Abstract
Erythropoietin (EPO) is a cytokine mainly induced in hypoxia conditions. Its major production site is the kidney. EPO primarily acts on the erythroid progenitor cells in the bone marrow. More and more studies are highlighting its secondary functions, with a crucial focus on its role in the central nervous system. Here, EPO may interact with up to four distinct isoforms of its receptor (erythropoietin receptor [EPOR]), activating different signaling cascades with roles in neuroprotection and neurogenesis. Indeed, the EPO/EPOR axis has been widely studied in the neurodegenerative diseases field. Its potential therapeutic effects have been evaluated in multiple disorders, such as Alzheimer’s disease, Parkinson’s disease, amyotrophic lateral sclerosis, spinal cord injury, as well as brain ischemia, hypoxia, and hyperoxia. EPO is showing great promise by counteracting secondary neuroinflammatory processes, reactive oxygen species imbalance, and cell death in these diseases. Multiple studies have been performed both
Introduction
Erythropoietin (EPO) is a 30-kDa molecular weight glycoprotein of 165 amino acids, coded by the EPO gene, located on Chromosome 7 (Law et al., 1986). EPO presents one small O-linked glycan and three tetra-antennary N-linked polysaccharide units, fundamental for its secretion, molecular stability, and binding to its receptor, erythropoietin receptor (EPOR; Tsuda et al., 1988). EPO is a type I cytokine essential for erythroid development and maturation (Brines and Cerami, 2005). Its expression is canonically induced by both hypoxia-inducible factor-1 (HIF-1) and HIF-2 (Keswani et al., 2011; Maiese et al., 2012), transduction factors sensitive to hypoxia and anemia. EPO expression is thus activated during the hematopoiesis process, where it coordinates the hormonal modulation on the hematopoietic system (Jelkmann, 2001). EPO’s main site of production changes throughout human life. During fetal development, blood cells are produced in the liver, and EPO is produced locally in this organ. After birth, the major EPO’s production site becomes the kidney peritubular cells (Jelkmann, 2001). Secondary EPO production sites have been described, and EPO’s expression in organs other than the kidney accounts for about 15% to 20% of the total production throughout the organism (Ponce et al., 2013). Although the kidney peritubular interstitial cells remain the main site of EPO production and secretion, a more pleiotropic role for EPO has been demonstrated in studies showing its expression in the endothelium and in the central nervous system (CNS; Anagnostou et al., 1990; Digicaylioglu et al., 1995; Marti et al., 1996). Within the CNS, EPO’s production and secretion has been demonstrated in the hippocampus, cortex, and midbrain. Furthermore, in the development of the brain and vascular system, EPO has been demonstrated to have a relevant function in promoting and stimulating neurogenesis (Shingo et al., 2001; Lombardero et al., 2011). Moreover, EPO limits cell injury and blocks the production of reactive oxygen species (ROS; Dang et al., 2010). Its neuroprotective effect and its role in counteracting oxidative stress indicate a potential beneficial effect for the molecule in the treatment of neurodegenerative diseases. EPO is currently the most sold biopharmaceutical product worldwide according to the Food and Drug Administration, and in particular, recombinant human EPO (rhEPO) is indicated in the treatment of anemic patients and diseases associated with low concentrations of EPO in plasma (European Medicines Agency; U.S. Food and Drug Administration; Ng et al., 2003). Remarkably, these patients show significant cognitive improvements, leading to the conjecture that EPO could be used for the treatment of neurodegenerative diseases (Sargin et al., 2011).
EPOR: Different Localizations for a Pleiotropic Function
EPOR presents a 225-amino acids extracellular domain, a single transmembrane section of 23 amino acids, and a cytoplasmic domain of 235 amino acids (Constantinescu et al., 1999; Marcuzzi et al., 2016). The extracellular component presents two domains, D1 and D2, both necessary for EPO’s binding (de Vos et al., 1992; Matthews et al., 1996). In the hematopoietic system, EPO’s binding to its receptor leads to homodimerization, with the activation of the associated Janus tyrosine kinase 2 (JAK2) and secondary signaling molecules, such as the signal transducer and activator of transcription 5 (STAT5), phosphatidylinositol 3-kinase (PI3K), and mitogen-activated protein kinase (MAPK; Darnell et al., 1994; Constantinescu et al., 1999). This signaling leads to differentiation and lineage commitment of erythroid colony-forming unit and burst-forming unit progenitors, indicating that EPO’s signaling is necessary for red cell maturation (Constantinescu et al., 1999; Brines and Cerami, 2005; Richmond et al., 2005).
In the past 20 years, many studies have reported various nonhematopoietic functions of EPO and EPOR in endothelial cells, myoblasts differentiation, colon, and in the CNS (Anagnostou et al., 1990; Ogilvie et al., 2000; Arcasoy et al., 2003; Brines and Cerami, 2005; Maiese et al., 2012). Interestingly, it has been demonstrated that biologically active EPO is synthesized in the brain with important roles in neurodevelopment and in the modulation of neurotransmission (Masuda et al., 1994; Digicaylioglu et al., 1995). As EPOR is detected at the embryonic state in neurons and astrocytes of the brain and spinal cord, EPO seems to have an important function in the brain already in the fetal stage (Juul et al., 1999). Linked to this finding, it has been demonstrated that EPOR is present in rats’ oligodendrocytes and astrocytes in culture and that rhEPO administration enhances their maturation and proliferation, suggesting that the EPO/EPOR interaction is important in tissue repair after damage (Sugawa et al., 2002).
The different functions exerted in the CNS with respect to the hematopoietic system could be explained by the fact that EPOR occurs in at least four different isoforms, expressed specifically in different tissues (Ostrowski and Heinrich, 2018; Figure 1).
