Abstract
Stroke is one of the leading causes of death and disability worldwide with limited therapeutic options. Melatonin can attenuate ischemic brain damage with improved functional outcomes. However, the cellular mechanisms of melatonin-driven neuroprotection against post-stroke neuronal death remain unknown. Here, distal middle cerebral artery occlusion (dMCAO) was performed in C57BL/6j mice to develop an ischemic stroke in vivo model. Melatonin was injected intraperitoneally immediately after ischemia, and 24 and 48 hours later. Melatonin treatment, with 5 to 20 mg/kg, elicited a dose-dependent decrease in infarct volume and concomitant increase in sensorimotor function. At the molecular level, phosphorylation of PTEN and Akt were increased, whereas PTEN activity was decreased in melatonin treated animals 72 hours after dMCAO. At the cellular level, oxygenglucose deprivation (OGD) challenge of neuronal cell line Neuro-2a (N2a) and primary neurons supported melatonin’s direct protection against neuronal cell death. Melatonin treatment reduced LDH release and neuronal apoptosis at various time points, markedly increased Akt phosphorylation in neuronal membrane, but significantly suppressed it in the cytoplasm of post-OGD neurons. Mechanistically, melatonin-induced Akt phosphorylation and neuronal survival was blocked by Wortmannin, a potent PIP3 inhibitor, exposing increased PI3K/Akt activation as a central player in melatonin-driven neuroprotection. Finally, PTEN knock-down through siRNA significantly inhibited PI3K/Akt activation and cell survival following melatonin treatment, suggesting that melatonin protection against ischemic brain damage, is at least partially, dependent on modulation of the PTEN/PI3K/Akt signaling axis.
Stroke caused by interruption of cerebral blood flow is a neurological disease with growing prevalence among aging adults, leading to severe neurological disabilities or death (Andrabi et al., 2015). Particularly in developed countries, stroke poses as major public health concern resulting in high morbidity, mortality, and disability rates (Ferreira et al., 2019). Clinically, stroke can be divided into ischemic and hemorrhagic stroke depending on pathological features; with ischemic stroke accounting for approximately 80% of all cases, leading to neuronal damage, neuroinflammation, oxidative stress, and cerebral edema with cognitive impairment (Ma et al., 2019).
At present, both clinical and experimental evidences suggest that neuroprotective strategies reducing neuronal injury are crucial for better clinical outcome (Radak et al., 2017; Zhang et al., 2019). Neuroprotective agents generally include drug protectors and physical protectants. Recombinant tissue plasminogen activator (rt-PA) is the only FDA-approved clinical drug, but its use as thrombolytic therapy is limited to 3–5% of stroke patients due of its narrow therapeutic window (Chen and Wang, 2016). Alternatively, other generic drugs, e.g. ginkgetin aglycone (Xu et al., 2019), propofol (Park et al., 2018), and β-Estradiol (Zhou et al., 2019) directly attenuate neuronal injury after stroke through modulation of multiple signaling pathways. Additionally, post-stroke treatment with argon (Liu et al., 2019), curcumin (Liu et al., 2017), or fingolimod (Shang et al., 2020) may indirectly protect against ischemic brain injury through modulation of microglia/macrophage polarization, favoring an anti-inflammatory M2 phenotype. Physical intervention, e.g. hypothermia (Sun et al., 2015), remote ischemic preconditioning (Liu et al., 2016), and electroacupuncture (Wang et al., 2020) have been reported to protect against neuronal injuries, improve neuroplasticity and motor recovery after stroke. Therefore, rational development of novel neuroprotectants that attenuate stroke-induced neuronal injury will likely result in better therapeutic approaches against ischemic stroke.
Melatonin, N-acetyl-5-methoxytryptamine, is an indole hormone secreted by the mammalian pineal gland, best known for its impact on the circadian rhythm (Yang et al., 2019). It is now widely accepted that melatonin is free radical scavenger with strong anti-oxidative properties (Zhang and Zhang, 2014). Furthermore, in vivo and in vitro studies revealed that melatonin has many other biological functions, including sleep regulation, anticancer, anti-aging, anti-inflammatory, and anti-apoptotic effects (Tordjman et al., 2017). Melatonin can easily cross the blood-brain barrier (BBB), and many pre-clinical and clinical studies hypothesize that it can act as a novel neuroprotective agent, and mitigate brain injuries in a variety of neurological diseases such as Alzheimer's disease (AD), traumatic brain injury, stroke and Huntington's disease (Wongprayoon and Govitrapong, 2017). During stroke, melatonin has been shown to alleviate infarct volume and promote behavioral recovery via reduction of neuronal injuries and modulation of microglia-mediated inflammatory responses (Liu et al., 2019).
In neuronal system, phosphatidylinositol 3-kinase (PI3K)/protein kinase B (Akt) pathway plays a critical role regulating cellular survival, apoptosis, proliferation and metabolism. PI3K/Akt activation is regulated by upstream signals including phosphatase and tensin homolog deleted on chromosome 10 (PTEN) (Liu et al., 2019) and Ca2+/calmodulin dependent kinase and cAMP/PKA complex (Song et al., 2005). In particular, PTEN plays a unique role in neuronal apoptosis and death both in vitro and in vivo stroke models (Miao et al., 2018; Xing et al., 2018). Melatonin was reported to alleviate brain injuries after cerebral ischemia via promotion of Akt phosphorylation by PI3K (Beker et al., 2015). However, whether PTEN-induced PI3K/Akt activation contributes to the neuroprotective effect of melatonin after cerebral ischemia remains unknown.
This study evaluated the potential benefits of melatonin in distal middle cerebral artery occlusion (dMCAO) stroke mouse model, and in N2a and primary neurons challenged with oxygen-glucose deprivation (OGD). Post-stroke administration of melatonin resulted in a significant decrease of infarct volume and a remarkable increase of neurological function. Melatonin also inhibited apoptosis in post-OGD neurons. Moreover, the present report provides evidence that melatonin inactivates PTEN, counteracting ischemia-driven PI3K/Akt inhibition. Consistently, a PI3K inhibitor and/or PTEN interference significantly reduced the melatonin’s neuroprotective effects. Thus, this work exposes one of the underlying mechanisms for melatonin mitigation of ischemic brain injury.
Materials and Methods
Animals
Adult male C57BL/6j mice (age 8–12 weeks, weight 23–25 g) were purchased from the Charles River laboratory (Beijing, China). Animals were given food and water ad libitum, and housed with controlled temperature (23 ± 2°C) and humidity (40–60%) conditions with a 12 hours light/dark cycle. All mice studies were approved by the Animal Ethical Committee of Beijing Rehabilitation Hospital, Capital Medical University (approval ID: 2019bkky-031), and the experiments procedures were performed in accordance with the guidelines of Institutional Animal Care and Use Committee (IACUC) for the care of laboratory animals. Animals were randomized into: sham, dMCAO plus vehicle, and dMCAO plus melatonin (5, 10, 20 mg/kg) groups. All mice were used for multiple outcome studies, including infarct volume, neurobehavioral tests, mRNA or protein expression.
Distal Middle Cerebral Artery Occlusion (dMCAO) Model
Distal middle cerebral artery occlusion (dMCAO) was induced by permanent focal cerebral ischemia in male mice, as previously described (10), Briefly, mice were anesthetized with 1.5% isoflurane in the 30% O2/70%N2 mixture. An approximately 1.5 cm skin incision was generated between the right ear and eye. A burr hole (diameter 2 mm) was drilled through the temporal bone. Under the microscope, the bilateral common carotid arteries (CCA) were separated and clamped by small artery clip for 15 minutes. Simultaneously, the right (MCA) was permanently electrocoagulated with bipolar electrocoagulation forceps (Goldbov Photoelectronics, Wuhan, China). A two-dimensional laser speckle (Perimed AB, Järfälla, Sweden) was used for detection of cerebral blood flow (rCBF). Rectal temperature was maintained at 37.0 ± 0.5°C with a heating blanket (Harvard Apparatus, Massachusetts, USA) throughout the surgery. Sham mice were operated in the same way except the occlusion of MCA and CCA.
Melatonin Preparation and Treatments
Melatonin was purchased from Sigma-Aldrich (Shanghai, China) and stored as a 40 mg/mL stock solution in dimethyl sulfoxide (DMSO, Sigma-Aldrich, Shanghai, China). Melatonin was further diluted to different concentrations in sterile saline, as described previously (Liu et al., 2019). After dMCAO, mice were immediately assigned at random to the vehicle or melatonin group. Melatonin was injected intraperitoneally at 5, 10, and 20 mg/kg following surgery, and this was repeated 24 and 48 hours after dMCAO. Vehicle control mice were subjected to intraperitoneal injection of the same volume of 0.5% DMSO solution in saline.
Sensorimotor Function Tests
To assess whether melatonin would improve the recovery of sensorimotor function, adhesive removal test, latency to move and Garcia score test were conducted prior to the surgery, 24, 48, and 72 hours after cerebral ischemia.
