XIST, mimic NC, miR-340-5p mimic, inhibitor NC, miR-340-5p inhibitor, pcDNA3.1, or pcDNA3.1-RAB1A (RiboBio Co., Ltd, Guangzhou, China) (lentivirus titer of 5 × 108 TU/ml) with microsyringe through the intrathecal catheter.
Cell Culture
Human embryonic kidney 293 cells (HEK293 T) were supplied by American Type Culture Collection (ATCC). Cells were immersed in Dulbecco's modified Eagle medium (DMEM, Thermo Fisher Scientific, MA, USA) covering 10% fetal bovine serum and 1% penicillin/streptomycin (Thermo Fisher Scientific) for incubation at 37°C with 5% CO2.
Behavioral Tests
The rats were tested for paw withdrawal threshold (PWT) and paw withdrawal latency (PWL) before plantar incision, and at days 1, 3, 7, 14, and 21, following the plantar incision. The method was as follows: Rats were placed solely in wire cages for 30 min in a quiet environment. Then, those adapted to the environment and the electronic Von Frey probe (IITC, Woodland Hills, CA, USA) was used to stimulate the soles of their feet three times at an interval of 10 min through the wire cages, and intensity was gradually increased. The pressure values displayed on the Von Frey electronic recorder were recorded when the avoidance reflex occurred in rats, and then the average of the three readings was regarded as the PWT of rats. PWL was regarded as the mean of three readings from the stimulus initiation time to the time of paw retraction in rats. After the pain behavior assessment on the 21st day, the rats were anesthetized with ketamine (100 mg/kg) and xylazine (12 mg/kg), and sacrificed by cervical dislocation. The spinal dorsal horn from L4 to L5 was isolated for follow-up studies.
RT-qPCR
TRIzol (Invitrogen, Carlsbad, CA, USA) was applied to extract the total RNA of the spinal dorsal horn from L4 to L5, and the cDNA template was synthesized by reverse transcription kit. The PCR reaction system was configured by using the fluorescence quantitative PCR kit (Takara, Dalian, China). The RT-qPCR assay was embarked on the ABI7500 real-time PCR instrument (Applied Biosystems, Shanghai, China). Following 10 min of predenaturation at 95°C, cDNA underwent 40 cycles of 10-s denaturing step at 95°C, 20-s annealing step at 60°C, and 34-s extension at 72°C. Subsequently, the expressions of XIST, miR-340-5p, and RAB1A were quantitated. The primer sequences are exhibited in
Primer Sequence Information.
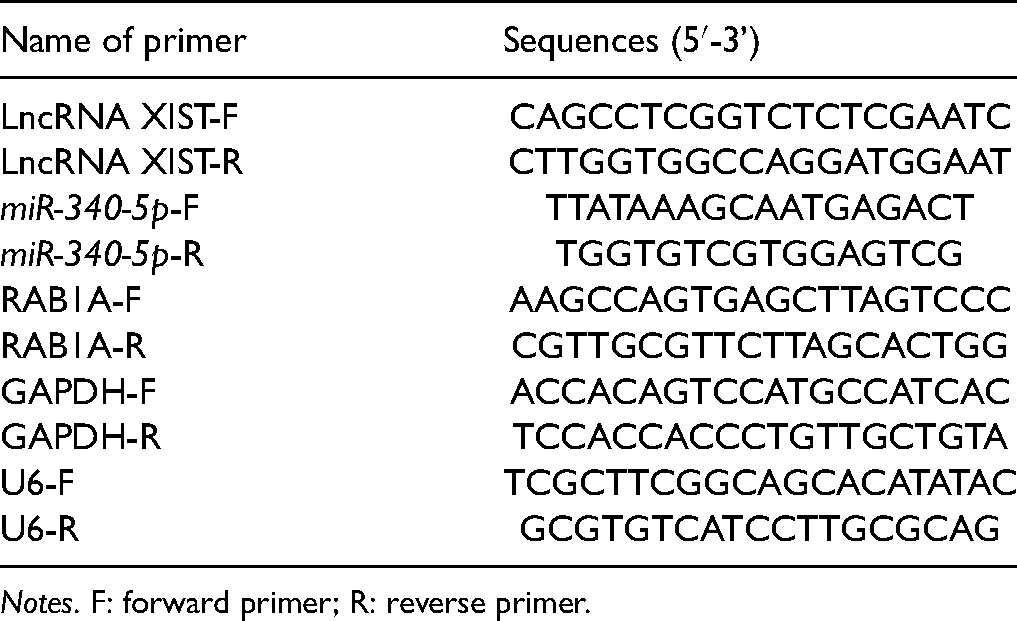
Notes. F: forward primer; R: reverse primer.
Western Blot
The tissue homogenate of the spinal dorsal horn in L4-L5 was obtained and washed thrice with precooled phosphate-buffered saline (PBS). Cells were lysed with RIPA cell lysis and subjected to 30 min of standing on ice at 4°C. After 10 min of centrifugation at 12,000 rpm at 4°C, the supernatant was aspirated to 0.5 mL centrifuge tubes for storage under −20°C or protein quantification using a BCA kit (Sigma, MO, USA). The proteins were then exposed to 6 × SDS loading buffer at 100°C for denaturation before SDS-PAGE electrophoresis for separation. The proteins were transferred onto membranes using a 4°C precooled transfer buffer for 1.5 h. The membranes were inactivated by 5% non fat dry milk-TBST for 1 h followed by incubation with TBST-diluted primary antibodies against rabbit-derived GAPDH (1:10000, ab181602), RAB1A (1:2000, ab228354), p-IκBα (ab133462, 1:1000), IκBα (ab32518, 1:1000), p-p65 (ab86299, 1:2000), and p65 (ab16502, 1:2000) (Abcam, MA, USA) overnight at 4°C. Before 2 h of incubation with the secondary antibody against goat anti-rabbit IgG or goat anti-mouse IgG (1:5000, Beijing ComWin Biotech Co., Ltd., Beijing, China), the membranes were washed with TBST for 3 × 10 min. The membrane was subjected to color development after another round of TBST washing. After the proteins were incubated with ECL chemiluminescence solution, the membranes were observed under the chemiluminescence imaging analysis system (GE Healthcare, Beijing, China).