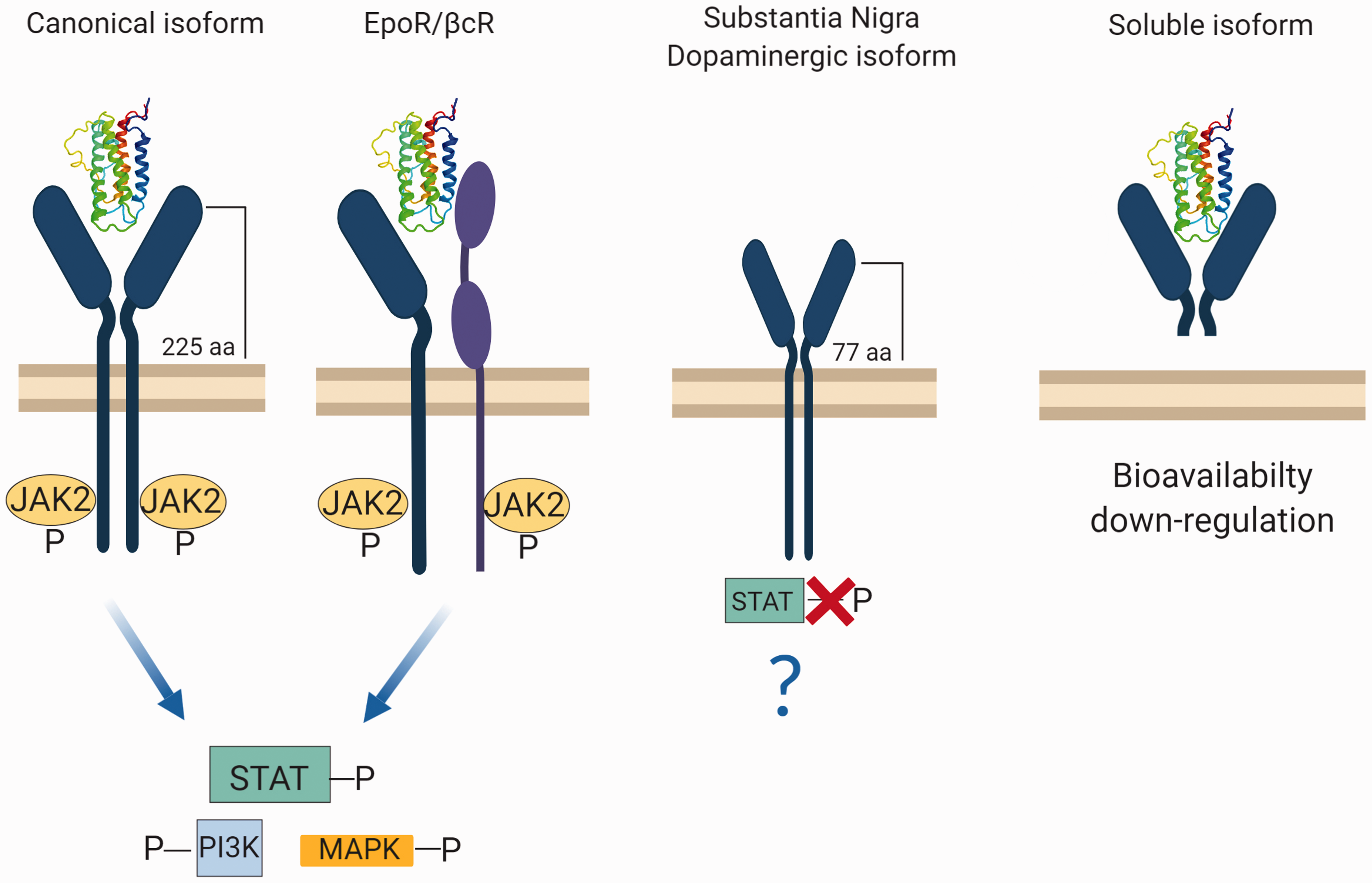
Different isoforms of EPOR in the CNS. In its canonical isoform, EPO’s binding leads to the homodimerization of the receptor and phosphorylation of JAK2 molecules. This activates specific intracellular pathways (STAT-PI3K-MAPK). In the second case, the EPOR monomer interacts with the beta common receptor βcR activating the JAK2 pathway. A third isoform of the receptor occurs in dopaminergic neurons of the substantia nigra, where EPOR results altered in the extra regional domain. In this case, the isoform is shorter than the full form, leading to the lack of STAT phosphorylation. Last, a soluble version of the receptor has been found in the murine brain. This isoform interacts with EPO, with no activation of any downstream pathway. This leads to a reduced availability of EPO, thereby reducing its interaction with other isoforms of EPOR. Made in ©BioRender—biorender.com
The canonical isoform, which is mainly present in the hematopoietic system, is present in the brain (Constantinescu et al., 1999). EPO’s binding to EPOR leads to the formation of a homodimer, which phosphorylates JAK2 and activates the subsequent intracellular pathway that enables erythroid differentiation and maturation (Constantinescu et al., 1999). In the brain, the canonical isoform activation by EPO mediates EPO’s action in neuroinflammation and hypoxia (Lombardero et al., 2011).
The second isoform of EPOR is involved in neural tissue protection (Brines et al., 2004). In this case, the EPOR monomer interacts with the beta common receptor βcR (CD131), a typical receptor subunit of interleukin 3, interleukin 5, and granulocyte-macrophage colony stimulating factor (Brines and Cerami, 2005). The most accredited hypothesis is that the dimer formation results in a specific tissue—protective receptor. The activation of this receptor seems to activate the same JAK2 signaling pathway as the canonical homodimer (Brines et al., 2004; Brines and Cerami, 2012).
The third isoform of the receptor has been found in dopaminergic neurons of the substantia nigra, where EPOR results altered in the extra regional domain, being shorter than the full form (Marcuzzi et al., 2016). EPOR truncated isoform does not implicate STAT phosphorylation, suggesting the presence of another mechanism of action not yet characterized (Marcuzzi et al., 2016). This could be useful for the identification of possible drugs targeting specifically this isoform.
