Abstract
Fifty years have passed since the discovery of glial fibrillary acidic protein (GFAP) by Lawrence Eng and colleagues. Now recognized as a member of the intermediate filament family of proteins, it has become a subject for study in fields as diverse as structural biology, cell biology, gene expression, basic neuroscience, clinical genetics and gene therapy. This review covers each of these areas, presenting an overview of current understanding and controversies regarding GFAP with the goal of stimulating continued study of this fascinating protein.
Glial fibrillary acidic protein, typically referred to as GFAP, is an intermediate filament protein that arose early in vertebrate evolution, coinciding with the development of different forms of glial cells in the central nervous systems of primitive fish (Wicht et al., 1994; Kálmán and Gould, 2001); reviewed in (Appel, 2013). Intermediate filaments are key components of the cytoplasmic cytoskeleton, with multiple roles encompassing structural support, scaffolding for enzymes and organelles, and mechanosensing of the extracellular environment (Lowery et al., 2015). Based on sequence homology, GFAP is classified as a type III intermediate filament, along with vimentin (expressed in multiple cell types), desmin (skeletal and cardiac muscle), and peripherin (neurons) (Geisler and Weber, 1983; Eriksson et al., 2009). Its discovery was reported by Lawrence Eng at a meeting of the International Society of Neurochemistry in September 1969. GFAP was quickly adopted as a marker of astrocytes in the central nervous system (CNS); and subsequently its elevated expression recognized as an indicator of gliosis associated with brain injury or disease. Since then, there has been a steady increase in our understanding of GFAP’s role in health and disease. Control of GFAP’s expression and measurement of its levels is now pursued in both basic and clinical neuroscience, with its influence now extending to genetic medicine and gene therapy.
In 2000, Eng et al. published a broad overview of research on GFAP, covering its first 31 years in the scientific literature (Eng et al., 2000). A count of publications per year that mention GFAP finds the trend noted by Eng in 2000 continuing to the present (Figure 1). As we now pass the 50th anniversary of GFAP’s discovery, we consider it appropriate to provide an updated account, concentrating on the most recent 20 years. Some may find this survey of current knowledge helpful, some may find the battle over controversies irritating, but hopefully everyone will find the unknowns an irresistible draw to future research. Much remains to be learned.
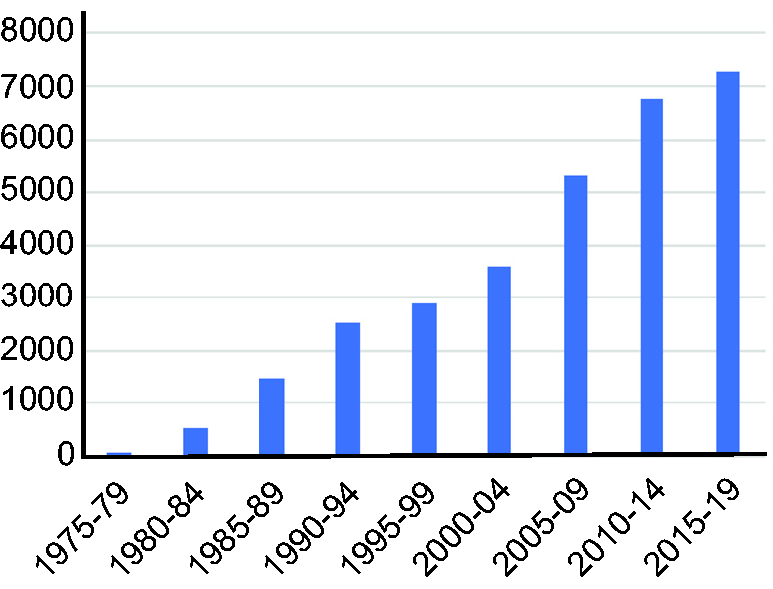
GFAP Publications. Number of publications listed in PubMed each year retrieved by a search consisting of “GFAP” or “glial fibrillary acidic protein.”
Discovery
The initial publication describing what would later be called GFAP derived from protein analysis of three different samples of gliotic scars in the CNS—plaques from individuals with multiple sclerosis, postsurgical scars, and the periventricular region from aged individuals with hydrocephalus ex vacuo (causes not further specified) (Eng et al., 1971; Figure 2). With assistance from the laboratory of Eric Shooter, the amino acid composition of the major band isolated by gel electrophoresis was defined, which along with its solubility properties clearly distinguished this protein from epithelial keratins and other filamentous proteins abundant in the CNS, such as microtubules and neurofilaments (then called “filarin”). The terms “GFA protein”, or more simply “GFAP”, came into common use after the publication of an immunological study by Uyeda et al. (1972).
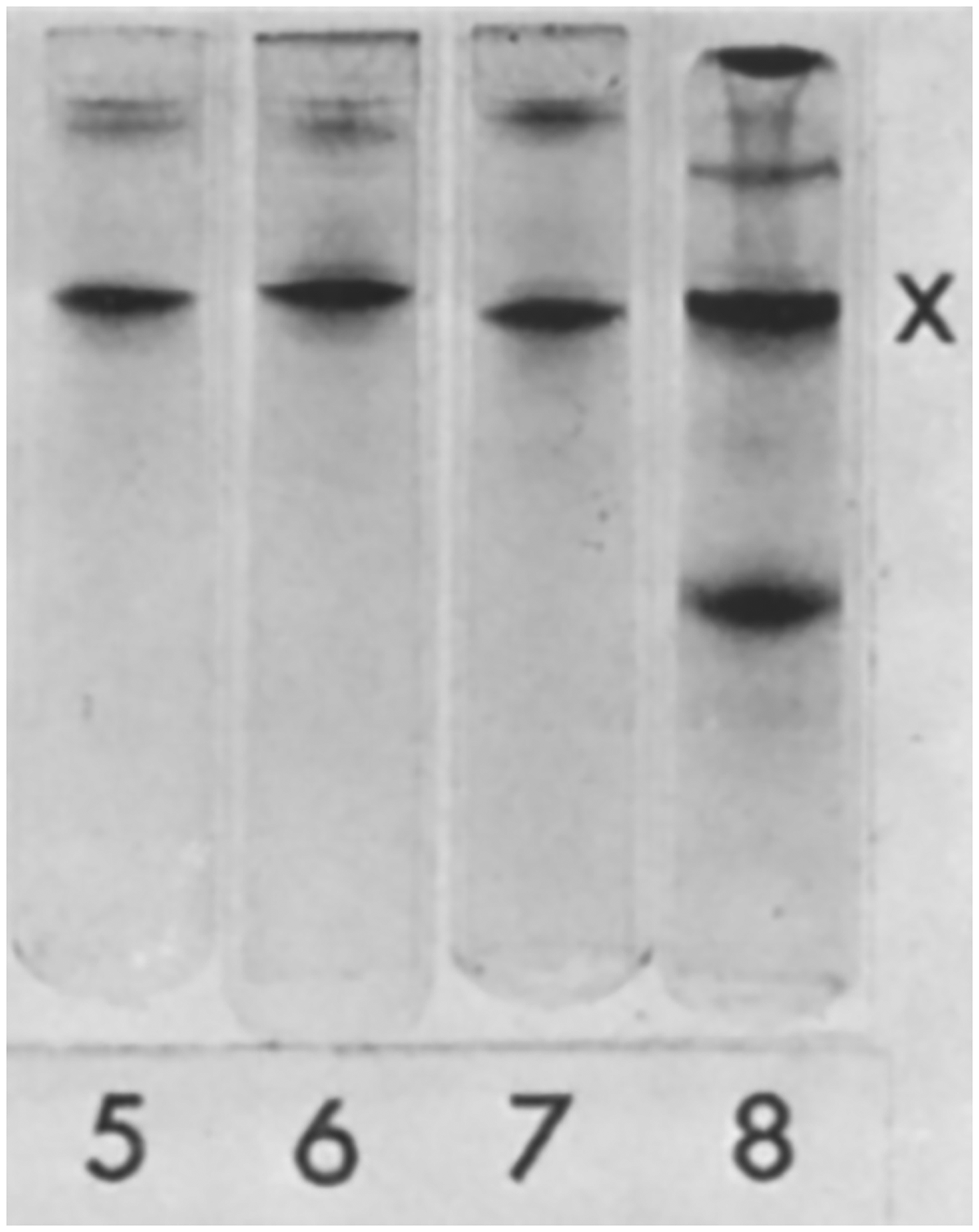
Western Blot Detection of GFAP. Samples shown in the figure are partial purifications of proteins from brain tissue containing a multiple sclerosis plaque (Lane 5), leukotomy scar (Lane 6), multiple sclerosis plaque + leukotomy scar (Lane 7), and periventricular layer in hydrocephalus ex vacuo (Lane 8). The common, major band marked by an X by the authors was identified by amino acid analysis as a novel acidic protein, now known as GFAP. Figure reprinted from Eng et al. (1971), with permission from Elsevier.