Adhesive Remove Test
The adhesive remove test was carried out as previously described (Liu et al., 2017). An adhesive tape (∼50 mm2) was applied to the palmar surface of the right forepaw as a tactile stimulus. Both the time to contact and the time to remove the tape were recorded within a maximum of 120 seconds per trial. Each mouse was tested 3 times on the day before and each day after dMCAO, up to 3 days.
Latency to Move
The test of latency to move was measured as previously described (Rosell et al., 2013). Mice were placed in the center of a circle with the body length of the animal as its radius, and the time of moving one body length (7 cm) was recorded in seconds.
Modified Garcia Scores
The modified garcia score test was performed as previously described (Liu et al., 2019). Five tests were performed: vibrissae touch, body proprioception, lateral turning, limb symmetry and forelimb walking. Each test was scored by a 3 points system according to the following criteria: 0, no deficits; 1, mild deficits; 2, moderate deficits; 3, severe deficits. The total score of the modified Garcia Score are 15 points, which ranges from 0 (serious injury) to 15 (no injury).
Analysis of Infarct Volume
For the evaluation of infarct volume, mice were anesthetized with sodium pentobarbital (50 mg/kg, i.p.) and sacrificed at 72 hours after dMCAO. Brains were rapidly removed, kept on ice and sliced into six equidistant brain sections, 1 mm-thick. Coronal sections were stained with 2% 2,3,5-triphenyltetrazoliumchloride (TTC, Sigma-Aldrich, Shanghai, China) according to previous report (Liu et al., 2019). Infarct volume was calculated by subtracting the non-infarcted volume of the ipsilateral region from that of the contralateral region, and further analyzed using the image analysis software (Image J).
Cell Culture and Oxygenglucose Deprivation (OGD)-Challenged Cell Model
N2a cells were purchased from cell resource center, Institute of Basic Medical Sciences, PUMC, Beijing, China. In vitro, OGD has been used to mimic in vivo cerebral ischemia (Liu et al., 2018).
For OGD/reoxygenation injury of N2a cells, cells were cultured in MEM-EBSS (Gibco, Life Technologies, USA) supplemented with 10% fetal bovine serum (FBS, Gibco, Life Technologies, USA) and 1% penicillin/streptomycin (PS, Gibco, Life Technologies, USA). N2a cells were further cultured in a glucose-free MEM-EBSS (Gibco, Life Technologies, USA) and placed in an anaerobic incubator containing 5% CO2 and 95% N2 at 37°C for 3 hours. Medium was then replaced by normal MEM-EBSS (Gibco, Life Technologies, USA). N2a cells were then immediately treated with vehicle or different concentration of melatonin (12.5, 25, 50 and 100 nM).
For primary neuron OGD/reoxygenation damage induction, primary cortical neurons were collected from brain tissue harvested from embryonic 18-day rat, as previously described (Liu et al., 2018). Isolated neuron cells were cultured in neuron basal medium containing 10% FBS, 1% PS, 2% B27 and 1% Glumax (Gibco, Life Technologies, USA). Primary neurons were then cultured without glucose in neuron basal medium (Gibco, Life Technologies, USA) at 37°C in an anaerobic incubator. After OGD for 40 minutes, the medium was changed for normal neuron basal medium (Gibco, Life Technologies, USA). Primary neurons were then immediately treated with vehicle or different concentration of melatonin (6.25, 12.5, 25, 50 nM).
Cells cultured in normal medium were used as control. The lactate dehydrogenase (LDH) assay and neuronal apoptosis was detected at 12, 24, or 48 hours post OGD. Western blotting and CCK-8 assay were performed 24hours after OGD.
Inhibition of Phosphatidylinositol 3-Kinase/AKT Signaling Pathway In Vitro
Wortmannin (Sigma Aldrich, Shanghai, China) was used to inhibit PI3K/AKT signaling pathway in vitro. The N2a cells were pretreated with vehicle (100% Dimethyl sulfoxide, DMSO) or the PI3K/Akt inhibitor Wortmannin (0.5 µM, dissolved in 100% DMSO). After 30 minutes incubation, the cells were exposed to OGD.
Detection of Cell Viability and Cell Death in Cultures
Cell viability was assessed with the cell counting kit-8 assay (CCK-8 assay, Beyotime, Shanghai, China). N2a cells were incubated with 10 µL of CCK-8 solution at 37°C for 2 hours. Absorbance was measured at 450 nm with a microplate reader (Biotek, Germany). Cell death was measured by the release of LDH into the culture medium with a LDH assay kit (Beyotime, Shanghai, China). Briefly, 120 µL of medium supernatant was taken from each sample, and mixed with 60 µL of LDH working solution for 30 minutes at 25°C in dark. Colorimetric changes were measured by absorbance at 490 nm with microplate reader (Biotek, Germany).
Detection of Cell Apoptosis
The cell apoptosis was measured by an annexin V-FITC/PI Apoptosis Kit following the manufacturer’s instruction (#556547, BD Biosciences, RRID: AB_2869082). Briefly, N2a cells were resuspended in binding buffer after three washes with cold phosphate buffer saline (PBS). Cells were further then with a combination of annexin V-FITC and propidium iodide (PI). All samples were analyzed within 1 hour by the flow cytometry on a BD FACS Calibur machine (BD Biosciences, USA).
TUNEL Staining
TUNEL assay was performed using one step TUNEL apoptosis assay kit from Beyotime (Shanghai, China) according to the manufacturer’s protocol. For the evaluation of neuronal apoptosis, primary neurons were washed with PBS, fixed with 4% paraformaldehyde and further permeabilized with 0.3% Triton X-100. After washing with PBS, the fixed cells were incubated with the TUNEL reagent containing Cyanine 3 (Cy3) labeled nucleotides at 37°C for 1 hour. 4′,6-diamidino-2-phenylindole (DAPI) was used to stain the nuclei. Fluorescent images of all samples were captured using a laser confocal microscope (Nikon, Japan). The ratio of TUNEL-positive nuclei to total cell nuclei was calculated from at least three randomly-selected images of each sample to determine the neuronal apoptosis.
PTEN siRNA Transfection
The PTEN siRNAs as well as their matched controls were synthesized by Gene Pharma (Suzhou, China). The sequences for PTEN siRNAs and negative control (NC) are available on Table 1. N2a cells were cultured in 60 cm2 culture dish at 1.2 × 106/dish for 24 hours in MEM-EBSS medium and transfected with PTEN siRNAs (siRNA-1 and siRNA-2) or negative control siRNA with lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA). After 24 hours, the endogenous PTEN expression was tested by western blotting.
Primers for PTEN Interference.

PTEN Activity Assay
The ability of melatonin to inhibit PTEN activity was detected by a Malachite Green based phosphatase assay kit (Echelon Biosciences, USA) according to the manufacturer’s instructions and as previously published (Wang et al., 2015). In a nutshell, PTEN activity is assessed through quantification of the phosphate liberated from PI (3,4,5) P3. A colored product with molybdate/malachite green is then added for 25 minutes at room temperature. Colorimetric change is measured through absorbance at 600 nm with a microplate reader (Biotek, Germany).
Western Blotting Analysis
Right brain injury tissues (n = 6 per group) were separated, homogenized on ice and stored at −80°C. In order to separate membrane and cytosol fractions, collected cells were treated with a membrane and cytoplasm protein extraction kit (Beyotime, Shanghai, China) according to the manufacturer’s instruction. Both tissue and cell samples were lysed with RIPA buffer containing the protease and phosphatase inhibitors (Roche, China). Full protein content was estimated with BCA reagents, and ran on 12% SDS/PAGE gels and transferred onto a PVDF membrane (Bio-Rad, USA). The membranes were blocked with 5% fat-free milk in Tris-buffered saline solution (TBST) for 1 hour at room temperature, and then incubated with the primary antibodies, including p-Akt (Ser473, ab38449, 1:1000, rabbit, RRID: AB_722678), Akt (ab8805, 1:1000, rabbit, RRID: AB_306791), p-PTEN (pSer380/pThr382/pThr383, SAB4300044, 1:1000, rabbit, RRID: AB_10622040), PTEN (ab32199, 1:1000, rabbit, RRID: AB_777535), GRP94 (ab108606, 1:1000, rabbit, AB_10863019), Ku86 (sc-515736, 1:1000, mouse) overnight at 4°C. On the next day, membranes were washed, and incubated with secondary detection horseradish peroxidase-labeled antibodies anti-mouse (#A28177, 1:4000, goat, RRID: AB_2536163) or anti-rabbit secondary antibody (ab6721, 1:2000, goat, RRID: AB_955447) for 1 hour at room temperature. At last, all protein bands were visualized by ECL detection systems (Bio-Rad). Both GAPDH (#14-9523-80, 1:5000, mouse, RRID: AB_11151704) and sodium potassium ATPase (ab76020, 1:5000, rabbit, RRID: AB_1310695) were used as an internal control. All experiments were repeated three times and relative protein expression determined by densitometry with Image J.
