Abstract
Nicotinic acetylcholine receptors (nAChRs) are cationic channels of the neuronal cell membrane, differentially expressed in the central nervous system which, when activated by endogenous acetylcholine or exogenous nicotine, are able to enhance cholinergic transmission. The aim of this study was to investigate in human perinatal age the immunohistochemical expression of the α7-nAChR subtype, given its involvement in neuronal differentiation and its significant vulnerability to the toxic effects of nicotine. Thirty fetuses (with a gestational age between 25 and 40 weeks) and 35 infants (1–6 months old), suddenly died of known (controls) and unknown causes (unexplained deaths), with smoking and nonsmoking mothers, were included in this study. A negative or low immunoexpression of α7-nAChRs, indicative of their inactivation, was observed in the granular layers of the cerebellar cortex in 66% of the sudden unexplained perinatal deaths and 11% of the controls. A high correlation was also observed between these findings and maternal smoking. Apart from the well-known adverse effects of nicotine exposure during pregnancy, it may also cause significant alterations in cerebellar cholinergic transmission in areas of the brain involved in vital functions. These events may give us insights into the pathogenetic mechanisms leading to sudden unexplained fetal and infant death.
Keywords
Introduction
Cigarette smoking is a major public health problem which is responsible for the death of approximately six million people worldwide every year. Tobacco exposure not only exerts deleterious effects on the health of adults but also on fetuses and newborns (Mathers et al., 2006; Prokhorov et al., 2006; Rogers, 2008; Cornelius and Day, 2009). In fact, nicotine absorption can cause profound damage during gestation and the early stages of life, especially to the brain, since it is still immature and particularly vulnerable to toxic agents (Lichtensteiger et al., 1988; Franco et al., 2000; Pusiol et al., 2016; Roncati, Piscioli, et al., 2016; Roncati, Termopoli, et al., 2016).
Smoke particles carry the nicotine into the lungs, where it is absorbed rapidly into the pulmonary circulation and then reaches the brain by crossing the blood–brain barrier, thanks to its high liposolubility (Dani and De Biasi, 2001; Benowits, 2010). Within the brain, nicotine binds to nicotinic acetylcholine receptors (nAChRs), which are widely distributed cation channels on the neuronal cell membrane, normally responding to endogenous neurotransmitter acetylcholine (ACh). These receptors are so named due to their ability to bind with exogenous nicotine agonists (Court and Perry, 1995; Dajas-Bailador and Wonnacott, 2004; Govind et al., 2009), even if other full and partial agonists have been identified, such as epibatidine, carbachol, DMXB-A, and so on (Jones et al., 2012).
A variety of experimental approaches, mainly using transgenic mice, enabled to identify some functionally different nAChR subtypes expressed in the brain, namely nine α subunits (α2 through α10) and three β subunits (β2 through β4), encoded by as many as 12 distinct genes (Lindstrom et al., 1996; Mineur and Picciotto, 2008; Albuquerque et al., 2009; Wu, 2009).
All of these nAChRs, when activated by one of the agonists, open their ion channels, thus allowing the entry of cations (Ca2+, Na+, and K+) through the cell membrane (Itier and Bertrand, 2001). One of the effects of this flow, especially of calcium, into neurons is the release of the neurotransmitter ACh, which is able to promote cholinergic transmission in specific brain areas. Through this mechanism, the nAChRs affect numerous biological processes and have a significant impact on cardiorespiratory functioning and nervous system development (Albuquerque et al., 2000). An ideal system for investigating the potential role of nAChRs on the differentiation of the nervous system is the cerebellum, given the long period required for it to reach full maturity, extending in humans from the early embryonic phases until the first postnatal years (Court et al., 1995). This is demonstrated by the presence, during this long period of time, of high levels of choline acetyltransferase, the enzyme that catalyzes ACh synthesis (Clos et al., 1989). Unfortunately this long-term developmental process makes the cerebellum, and above all its surface, the cerebellar cortex (CC), particularly vulnerable to a broad spectrum of extrinsic environmental injuries, including nicotine (Fonnum and Lock, 2000).
In previous studies, we reported the morpho-functional alterations of the CC in victims of sudden intrauterine unexplained death syndrome (SIUDS) and sudden infant death syndrome (SIDS) with a smoking mother (Lavezzi, Ottaviani, Mauri, et al., 2006; Lavezzi, Ottaviani, Terni, et al., 2006; Lavezzi et al., 2007; Lavezzi et al., 2013; Lavezzi et al., 2016). In particular, we highlighted the presence of neuronal immaturity with altered apoptotic programs in the most superficial layers, and a strong Purkinje cell (PC) vulnerability, consisting in decreased argyrophilic nucleolar organizer region, neuronal nuclear antigen, and somatostatin expression.