Structure
In 1984, Lewis et al. reported the first cloning of a
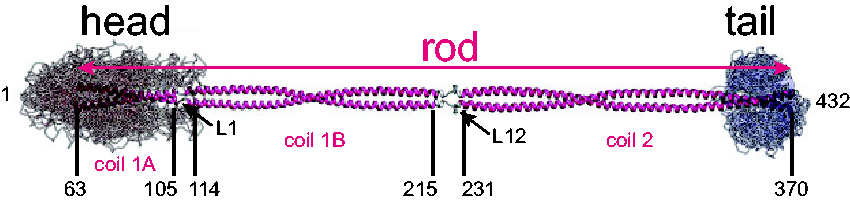
Model of the GFAP Dimer. Shown is a model of the GFAP coiled-coil dimer, based on that of vimentin, showing the α-helical central rod domain flanked by disordered head and tail domains (adapted from Figure 2(E) of Chernyatina et al., 2015). The coordinates for the boundaries of each coil domain are based on amino acid homology between human GFAP and vimentin. Note that GFAP coil 2 may extend further than shown, to position 378 (Roy Quinlan, personal communication, June 4, 2020).
Under normal circumstances, the type III intermediate filaments assemble in a multistep process that begins with parallel binding of monomers to form dimers, which then associate in an antiparallel fashion to form tetramers, with succeeding steps of lateral associations to produce octamers, oligomers, and finally the structures visible by electron microscopy termed unit length filaments that contain anywhere from 30 to 59 monomers in cross section (depending on the filament) (Herrmann et al., 1999). The unit length filaments anneal in a nonpolar fashion (unlike microtubules and actin) to generate mature intermediate filaments (for review, see Herrmann and Aebi, 2004). The association of dimers to form tetramers is primarily mediated through the rod domains. The roles of the head and tail domains appear to vary considerably among the different groups of intermediate filaments and are poorly understood. Using truncation mutants and cell culture as well as cell-free systems, both the head and tail domains of GFAP or its close relative, vimentin, were found important for proper assembly into mature 10-nm filaments (Chen and Liem, 1994a; Ralton et al., 1994; Herrmann et al., 1996). However, these studies did not did not consider the role of binding partners such as αB-crystallin (Nicholl and Quinlan, 1994).
Posttranslational modifications occur at multiple sites in GFAP and may impact its properties and function (Figure 4; for a general review of posttranslational modifications and intermediate filament biology, see Snider and Omary, 2014). Phosphorylation has received the most attention. It is performed by several kinases, including protein kinase A, protein kinase C, calcium/calmodulin-dependent protein kinase II, Rho-kinase, and Cdc2 kinase (summarized in Sullivan et al., 2012; Battaglia et al., 2019). Nothing is known about the relevant phosphatases that reverse these modifications. Phosphorylation of Ser8 and Ser13 (human sequence numbers) in particular has been implicated in regulating assembly and disassembly of GFAP (Inagaki et al., 1990), which might be important during mitosis (Nakamura et al., 1992). Phosphorylation of Ser8 also promotes binding to 14-3-3γ (Li et al., 2006), although the functional significance of this binding is not known. Increased phosphorylation of Ser13 occurs in response to hypoxic injury (Sullivan et al., 2012), and in the conditions of frontotemporal lobar dementia (Herskowitz et al., 2010) and Alexander disease (Battaglia et al., 2019; discussed in the Danger section). Phosphorylation of an abundant protein such as GFAP may also impact the intracellular pools of phosphate, thereby regulating diverse signaling pathways, as has been suggested for keratins as the “sponge hypothesis” (Ku and Omary, 2006).
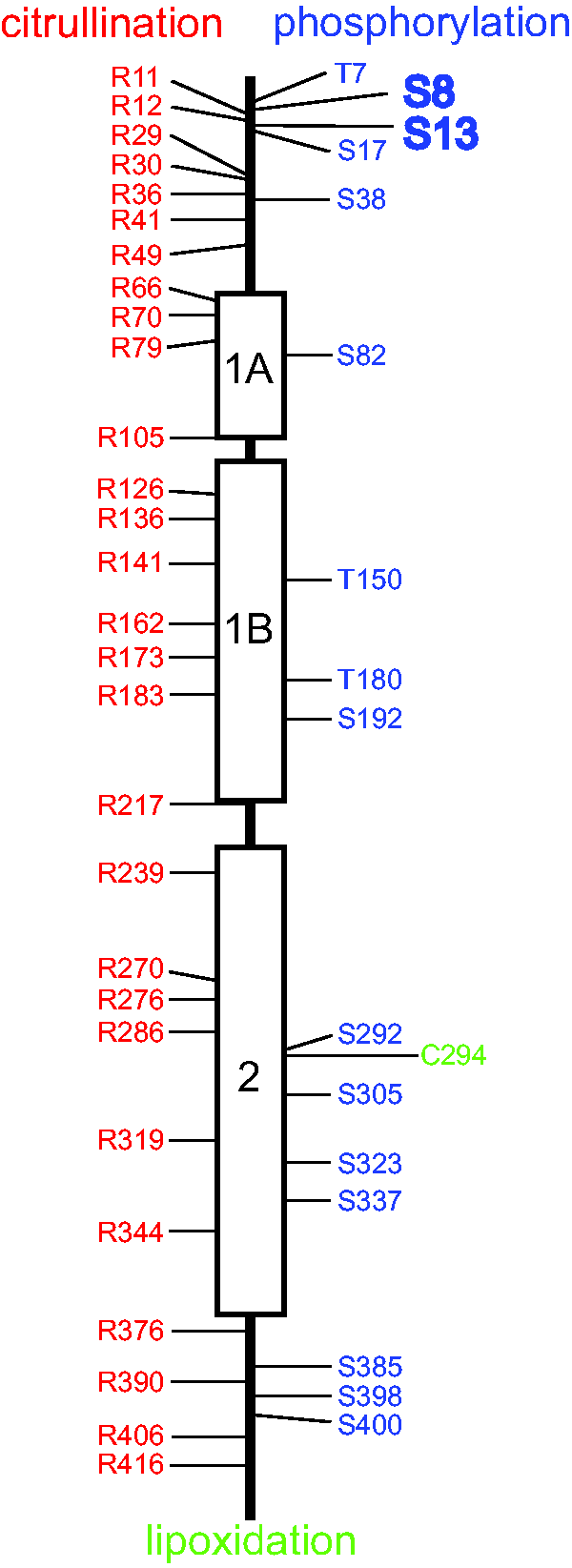
Posttranslational Modifications of GFAP. The three α-helical regions of the GFAP central rod domain are indicated by boxes separated by nonhelical linker regions, and the N-terminal head and C-terminal tail domains are indicated by straight lines. Amino acids identified as sites of citrullination are shown in red (Jin et al., 2013; Brenner and Nicholas, 2017; Faigle et al., 2019), sites of phosphorylation are shown in blue (Inagaki et al., 1994; Battaglia et al., 2019), and the single site for lipoxidation is shown in green (Viedma-Poyatos et al., 2018). Evidence also exists for N-glycosylation and O-glycosylation of GFAP, but none of the specific sites for these modifications is known (Kanninen et al., 2004; Korolainen et al., 2005). Phosphorylation of Ser8 and Ser13 are displayed in larger font because multiple studies have implicated them in regulation of filament assembly or responses to injury or disease (Sullivan et al., 2012; Battaglia et al., 2019). The functional significance of the other modifications has yet to be established.
Citrullination, the enzymatic conversion of peptidyl-arginine to peptidyl-citrulline through deimination, is another posttranslational modification of potential significance (reviewed in Brenner and Nicholas, 2017). Citrullination has been observed at multiple arginine residues in neurologically normal individuals (Jin et al., 2013; Faigle et al., 2019); both the number of sites and/or amount increases in several diseases, including multiple sclerosis (Nicholas et al., 2004; Faigle et al., 2019), experimental allergic encephalomyelitis (Nicholas et al., 2005), Alzheimer’s disease (Ishigami et al., 2005, 2015), and hepatic fibrosis (Kim et al., 2018b).
The predominant isoform in astrocytes is GFAPα, which is the 432 amino acid (for the human sequence) protein that is the subject of most publications. Two other GFAP isoforms, GFAPβ and GFAPγ, derive from alternative RNA start sites (Figure 5). The start site for GFAPβ mRNA is upstream of that of GFAPα, being reported as 169 or 79 nucleotides further 5′ (Feinstein et al., 1992; Lim et al., 2008). In contrast, transcription of GFAPγ commences in GFAPα intron 1, about 130 nucleotides from the end of this intron (Zelenika et al., 1995). Both the GFAPβ and the GFAPγ transcripts contain the GFAPα exons downstream of their start points. GFAPβ is the primary GFAP transcript in the rodent peripheral nervous system (PNS) and also is present in rodent brain and hepatic stellate cells, and in human gliomas and lymphocytes (Galea et al., 1995; Riol et al., 1997). The ability of certain monoclonal antibodies to stain GFAP in the CNS but not in the PNS suggests a difference between the GFAPα and GFAPβ proteins, but it has yet to be established whether this difference is in the primary sequence or posttranslational modifications. This question and other properties of GFAPβ are discussed in detail in the accompanying supplemental file, “GFAPβ.” GFAPγ transcripts are present in mouse and human brain, and in mouse bone marrow and spleen. The longest open reading frame starts in exon 4, which is near the end of rod domain 1B and would thus be expected to produce a polymerization-incompetent protein. The single attempt to detect a protein encoded by the GFAPγ mRNA failed (Zelenika et al., 1995).