Immunofluorescence Staining
Mice were anesthetized with pentobarbital sodium (50 mg/kg, i.p.) and sacrificed at 72 hours after dMCAO. The brains were perfused with 0.9% saline, removed, which were further fixed with 4% paraformaldehyde for 48 hours and sank in 30% sucrose. The brains were cut into 20 µm slices by a cryotome (Leica, Germany) and subjected to immunofluorescent staining. The samples were attached to slides and blocked with 10% normal goat serum (ZSGB-BIO, China). For double (p-PTEN/NeuN, p-Akt/NeuN) immunofluorescence staining, the sections were treated in the mixture of primary antibodies containing anti-NeuN antibody (#94403, 1:100, mouse, Cell Signaling), anti-p-PTEN antibody (pSer380/pThr382/pThr383, SAB4300044, 1:100, rabbit, RRID: AB_10622040)/anti-p-Akt antibody (Ser473, ab38449, 1:1000, rabbit, RRID: AB_722678) for 24 hours at 4°C, and then incubated with the mixed solution of fluorescent second antibodies at room temperature for 2h, including mouse anti-rabbit IgG-CFL 594 (sc-516250, 1:100, Santa Cruz) and mouse-IgGκ BP-CFL 488 (sc-516176, 1:50, Santa Cruz). And then the samples were counterstained by DAPI fluoromount-G (Southern Biotech, USA) and covered with coverslips. All samples were captured by a laser confocal microscope (Nikon, Japan), and the numbers of positive cells in cortex was calculated from three randomly-selected microscopic fields with software of the laser confocal microscope.
Statistical Analysis
All data are expressed as mean ± standard error of mean (SEM). The statistical analyses were performed by one-way analysis of variance (ANOVA) or two-way ANOVA followed by the Bonferroni post hoc test. A value of p < 0.05 was set as statistically significant.
Results
Melatonin Treatment Ameliorates Infarct Volume and Promotes Sensorimotor Functional Recovery After dMCAO
To verify the neuroprotective effects of melatonin, various concentrations of melatonin (5, 10, 20 mg/kg) were injected intraperitoneally immediately after dMCAO, and 24 and 48 hours after surgery. We further examined sensorimotor function deficits at 24, 48, 72 hours after stoke, and infarct volume with TTC staining at 72 hours (Figure 1A). Melatonin treatment (10, 20 mg/kg) significantly reduced infarct volumes compared to vehicle (Figure 1B and C). However, no significant changes in infarct volume were observed with 5 mg/kg melatonin treatment. These data suggest that melatonin can mitigate cerebral ischemia injury in a dose-dependent manner.
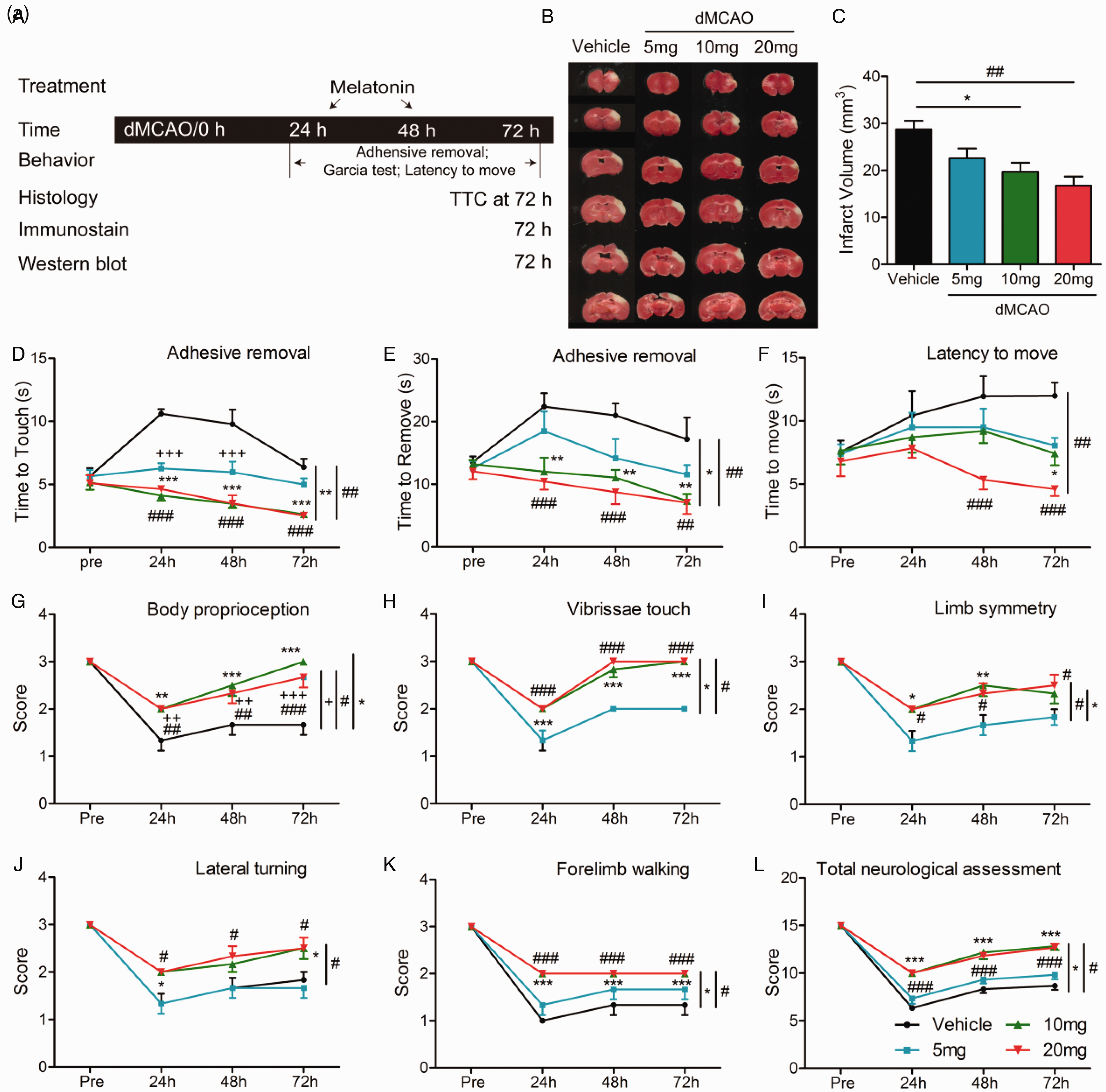
Melatonin Significantly Attenuates Infarct Volume and Improves Sensorimotor Function Outcomes After Ischemic Stroke. Mice were subjected to dMCAO and treated with melatonin (5, 10, 20 mg/kg) at 24, 48, 72 hours after dMCAO. A: Illustration of the experimental timelines. (B) Representative TTC staining and (C) quantification of infract volume 72 hours after dMCAO. n = 6 for each group. *p<0.05; ##p<0.05 vs. vehicle. One-way ANOVA followed by Bonferroni post hoc test. Latency to move (D), adhesive removal (E and F), modified Garcia scores (G and L) were used to evaluate sensorimotor deficits at pre-surgery, 24, 72 hours after operation. (E) Time to touch adhesive tape, (F) Time to remove adhesive tape, (G) Body proprioception, (H) Vibrissae touch, (I) Limb symmetry, (J) Lateral turning, (K) Forelimb walking, (L) Total neurologic assessment score. n = 6 for each group. Data are presented as means ± SEM. #p<0.05, ##p<0.01, ###p<0.001; *p<0.05, **p<0.01, ***p< 0.001; ++p<0.01, +++p<0.001 vs. vehicle. Two-way repeated measures ANOVA followed by Bonferroni post hoc test.
To evaluate the effects of melatonin treatment on functional outcomes after dMCAO, sensorimotor deficits were measured by the adhesive removal test, latency to move, and modified Garcia score system at 24, 48, 72 hours post-stroke. In the adhesive removal test, the administration of 10 or 20 mg/kg melatonin consistently reduced both the times to touch and to remove the tape from the compromised limb at 24, 48, 72 hours in ischemic mice (Figure 1D and E). Treatment with a low dose of melatonin (5mg/kg) only decreased the time to touch at 24 and 48 hours compared to the vehicle group (Figure 1D). As shown by latency to move (Figure 1F), 20 mg/kg melatonin markedly reduced the time to move at 48, 72 hours after dMCAO. Melatonin (10 mg/kg) treatment only significantly decreased the time to move at 72 hours after dMCAO. But there were no significant differences between vehicle and treatment with low melatonin concentrations (5 mg/kg). For the modified Garcia scores, melatonin (10, 20 mg/kg) significantly increased scores at 24, 48, 72 hours after stroke, including body proprioception, vibrissae touch, limb symmetry, lateral turning, forelimb walking, and total neurological assessment in comparison to vehicle group (Figure 1G to L). Consistent with the infarct volume findings, these data suggest that post-stroke melatonin treatment remarkably improves sensorimotor functional deficits at early time points after stroke. Particularly, treatment with 20 mg/kg melatonin afforded a more significant neuroprotection. Therefore, 20 mg/kg melatonin dose were selected for further in vivo experiments.