Last, an extracellular soluble version of the receptor has been shown to occur in the murine brain. This isoform interacts with EPO, with no activation of any downstream mediator. This leads to a reduced bioavailability of EPO, thereby limiting its interaction with other isoforms of EPOR. The expression of this isoform results downregulated during hypoxia, which triggers a mechanism that opposes full-length EPOR (Soliz et al., 2007).
Even if the presence of all these different isoforms paves the way for new interesting mechanisms through which EPO could exert its function in the brain, many questions remain unanswered. Indeed, it is necessary to investigate how EPO’s binding to each isoform activates a specific intracellular pathway, modulating the expression of target genes. Once these mechanisms have been elucidated, it will be possible to develop new isoform-selective drugs, resulting in a more specific therapy.
EPO Is Involved in Canonical Cellular Pathways of Neurodegeneration
EPO’s production and function in the brain are now widely investigated. Multiple studies
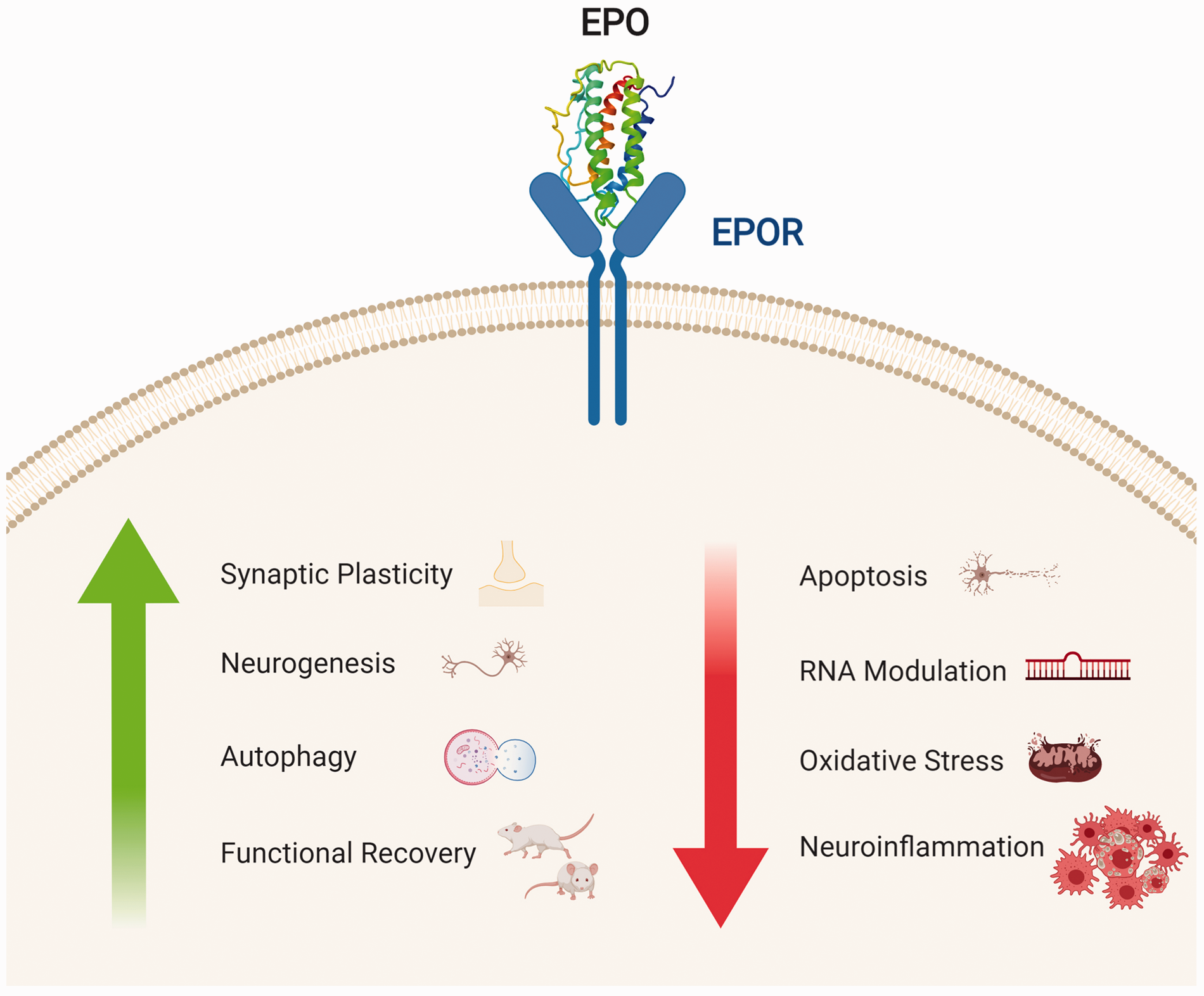
Neurotrophic role of EPO’s binding with EPOR. In the CNS, EPO’s binding to its receptor leads to an increase in neuroprotective actions (synaptic plasticity, neurogenesis, autophagy) with an overall functional recovery in animal models. Conversely, the binding leads to a decrease in apoptosis, expression of miRNAs regulating the apoptotic process, oxidative stress, and neuroinflammation. Made in ©BioRender—biorender.com
Regulation of Cellular Proliferation: Effects on Neurogenesis and Apoptosis
Considering the fact that EPO’s primary function is the regulation of the development and maturation of erythroid cells, it is worth investigating whether a similar function could be exerted by targeting neuronal cells. EPO’s effects on neurogenesis are now widely studied, representing a key pathway through which EPO could exert its neuroprotective roles in neurodegenerative diseases (Chong et al., 2013). Furthermore, the ablation of the canonical isoform of EPOR in knockout murine models results in massive apoptosis, associated with a reduction of neural progenitor cells and death at Embryonic Day 13.5 (Yu et al., 2002). As EPO’s effects on neurogenesis are pleiotropic, they may concern neural precursors cells (NPCs) of different origin as well and may include increased proliferation of progenitors with accelerated differentiation, a process described in hypoxic mesencephalic precursors cells (Studer et al., 2000). Likewise, similar effects of EPO in neural stem cells isolated from both murine E14 ganglionic eminence and adult subventricular zone have been characterized (Shingo et al., 2001). Furthermore, EPO seems to play a pivotal role in the enhancement of hippocampal neurogenesis (Osredkar et al., 2010; Hassouna et al., 2016). EPO-led neurogenesis was also observed in spinal cord-derived neural stem cells (Zhang et al., 2018). EPO can also indirectly promote neurogenesis through the activation of brain-derived neurotrophic factor (BDNF; Wang et al., 2004). Besides increasing neurogenesis, EPO can inhibit the apoptosis process (Merelli et al., 2015). The activation of EPOR downstream molecules JAK2 and PI3K, together with the regulation of the apoptotic proteins Bad and Bcl-xL, leads to a decrease in the apoptosis process (Bartesaghi et al., 2005; Shang et al., 2012; Hernández et al., 2017).