ELISA
The levels of TNF-α, IL-1β, and IL-6 in the spinal dorsal horn from L4 to L5 were determined by ELISA. The tissue homogenate of the spinal dorsal horn was prepared, and the absorbance was measured according to the instructions of the kit (MultiSciences (Lianke) Biotech Co., Ltd., Hangzhou, China). The standard curve was drawn according to the concentration of the standard substance and its absorbance value. The concentration of each sample was calculated according to the optical density (OD) value of the specimen (subtracted from the OD value of the background calibration hole) and the standard curve.
Hematoxylin and Eosin Staining
Rats were anesthetized with ketamine (90 ∼ 150 mg/kg) and xylazine (7.5 ∼ 16 mg/kg) and then sacrificed by cervical dislocation. The tissues of the spinal dorsal horn in L4-L5 were extracted to prepare 4 μm paraffin sections. Histopathological changes of spinal cord tissues were observed under the microscope after H and E staining. H and E staining was carried out in strict accordance with the instructions of the H and E staining kit (Beyotime Biotechnology Co., Ltd., Shanghai, China). Briefly, the paraffin sections were deparaffinized by exposure to xylene and then orderly underwent gradient ethanol hydration. Following 5 min of hematoxylin staining, the paraffin sections were subjected to 30 s of differentiation with hydrochloric acid ethanol, 15 min of immersion in tap water, and 3 min of eosin staining. Pictures were captured under a microscope after sections were given dehydration, permeabilization, and seal.
TUNEL Staining
The paraffin sections were dewaxed, hydrated, and permeabilized. Tissues were digested with 20 mg/L proteinase K for 15 min at room temperature and reacted with 500 μL TUNEL reaction solution (a mixture of 50 μL TdT solution and 450 μL fluorescein-labeled dUTP solution) for 1 h at room temperature in a wet box. This process of staining was conducted strictly according to the instructions of the TUNEL kit (In Situ Cell Death Detection Kits, Roche, Indianapolis, IN, USA). Then, cells were incubated with a 50 μL converter-POD for 30 min and exposed to 100 μL DAB substrate for 10 min. After being counterstained with hematoxylin, sections were observed under an optical microscope. The apoptotic rate was calculated. Apoptotic rate = number of apoptotic nucleus /total number of the nucleus.
Immunohistochemistry
Sections were baked for 20 min, dewaxed with conventional xylene, and washed with distilled water. After three times of PBS washing, they were treated with 3% H2O2 for 10 min, washed thrice with PBS, and subjected to antigen retrieval, and three times of PBS wash. Then, the sections were blocked with normal goat serum for 20 min. After removal of excess liquid, they were incubated with primary antibody against RAB1A (1:500, ab192446) overnight at 4°C, washed thrice with PBS, and treated with the secondary antibody for 1 h at room temperature. Subsequently, the sections were exposed to DAB for 1-to 3-min color development. This progress was then terminated by three times of PBS washing. Following 3 min of hematoxylin staining, they were dehydrated, permeated, and sealed.
Dual-Luciferase Reporter Assay
The binding site of miR-340-5p and lncRNA XIST or RAB1A was predicted by online prediction software StarBase. The mutated and wild-type sequences in the binding sites were designed and cloned into a pmirGLO vector, subsequently renamed as MUT-XIST, WT-XIST, MUT-RAB1A, and WT-RAB1A. Then the vectors were cotransfected with MUT-XIST, WT-XIST, MUT-RAB1A, or WT-RAB1A and miR-340-5p mimic (50 nM, GenePharma) or miR-340-5p inhibitor (50 nM, GenePharma), respectively, into HEK-239 T cells. Cells were incubated in constant temperature DMEM at 37°C with 5% CO2 for 48 h, and the fluorescence intensity of cells in each group was assessed by a dual-luciferase reporter kit (Promega, WI, USA).
Statistical Analysis
Statistical analysis of data was performed by SPSS 20.0 (IBM Corp., Armonk, NY, USA) and GraphPad Prism 6.0 (GraphPad Software Inc.). Data were displayed as mean ± standard deviation (SD). Mann–Whitney U-test was adopted for the comparison between two groups. Comparisons among multiple groups were analyzed by Kruskal–Wallis test. P < .05 was considered statistically significant.