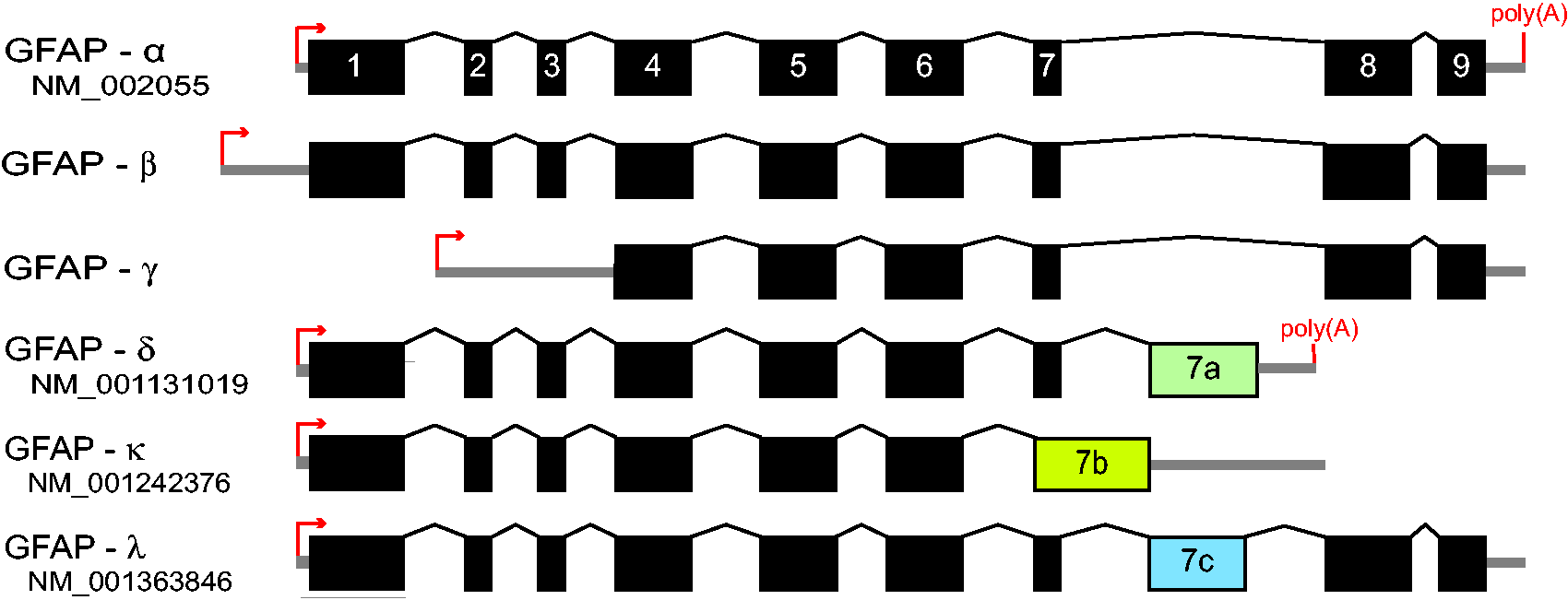
Additional GFAP isoforms are produced by alternative splicing, a process unique to GFAP among cytoplasmic intermediate filaments other than synemin (reviewed in Hol and Pekny, 2015; Figure 5). In 1999, Condorelli et al. reported a novel transcript in rat hippocampus, designated GFAPδ (Condorelli et al., 1999). Sequencing of the GFAPδ transcript revealed that it contained a previously undetected exon within GFAPα intron 7, and so termed exon 7a, whose presence in GFAPδ replaces exons 8 and 9, resulting in a different sequence for the C-terminal tail domain. This exon, defined by a new splice acceptor site and polyadenylation signal, is present in mammals but not lower vertebrates such as birds and fish (Singh et al., 2003). Nielsen et al. (2002) independently detected this isoform through a yeast two-hybrid assay as a binding partner for presenilin, and gave it the name GFAPε. Despite efforts to settle on only GFAPδ for terminology (Roelofs et al., 2005), its designation as GFAPε continues in the clinical literature (here we will use GFAPδ). Another isoform of interest, also generated by alternative splicing in intron 7 but different from that of GFAPδ, is GFAPκ (Blechingberg et al., 2007). While GFAPα can assemble into mature intermediate filaments on its own, GFAPδ and GFAPκ cannot achieve this feat but can coassemble with GFAPα or vimentin (Nielsen and Jørgensen, 2004; Blechingberg et al., 2007). By varying the proportion of α and δ in cell-free assays or transfected cell lines, both Nielsen and Jørgensen (2004) and Perng et al. (2008) found that GFAPδ by itself is prone to aggregation and prevents normal filament assembly if above a threshold concentration (10%–30% of total GFAP, depending on the assay). Based on the relative level of its mRNA, it constitutes about 4% of the total GFAP in normal mouse brain (Thomsen et al., 2013). Similar coassembly titrations have not been performed for GFAPκ, which is present at less than 1% of total GFAP in normal mouse brain (Blechingberg et al., 2007).
Unlike GFAPα, the C-terminal sequences of both GFAPδ and GFAPκ vary considerably among species. Singh et al. (2003) observed that the sequence of exon 7a is strongly conserved among higher primates (e.g., human, chimpanzee, gorilla, orangutan, baboon), but not among other mammals (e.g., pig, rat, mouse). In addition, the conserved sequence among higher primates contains an unusually high number of nonsynonymous sequence changes compared with other species. This led them to suggest that there has been positive selection for a novel (though as yet unspecified) function in the higher primates. Another isoform that contains sequence from exon 7a is GFAPλ, which is a hybrid between GFAPα and GFAPδ (Helman et al., 2020). It includes nearly all of exon 7a, but just before the last 7a codon, it is spliced to include GFAPα exons 8 and 9 (Figure 5). Point mutations in exon 7a that result in overexpression of this isoform are associated with Alexander disease (see later).
Expression
GFAP is predominantly but not exclusively expressed in astrocytes of the CNS (Figure 6). Early evidence for this specificity came from standard immunostaining of both cultured cells and tissues (Bignami et al., 1972; Raff et al., 1979). It is widely accepted that GFAP is much higher in subpial and white matter astrocytes than gray matter astrocytes (Lundgaard et al., 2013; Olabarria and Goldman, 2017), a pattern noted in early studies from the Eng lab (Ludwin et al., 1976). It also coincides with analysis of mRNA by in situ hybridization (Lewis and Cowan, 1985) as well as ultrastructural observations that intermediate filaments are more abundant in astrocytes of these regions compared with most of those in the gray matter (Peters et al., 1991). However, immunocytochemistry must be interpreted with caution, as it can be highly sensitive to the details of processing and fixation (Shehab et al., 1990).

Tissue-Specific Expression of GFAP. GFAP expression in different tissues reflected by mRNA levels, illustrated in the Consensus Dataset by the Human Protein Atlas (data available from v19.3.proteinatlas.org; https://www.proteinatlas.org/ENSG00000131095-GFAP/tissue; Uhlén et al., 2015).
Developmentally, GFAP first appears in radial glia (Levitt and Rakic, 1980), which are progenitors for both astrocytes and neurons. The subsequent rise in GFAP expression as astrocytes differentiate is often viewed as a defining feature of astrocyte maturation. Mouse cortical astrocytes grown in primary culture display monoallelic rather than the more typical biallelic expression of GFAP (Takizawa et al., 2008). Monoallelic expression was not found for mouse hippocampal astrocytes and has not yet been verified to occur
In addition to its baseline expression, GFAP levels increase as a nearly universal response to injury and disease, when astrocytes enter a different state (or states) of “reactive gliosis” that itself defies simplistic definition and likely varies depending on the insult (for recent reviews, see Anderson et al., 2014; Pekny et al., 2016; Liddelow and Barres, 2017; Escartin et al., 2019). Single-cell expression analysis both
Among the most convincing examples of GFAP expression in nonastrocytic cells are nonmyelinating Schwann cells, enteric glia, and the neurogenic stem cells of the subgranular zone in the hippocampus and the subventricular zone surrounding the lateral ventricles. Other sites of GFAP expression, some still speculative, include stellate cells of the liver, pancreas, and vocal fold, Leydig and Sertoli cells in the testis, lens epithelium, lymphocytes, and vertebral and tracheal cartilage (for review, see Messing, 2018b). In the setting of cancer, several surprising examples occur, such as salivary gland tumors and myoepithelial tumors of soft tissue (Huang et al., 1996; Jo and Fletcher, 2015). Clearly, the utility of GFAP as a marker for astrocytes is context-dependent.
The role of GFAP expression as a marker for astrocytes and for their differentiation and response to injury has prompted multiple studies of its transcriptional regulation (Besnard et al., 1991; Mucke et al., 1991; Brenner et al., 1994; Johnson et al., 1995; Bernardos and Raymond, 2006; Yeo et al., 2013; Brenner et al., 2019). More than a dozen transcription factors have been proposed to directly affect GFAP expression. Paramount among these are STAT3, which is critically important for the developmental onset of expression and for its maintenance in the resting state, and AP-1, which appears required for GFAP upregulation following injury (an extensive discussion of regulation of GFAP expression will be published separately. An important outcome of these promoter studies has been development of cassettes to target expression to astrocytes. These have been extensively used both to create transgenic mice and to direct cell-specific viral activity for both basic research and therapeutics (von Jonquieres et al., 2013; Vagner et al., 2016). Descriptions of the more commonly used GFAP promoters are provided in the accompanying supplemental file, “GFAP Transgenes.”