Modulation by Oxygen: Implications of Hypoxia and ROS Formation
EPO is perhaps the first ever protein whose expression was reported to be oxygen-dependent (Jelkmann, 2007). The discovery of the HIF-1α was indeed made possible by targeting EPO itself as downstream protein regulated by hypoxia (Semenza et al., 1991). Today, however, HIF-2α, also known as EPAS-1 (endothelial PAS domain protein 1), seems to be more critical than HIF-1 in overexpressing EPO during hypoxia (Chavez et al., 2006; Haase, 2013). Although the classical hypoxia-dependent EPO/EPOR system was long believed to target the kidney-erythroid axis, today, the brain axis is highlighted as an important protective mechanism, based on the observation that EPO and EPOR are expressed in those regions of the brain that are most sensitive to hypoxia, that is, the hippocampus and the telencephalon (Digicaylioglu et al., 1995). Thus, it is tentative to speculate that EPO’s physiological role in the brain is to act as a protective agent against hypoxia (Rabie and Marti, 2008) and, possibly, ischemia. To this end, the role of EPO in brain ischemia has been widely studied and will be discussed in depth in a subsequent section. Even so, it is worth noting that ischemia-preconditioned neurons protect neighboring astrocytes through an EPO-mediated activation of antioxidant proteins and enzymes (Wu et al., 2017). ROS also have a role in perpetrating tissue damage and neuronal death in numerous other neurodegenerative disorders (Kim et al., 2015; Liu et al., 2017b). More specifically, EPO antioxidant mechanisms reduce beta-amyloid-induced cell death (Li et al., 2008) and increases tyrosine hydroxylase (TH)-positive neurons in a rat model of Parkinson’s disease (PD; Erbaş et al., 2015).
If EPO and HIF-1 respond to hypoxia, it is tempting to believe that excess oxygen, or hyperoxia, has opposite outcomes. Despite being a straightforward paradigm, literature is, however, very controversial. Although a few early studies support this view (Fletcher et al., 1973; Kokot et al., 1994a, 1994b), more recent studies testing the effects of transient hyperoxia on EPO blood levels show increased, not decreased, circulating EPO (Balestra et al., 2006, 2004; Donati et al., 2017; Kiboub et al., 2018). This has also been confirmed in a clinical trial (Hyperoxia, Erythropoiesis and Microcirculation in Critically Ill Patient, NCT02481843). Still another study shows a significant increase of EPO production after a short-term hyperoxic stimulus, only when preceded by antioxidants administration (Momeni et al., 2011; Rocco et al., 2014). This apparently paradoxical response has a strong mechanistic background. Indeed, despite their name, HIFs are stabilized not only by hypoxia but also by hyperoxia. This feature was observed in rats’ brains exposed to 50% oxygen for 3 weeks (Benderro et al., 2012) as well as in mice exposed to 30% oxygen (Terraneo et al., 2017). These key findings are indirectly confirmed in growing prostate cancer (Terraneo et al., 2014), myocardium (Wikenheiser et al., 2009; Zara et al., 2012; Gyongyosi et al., 2018) as well as newborn rats hepatocytes and liver hemopoietic cells (Marconi et al., 2014). It has been pointed out that the main reason for the high sensitivity of HIF’s to hyperoxia may not be related to HIF mRNA but rather to a decreased expression of the prolyl hydroxylases that degrade and address HIF’s toward ubiquitination (Terraneo and Samaja, 2017). The generation of oxidative stress in both hypoxia and hyperoxia may play an additional pivotal role in HIF stabilization and hence EPO production. Although ROS blockade is clearly associated with increased prolyl hydroxylation and therefore decreased HIF activity (Chandel et al., 2000; Brunelle et al., 2005; Guzy et al., 2005; Kaelin, 2005), it is not clear in which direction lack or excess oxygen modulates the oxidative stress in the brain. For example, long-term 30% oxygen cause less oxidative stress than 10% oxygen (Terraneo et al., 2017). Nevertheless, the recent finding of a molecular mechanisms for HIF-1α activation alternative of hydroxylation, which depends on the cAMP-PKA pathway, could provide an explanation for this apparently contradictory behavior (Bullen et al., 2016). The mechanism through which EPO/EPOR interaction modulates oxidative stress has been summarized in Figure 3.