Results
High Expression of XIST and RAB1A and Low Expression of miR-340-5p in PIP Rat Models
PWT and PWL were determined before plantar incision surgery and on days 1, 3, 7, 14, and 21 after surgery, and the sham group was used as the control group. Compared with the sham group, the PIP group had more sensitive postoperative pain (
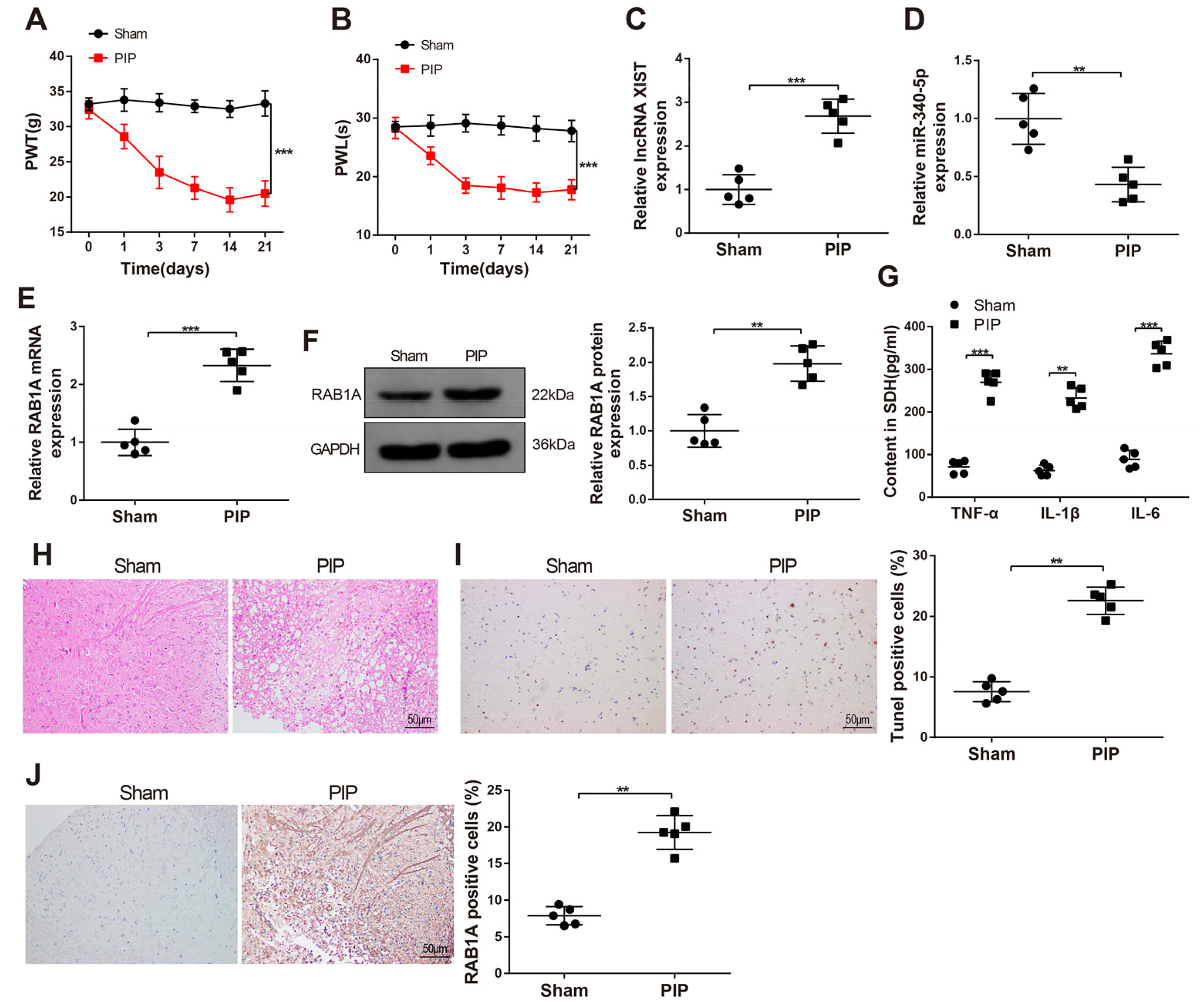
Activated Postoperative Pain Sensitivity and Inflammation in PIP Rat Models
Results of ELISA addressed the rises in the levels of inflammatory cytokines (IL-6, TNF-α, and IL-1β) in the spinal dorsal horn of the PIP group (
Inhibition of XIST Ameliorates the Postoperative Pain and Inflammation of PIP Rat Models
To investigate the effect of lncRNA XIST on pain and inflammation in PIP rats, sh-NC or sh-XIST was intrathecally injected into PIP rat models. RT-qPCR detection displayed that the PIP + sh-XIST group possessed lower XIST expression than the PIP + sh-NC group (
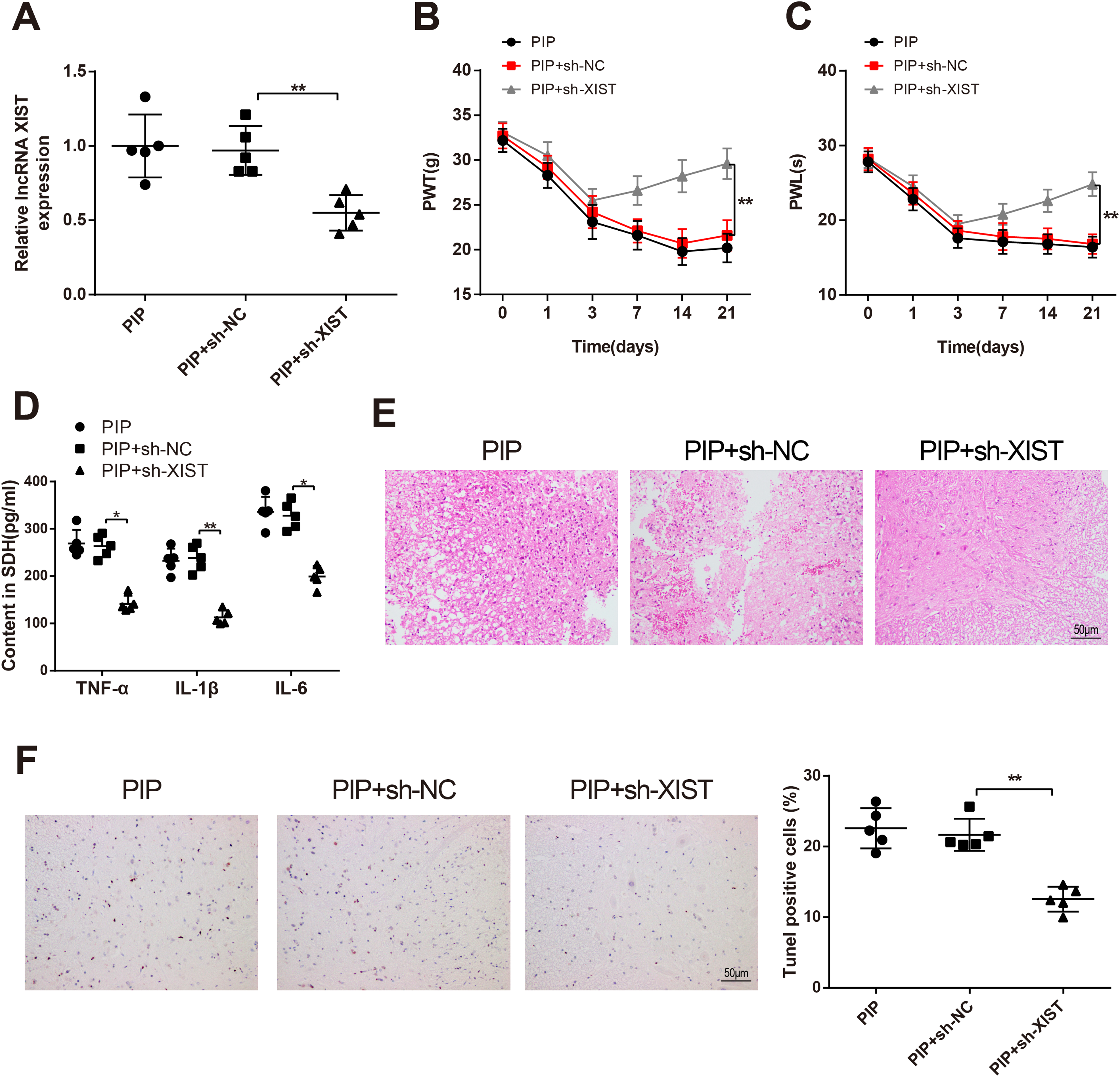
The Postoperative Pain and Inflammatory Response of PIP Rat Models Can be Mitigated by lncRNA XIST
The Pain and Inflammation of PIP Rat Models is Mitigated by miR-340-5p Mimic