Melatonin Protects Against Ischemic Neuronal Death After Stroke In Vitro
Our in vivo results showed that melatonin attenuated the infarct volume and ameliorated motor function deficits after dMCAO. Next, we measured whether melatonin could exert a protective effect on post-OGD neurons in vitro. To assess the impact of melatonin on cell death, the levels of LDH released in the culture supernatant were quantified with an LDH. After OGD stimulation, the LDH levels released from N2a cells were significantly increased at 24 hours. Treatment with 12.5, 25, 50, 100 nM melatonin markedly inhibited LDH release from post-OGD N2a cells in a dose-dependent manner (Figure 2A). To further confirm that melatonin could exert anti-apoptotic effect on OGD-stimulated N2a cells, flow cytometry analysis was performed. Administration of 25, 50, 100nM melatonin greatly reduced the percentage of apoptotic cells compared to the vehicle OGD group (Figure 2B and C). However, treatment with 12.5nM melatonin showed no significant effect on apoptotic rate of post-OGD N2a cells. We further measured whether melatonin specifically protected the post-OGD neurons from LDH leakage and apoptosis at various time points. As shown in Figure 2D to F, melatonin markedly suppressed the LDH levels, and exerted the significantly anti-apoptotic effects at 12, 24, 48 hours after OGD stimulation. Collectively, these in vitro results indicate that melatonin can protect against OGD-induced cell death in N2a cells.
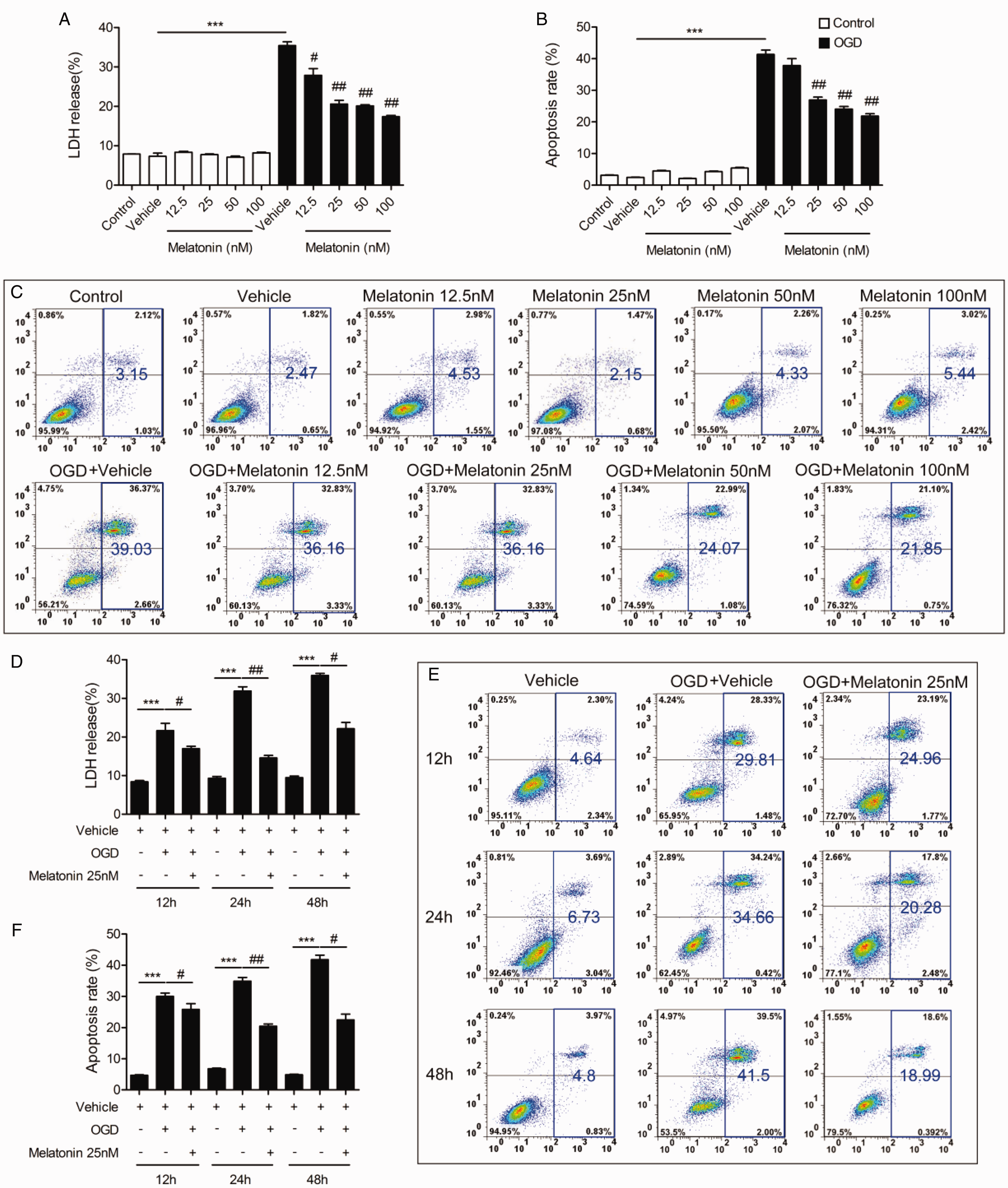
Melatonin Treatment Remarkably Inhibits LDH Release and Cell Apoptosis of Post-OGD N2a Cells in a Dose-Dependent Manner. A-C: N2a cells were subjected to OGD and treated with melatonin (12.5, 25, 50, 100 nM) for 24 hours. A: LDH release at 24 hours after OGD. B: Quantification of cell apoptosis at 24 hours after OGD. C: Representative images of cell apoptosis detected by flow cytometry at 24 hours after OGD. D-F: N2a cells were treated with 25 nM melatonin for 12, 24, 48 hours after OGD. D: LDH release. E: Representative images of cell apoptosis detected by flow cytometry. F: Quantification of cell apoptosis. Samples were collected from three independent experiments, each performed in duplicate. Values are presented as mean ± SEM. ***p<0.001 vs. vehicle, #p<0.05, ##p<0.01 vs. vehicle plus OGD. One-way ANOVA followed by Bonferroni post hoc test.
Melatonin Treatment Alleviates Cerebral Ischemia Injury via Activating PI3K/Akt Pathway In Vivo and In Vitro
We have demonstrated that melatonin treatment inhibited the post-OGD neuronal death, as indicated by decreased LDH release and cell apoptosis results (Figure 2). We further confirmed that melatonin could enhance the cell survival in post-OGD N2a cells (Figure 3A). To determine the molecular mechanisms underlying the protective effect of melatonin on ischemic stroke, pro-survival PI3K/Akt pathway was blocked. Wortmannin, a potent PI3K/Akt signal inhibitor, significantly reduced melatonin-mediated protection on cell viability of post-OGD N2a cells (Figure 3A).