Functions
Because GFAP arose early in vertebrate evolution and has remained relatively unchanged over an extraordinarily long period of time, one might surmise that it is necessary for one or more critical functions (an extensive review of putative functions for GFAP can be found in Brenner, 2014). The expectation that clarity on this topic would come from generation of mouse knockouts was upended by the results from four groups that independently produced such animals, with essentially the same result. Mice rendered completely deficient for GFAP were born in normal numbers, grew to adulthood, and reproduced well (reflecting a complex behavior) (Gomi et al., 1995; Pekny et al., 1995; Liedtke et al., 1996; McCall et al., 1996). Similar vitality has been observed by us for a GFAP knockout rat (Hagemann, unpublished data). However, although the rodent knockouts appear overtly normal, aspects of both hippocampal and cerebellar physiology, such as long-term potentiation and long-term depression, may be defective (McCall et al., 1996; Shibuki et al., 1996).
More significant deficiencies become apparent following injury. As one example, based on the general concept that intermediate filaments in other cell types provide tensile strength and support intercellular connections, Nawashiro et al. (1998) studied the effects of closed head injury using a weight-drop device with anesthetized animals. In this model, most of the GFAP-null mice suffered mortality, whereas all wild-type mice survived. Death was likely due to whiplash injury to the cervical spinal cord that occurred when the foam cushioning allowed head movement following impact (Figure 7). When the heads were fixed in position during impact, no mortality occurred. In another injury model, regeneration of axons in the PNS was impaired in the GFAP-null mice after crush injury to the sciatic nerve (Triolo et al., 2006), although it is not clear whether this reflects changes in interactions between neurons and astrocytes within the CNS, or between axons and nonmyelinating Schwann cells in the PNS. GFAP-null mice are also impaired in their response to both inflammation (experimental allergic encephalomyelitis) and infection (
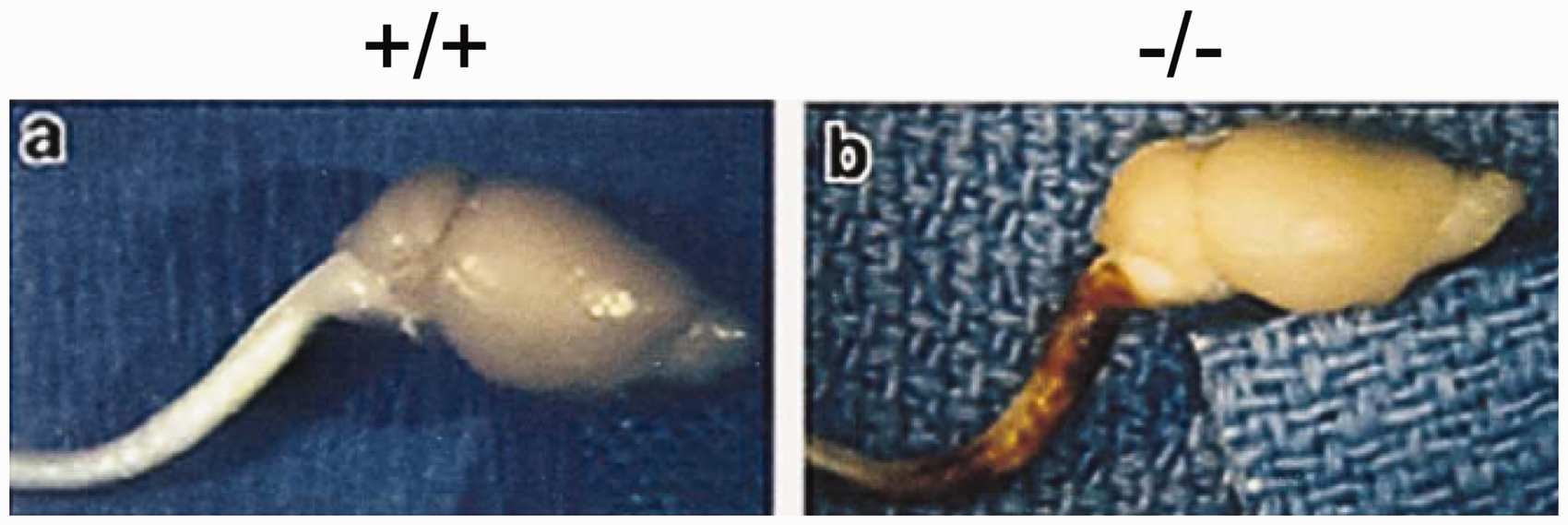
Hypersensitivity of GFAP-Null Mice to Traumatic Cerebrospinal Injury. Neuropathology following trauma from closed head injury is concentrated in the cervical spinal cord of GFAP-null mice (–/–), whereas wild-type mice (+/+) subjected to the same injury are relatively unaffected. Figure reproduced from Nawashiro et al. (1998), with permission.
The generally mild phenotype of GFAP-null animals in the absence of injury may derive from the continued presence of vimentin. When both GFAP and vimentin are absent, several functional deficits are observed not seen for either of the single knockouts, such as release of taurine from cultured astrocytes exposed to hypotonic stress (Ding et al., 1998), regeneration following lesioning of the entorhinal cortex (Wilhelmsson et al., 2004), and infarct volume after ischemia induced by transection of the middle cerebral artery (Li et al., 2008). Although the double knockout experiments suggest participation of GFAP in these additional functions, further studies are required for establishing causality. Other claims related to GFAP functions have been based on studies of cultured astrocytes, but the validity of these are questionable because these cells deviate considerably from the phenotype of astrocytes
The early era of knockout experiments suffered from important limitations, particularly related to the lack of temporal control. To date, all published studies on GFAP knockouts have used one of the four strains generated in the 1990s, none of which employed targeting vector designs that allow for inducible deletion after administration or withdrawal of drugs such as tetracycline or tamoxifen. Hence, all existing GFAP knockouts are GFAP deficient from the moment of conception, raising the possibility that compensatory changes mask the consequences of GFAP deficiency. Nevertheless, the obvious candidates for compensation (other intermediate filaments such as vimentin and nestin) do not change their levels of mRNA and/or protein (McCall et al., 1996; Triolo et al., 2006; Kamphuis et al., 2015). One study compared gene expression profiles of nulls versus wild types, finding changes in expression of 392 genes. None of the changes was identified as possibly being compensatory for the loss of GFAP. In addition, some of these differences could derive from the mixed genetic backgrounds of the mice used, and it was not stated if the animals were sex-matched (Kamphuis et al., 2015). Removal of GFAP in an adult, after the initial period of development has already taken place, might reveal functions of which we are currently unaware. Recent progress in the development of antisense methods for suppression of gene expression, described below in the Alexander disease section, will allow a new set of approaches for addressing these questions.
It is interesting that some vertebrate species have naturally undergone GFAP gene deletion during their evolution (Martinez-De Luna et al., 2017). These include frogs, toads, and some caecilians, whereas salamanders and newts retain the gene. Whether functional knockouts of the
The subcellular localization of GFAP mRNA and protein might be relevant for function, but few studies have examined this topic, and none has established a causal relationship. A general finding for GFAP mRNA is that it colocalizes with the protein, being particularly present within processes (Sarthy et al., 1989; Erickson et al., 1992; Medrano and Steward, 2001; Thomsen et al., 2013). This might minimize the need for transport of the protein to its assembly site. A subplasmalemmal network of GFAP filaments, including association with hemidesmosomes, has been described in the end feet of subpial and perivascular astrocytes of the rat by Nakazawa and Ishikawa (1998). Extrapolating from a similar arrangement for keratins in epithelial cells, and the possibility that links extend from the plasma membrane to the nuclear membrane, Quinlan et al. (2017) proposed a general model in which the network of intermediate filaments has a role in mechanotransduction. The effect on tissue mechanics of conditions that change GFAP levels has been mixed. In both
A novel role for GFAP as a regulator of chaperone-mediated autophagy (CMA) by lysosomes was reported by Bandyopadhyay et al. (2010), who found that GFAP binds to the lysosome-associated membrane protein type 2A (LAMP-2A), a protein involved in CMA substrate transport into lysosomes. In a follow-up study, phosphorylation of GFAP was proposed as a switch to temporally inactivate CMA activation (Arias et al., 2015). These studies were performed in mouse and rat fibroblasts and hepatocytes with GFAP being identified by multiple methods. There are several curiosities about these results, in addition to the studies being confined to two cell types that are only minor sites of expression. Contrary to expectation, CMA was increased by both the addition of purified GFAP to isolated lysosomes
Most of the preceding section reflects focus on the major isoform, GFAPα. The functions of the minor isoforms, as for GFAPα, are poorly understood. Speculation has focused on potential roles for GFAPδ in stem cells (Roelofs et al., 2005; van den Berge et al., 2010) and gliomas (Moeton et al., 2016; Stassen et al., 2017; van Bodegraven et al., 2019b). No animal models have yet been made to selectively express or delete a specific isoform. Possibly complicating such an approach in animal models is evidence that GFAPδ is expressed in different populations of cells in humans versus mouse (Kamphuis et al., 2012; Mamber et al., 2012); and as noted earlier, the marked divergence between lower and higher mammalian species in the amino acid sequence encoded by exon 7a may correspond to functional differences as well.