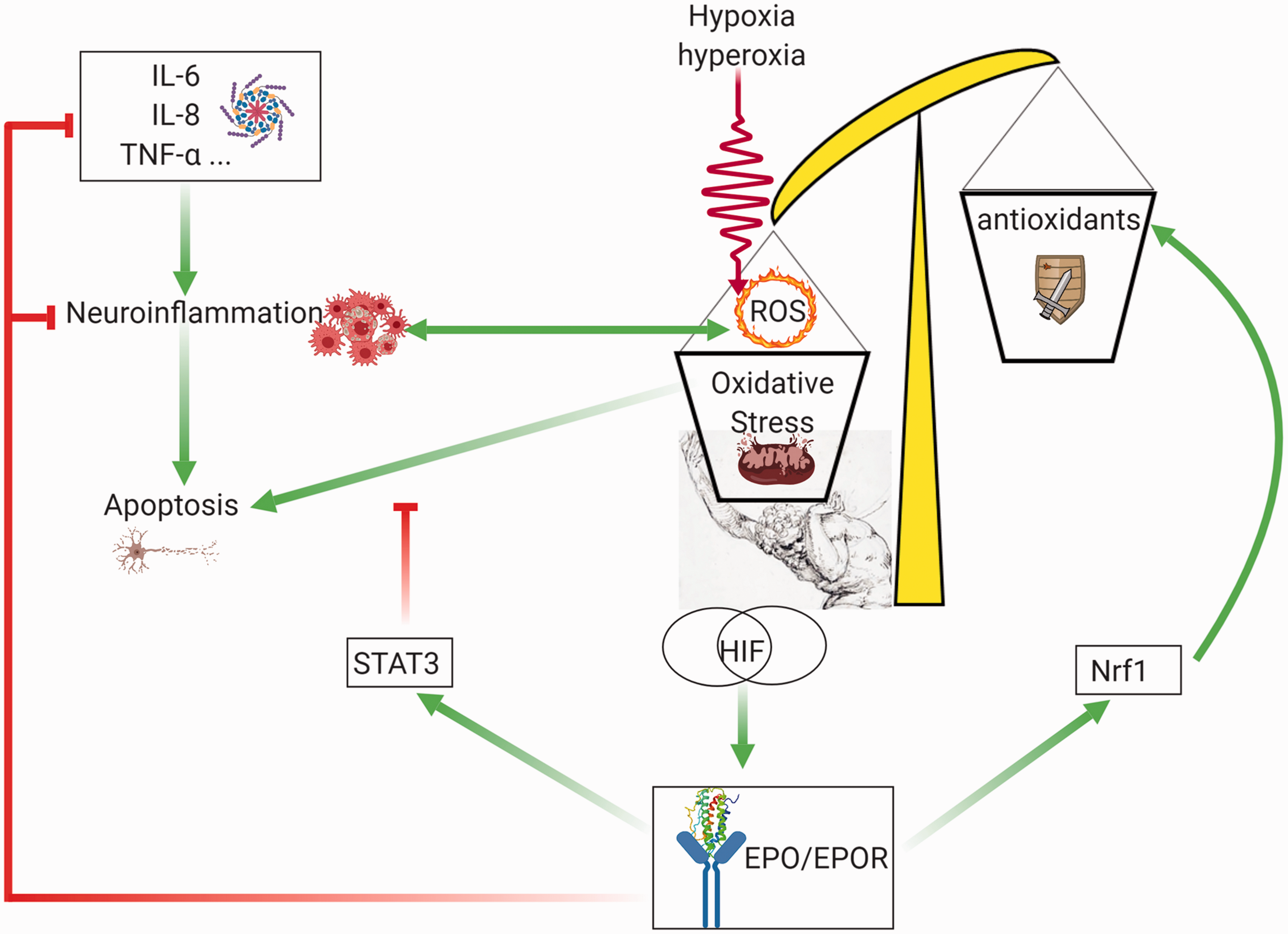
Role of the EPO/EPOR pathway in opposing the oxidative stress induced by hypoxia or hyperoxia. Both hypoxia and hyperoxia are sources of oxidative stress and activate the HIF pathway, which leads to increased EPO expression and EPO/EPOR interaction, which activates the antiapoptotic STAT3 pathway, and increases the antioxidant enzymes expression through the Nrf1 pathway. Green arrows: activation. Red lines: inhibition. Made in ©BioRender—biorender.com
EPO’s Function in Suppressing Neuroinflammation
EPO has emerged as a multifunctional tissue-protective cytokine, and this is partially due to its anti-inflammatory effects (Wang et al., 2011; Sanchis-Gomar et al., 2014). Indeed, when rhEPO is administered in the setting of focal brain ischemia in rats, it crosses the blood–brain barrier and reduces injury severity by 50% to 75% (Brines et al., 2000). rhEPO appears effective in preclinical models of other types of injury in the nervous system whose gravity is exacerbated by inflammation (Sugawa et al., 2002). In a preclinical model of multiple sclerosis (consisting in experimental autoimmune encephalitis), rhEPO reduced the production and release of proinflammatory cytokines and chemokines, and the influx of inflammatory cells into the injured tissue (Agnello et al., 2002). EPO’s neuroprotective effect is mediated by EPOR; this is particularly relevant during hypoxia, where it has been proposed to contribute to endogenous neuroprotection during carotid endarterectomy (Fantacci et al., 2006; Carelli et al., 2016a; Terraneo et al., 2017; Terraneo and Samaja, 2017). The mechanism whereby EPO exerts its neuroprotective action might involve activation of the antiapoptotic STAT3 pathway (Ponce et al., 2013; Jia et al., 2014), the increase of antioxidant enzymes expression, and the reduction of ROS production (Bartesaghi et al., 2005; Chavez et al., 2006).
Novel Potential Targets: Modulation of RNA Expression
In recent years, the role of the noncoding epigenome in disease mechanisms is gaining more and more importance. Among the multiple mechanisms implicated in neurodegeneration, a new attention has been posed on the alteration of RNA processing and function in neurodegenerative diseases (Liu et al., 2017a). New studies have implicated more and more different classes of noncoding RNAs (ranging from microRNAs to long noncoding RNAs) in neurodegeneration (Salta and De Strooper, 2017). An epigenetic activity for EPO has been reported for erythroid cells proliferation (Perreault et al., 2017), but a similar regulation in the brain is yet to be fully characterized. Interestingly, EPO downregulates two microRNA molecules (miR-451 and miR-855-5p) in the SH-SY5Y neuroblastoma cell line (Alural et al., 2014). This downregulation seems to be responsible for the neurotrophic, neuroprotective, antioxidant, and antiapoptotic effects observed after EPO’s administration in SH-SY5Y (Alural et al., 2014). EPO’s downregulation of miR-451 also has a possible implication in the progression of glioblastoma (Alural et al., 2017). Further studies by transcriptomic approach investigated the deregulated RNAs which could be associated with EPOR’s signaling (Sollinger et al., 2017). Indeed, EPOR deletion in rodent fetal NPCs is associated with altered expression of key transcriptional and epigenetic regulators in the developing CNS. Multiple transcriptomic analysis revealed different factors involved, such as REST and NRF1. REST is an epigenetic modifier involved in neural differentiation and plasticity (Gao et al., 2011). NRF1 is a key regulator of antioxidant response and mitochondrial biogenesis (Lee et al., 2011). These two factors could act mediating EPO’s downstream pathway in the developing CNS (Sollinger et al., 2017).