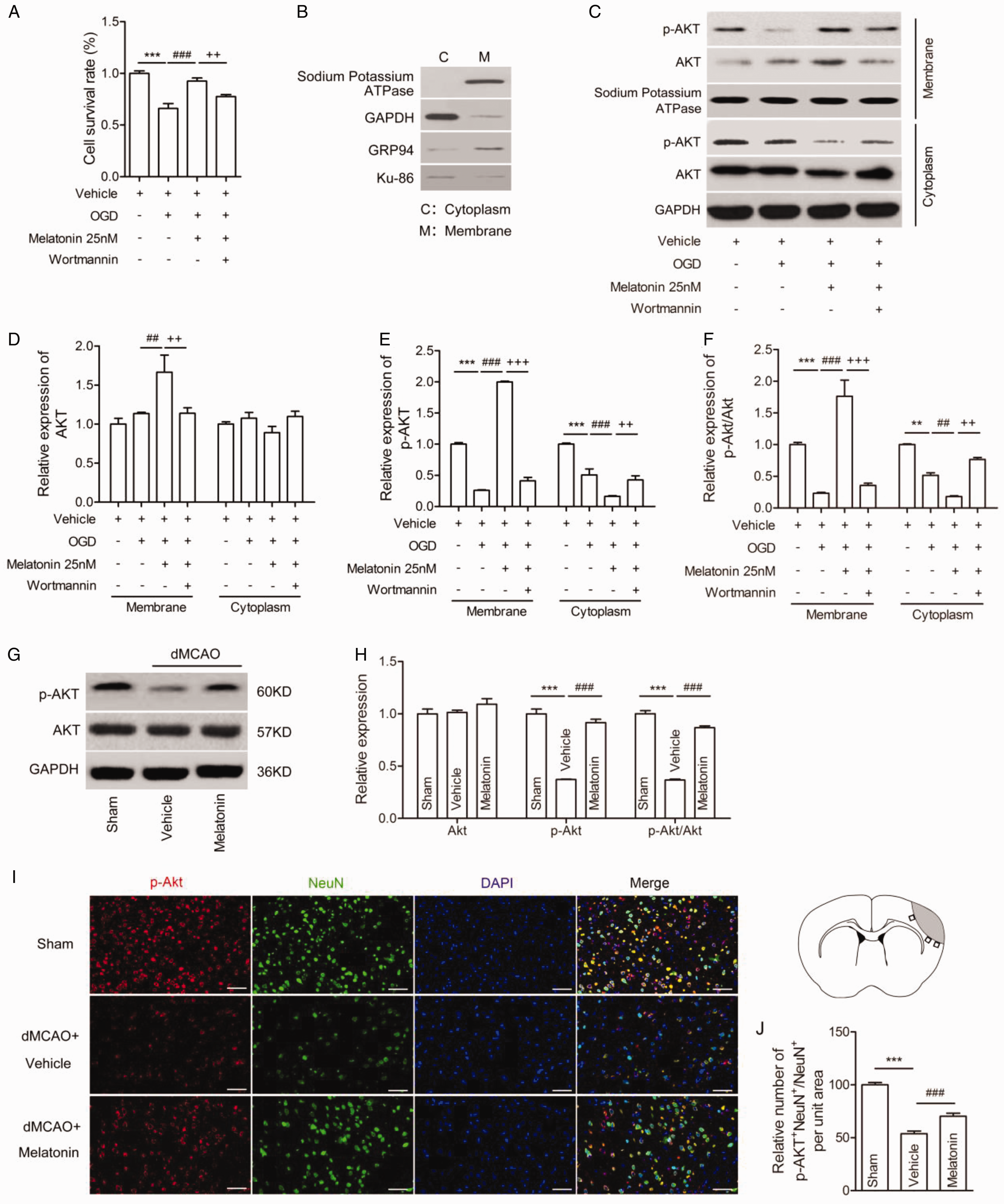
Melatonin Treatment Significantly Improves Cell Survival via the Activation of PI3K/Akt Pathway After Ischemic Stroke In Vitro and In Vivo. A-F: In vitro, N2a cells were treated with 25 nM melatonin for 24 hours after OGD in the presence or absence of wortmannin. A: Cell survival. B: Evaluation of the purity of the cell membrane and cytoplasm isolation of N2a cells. Western blots analysis of the different protein markers sodium potassium ATPase (plasma membrane), GRP94 (endoplasmic reticulum), Ku86 (nuclear) and GAPDH (cytoplasm). C: Representative western blots of p-Akt and Akt in the membrane and cytoplasm fractions. Quantification of western blots data of Akt (D), p-Akt expression (E), p-Akt/Akt ratio (F), respectively. The data resulted from Figure 3C. Values are mean ± SEM. ***p < 0.001 vs. vehicle; ##p < 0.01, ###p < 0.001 vs. vehicle plus OGD; ++p < 0.05, +++p < 0.01 vs. vehicle plus OGD plus 25 nM melatonin. One-way ANOVA followed by Bonferroni post hoc test. Samples were collected from three independent experiments, each performed in duplicate. G-J: In vivo, mice were subjected to dMCAO and treated with 20 mg/kg melatonin at 72 hours after ischemic stroke. G: Representative western blots of p-Akt and Akt in mice. H: Quantification of Akt, p-Akt expression, and p-Akt/Akt ratio. The data resulted from Figure 3G. I: Representative co-immunostaining of p-Akt (red) and NeuN (green). Nuclei were labeled with DAPI (blue) in each group. Scale bar=50 µm. Schematic diagram illustrating the anatomical location of images in the ipsilateral peri-lesion cortex (black). J: Quantification of the percentage of p-Akt +NeuN+ cells among total NeuN+ cells in the peri-lesion cortex. Values are mean ± SEM. ***p < 0.001 vs. sham; ###p < 0.001 vs. dMCAO plus Vehicle. n = 6 mice per group. One-way ANOVA followed by Bonferroni post hoc test. M denotes membrane. C denotes cytoplasm. p-Akt denotes phosphorylated Akt.
The membrane and cytoplasm fractions were further isolated from the N2a cells. Sodium Potassium ATPase, GAPDH, GRP94 and Ku-86 represent plasma membrane, cytoplasm, endoplasmic reticulum and nuclear markers, respectively. To test for the purity of their separation, the different markers were detected in the two fractions (membrane and cytoplasm) using western blot (Figure 3B). Sodium Potassium ATPase was only detected in membrane, but GAPDH, GRP94 and Ku-86 were present in both fractions. GAPDH protein was mainly expressed in cytoplasm. GRP94 protein in membrane was higher than that in cytoplasm, while Ku-86 protein was slightly higher in cytoplasm compared to membrane.
To analyze the effect of melatonin on activation and translocation of Akt, we measured the protein levels of total Akt and phosphorylated Akt (p-Akt) in normal N2a cells (Suppl. Fig. 1), and membrane and cytoplasm of post-OGD N2a cells, respectively (Figure 3C). In normal N2a cells, no significant changes of Akt (Suppl. Fig. 1A and B), p-Akt exprtession (Suppl. Fig. 1A and C), p-Akt/Akt ratio (Suppl. Fig. 1A and D) were observed in melatonin-treated group compared to the vehicle group. The protein expression of Akt in membrane was dramatically elevated with melatonin treatment, which could be reversed by Wortmannin treatment (Figure 3C and D). Interestingly, no significant changes in Akt levels were observed in cytoplasm following melatonin treatment in OGD-stimulated N2a cells (Figure 3C and D). In this case, melatonin markedly enhanced the p-Akt protein levels and p-Akt/Akt ratio in membrane, but significantly reduced p-Akt (Figure 3C and E) and p-Akt/Akt ratio (Figure 3C and F) in cytoplasm. Wortmannin inhibited melatonin-induced changes of p-Akt (Figure 3C and E) and p-Akt/Akt ratio (Figure 3C and F) in OGD-treated neurons. These in vitro findings suggest that melatonin treatment results in the translocation of activated Akt from cytoplasm to cell membrane.
This hypothesis was further supported by in vivo studies examining Akt phosphorylation in mice brain. Consistent with in vitro results, melatonin treatment dramatically promoted Akt phosphorylation and enhanced the ratio of p-Akt/Akt in peri-infarction cortex (Figure 3G and H). To substantiate this result, we further performed co-immunostaining of NeuN and p-AKT on the cortex from the sham, vehicle, and melatonin-treated groups (Figure 3I). As expected, there were more p-Akt positive cells co-localized with NeuN in the melatonin-treated group compared to the vehicle group (Figure 3J). Overall, our in vivo and in vitro results identified a neuroprotective effect of melatonin against ischemic stroke through PI3K/Akt pathway activation.
Melatonin Activates PI3K/Akt Pathway Through Suppression of PTEN Activity
To determine whether melatonin-mediated the increase of neuronal survival was related to PTEN expression, we knocked down PTEN gene in N2a cells using siRNA. Western blot results revealed that both siRNA-PTEN1 and siRNA-PTEN2 dramatically inhibited the expression levels of PTEN (Figure 4A and B). As shown in Figure 4C, melatonin markedly promoted the cell survival in post-OGD N2a cells compared to siRNA-Con group, while siRNA-PTEN treatment significantly inhibited the melatonin-induced increase in cell viability of post-OGD N2a cells.