Degradation
Although there is a sizable literature on synthesis of GFAP and the factors that regulate this process, relatively little attention has been given to pathways for its degradation. Early studies from the Eng lab found that GFAP was particularly susceptible to degradation by a calcium-mediated protease (later called “calpain”) (DeArmond et al., 1983), with predominant cleavage sites after residues Asn59 and Thr383, yielding a central 37-kDa fragment consisting of residues 60–383 that essentially consists of just the rod domain (Dahl and Bignami, 1975; Zhang et al., 2014). These cleavages may be responsible for the degradation product of about 37 kDa often seen in immunoblots of GFAP isolated from tissue (discussed in Heaven et al., 2019). Subsequently, cleavage sites have been identified for caspase 3 after Asp266 (Mouser et al., 2006; Acarin et al., 2007) and caspase 6 after Asp142 (Jonesco et al., 2019) and D225 (Chen et al., 2013). These cleavage sites are shown diagrammatically in Figure 8. It is not yet known whether any of the degradation products acquires new and deleterious functions, as has been suggested for the huntingtin protein (Orr et al., 2008) and β-amyloid (Yankner et al., 1989). None of these degradation fragments appears likely to be capable of self-assembly, although the 1–225 fragment that is one product caspase 6 cleavage is aggregation prone (Chen et al., 2013). Whether any of these cleavages occur at the level of the filament, and if so, causes disassembly, is not known. In addition to sequence-specific proteases, evidence exists for the involvement of both the proteasome and autophagy in GFAP degradation (Tang et al., 2008; Middeldorp et al., 2009; Tang et al., 2010).
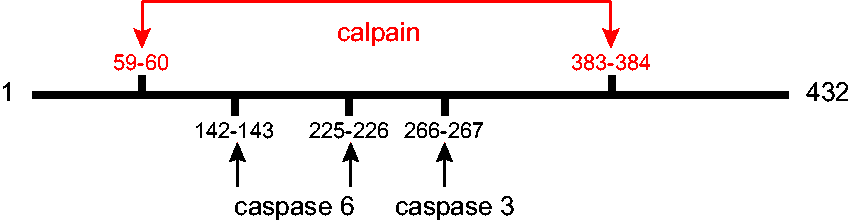
Protease Cleavage Sites in GFAP. Diagram showing the major cleavage sites by proteases that contribute to degradation of GFAP. Calpain cleavage produces a ∼37-kDa product (Zhang et al., 2014). Caspase 3 has one cleavage site, producing 31- and 19-kDa products (Mouser et al., 2006). Caspase 6 has two alternative cleavage sites, producing either 26- and 24-kDa bands (Chen et al., 2013) or 16- and 34-kDa bands (Jonesco et al., 2019).
However GFAP degradation is achieved, it is a slow process; similar to other intermediate filaments, GFAP has a relatively long half-life
Applications
As noted earlier, GFAP expression was predominantly found in astrocytes of the CNS, and thus GFAP was quickly adopted by the neuroscience community as a convenient marker for cell identification (Bignami et al., 1972; Raff et al., 1979; Levitt and Rakic, 1980). Subsequently, the value of GFAP detection in tumor diagnosis quickly emerged. The presence of GFAP complemented previous morphological assessments that related the neoplastic cells in astrocytomas to their presumed normal counterparts and provided a very helpful tool for distinguishing astrocytomas from other gliomas and tumors involving the CNS (Duffy et al., 1977; reviewed by Eng and Rubinstein, 1978). While the presence of GFAP supported the astrocytic nature of such cells, it has since proven difficult to relate the levels of expression with degree of malignancy (recently reviewed by van Bodegraven et al., 2019a). Tumor diagnosis and classification has now moved into a new era that adds more specific immunohistochemical tests and molecular and genetic phenotyping to traditional morphological characterization for subtyping and grading of CNS neoplasms to predict outcomes and guide treatment (Louis et al., 2016; DeWitt et al., 2017; Yeaney and Brat, 2019).
Although GFAP continues to be used as a marker of astrocytes, especially in their reactive state, this use should be tempered by findings that not all astrocytes express detectable levels of GFAP, whereas many nonastrocytic cells both inside and outside of the CNS do (see the earlier section on Expression). In addition, although standard light microscopic immunostaining for GFAP is commonly used to identify gross astrocyte morphology, intracellular dye filling reveals a much more elaborate architecture of fine processes (Bushong et al., 2002; Figure 9).
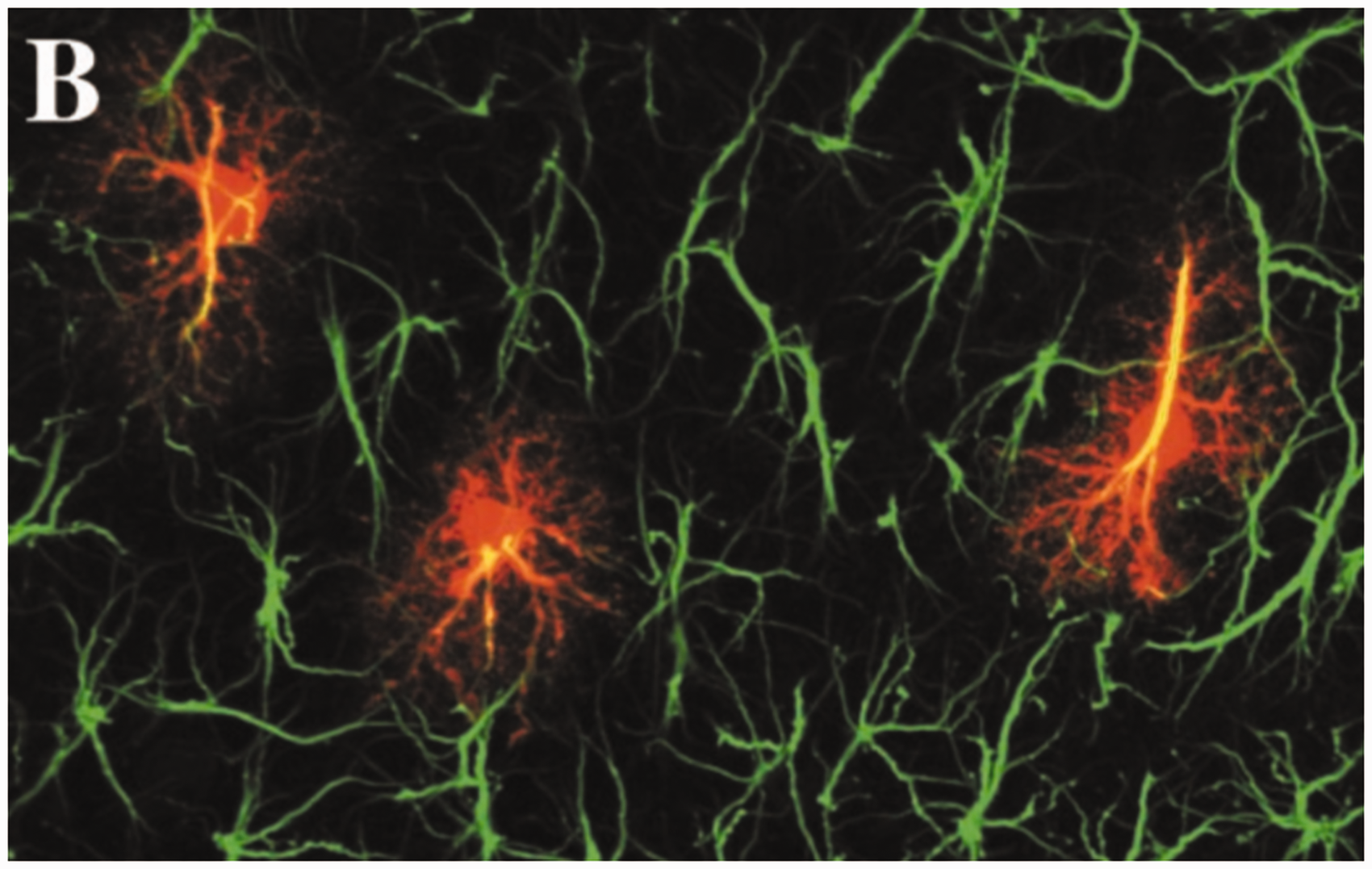
GFAP Immunolabeling and Astrocyte Morphology. Astrocytes in the CA1 region (stratum radiatum) of rat hippocampus, stained for GFAP by immunolabeling and dye-filled with Lucifer yellow to reveal the complexity of fine processes. Figure 2(B) from Bushong et al. (2002)(Copyright 2002 Society for Neuroscience).
In addition to serving as a marker for astrocytes, the presence of GFAP in cerebrospinal fluid (CSF) and blood has been receiving increasing attention as a diagnostic marker of brain injury. One can imagine several mechanisms by which GFAP escapes its intracellular location, especially cell death with subsequent release of normal cytoplasmic contents. Halford et al. (2017) have proposed that the presence of 18- to 25-kDa degradation products in the CSF appear late after traumatic brain injury, and signify cell death, but did not specify from which parts of the protein these fragments derive. GFAP may also be contained within exosomes, whose release is under active control (Lee et al., 2016; Willis et al., 2017; Manek et al., 2018). In the setting of injury or disease, GFAP levels in CSF and blood rise, sometimes to striking levels, particularly in traumatic brain injury, stroke, subarachnoid hemorrhage, and neuromyelitis optica (for reviews, see Liem and Messing, 2009; Petzold, 2015). Monitoring of CSF and blood levels could have particular value in Alexander disease (discussed later), where GFAP is the instigator of the disease process and is also elevated (Kyllerman et al., 2005; Jany et al., 2013).