EPO’s Implications in Neurodegenerative Diseases
To this day, there is no curative therapy for the treatment of neurodegenerative diseases. Along with the canonical first line therapies used for each specific neurodegenerative disease, new therapies are now being investigated. Epigenetic therapy, gene therapy, stem cells therapy, and even 3-D organoids are gaining more and more relevance in this context (Sakthiswary and Raymond, 2012; Coppedè, 2014; Piguet et al., 2017; Bordoni et al., 2018). Even so, the potentiality of a single-drug therapy is not to be underestimated, and EPO’s role as a therapeutic agent in neurodegenerative diseases is clearly worth investigating (Brines and Cerami, 2005).
Parkinson’s Disease
PD is the second most common neurodegenerative disorder. Its symptoms can be both motor (bradykinesia, rigidity, and tremor) and nonmotor (these can precede the manifestation of the disease; Tysnes and Storstein, 2017). The two main pathological hallmarks are the death of dopaminergic neurons in the substantia nigra pars compacta (Wirdefeldt et al., 2011) and the presence of alpha synuclein aggregates, noted as primary components of Lewy bodies (Benskey et al., 2016). These aggregates lead to cellular toxicity with an increase in oxidative stress and neuroinflammation (Benskey et al., 2016). Dopaminergic agonists (such as L-3,4-dihydroxyphenylalanine, L-DOPA) are the first line of therapy for PD treatment, but they are only a symptomatic remedy as they do not provide permanent or disease-altering effects (Nutt and Wooten, 2005). Various studies have reported EPO’s ability to counteract numerous processes altered in PD, such as oxidative stress, inflammation, and mitochondrial dysfunction (Jang et al., 2016). Neuroprotective effects have been demonstrated in numerous
EPO is involved in neuroinflammation modulation with different neurotrophic actions on astrocytes, microglia, and neurons (Bond and Rex, 2014). Indeed, EPO is able to preserve the blood–brain barrier through the inhibition of microvascular endothelial cells’ apoptosis and astrocytes’ activation (Maiese et al., 2012). Specifically, astrocytes respond to EPO’s presence through the activation of JNK and p38-MAPK, a decrease of glial swelling, and a reduction of blood–brain barrier permeability (Gunnarson et al., 2009; Tang et al., 2013). It has also been shown that the upregulation of the stress-induced intermediate filament protein is associated with glial hypertrophy, and it is decreased by EPO, independently of the MAPK pathway (Gonzalez et al., 2013). Moreover, EPO may directly influence the reactive microglia, which has the ability to engulf dying neurons through the increased expression of phosphatidylserine receptor, which specifically targets apoptotic neurons (De et al., 2002; Bond and Rex, 2014). EPO decreases the
Different signaling effects of EPO as a neuroprotective molecule have been tested
Alzheimer’s Disease
Alzheimer’s disease (AD) is the most common form of dementia, clinically characterized by a progressive loss of memory and a decline in cognitive functions (Karch et al., 2014). The first evidences indicating that EPO could have a role in improving cognitive functions arose from observations relating to nonneurological diseases, where patients in hemodialysis treated with EPO demonstrated an improvement in cognitive functions (Grimm et al., 1990; Temple et al., 1995). These early observations led to more in-depth studies aiming to characterize the pleiotropic effects of the molecule and its potential benefits in diseases where cognitive function was impaired, such as AD (Rabie and Marti, 2008; Hernández et al., 2017). At a cellular level, the hallmarks of the disease are the presence of extracellular neuritic plaques (due to Aβ aggregation) and intracellular neurofibrillary tangles (characterized by a hyperphosphorylation of the tau protein; Blennow et al., 2006). So far, no curative treatment has been found for AD, and classic pharmacological therapy includes acetylcholinesterase inhibitors, N-methyl-D-aspartate antagonists, and their possible combination (Madav et al., 2019). For this reason, it is necessary to evaluate the therapeutic potentials of new approaches and molecules, such as EPO. Indeed, multiple studies have already been performed, showing promising results (Sun et al., 2019). The therapeutic effects of EPO have been first studied at a cellular level, using both stable cell lines, such as PC12 (Li et al., 2008), and primary hippocampal neurons (Viviani et al., 2005). The molecule seems to exert its effect through the blockade of the apoptotic pathway and the protection from Aβ toxicity (Ma et al., 2009; Esmaeili Tazangi et al., 2015). Moreover, it is possible to observe a taming of the neuroinflammatory response (Shang et al., 2011, 2012) and an increase of antioxidant mechanisms (Li et al., 2008). Interestingly, EPO also shows a neurotrophic function in primary hippocampal neurons through the expression of BDNF (Viviani et al., 2005).
Most of these neuroprotective effects observed
Amyotrophic Lateral Sclerosis
Out of all the motor neuron diseases, amyotrophic lateral sclerosis (ALS) is the most prevalent. It is characterized by the progressive loss of upper and lower motor neurons, leading to muscle atrophy, paralysis, and death 2 to 5 years after the first diagnosis (Wijesekera and Leigh, 2009). Perhaps the most tragic effect of ALS, which eventually leads to patients’ death, is the respiratory muscle weakness, which causes ventilator insufficiency and tissue hypoxia (Carilho et al., 2011). This observation led to the investigation of the role of EPO in ALS. Initial studies focused on the expression levels of EPO in cerebrospinal fluid, serum, and plasma of ALS patients. Studies demonstrated a decrease of EPO levels in ALS patients’ cerebrospinal fluid but not in patients’ serum (Brettschneider et al., 2007; Widl et al., 2007). Further studies indicated that EPO levels were decreased in the plasma of only a subset of ALS patients, which are those that presented with respiratory failure (Carilho et al., 2011).