PIP rats were given mimic NC or miR-340-5p mimic by intrathecal injection to explore the role of miR-340-5p in the postoperative pain of PIP rats. Then, the efficiency of miR-340-5p mimic was examined by RT-qPCR. We found that intrathecal injection of miR-340-5p mimic dramatically elevated miR-340-5p level in PIP rats (
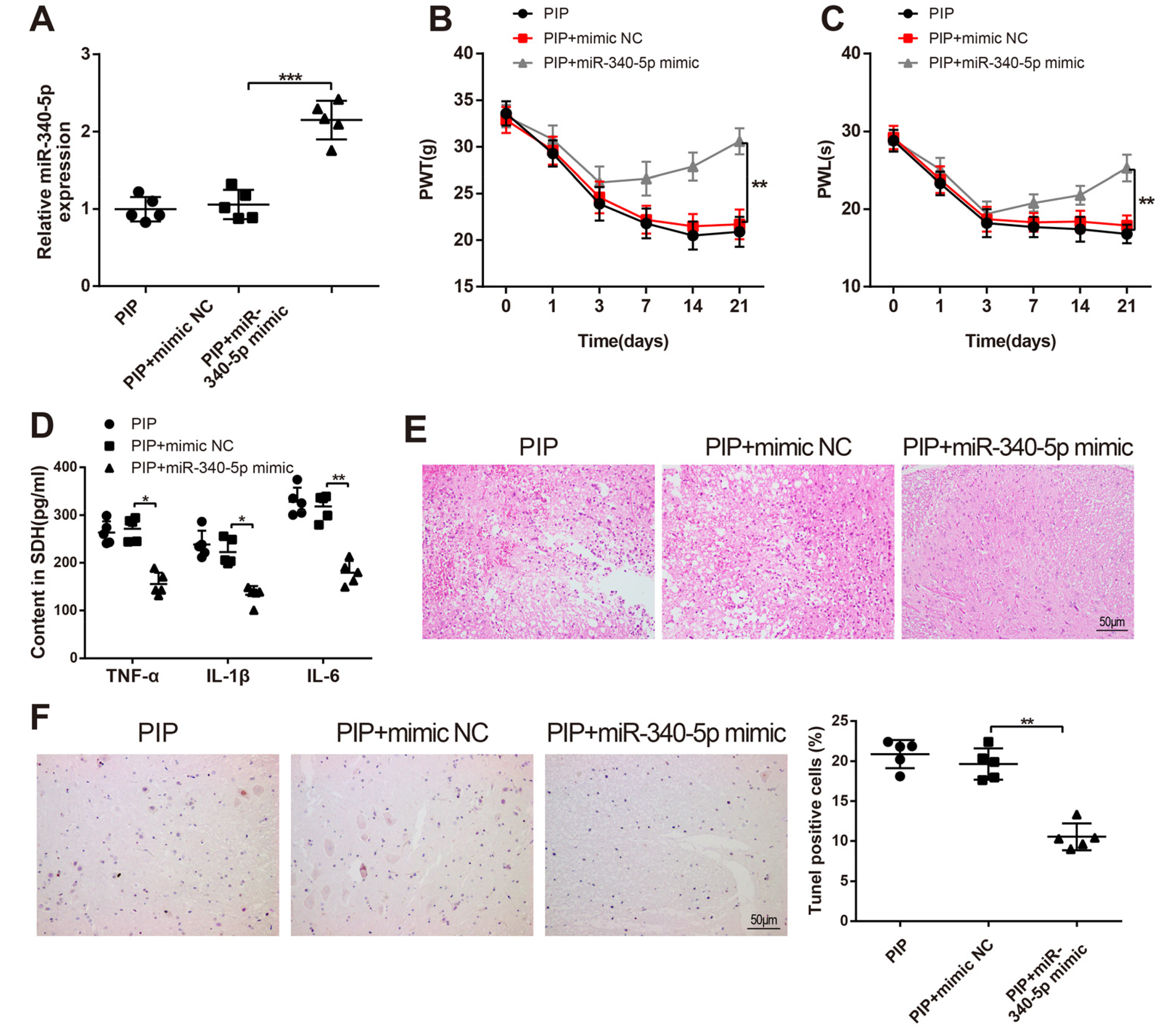
Overexpression of miR-340-5p Relieves the Postoperative Pain and Inflammatory Response of PIP Rat Models
The Negative Relationship Between XIST and miR-340-5p, as well as miR-340-5p and RAB1A
According to the regulatory mechanism of ceRNA, we speculated that XIST may mediate the expression of RAB1A through a miRNA miR-340-5p. Subsequently, the StarBase database was employed to predict the relationship between miR-340-5p and XIST, as well as miR-340-5p and RAB1A. We found that miR-340-5p may be a potential target of XIST, and there was also a binding site of miR-340-5p and RAB1A. Therefore, we further speculated that XIST regulates RAB1A by regulating the expression of miR-340-5p, thereby playing a crucial role in PIP rats.