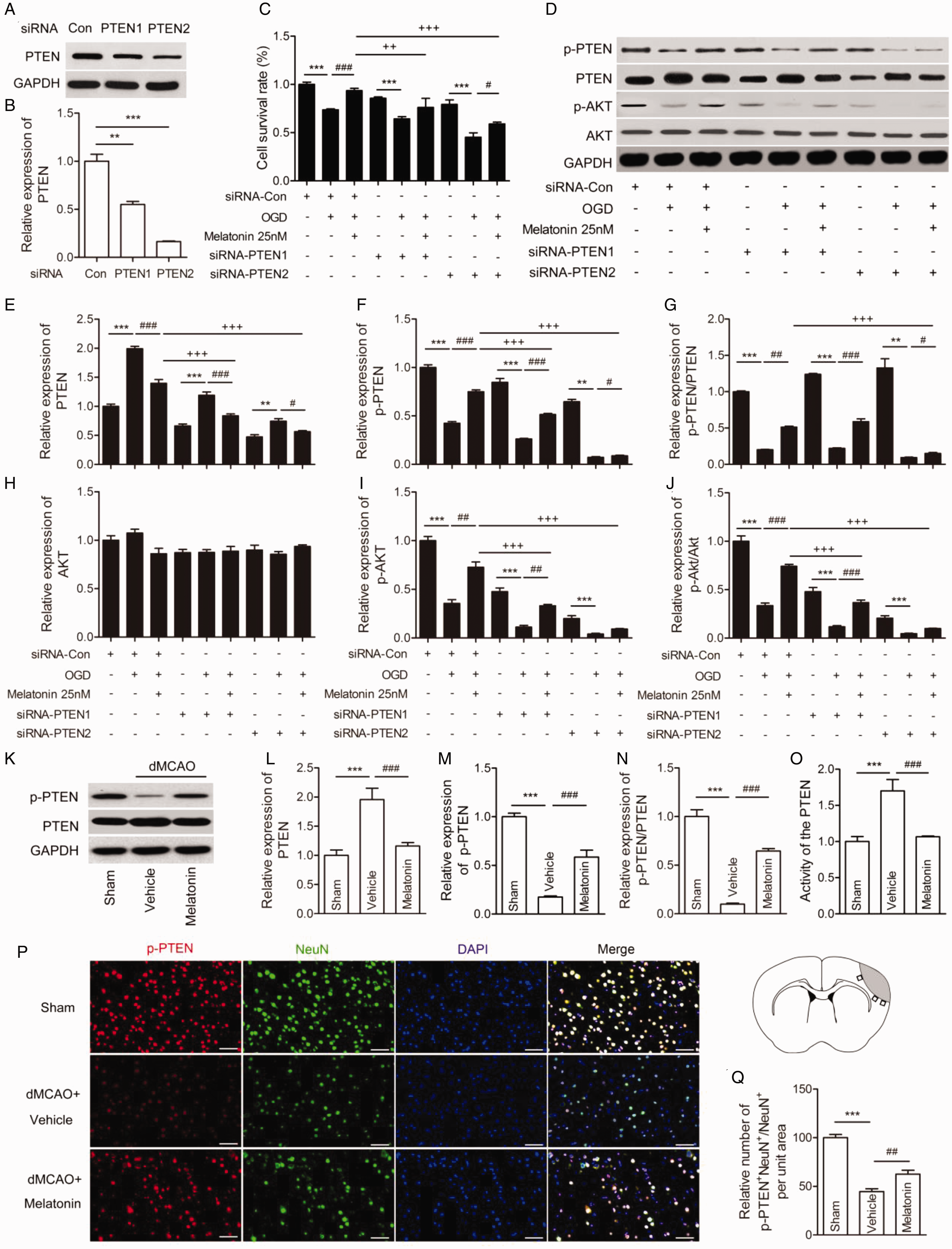
Melatonin Treatment Markedly Increases the Activation of PI3K/Akt via Suppression of PTEN Activity In Vitro and In Vivo. A-J: In vitro, N2a cells were treated with 25 nM melatonin for 24 hours after OGD in the presence or absence of PTEN interference (siRNA1/2). A: Knock down of PTEN gene with siRNA in N2a cells. Representative western blots of p-PTEN and PTEN. B: Quantitative analysis of western blots data of PTEN expression. The data resulted from Figure 4A. C: Cell survival. ***p < 0.001 vs. siRNA-Con. One-way ANOVA followed by Bonferroni post hoc test. D: Representative western blots of p-PTEN, PTEN, p-Akt, and Akt in the whole cell. Quantification of PTEN (E), p-PTEN (F) expression, p-PTEN/PTEN ratio (G), Akt (H), p-Akt (I) expression, p-Akt/Akt ratio (J). The data resulted from Figure 4D. Values are mean ± SEM. ***p < 0.001 vs. siRNA-Con, siRNA-PTEN1, or siRNA-PTEN2; #p < 0.05, ##p < 0.01, ###p < 0.001 vs. OGD plus siRNA-Con, OGD plus siRNA-PTEN1, or OGD plus siRNA-PTEN2; ++p < 0.01, +++p < 0.001 vs. OGD plus siRNA-Con plus 25 nM melatonin. One-way ANOVA followed by Bonferroni post hoc test. Samples were collected from three independent experiments, each performed in duplicate. In vivo, (K-Q) Mice were subjected to dMCAO and treated with 20 mg/kg melatonin at 72 hours after ischemic stroke. K. Representative western blots of p-PTEN and PTEN in mice. Quantification of western blots data of PTEN (L), p-PTEN (M) expression, and p-PTEN/PTEN ratio (N). The data resulted from Figure 4K. O: PTEN activity detected by phosphatase assay kit. P: Representative co-immunostaining of p-PTEN (red) and NeuN (green). Nuclei were labeled with DAPI (blue) in each group. Scale bar = 50 µm. Schematic diagram illustrating the anatomical location of images in the peri-lesion cortex (black). Q: Quantification of the percentage of p-PTEN+NeuN+ cells among total NeuN+ cells in the peri-lesion cortex. Values are mean ± SEM. ***p < 0.001 vs. sham; ###p < 0.001 vs. dMCAO plus Vehicle. n = 6 mice per group. One-way ANOVA followed by Bonferroni post hoc test. p-PTEN denotes phosphorylated PTEN. p-Akt denotes phosphorylated Akt.
To elucidate whether melatonin-mediated PI3K/Akt signal activation was due to increased PTEN phosphorylation, we further analyzed the phosphorylation states of the proteins in normal N2a cells and post-OGD N2a cells treated with siRNA of PTEN. In normal N2a cells, melatonin markedly elevated the expression levels of PTEN in normal N2a cells compared to the vehicle group (Suppl. Fig. 1A and E), while melatonin had no significant effect on PTEN levels (Suppl. Fig. 1A and F) and p-PTEN/PTEN ratio (Suppl. Fig. 1A and G). Compared to siRNA-Con group, melatonin treatment significantly inhibited PTEN protein levels (Figure 4D and E), increased the levels of p-PTEN (Figure 4D and F), p-PTEN/PTEN ratio (Figure 4D and G), p-Akt expression (Figure 4D and I), p-Akt/Akt ratio (Figure 4D and J) in post-OGD N2a cells. Knock down of PTEN with siRNA significantly reversed the melatonin-induced reduction of PTEN levels (Figure 4D and E), and inhibited melatonin-stimulated elevation of p-PTEN (Figure 4D and F), p-Akt (Figure 4D and I), p-Akt/Akt ratio (Figure 4D and J) in post-OGD N2a cells. Furthermore, siRNA-PTEN2 also markedly suppressed the increase of p-PTEN/PTEN ratio induced by melatonin in post-OGD N2a cells (Figure 4D and G). But no significant changes in Akt levels were observed in OGD-stimulated N2a cells treated with melatonin in the presence or absence of PTEN siRNA1/2 (Figure 4D and H). These data suggest that inhibiting PTEN activity, leads to increased Akt phosphorylation which in turn mediates melatonin's neuroprotective activity after stroke.
To validate the central role of PTEN in protection against stroke-induced brain injury in vivo, the protein level of phosphorylated PTEN and PTEN-activity were assessed in the peri-infarction cortex of mice (Figure 4K to N). Consistent with in vitro observations, PTEN levels dramatically elevated (Figure 4K and L), whereas both PTEN phosphorylation (Figure 4K and M) and p-PTEN/PTEN ratio (Figure 4K and N) were remarkably inhibited after brain ischemia. In contrast, administration of melatonin reversed these changes in the ischemic brain and thereby exerted neuroprotective effect. Furthermore, melatonin markedly reversed ischemic stroke-induced the enhancement of PTEN activity (Figure 4O). Consistent with the western blot analysis, there were a higher number of p-PTEN positive neurons co-localized with NeuN in the melatonin-treated group than the vehicle group (Figure 4P and Q). Taken together, PTEN, an upstream regulator of Akt phosphorylation, played an important role in melatonin-mediated neuroprotection against ischemic stroke.
Melatonin Inhibited the OGD-Induced Primary Neuronal Apoptosis Through PTEN/Akt Pathway
To further investigate the effects of melatonin on primary neuron exposed to OGD, cell survival and cytotoxicity in primary neuron cultures were examined (Figure 5A and B). OGD stimulation markedly decreased cell viability (Figure 5A) and promoted LDH release (Figure 5B) compared with the vehicle group. After treatment with 12.5, 25, 50 nM melatonin, post-OGD neuronal death was significantly ameliorated, as shown by the increased cell viability (Figure 5A) and decreased LDH release (Figure 5B). We further tested the effect of melatonin on apoptosis in post-OGD primary neurons using a TUNEL assay (Figure 5C-E) and flow cytometry (Figure 5F and G). Consistent results were obtained that melatonin treatment (6.25, 12.5, 25, 50 nM) inhibited OGD-induced primary neuronal apoptosis compared to the vehicle group. OGD stimulation significantly led to the increase of PTEN protein levels (Figure 5H and I), reduced the p-PTEN levels (Figure 5H and J), p-PTEN/PTEN ratio (Figure 5H and K), p-Akt levels (Figure 5H and M), and p-Akt/Akt ratio (Figure 5H and N) in primary neurons, which could be reversed by melatonin treatment. But melatonin had no significant effect on the expression of Akt protein levels in OGD-induced N2a cells (Figure 5H and L). Furthermore, PTEN activity was greatly enhanced post-OGD compared with untreated cells, but again, this increase was greatly inhibited after melatonin treatment (Figure 5O). These results further demonstrate that melatonin treatment inhibits OGD-induced neuronal cell apoptosis through modulation of PTEN/Akt pathway.