This study provides new insight to this topic by evaluating whether smoke exposure can also lead to the defective expression of nAChRs in CC development, thus causing a dysregulation of cholinergic activity. We specifically focused our study on the α7-nAChR subtype, due to its high expression during neuronal differentiation and high vulnerability to the toxic effect of nicotine, with serious consequences on synaptogenesis and the formation of brain circuits in perinatal life (Broide and Leslie, 1999; Falk et al., 2002). Moreover, we have already reported a defective expression of α7-nAChR in nuclei of the brainstem checking the vital functions related to maternal smoking in both SIUDS and SIDS (Lavezzi et al., 2015).
Therefore, the main goal of this study was to compare the expression of α7-nAChRs in the CC in a large group of fetuses and infants who suddenly died from both known and unknown causes, with smoking and nonsmoking mothers, in order to determine a possible correlation between a dysregulation of these specific cholinergic receptors, nicotine absorption, and sudden unexplained perinatal death.
Materials and Methods
Study Subjects
The study was performed on 65 cases of sudden perinatal death, 30 fetuses, 16 males, and 14 females, ranging between 25 and 40 weeks of gestation (mean age 35 ± 2.3 gestational weeks), and 35 infants who died between 1 and 6 months of age (20 males and 15 females, mean age 3.4 ± 4.2 months), in accordance with the Italian law n.31/2006 “Regulations for Diagnostic Post Mortem Investigation in Victims of Sudden Infant Death Syndrome (SIDS) and Unexpected Fetal Death.” This law states that all infants who die suddenly within the first year of age, as well as all fetuses who die after the 25th week of gestation without any apparent cause, must be submitted to diagnostic postmortem investigation, including the examination of the placental disk, umbilical cord, and membranes in fetuses, and that the findings of the in-depth autopsy and the information concerning the pregnancy, fetal development, delivery, and risk factors must be collected by the medical personnel involved in each case (obstetricians, gynecologists, pathologists, etc.) and recorded in the registry of a National Data Bank.
The subjects were selected from a large series of cases sent to our center over a 10-year period for whom we already had all of the necessary data.
In 47 cases, death was classified as totally unexplained, after having ruled out any possible underlying causes. A diagnosis of SIUDS was established for 21 fetuses and a diagnosis of SIDS for 26 infants. In the remaining 18 cases (nine fetuses and nine infants), the cause of death was established at autopsy (congenital cardiomyopathy and severe chorioamnionitis in fetal deaths; respiratory infection, congenital cardiomyopathy, malaria, and aspergillosis in infant deaths (Roncati et al., 2010; Pusiol et al., 2015). These cases were used as controls.
Clinical histories, which were collected for every subject of the study according to the Italian law, included a questionnaire to be filled in by the mothers regarding their smoking habits. Furthermore, a lock of each victims’ hair was removed to search for xenobiotics and especially for cotinine, which is the main nicotine metabolite characterized by a long half-life and great stability. A qualitative cotinine test kit was routinely used to detect the presence or absence of the substance on the hair structure. In addition, a quantitative analysis by liquid chromatography-mass spectrometry system was carried out, whenever possible, in order to determine cotinine concentration, according to the method used by Tzatzarakis et al. (2012).
Case Profiles of the Study.

gw = gestational weeks; pm = postnatal months; SIUDS = sudden intrauterine unexplained death syndrome; SIDS = sudden infant death syndrome.
Consent
The parents of all of the victims considered in the study provided written informed consent to autopsy, according to the protocol approved by the “L.Rossi” Research Center of the Milan University.
Ethics statement
Permission from the Ethics Committee was not required for this study as the “L.Rossi” Research Center is the national referral center for sudden unexplained fetal and infant death, in accordance with the above mentioned Italian law n. 31/2006. Every effort has been made to ensure the anonymity of the subjects included in the study. Therefore, as no identifying information was collected during our research, none of the authors had access to potentially identifiable information.
In-Depth Anatomopathological Examination of the Cerebellum and Brainstem
In all cases, we carried out an in-depth histological examination of the autonomic nervous system, according to the guidelines specifically developed by the “Lino Rossi” Research Center paying particular attention to the cerebellum, which was the primary aim of this study, as well as the brainstem. The cerebellum was excised from the brainstem by cutting the peduncles. Transversal samples of 60 µm of both cerebellar hemispheres, including all of the lobules (anterior, posterior, and flocculonodular lobule) were then obtained. Samples of the same thickness were also obtained from the midbrain, pons, and medulla oblongata, where the main brainstem structures involved in the control of the vital functions are located. All of the samples were fixed in 10% phosphate-buffered formalin, embedded in paraffin, and serially sectioned. For each level, six to seven 5 µm sections were obtained, two of which were assigned to histological examination using hematoxylin-eosin and Klüver-Barrera stains; adjacent paraffinized sections were submitted to immunohistochemical procedures for the evaluation of the nicotinic receptors. The remaining sections were saved and stained as deemed necessary for further investigations.