Danger
Alexander Disease
Surprisingly, although the complete absence of GFAP produces only subtle effects (at least in laboratory rodents), mutations that change only a single amino acid can be lethal. This was revealed through investigation of the devastating leukodystrophy, Alexander disease. The disease occurs in all ethnic groups, affects both sexes, and has ages of onset ranging from fetal through the eighth decade (Prust et al., 2011). Most patients have deficits in white matter (thus the classification among the leukodystrophies), but the degree and distribution of these lesions vary by age. Depending on age of onset and locations of pathology, symptoms include seizures, developmental delays, intellectual disability, difficulty in speaking and swallowing, vomiting, pain, abnormal gait, and autonomic dysfunction. Prevalence was calculated at 1 in 2.7 million for the population of Japan, although this is certain to be an underestimate (Yoshida et al., 2011). Extensive reviews have appeared that are comprehensive (Brenner et al., 2009; Flint and Brenner, 2011; Messing, 2018b) or limited to particular aspects of the disease, such as clinical features (Pareyson et al., 2008; Balbi et al., 2010), neuroinflammation (Olabarria and Goldman, 2017), or genetics (Messing, 2018a). Here, we focus on the most recent findings, which mainly represent attempts to determine mechanisms of the disease.
Variants in GFAP are found in more than 90% of patients clinically diagnosed with Alexander disease; the cause in the other ∼10% is currently unknown (Figure 10). One of the striking features of the genetics is that pathogenic variants have been identified in 87 of the 432 amino acids of the major isoform, GFAPα, and variants in only 5 amino acids are currently considered benign (Pro47Leu, Thr110Ser, Glu223Gln, Asp157Asn, and Asp295Asn) (Messing, 2018a). All known pathogenic variants are genetically dominant and appear to cause a toxic gain of function. Thus, Alexander disease is unlikely to result from loss of any of the “normal” GFAP functions, a conclusion consistent with the absence of Alexander disease-related symptoms in GFAP-null animals. A recent report of putative recessive inheritance has appeared (Fu et al., 2020), but given that the same mutation had previously been shown to be dominant in several patients, the results are more plausibly explained by dominant inheritance along with gene dosage effects. Pathogenic variants are distributed throughout the protein (though few in the nonhelical head domain), suggesting that the disorder is caused by a global property of the protein rather than alteration of a local interaction site. One variant within the head domain has been described, Gly18Val, for which pathogenicity is strongly supported by segregation analysis within the family, although with variable expressivity (Casasnovas et al., 2019). Approximately two thirds of the published mutations are “private” (i.e., found in only one patient or family), thus limiting the potential for genotype–phenotype correlations. Because cases are generally not published if the mutation has already been described, this number for private mutations must be an overestimate; we cannot judge by how much without population-based studies.
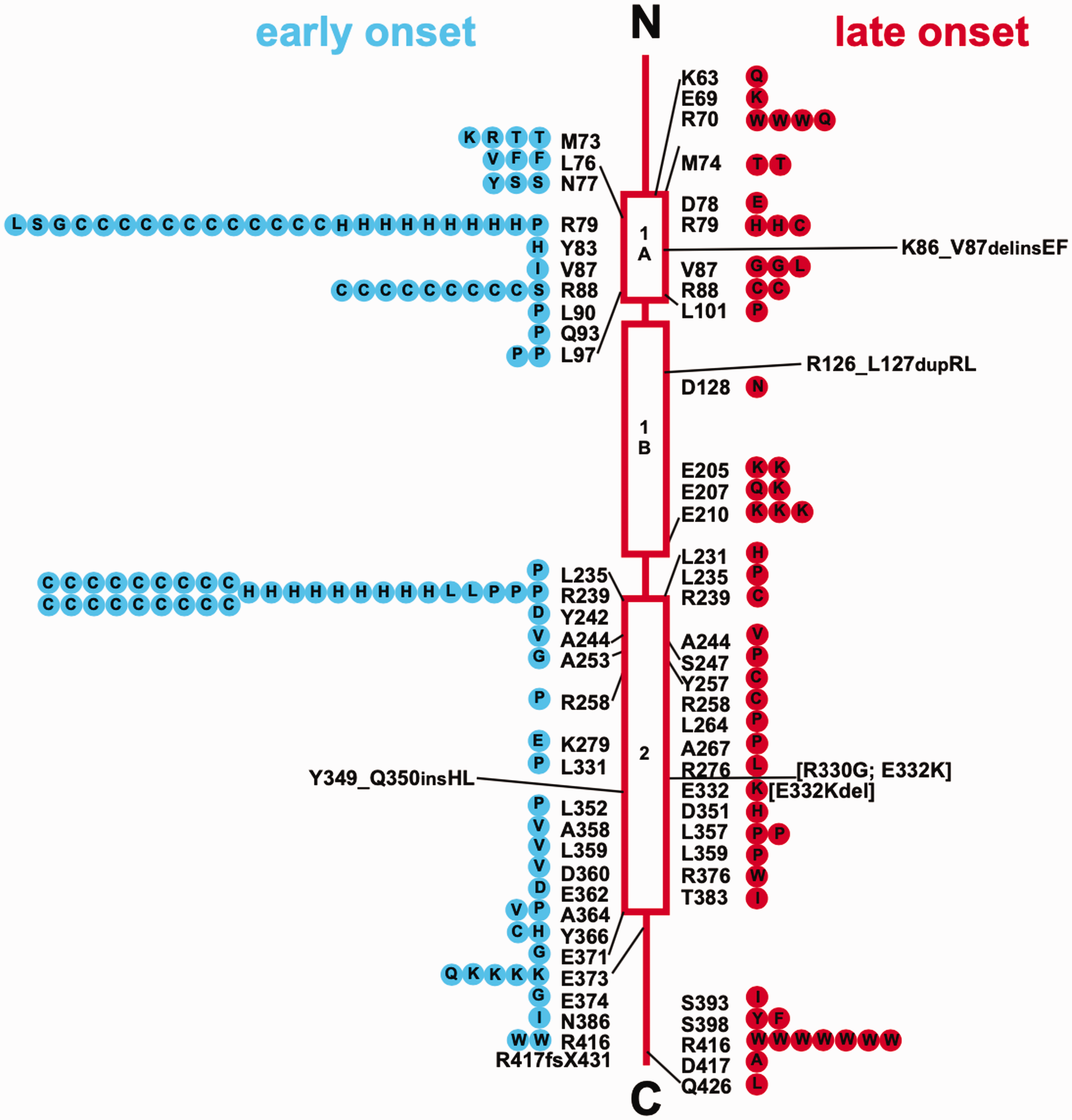
GFAP Mutations in Alexander Disease. Shown is a compilation of GFAP mutations in Alexander disease, prepared in 2012, with their locations in relation to protein structure. The wild-type amino acid is indicated next to the structure, and amino acid replacements within symbols on either side. Early-onset cases (first symptom before the age of 2 years) are on the left, shown as blue circles, and late-onset cases (first symptom after the age of 2 years) are on the right, shown as red circles. Each symbol represents a single patient, except that familial cases, including identical twins, are represented by a single symbol coded for the onset type of the proband. A continually updated list of published mutations is maintained at https://alexander-disease.waisman.wisc.edu. Reprinted with permission from (Messing et al., 2012).