To confirm our conjecture, PIP rat models were intrathecally injected with sh-NC, sh-XIST, mimic NC, or miR-340-5p mimic. The RT-qPCR results manifested the progressive increase in miR-340-5p expression in PIP rats treated with sh-XIST (
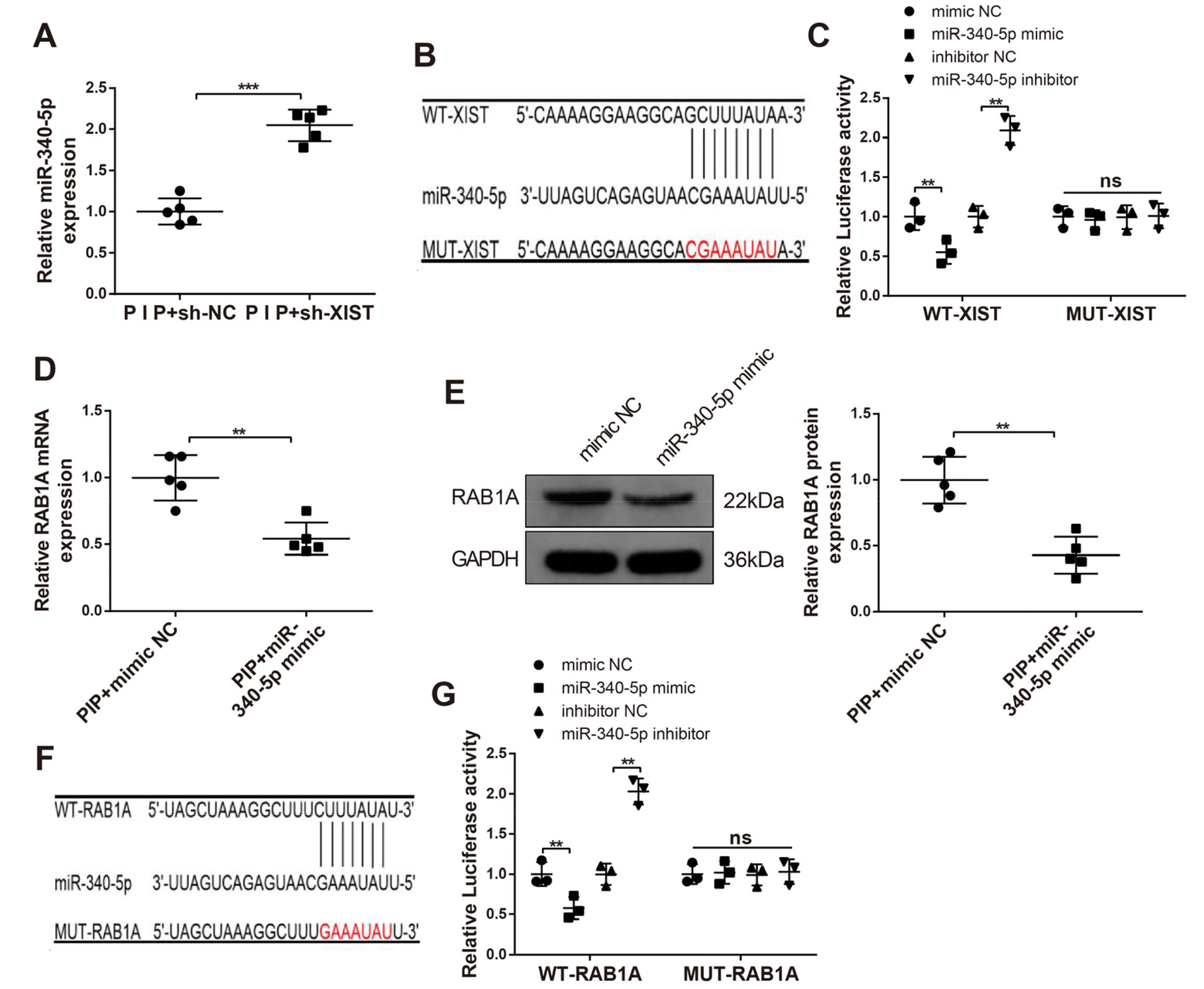
XIST Targets miR-340-5p, and miR-340-5p Negatively Regulates RAB1A
Additionally, the analyses of RT-qPCR and Western blot expounded that the PIP + miR-340-5p mimic group had diminished mRNA and protein expressions of RAB1A (
LncRNA XIST Intensifies the Postoperative Pain of PIP Rat Models via the miR-340-5p/RAB1A Axis
To investigate whether XIST could influence PIP rats by regulating the miR-340-5p/RAB1A axis, PIP rat models were solely or simultaneously injected with sh-NC, sh-XIST, inhibitor NC, miR-340-5p inhibitor, pcDNA3.1, or pcDNA3.1-RAB1A. The results of RT-qPCR and Western blot exhibited that treatment with sh-XIST decreased the level of RAB1A in PIP rat models (P < .01, the PIP + sh-XIST group vs. the PIP + sh-NC group), while the following injection with miR-340-5p inhibitor (the PIP + sh-XIST + miR-340-5p inhibitor group vs. the PIP + sh-XIST + inhibitor NC group), or pcDNA3.1-RAB1A reinforced RAB1A expression (
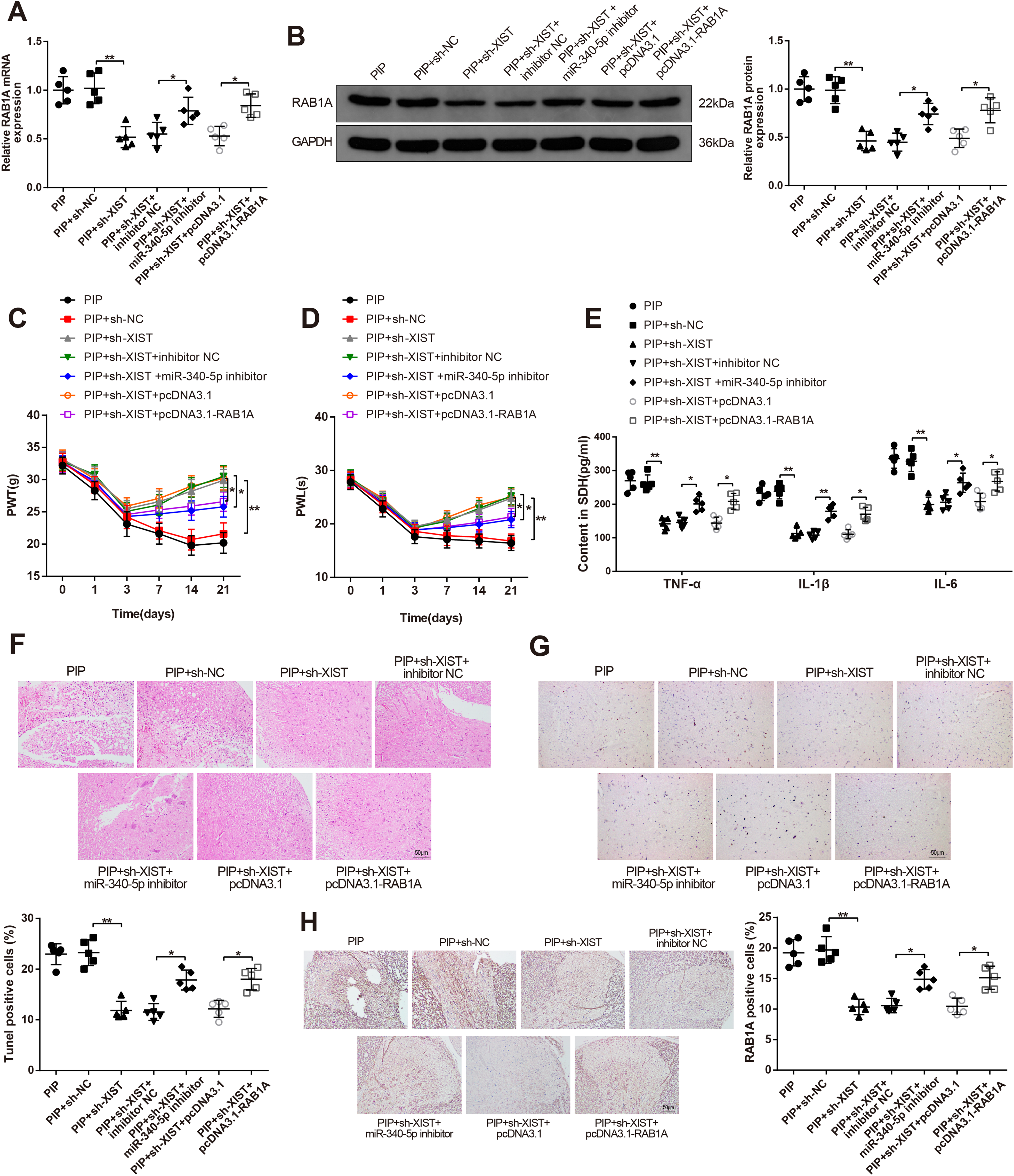