Spinal Cord Injury
Spinal cord injury (SCI) is a debilitating condition caused by high-energy trauma. It is characterized by severe neurological dysfunctions ranging from sensation loss to partial or complete limb paralysis (Profyris et al., 2004). The trauma leads to the release of toxic substances such as free radicals, lipid peroxidase, eicosanoids, and glutamate, causing neuronal and glial apoptosis (Hock, 1998; Profyris et al., 2004; Mofidi et al., 2011). The main strategy to promote recovery following SCI is the reduction of secondary degeneration occurring after the initial injury. There is now no curative therapy, and the current main therapeutic strategy is the reduction of secondary degeneration through the administration of high-dose corticosteroids, surgical stabilization, and decompression (King et al., 2007; Furlan et al., 2011). Various pharmacological agents are currently being evaluated in humans and animals, aimed at reducing SCI consequences, and EPO is resulting to be a very promising candidate (Celik et al., 2002; Leist et al., 2004; Gorio et al., 2005; Cetin et al., 2006; Carelli et al., 2014, 2015, 2017a). The beneficial neuroprotective and neurotrophic effects have been demonstrated
Stem cells and stem cell-derived molecules have shown beneficial effects to a variable extent in the treatment of SCI (Martino and Pluchino, 2006; Romanko et al., 2007). Er-NPCs also show a beneficial effect when applied to SCI, leading to a promotion of axonal regeneration and an increase in TH-positive fibers in the ventral portion of the lumbosacral cord of the treated mice (Carelli et al., 2014, 2015, 2017a). Furthermore, Er-NPCs modify the microenvironment reducing the production of proinflammatory cytokines, such as tumor necrosis factor alpha and IL-6, and the infiltration of inflammatory cells (Carelli et al., 2014, 2015, 2017a). EPO also induces neurogenesis increasing BDNF’s activity (Mofidi et al., 2011; Carelli et al., 2015). To this day, only one clinical trial has been designed to study the role of rhEPO in SCI (Costa et al., 2015) in which it has been shown that the administration of rhEPO in acute SCI is safe. Unfortunately, the limited number of patients recruited and the short observational period were not sufficient to demonstrate EPO’s efficacy (Costa et al., 2015).
Brain Ischemia
Ischemic stroke is due to transient or permanent reduction in cerebral blood flow, generally caused by the occlusion of a cerebral artery, an embolus, or local thrombosis, which cause acute ischemia stroke (AIS; Dirnagl et al., 1999). The most common consequence of AIS is hypoxia, or the imbalance between oxygen supply and consumption (Dirnagl et al., 1999). Activation of HIF’s upregulates downstream factors, such as the vascular endothelial growth factor and EPO, which exert a neuroprotective action against ischemic damage (Dirnagl et al., 1999). Further consequences of ischemic strokes are the increase in ROS, as well as the inflammatory response by means of induction of proinflammatory genes, as tumor necrosis factor alpha and interleukin 1 beta (Ruscher et al., 1998). In
Several experimental works demonstrated the involvement of the EPO/EPOR system in neuroprotection (Rothwell and Hopkins, 1995). rhEPO administration prevented ischemia-induced learning disability by rescuing hippocampal CA1 neurons from lethal ischemic damage (Lewczuk et al., 2000), while another study showed that the endogenous EPO/EPOR system helps protecting hypoxic astrocytes and oligodendrocyte precursor cells, showing that the suppression of endogenous EPO in astrocytes implies decreased protection of oligodendrocyte precursor cells and cell death (Bernaudin et al., 2000). This hypothesis is further supported by the fact that in the mammalian brain exposed to ischemic infarct or hypoxic damage, the EPO/EPOR system results activated and EPOR is expressed in neuronal fibers (Shingo et al., 2001). Other studies demonstrate that even though EPOR transcription is not directly influenced by hypoxia
A few clinical trials tested the potential effects of EPO in AIS patients. The safety of rhEPO was demonstrated, showing that rhEPO, when infused intravenously 3 days after stroke, can pass the blood–brain barrier reducing the infarct size (Ehrenreich et al., 2002). Another study tested EPO’s effects on long-term neurological outcomes in AIS patients and evaluated EPO’s safety and benefits (Tsai et al., 2015). By contrast, a meta-analysis supporting the view that the efficacy evidence is inconsistent indicates that EPO’s administration in AIS patients is not recommended (Yao et al., 2017). It is thus evident that more studies are needed to assess rhEPO efficacy in AIS patients.
Brain Hypoxia and Hyperoxia
The discovery that HIF-1α binds to the EPO enhancer sequence in response to hypoxia (Semenza and Wang, 1992) univocally addresses the key role of EPO/EPOR to protect the neurological functions in brain tissue challenged not only by hypoxia (Sanchez et al., 2009; Terraneo et al., 2017), but also by ischemia (Sakanaka et al., 1998), carotid clamping (Carelli et al., 2016a), and hypoxia/reoxygenation (Kato et al., 2011). The most relevant mechanisms underlying neuroprotection involve either the prevention of neuronal apoptosis through upregulation of Akt phosphorylation (Chong et al., 2002; Caretti et al., 2008) or the increase of vascular NO availability through activation of eNOS (Beleslin-Cokic et al., 2004). In support of these mechanisms, treatment with rhEPO may provide clues for EPO-based therapy in pediatric chronic hypoxia by rescuing hypoxia-induced depression in hippocampal neurogenesis and oligodendrocyte progenitor populations (Chung et al., 2015), as well as in improving neurologic outcomes in newborns affected with hypoxic-ischemic encephalopathy (Zhu et al., 2009). To prevent erythropoiesis, carbamylated EPO, a derivative devoid of erythropoietic activity, represents a solid pharmacological tool to protect brain in severely hypoxic mice (Fantacci et al., 2006).