The hallmark pathologic feature, required for the diagnosis, is the presence of cytoplasmic protein aggregates within astrocytes known as “Rosenthal fibers,” named after Werner Rosenthal, the 19th century neuropathologist who first described them as part of a case report on another condition (Rosenthal, 1898; for review, see Wippold et al., 2006; Figure 11). Although Rosenthal fibers are present in several neurological conditions, they are particularly abundant in Alexander disease. These aggregates are complex mixtures of GFAP, other intermediate filaments including vimentin and synemin, stress proteins including αB-crystallin and heat shock protein 27, and other recently described components such as cyclin D2 (Perng et al., 2006; Heaven et al., 2016; Messing, 2018b). The early stages of Rosenthal fiber formation are not yet clear, but they may arise from stress granules (Heaven et al., 2016) or involve focal deposits on existing intermediate filaments along with αB-crystallin (Sosunov et al., 2017). Initially, Rosenthal fibers were viewed as static, and perhaps resistant to degradation, but studies in cultured cells transfected with fluorescently tagged GFAP revealed them as dynamic structures that can disappear over the course of 1 to 2 days (Mignot et al., 2007). Loss of Rosenthal fibers has also been demonstrated
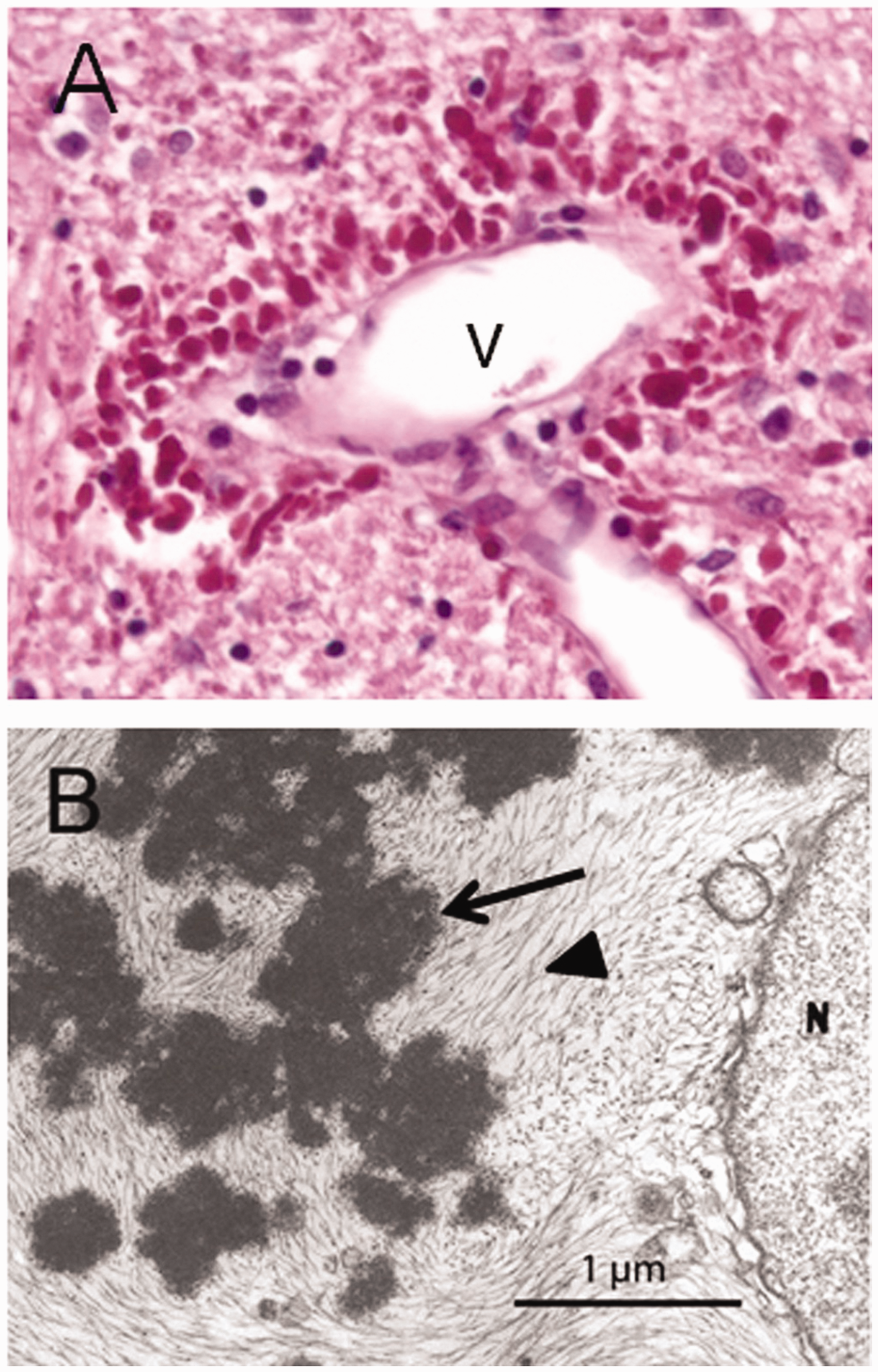
Rosenthal Fibers. Rosenthal fibers in humans with Alexander disease as seen by (A) light microscopy (bright red globular structures surrounding a vein [v]) and (B) electron microscopy (arrow). Figure reprinted from (Messing et al., 2012), used with permission.
The level of GFAP accumulation in Alexander disease roughly correlates with disease severity (Jany et al., 2013; Walker et al., 2014; reviewed in Messing, 2019), but the mechanisms leading to that accumulation are the subject of debate. At least one contributing factor is an increase in GFAP synthesis, prompted by activation of the GFAP promoter that occurs after onset of expression of the mutant GFAP (Jany et al., 2013). Precisely what pathway mediates this activation is not known. Whether decreased degradation also contributes to accumulation of GFAP is even less clear. On the one hand, evidence exists for mutant GFAP impairing proteasome activity (Tang et al., 2006), for which the most likely culprits are soluble GFAP oligomers rather than Rosenthal fibers (Tang et al., 2010). Yet at the same time there is an increase in autophagy (Tang et al., 2008) and activation of caspases 3 and 6 (Chen et al., 2011, 2013). When the knockin mouse model was studied during adulthood using methods of isotope turnover, the half-life of GFAP was observed to be actually decreased (Moody et al., 2017). Furthermore, recent studies on autopsy tissues from three patients found that the mutant form of the protein is not the majority of the GFAP present in the brain, suggesting that it is less stable than the wild type (Heaven et al., 2019). Cleavage of GFAP by caspase 6 has recently been linked to increased phosphorylation of Ser13, with the latter change being most prominent in the more severe, early-onset forms of disease (Battaglia et al., 2019).
How mutant GFAP impacts astrocyte function is not understood. Plausible suspects include decreased levels of glutamate transporters (specifically Glt-1) (Hagemann et al., 2009; Tian et al., 2010), perhaps mediated through caspase 3 (Boston-Howes et al., 2006). Another candidate is change in the potassium channel Kir4.1. Its levels decrease substantially in the caudal CNS and spinal cord of Alexander disease model mice but actually increase in rostral regions (Sosunov et al., 2013; Minkel et al., 2015; Canals et al., 2018). Loss of function mutations in both Glt-1 and Kir4.1 have been linked to epilepsy, ataxia, and other neurological phenotypes in mice and humans (Tanaka et al., 1997; Neusch et al., 2001; Djukic et al., 2007; Bockenhauer et al., 2009; Scholl et al., 2009). Other astrocyte proteins that may be relevant and are the subject of current investigation include aquaporin-4 and connexin 43, given their involvement in other white matter disorders (Hinson, 2008; Lutz et al., 2009; Magnotti et al., 2011; Masaki et al., 2013). However, other changes have been noted that themselves could have wide-ranging downstream effects, including mislocalization and phosphorylation of TAR DNA binding protein of 43 kDa (TDP-43) (Walker et al., 2014) and upregulation of the nuclear lamins A/C (Wang et al., 2018). Studies originating from the
Model systems using
Minor Isoforms and Disease
Until recently, the question of whether any of the minor isoforms have a role in disease has been unclear. GFAPκ is the major isoform in enteric glia, where it is differentially upregulated in patients with Parkinson’s disease, but a direct connection of GFAP to the gut phenotypes associated with this disorder has yet to be established (Clairembault et al., 2014). Although GFAPδ is usually coordinately upregulated with GFAPα in the CNS, as demonstrated in a mouse model of Alzheimer’s disease with prominent gliosis (Kamphuis et al., 2014), in two neurological diseases, the ratio of GFAPδ to GFAPα rises. One of these is vanishing white matter disease, which like Alexander disease is a primary disorder of astrocytes (Bugiani et al., 2011; Huyghe et al., 2012; Dooves et al., 2016; Zhou et al., 2019). A second is a subset of astrocytoma patients with poor outcome (van Bodegraven et al., 2019b). The authors suggest that the poor prognosis is due to the elevated GFAPδ/GFAPα ratio causing upregulation of the phosphatase dual-specificity phosphatase-4 (DUSP4), with speculation involving mitogen-activated protein kinase signaling pathways and interactions with the extracellular matrix. A sequence variant present in GFAPδ but not in GFAPα that predicts an Arg430His amino acid change has been causally linked to Alexander disease (Melchionda et al., 2013; Karp et al., 2018). However, Helman et al. (2020) recently reported that this variant (as well as a silent R430R variant found in a different family) causes disease not through the predicted amino acid change but by altering splicing to increase the level of the GFAPλ isoform (described earlier in the Structure section; Figure 5).
Autoimmunity to GFAP
GFAP may also pose danger by acting as a target in autoimmune disease. Autoantibodies to a variety of neural proteins, including GFAP, occur in many different neurological diseases but are likely nonspecific responses that have little bearing on the course of disease. Examples where presumably incidental GFAP autoantibodies are seen include traumatic brain or spinal cord injury (Zhang et al., 2014), lead poisoning (Moneim et al., 1999), Alzheimer’s disease (Tanaka et al., 1989), and gliomas (Wei et al., 2013). GFAP autoantibodies also occur in a small percentage of patients with multiple sclerosis and in rodent experimental allergic encephalomyelitis, both clearly immune-mediated diseases; however, here also, whether GFAP is necessary or sufficient as a target for disease is not known (Pekovic et al., 1990; Kaiser et al., 1997; Lambracht-Washington et al., 2007).
The best case for a pathogenic immune response aimed at GFAP exists for a condition first described in 2016 and generally referred to as “autoimmune GFAP astrocytopathy” (for review, see Kunchok et al., 2019). These patients suffer from a steroid-responsive meningoencephalitis, sometimes including myelitis, and display a broad range of symptoms affecting vision, gait, and cognition, but not seizures (Fang et al., 2016). One third also have some form of neoplasia, suggesting classification of autoimmune GFAP astrocytopathy among the paraneoplastic syndromes. Studies using mouse models implicate cytotoxic T cells as having a key role in pathogenesis (Sasaki et al., 2014). GFAP autoantibodies are also prominent in a canine disorder, especially seen in Pugs, referred to as necrotizing meningoencephalitis (Uchida et al., 1999). Like the human disease, this canine version displays GFAP autoantibodies in blood and CSF, where their levels may serve as useful biomarkers of severity and progression (Shibuya et al., 2007; Miyake et al., 2013). What exactly initiates this autoimmune attack, and the significance of the GFAP-targeted immune response in driving disease progression, is not yet clear. Most patients have an immune response directed against the GFAPα isoform (Flanagan et al., 2017), though a subset shows specificity for the minor GFAPδ isoform (Long et al., 2018).