LncRNA XIST Regulates the miR-340-5p/RAB1A Axis to Aggravate the Inflammatory Response and Postoperative Pain of PIP Rat Models
In addition, compared with the PIP + sh-NC group, the PIP + sh-XIST group possessed higher values of PWT and PWL (
H and E staining observed that the neurons in the PIP + sh-NC group were obviously swollen, disordered, and widely loose. However, the neurons in the PIP + sh-XIST group were disordered, and neuronal swelling was reduced (
TUNEL staining results addressed that more TUNEL-positive cells were noticed in the PIP + sh-NC group (
Then, the expression of RAB1A in spinal dorsal horn neurons was measured by immunohistochemistry. The results displayed that injection with sh-XIST repressed the level of RAB1A in PIP rat models (P < .01, the PIP + sh-XIST group vs. the PIP + sh-NC group), while the following exposure to miR-340-5p inhibitor (the PIP + sh-XIST + miR-340-5p inhibitor group vs. the PIP + sh-XIST + inhibitor NC group) or pcDNA3.1-RAB1A heightened RAB1A expression (
XIST Modulates the NF-κB Signaling Pathway via miR-340-5p/RAB1A Axis
To further study whether NF-κB could be the downstream signaling pathway of XIST/miR-340-5p/RAB1A axis, PIP rats were solely or simultaneously injected with sh-NC, sh-XIST, inhibitor NC, miR-340-5p inhibitor, pcDNA3.1, pcDNA3.1-RAB1A, and