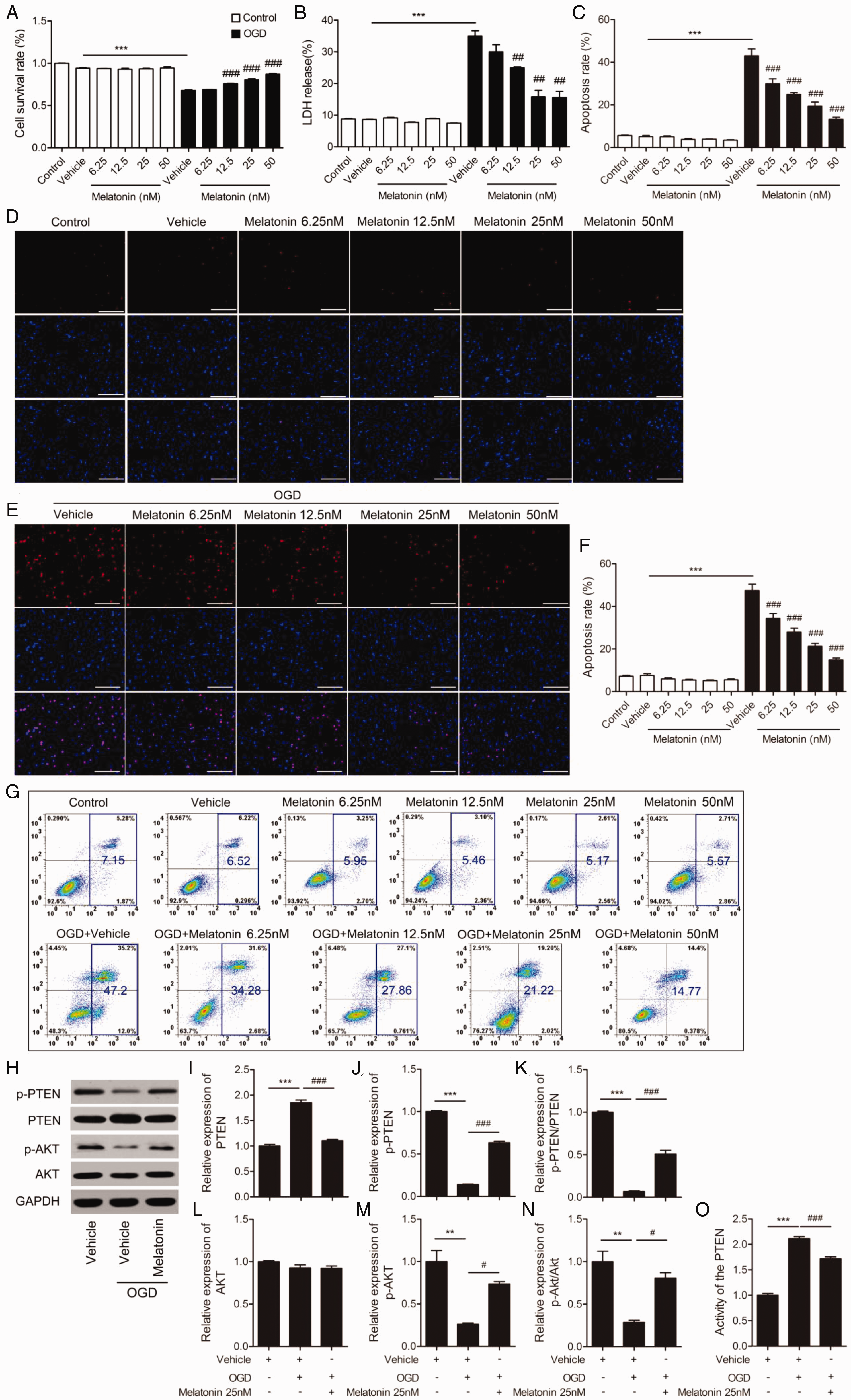
Melatonin Dramatically Increases Cell Survival and Decreases Cell Apoptosis Post-OGD Primary Neurons. Primary neurons were subjected to OGD for 40 minutes and treated with melatonin (6.25, 12.5, 25, 50 nM) for 24 hours. A: Cell survival. B: LDH release. C: Quantification of cell apoptosis detected by TUNEL staining. D, E: Representative images of cell apoptosis by a TUNEL assay. The total number of cells was stained by DAPI and apoptotic cells were scored by TUNEL staining. Scale bar=50 µm. F: Quantification of cell apoptosis detected by flow cytometry. G: Representative images of cell apoptosis by flow cytometry analysis. ***p < 0.001 vs. vehicle; ##p < 0.01, ###p < 0.001 vs. OGD plus vehicle. One-way ANOVA followed by Bonferroni post hoc test. H-O: The effects of melatonin (25 nM) on PTEN/Akt pathway in OGD-induced primary neurons at 24 hours. H: Representative western blots of p-PTEN, PTEN, p-Akt, and Akt in the whole cell. Quantification of PTEN (I), p-PTEN (J) expression, p-PTEN/PTEN ratio (K), Akt (L), p-Akt (M) expression, p-Akt/Akt ratio (N) from Figure 5H. O: PTEN activity. Samples were collected from three independent experiments, each performed in duplicate. Values are mean ± SEM. **p < 0.01, ***p < 0.001 vs. vehicle; #p < 0.01, ###p < 0.001 vs. OGD plus vehicle. One-way ANOVA followed by Bonferroni post hoc test.
Discussion
Melatonin is a neurohormone secreted by pineal and initially recognized as an antioxidant and a powerful free radical scavenger implicated in various physiological and pathological processes (Tang et al., 2020). Its ability to readily cross the blood-brain barrier (BBB) enables a neuroprotective effect on central nervous system in a myriad of neurological diseases; e.g. Alzheimer, Parkinson, spinal cord injury, traumatic brain injury, and stroke (Grima et al., 2018; Cardinali 2019; Li et al., 2019). Accumulating evidence suggests that melatonin can directly afford neuroprotection against stroke-induced brain damage and improve functional outcomes (Andrabi et al., 2015). In a previous study, we revealed that melatonin indirectly alleviated ischemic stroke injury by modulating microglia/macrophage polarization toward anti-inflammatory phenotype (Liu et al., 2019). In the present study, we analyzed the protective effect of melatonin using an in vivo model of dMCAO, in vitro models of N2a cell line and primary neuron subjected to OGD. Overall, melatonin treatment not only diminished infarct volume, but also improved several aspects of neurological function after ischemic stroke in a dose-dependent manner. In agreement with our results, several recent studies showed that melatonin alleviated brain tissue loss and neurological dysfunction in transient MCAO mice (Liu et al., 2019; 2019). Our in vitro results further revealed that melatonin treatment significantly increased cell survival, suppressed LDH release, an inhibited cell apoptosis in post-OGD neurons. This study provides the new evidence that melatonin maybe be a promising therapeutic strategy for stroke.
A multitude of different pathways have been associated with melatonin’s protection on neurological damage, including maintaining Ca2+ homeostasis (Onaolapo et al., 2019), decreasing oxidative stress (Watson et al., 2016), and attenuating endoplasmic reticulum stress (Lin et al., 2018). Melatonin was reported to play important roles in the pathophysiological components of cellular injury after ischemic stroke via modulating oxidative stress, mitochondria, apoptosis, inflammatory response, melatonin receptors 1/2, circular RNA in ischemic injury (Kilic et al., 2020). For example, in a focal cerebral ischemic injury rat model, melatonin reduced brain infarction associated with sequentially rescued neuronal apoptosis and attenuated neuroinflammatory response by modulating oxidative stress signaling (NF-κB/COX2/iNOS) and enhancing anti-oxidative systems such as Nrf2/HO-1/Trx (Ling et al., 2020). But, the cellular mechanisms whereby melatonin protects against stroke-induced neuronal damage are not yet fully clear. This present study suggests that melatonin alleviates ischemic stroke injury by increasing neuronal viability and inhibiting apoptosis. PI3K/Akt signaling is known to be one of the most critical pathways in regulation of cellular survival (Manning and Toker, 2017). Notably, PI3K/Akt inhibition with Wortmannin reversed melatonin's neuroprotection, as manifested by a significant reduction in post-OGD neuronal survival. Moreover, melatonin suppressed OGD-challenged neuronal apoptosis by promoting Akt activation and translocation from cytoplasm to membrane. Thus, PI3K/Akt pathway directly contributed to melatonin's neuroprotective effect after ischemic stroke. In addition, Akt activation has been involved in melatonin-mediated protective effect in the models of AD (Shukla et al., 2017) or spinal cord injury (Li et al., 2019). In line with our results, several studies have demonstrated that melatonin increases phosphorylation of Akt through PI3K activation after stroke (Beker et al., 2015; Kilic et al., 2017; Zhi et al., 2020). For instance, Zhi et al. (2020) revealed that Akt activation was involved in melatonin’s protective effect against oxygen-glucose deprivation/reoxygenation (OGD/R)-induced neuronal injuries. Moreover, melatonin attenuated glutamatergic impairment, synaptic dysfunction, and neuronal apoptosis in ischemic brain, and further protected MCAO-induced neuronal loss through promoting γ-enolase/PI3K/AKT/GSK-3β/CRMP2 pathway (Shah et al., 2019). Buendia et al. also provided the evidence that the role of melatonin derivative Neu-P11 in neuroprotction was associated with PI3K/AKT activation after ischemia stroke (Buendia et al., 2015). Our study and others indicated that Akt signaling plays a crucial role in melatonin-mediated neuroprotection after stoke. In addition to stroke model, melatonin was reported to improve the recovery of locomotor function by enhancing autophagy and reducing apoptosis after SCI, partially via the PI3K/AKT/mTOR signaling pathway in a rat model of spinal cord injury (SCI) and lipopolysaccharide (LPS)-stimulated primary neurons (Li et al., 2019).