Two additional examples of potential GFAP autoimmunity, surprisingly outside the nervous system, are type I diabetes and arthritis. In the pancreas, peri-islet stellate cells express GFAP, similar to the perisinusoidal stellate cells in the liver (Buniatian et al., 1996; Apte et al., 1998). Both T- and B-cell responses to GFAP have been reported as potential early events in the evolution of diabetes in the mouse and human (Winer et al., 2003), with responsible epitopes at several sites within the protein (Standifer et al., 2006; Tsui et al., 2008; Pang et al., 2017b). In the nonobese diabetic mouse model of diabetes, vaccination against GFAP at 9 weeks of age with the goal of inducing tolerance reduced the number of mice with hyperglycemia at 1 year (Pang et al., 2017a). In rheumatoid arthritis, patients may have elevated levels of GFAP in their synovial fluid and antibodies to GFAP in their plasma (Biswas et al., 2013). This finding may relate to reports from the early literature of expression of GFAP in cartilage of mouse and human, although these initial conclusions were based on limited immunohistochemical data (Viale et al., 1988; Kasper and Stosiek, 1990).
Relief
If we focus on Alexander disease, can the danger posed by mutant GFAP be addressed? Genetic studies in both mouse and
The early finding that elevated levels of even wild-type GFAP could lead to both Rosenthal fibers and death in mice led to the suggestion that reducing the expression of GFAP could be a treatment for Alexander disease (Messing et al., 1998). As an initial (and unbiased with respect to mechanism) attempt to reduce GFAP, Cho et al. (2010) screened a library of Food and Drug Administration-approved and other compounds for their ability to reduce expression of GFAP in primary cultures of mouse astrocytes. Compounds identified in this screen, such as clomipramine, reduced GFAP levels in both cultured cells and wild-type mice, but subsequent attempts to extend these findings to mice expressing mutant GFAP failed (unpublished observations). Lithium was administered to mice via the oral route with the goal of reducing GFAP by stimulating autophagy (as in Sarkar et al., 2005). This did achieve significant reductions in GFAP in multiple areas of the nervous system (although likely through effects on Stat3 rather than autophagy) (LaPash Daniels et al., 2015). However, lithium had a very narrow window of safety and is not being pursued for clinical use.
Antisense suppression as a strategy to reduce GFAP was tried in cell culture models decades ago with either the broad goal of reducing gliosis (Weinstein et al., 1991; Yu et al., 1991, 1993; Ghirnikar et al., 1994; Lefrançois et al., 1997) or to alter migration and proliferation of gliomas (Chen and Liem, 1994b; Rutka et al., 1994). Others have used lentiviral vectors to deliver GFAP-targeted antisense
Most recently, Hagemann et al. (2018) reported the dramatic ability of ASOs to suppress expression of GFAP and change the course of Alexander disease in a mouse model (Figure 12). Single intracerebroventricular injections of GFAP-targeted ASOs reduced mRNA and protein throughout the CNS to essentially the null levels found in genetic knockouts of the gene for at least 8 weeks duration (how long the suppression lasts is the subject of current investigation). Even in the Alexander disease mouse models that express mutant GFAP and have elevated GFAP levels, nearly all the GFAP was eliminated by 8 weeks after injection. Rosenthal fibers disappeared, and other downstream markers of disease, such as Lcn2, Ccl2, Cxcl1, and ceruloplasmin, were normalized. The elimination of GFAP in brain parenchyma was also tracked by a drop in the CSF to undetectable levels. Of particular interest for clinical application of GFAP-targeted ASOs, treatment of these mice achieved both prevention and reversal of disease based on both cellular and clinical phenotypes (e.g., Rosenthal fibers disappeared, and body weight was restored). Attempts are now underway to extend these findings to humans in a formal clinical trial.
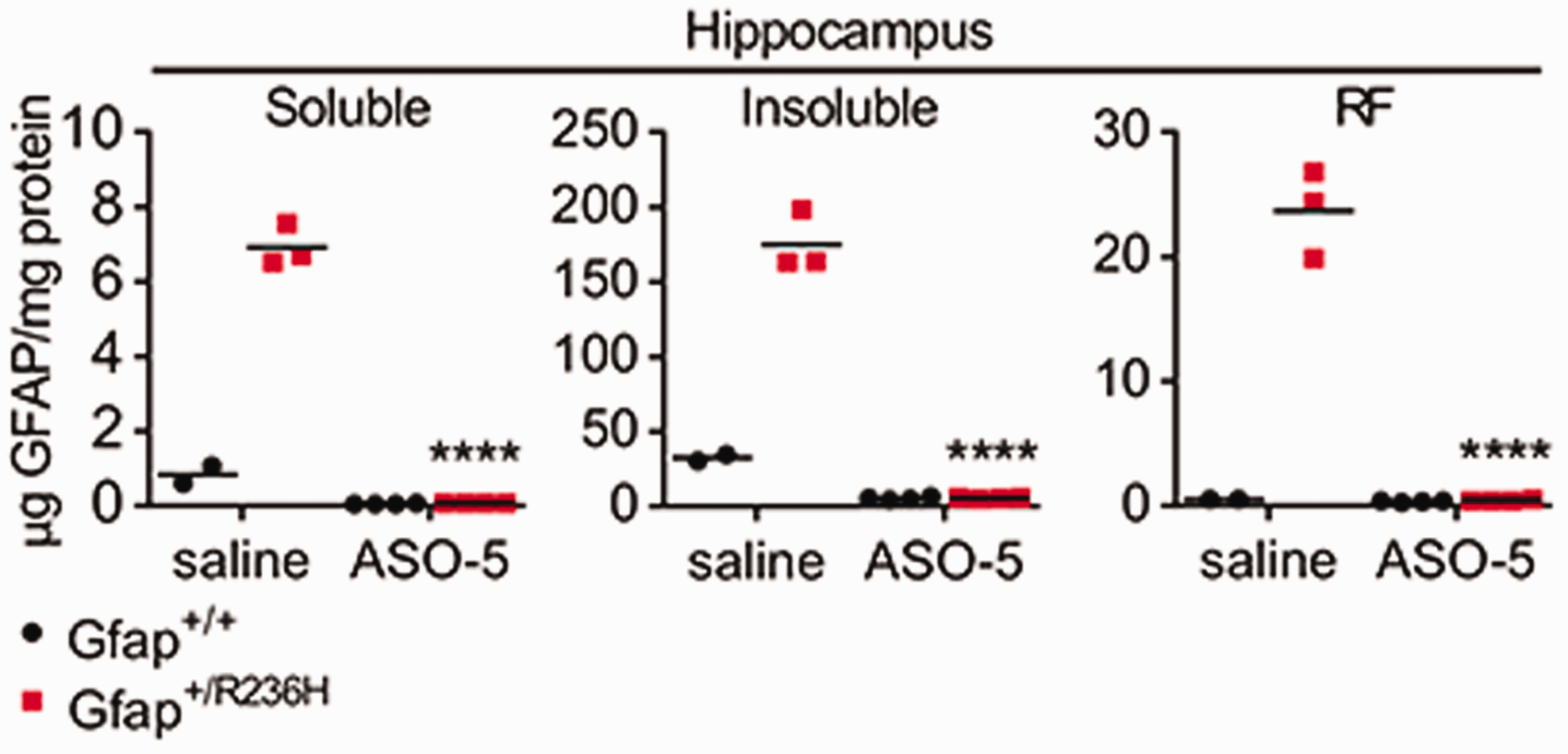
ASO Suppression of GFAP Expression. A single injection of antisense oligonucleotides into the lateral ventricle of adult mice leads to nearly complete elimination of GFAP throughout the CNS (GFAP quantitation by ELISA). Mice given saline as a control show the natural elevation of GFAP that occurs in the mutant mice. Mutants (red), wild type (black), 8 weeks posttreatment. GFAP levels were measured in three biochemically defined pools of intermediate filaments, defined by solubility in Triton-X-100 (left and middle, representing monomers through small oligomers and mature filaments, respectively) and SDS (right, representing an enrichment for Rosenthal fibers). ****p<0.0001, unpaired 2-tailed
Future
We anticipate fruitful future investigations into a number of topics, including the role of minor isoforms, the question of whether variants in the gene influence normal biology or cause pathologies other than Alexander disease, and the question of whether GFAP has currently unknown functions, particularly in higher primates, given that they have more extensive astrocyte networks than lower vertebrates. The full collection of binding partners for GFAP has yet to be identified, and even potential structural relationships remain to be discovered, such as with the nuclear lamins and the recently described subplasmalemmal rim (Quinlan et al., 2017). A previous review by Brenner (2014) cites instances in which there is uncertainty about multiple possible functions of GFAP because of conflicting reports in the literature. It points out that in many instances, these apparent conflicts could be due to GFAP performing different functions in different brain regions. Pursuing comparative regional studies of GFAP function could thus also be a fruitful area for future research. The past 50 years of research on GFAP has yielded many insights over a continuously changing landscape of areas in modern biology and medicine. We expect no less of the next 50.
Supplemental Material
sj-pdf-1-asn-10.1177_1759091420949680 - Supplemental material for GFAP at 50
Supplemental material, sj-pdf-1-asn-10.1177_1759091420949680 for GFAP at 50 by Albee Messing and Michael Brenner in ASN Neuro
Supplemental Material
sj-pdf-2-asn-10.1177_1759091420949680 - Supplemental material for GFAP at 50
Supplemental material, sj-pdf-2-asn-10.1177_1759091420949680 for GFAP at 50 by Albee Messing and Michael Brenner in ASN Neuro
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Work in our laboratories has been funded by the National Institutes of Health (HD076892, HD090256, NS110719, NS42803, NS39055, and NS060120), the United Leukodystrophy Foundation, Children Living with Inherited Metabolic Diseases, the Palamaro Fund, the Juanma Fund, the Jelte Rijkaart Fund, and Elise’s Corner.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