then the expressions of NF-κB signaling pathway-related proteins (p-IκBα, IκBα, p-p65, and p65) were measured. Western blot presented that rats in the PIP group had higher expressions of p-IκBα and p-p65 than rats in the sham group (P < .01). In addition, intrathecal injection of sh-XIST inhibited the protein levels of NF-κB pathway-related proteins (
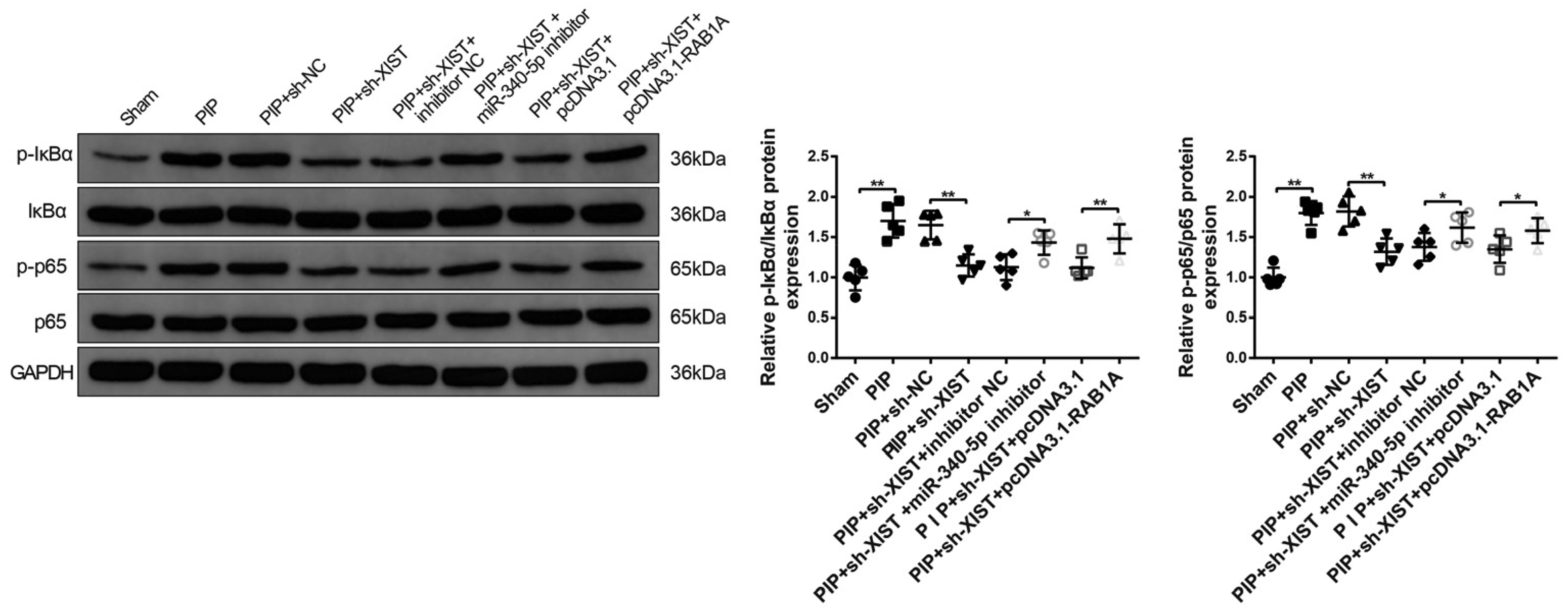
The XIST/miR-340-5p/RAB1A Axis Activates the NF-κB Signaling Pathway
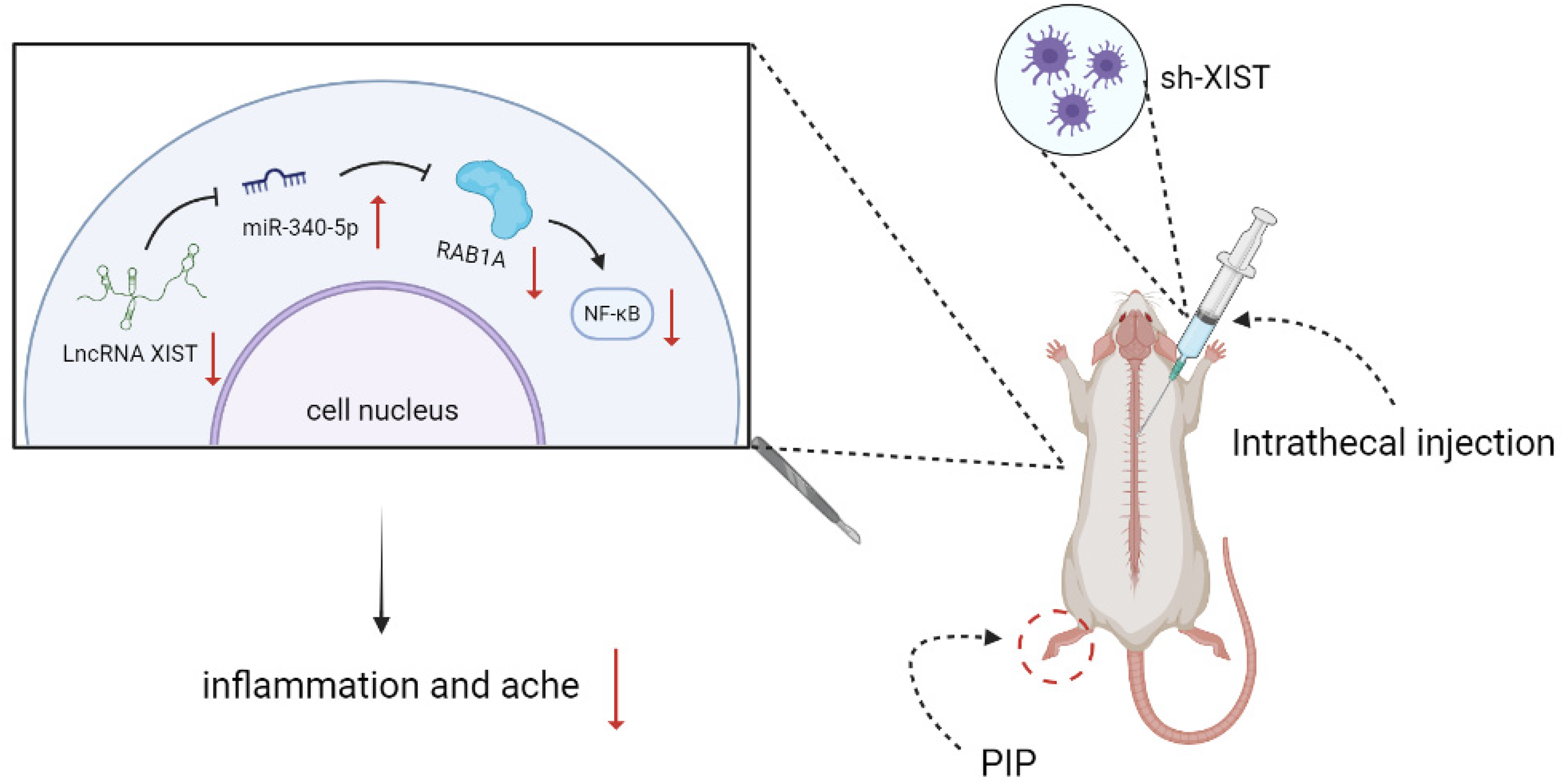
The Mechanism Diagram of LncRNA XIST/miR-340-5p/RAB1A Axis Activating NF-κB Signal Pathway in Enhancing Inflammatory Response and Postoperative Pain of PIP Rat Models
Discussion
Patients undergoing surgical operation have clinical postoperative pain even when adjuvant treatments were optimized (Brennan, 2011). Therefore, appropriate postoperative pain therapy still remains a challenge. The animal model of postoperative pain established by making an incision at the plantar hind paw has similarities to the time course for pain in postoperative patients (Xing et al., 2018). Toward this end, the PIP model in the rat hind paw was used in our study to investigate the role of the XIST/miR-340-5p/RAB1A axis in the course of postoperative pain and to identify the novel therapy target.