As stated earlier, hyperoxia may represent a valuable bench to test the hypothesis linked to the endogenous hypoxia-induced, EPO-mediated neuroprotection. The effect of experimental hyperoxia on brain EPO overexpression was examined in two studies with controversial outcomes. While breathing 50% oxygen for 3 weeks upregulates HIF-2α and EPO expressions in mouse brain (Benderro et al., 2012), breathing 30% oxygen for 4 week increases EPOR, but not EPO expression (Terraneo et al., 2017). As the EPO/EPOR system of neuroprotection requires the availability of both EPO and EPOR, it remains to be understood whether the recruitment of this system depends only on oxygen availability or other components are to be ruled in.
Future Challenges
A critical attention needs to be put to avoid the risk of adverse reactions, which would involve the hematopoietic system when EPO is chronically administered. To this end, EPO analogues are currently being investigated (Sun et al., 2019). These can present reduced erythropoietic activity or be posttranscriptionally modified to yield a molecule with enhanced clearance or selectively target the EPOR receptor (King et al., 2007; Armand-Ugón et al., 2015; Collino et al., 2015; Culver et al., 2017; Yoo et al., 2017). The development of novel EPO analogues with a more specific action and fewer side effects necessarily requires deeper investigation to decipher the relationship between the different isoforms of the receptor and the different functions of EPO.
Conclusions
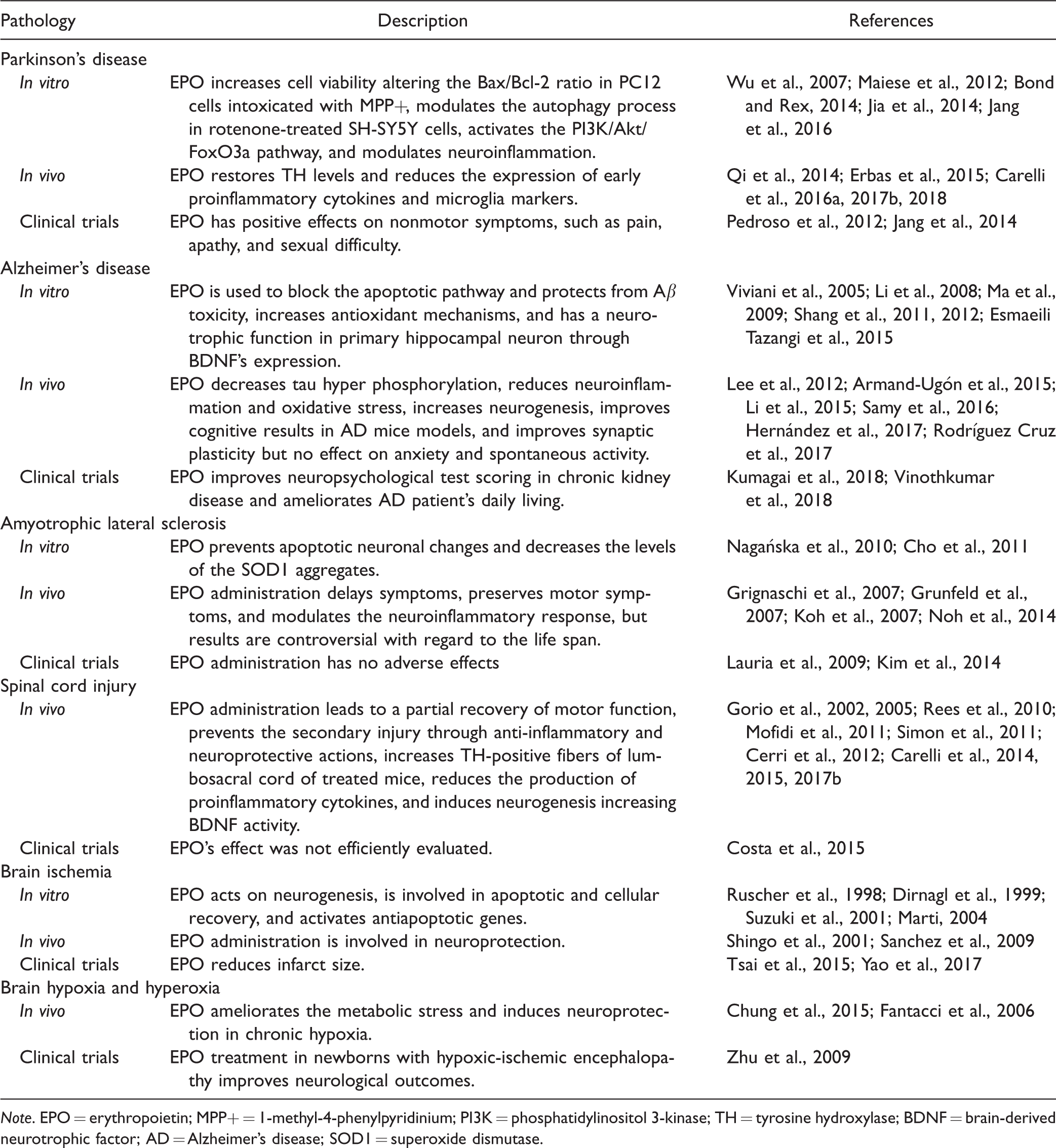
Considering all the advances made in the recent years, EPO has shown great promise in the treatment of neurodegenerative diseases. It is able to target multiple pathways, which are deregulated in neurodegeneration, leading to an overall neuroprotective effect (Table 1).
Summary of the Studies Describing EPO’s Neurotrophic Effect in Neurodegenerative Diseases.
Summary
EPO is a cytokine with a broad action on the CNS, where it is able to target multiple pathways that are deregulated in neurodegenerative diseases, leading to an overall neuroprotective effect.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the economic support of the “Neurogel-en-Marche” Foundation (France), AUS Niguarda Onlus (Italy) to S.C., and Fondazione “Romeo and Enrica Invernizzi” to A. M. D. G.