Our results identified the activation of PI3K/Akt and suppression of PTEN activity by melatonin after stroke. Pro-survival Akt pathway can be indirectly regulated by PTEN through changes in its phosphorylation status. PTEN is a major up-stream negative regulator of the PI3K/AKT signal transduction via antagonizing PI3K function, i.e. catalyzing the dephosphorylation of PIP3 (Wang et al., 2015). Phosphorylation of PTEN inherently inhibits its phosphatase activity (Worby and Dixon, 2014). The decrease in phosphorylated PTEN by stroke denotes an increase in active PTEN, which then indirectly inhibits the activation of Akt. However, in the presence of melatonin, phosphorylated PTEN levels were increased, decreasing PTEN activity and thereby reversing the PI3K/Akt pathway inhibition. It has been previously reported that suppression of PTEN activity could protect against neuronal death against OGD in vitro and ischemic stroke in vivo models via enhancing Akt phosphorylation (Howitt et al., 2012). Here, our results supported the previous report that melatonin promoted cell survival in OGD induced neurons via enhancing phosphorylation of Akt and PTEN (Beker et al., 2019). The phosphorylation levels of both PTEN and Akt were increased by melatonin treatment both in in vivo and in vitro stroke models (Kilic et al., 2017). Yet, whether the elevation of activated PTEN mediated melatonin-driven protection and activation of Akt remained unknown and required further clarification. PTEN knock down with siRNA in N2a cells showed that siRNA of PTEN significantly diminished melatonin-mediated Akt activation. Additionally, PTEN-inhibition with siRNA reversed melatonins’ neuroprotective effects post-OGD on N2a cells. Furthermore, melatonin promoted phosphorylated Akt’s translocation from cytoplasm to membrane, suggestive of enhanced PTEN phosphorylation in post-OGD neurons. These results indicated that melatonin-mediated phosphorylation or inactivation of PTEN resulted in the suppression of PI3K/Akt signal transduction cascades, exerting pro-survival and anti-apoptotic effects on post-OGD neurons.
Conclusions
Our study demonstrates that melatonin post-treatment can effectively reduce infarct volume and improve sensorimotor functional outcomes after ischemic stroke. Additionally, melatonin afforded neuroprotection, at least in part, through the PTEN/PI3K/Akt regulatory axis to increased neuronal survival (Figure 6). These findings expose melatonin as a promising novel therapeutic strategy for ischemic stroke.
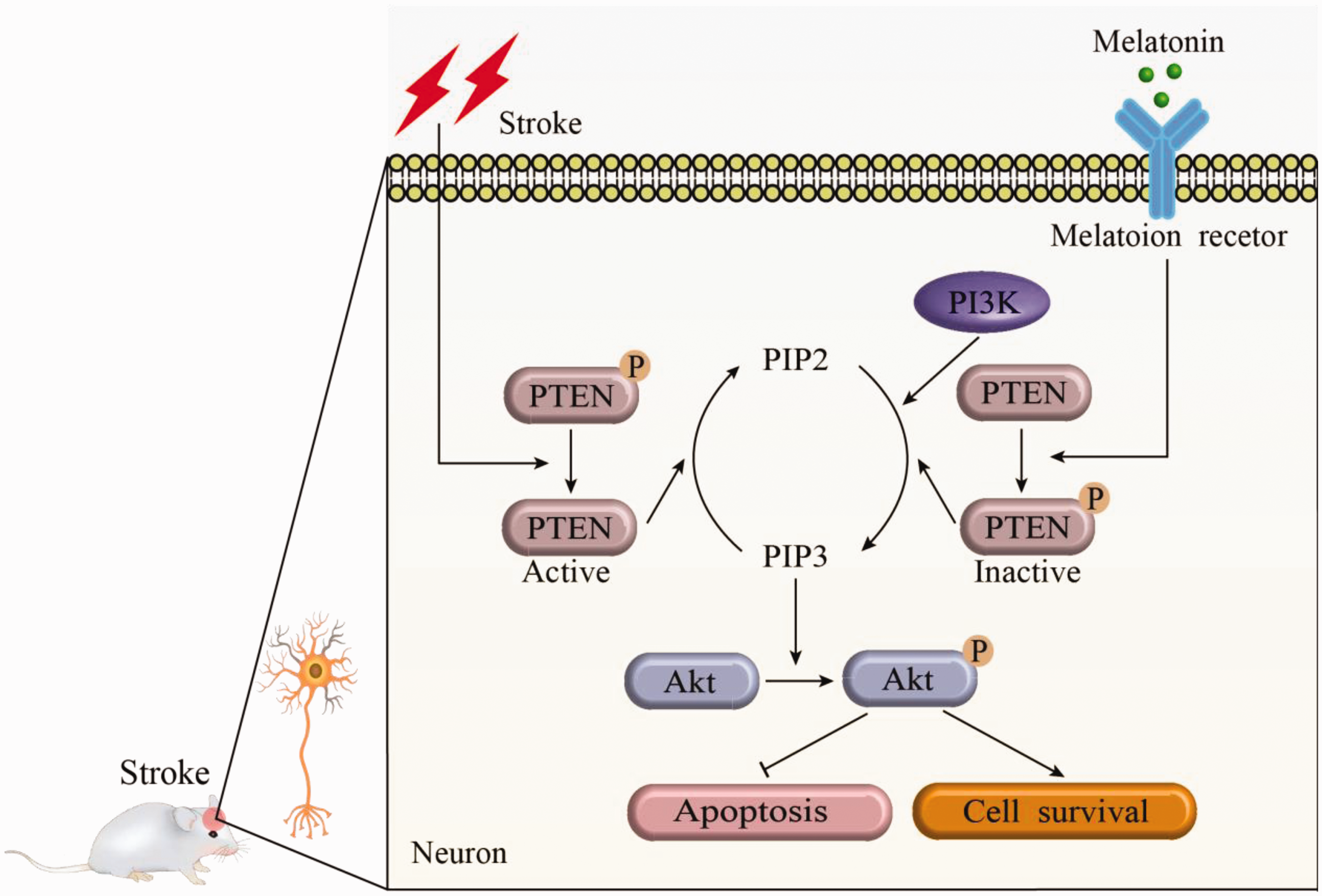
Schematic Diagram Illustrating the Inhibitory Effect of Melatonin on Neuronal Damage and Brain Injury by PI3K/AKT Activation via PTEN Activity Suppression After Stroke. Ischemic stroke results in PTEN dephosphorylation and triggers the activation of PTEN. Activated PTEN subsequently converts phosphatidylinositol (3,5)-triphosphate (PIP3) to phosphatidyl (4,5)-biphosphate (PIP2), leading to the inactivation of pro-survival Akt pathway followed by neuronal toxicity. In the presence of melatonin, the PTEN activity is reversed, thereby leading to the convert of PIP2 to PIP3 catalyzed by phosphatidylinositol 3-kinase (PI3K). Thus, the activation of Akt is increased. Activated Akt inhibits neuronal apoptosis and promotes cell survival after stroke.
Supplemental Material
sj-pdf-1-asn-10.1177_17590914211022888 - Supplemental material for Melatonin Protects Against Ischemic Brain Injury by Modulating PI3K/AKT Signaling Pathway via Suppression of PTEN Activity
Supplemental material, sj-pdf-1-asn-10.1177_17590914211022888 for Melatonin Protects Against Ischemic Brain Injury by Modulating PI3K/AKT Signaling Pathway via Suppression of PTEN Activity by Yuanyuan Ran, Lin Ye, Zitong Ding, Fuhai Gao, Shuiqing Yang, Boyan Fang, Zongjian Liu and Jianing Xi in ASN Neuro
Footnotes
Author Contributions
Y. R. performed the animal experiments, acquired and analyzed data, and drafted the work. B. F. is responsible for conception and critical revision. Z. D., F. G., and S. Y. performed the in vitro experiments. L. Y. edited the manuscript. Z. L. and J. X. are responsible for the conception and design of this work. All authors drafted the work, made critical revision, and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Natural Science Foundation of China (81671161 to Z. L.) and Beijing Natural Science Foundation (7182077 to B. F.).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