Postoperative pain is also influenced by an inflammatory response in local injured tissue and the spinal cord (Brennan et al., 1996). In the present study, plantar incision led to enhanced levels of TNF-α, IL-6, and IL-1β, increased number of apoptotic cells in tissues of the spinal dorsal horn, and decreased values of PWT and PWL. Our data indicated that postoperative pain sensitivity and inflammation may be activated in the PIP rat models. XIST is a lncRNA that mediates the silencing of gene transcription on the X chromosome (Patil et al., 2016). In this study, we analyzed the behavior of lncRNA XIST in the tissues of the spinal dorsal horn. Initially, we observed that PIP rats had higher XIST expression than rats that did not undergo surgery. XIST was therefore speculated to be related to postoperative pain. Subsequently, loss-of-function for XIST proved that deficiency of XIST attenuated the inflammation and postoperative pain of rats following plantar incision. Of note, there has been convincing evidence supporting the role of XIST in neuropathic pain. A study conducted by Yan et al., expounded that XIST can induce neuropathic pain through the upregulation of miR-150 and ZEB1 (Yan et al., 2018). Downregulation of XIST represses the neuroinflammation of CCI rat models (Jin et al., 2018).
LncRNAs can serve as endogenous RNA and sponge miRNAs to alter their target gene expression at the posttranscriptional level (Chen et al., 2018). As such, we are prompted to further look into the molecular actions of XIST in regulating postoperative pain from the lncRNA-miRNA-mRNA aspect. Herein, we identified miR-340-5p as a direct target of XIST by using StarBase software and dual-luciferase reporter assay. Moreover, lowly expressed miR-340-5p was found in PIP rats. Results of RT-qPCR addressed that XIST negatively mediated miR-340-5p expression in the tissues of the spinal dorsal horn. Toward this end, the potential of miR-340-5p in postoperative pain of PIP rats was explored by intrathecal injection of miR-340-5p mimic. More interestingly, the findings of behavior test, ELISA, H and E staining, and TUNEL staining demonstrated that the overexpression of miR-340-5p relieved the postoperative pain of PIP rats. Consistent with our findings, a previous study also uncovered the performance of miR-340-5p in neuropathic pain (Gao et al., 2019). Simultaneously, we found high expression of RAB1A in rats that had undergone plantar incision surgery. RAB proteins modulate membrane transport and impact cell pathways by binding to effector proteins (Li et al., 2017). RAB1A, a member of the RAB family, is closely related to inflammation response (Zhang et al., 2019). In the current study, the targeting relationship between miR-340-5p and RAB1A was also identified. Considering the regulatory mechanism of ceRNA, we speculated that XIST may mediate RAB1A by regulating the expression of miR-340-5p, thereby playing a crucial role in PIP rats. Then, our conjecture was confirmed by findings that silencing of miR-340-5p or the upregulation of RAB1A partly reversed the role of sh-XIST in PIP rats. Our study proposed that XIST reinforced the pain and inflammation of rats following plantar incision via the miR-340-5p/RAB1A axis. Thus, we provided evidence of the ceRNA-regulatory network in which XIST functioned as a sponge for miR-340-5p.
As a key transcriptional factor, NF-κB is widely expressed in various cells and acts as a key regulator of inflammatory responses (Pei et al., 2018). Activation of NF-κB begins with the phosphorylation of IκBα, and once IκBα is degraded, NF-κB translocates to the nucleus and facilitates the transcription of its target genes, such as IL-1β, IL-6, and TNF-α (Fuentes et al., 2016; Schuster et al., 2013). Toward this end, the NF-κB pathway was selected and the protein expressions of NF-κB signaling pathway-related proteins were measured in our study. We revealed that NF-κB was the downstream pathway of the XIST/miR-340-5p/RAB1A axis. A former study expounded that NF-κB can control the expressions of proinflammatory and pain mediators (Liang et al., 2017). Additionally, the NF-κB pathway plays an indispensable role in inflammatory response modulated by lncRNA XIST (Ma et al., 2019). Here, we further proposed that lncRNA XIST activated the NF-κB pathway via the miR-340-5p/RAB1A axis to intensify postoperative pain and the inflammatory response of PIP rats.
There are two limitations of this study that we should bear in mind. Currently, pain research has greatly relied on animal models to understand mechanisms and to advance new therapies. However, due to the differences between rodents and humans, it is often difficult to come to clear conclusions. In addition, even within the species, pain-related behaviors are also influenced by variables such as sex and the nociceptive test used. Therefore, more researches and verifications on the result of this study are still needed. These limitations are inevitable in a study, but it also provides potential directions for our future studies.
In summary, we report the roles of XIST in rats after plantar incision surgery. Our findings elucidate that XIST reinforces pain and inflammation of PIP rats, by functioning as a ceRNA for miR-340-5p, and subsequently, activating the NF-κB pathway; thus emphasizing the potentials of the XIST/miR-340-5p/RAB1A axis in the therapy for postoperative pain.