The microscopic analysis of the cerebellum was carried out on the cortex layers, that is, the external granular layer (EGL), molecular layer (ML), Purkinje cell layer (PCL), internal granular layer (IGL), and the medullary deep nuclei (dentate nucleus, fastigial nucleus, globose nucleus, and emboliform nucleus).
The histological assessment of the brainstem was performed on the locus coeruleus and the Kölliker-Fuse nucleus in the caudal mesencephalon or rostral pons; the retrotrapezoid nucleus, superior olivary complex, and facial/parafacial complex in the caudal pons; the hypoglossus, dorsal motor vagus, tractus solitarius, ambiguus, pre-Bötzinger, inferior olivary, raphé and arcuate nuclei in the medulla oblongata.
α7-nAChR Immunohistochemistry
The immunohistochemical method for evaluating the expression of α7-nAChR was applied using a specific rabbit polyclonal antibody (aa 22–71, Abcam Ltd, UK, cod. ab10096) on selected transverse brain sections. After dewaxing, rehydration, endogenous peroxidase blocking by 3% hydrogen peroxide treatment, the tissue sections were immersed and boiled in Tris-EDTA buffer for antigen retrieval in a microwave oven. Subsequently, they were incubated with diluted 1:167 primary antibody overnight in a wet chamber. The samples were then washed with phosphate-buffered saline buffer, incubated with a biotinylated goat anti-rabbit IgG secondary antibody (PK-6101, Vector Laboratories, CA, United States) and processed using the avidin-biotin-immunoperoxidase technique (VEDH-4000, Vector Laboratories, CA, United States). Finally, each section was counterstained with Mayer’s hematoxylin and coverslipped. The controls lacking primary antiserum were used to determine the specificity of the reaction. The specificity of the staining was ascertained using an isotype and concentration matched control for the primary antibody (polyclonal rabbit IgG isotype control-Abcam ab27478).
nAChR immunohistochemistry quantification
The IGL of the CC was selected for immunostaining evaluation, due to its high cellular intensity. To quantify the immunohistochemical results, we applied semiautomated criteria. For each case, approximately 30 adjacent fields in the IGL were examined under a Nikon Eclipse E800 light microscope (Nikon Corporation, Tokyo, Japan) equipped with an ocular micrometer. The images of interest were captured using a 40X objective lens and a Nikon Coolpix 8400 digital camera attached to the microscope, at the same settings and exposure times. More precisely, prior to image capturing, the camera was white balanced, and the exposure times were standardized to 0.055 ms. For each image, the following 4-point rating system was used to quantify the degree of immunoreactivity which was expressed as “nAChR-Index” (number of neuronal cells with dark brown color prevalently localized in the cytoplasm, divided by the total number of neurons, and expressed as percentage):
“Class 0” of the nAChR-index for no or very light staining (negativity); “Class 1” when the index was <10% (weak positivity); “Class 2” with a percentage of immunopositive cells ranging between 10% and 40% (moderate positivity); “Class 3” with an index > 40% of the counted cells (strong positivity).
Statistical Analysis
The observations of two independent and blinded pathologists were compared using Kappa statistics in order to evaluate the interobserver reproducibility, according to the Landis and Koch (1977) system. More precisely, a Kappa Index (KI) value of 0 to 0.2 corresponds to “slight agreement”; from 0.21 to 0.40 “fair agreement”; from 0.41 to 0.60 “moderate agreement”; 0.61 to 0.80 “strong agreement”; 0.81 to 1.00 = “very strong” or “almost perfect agreement”, with a value of 1.0 corresponding to “perfect agreement.” The overall KI obtained in this study was highly satisfactory (KI = 0.82).
The statistical significance of direct comparisons between the groups of victims was determined with the one-way analysis of variance. The statistical calculations were carried out with SPSS (statistical package for social science) statistical software (SigmaPlot®, version 13, Systat Software Inc., Chicago, IL). Differences were regarded as statistically significant if the p value was <.05.
Results
According to the purpose of this study, first we report the results obtained from the in-depth histological and immunohistochemical analyses performed on the CC.
Morphological Features of the CC
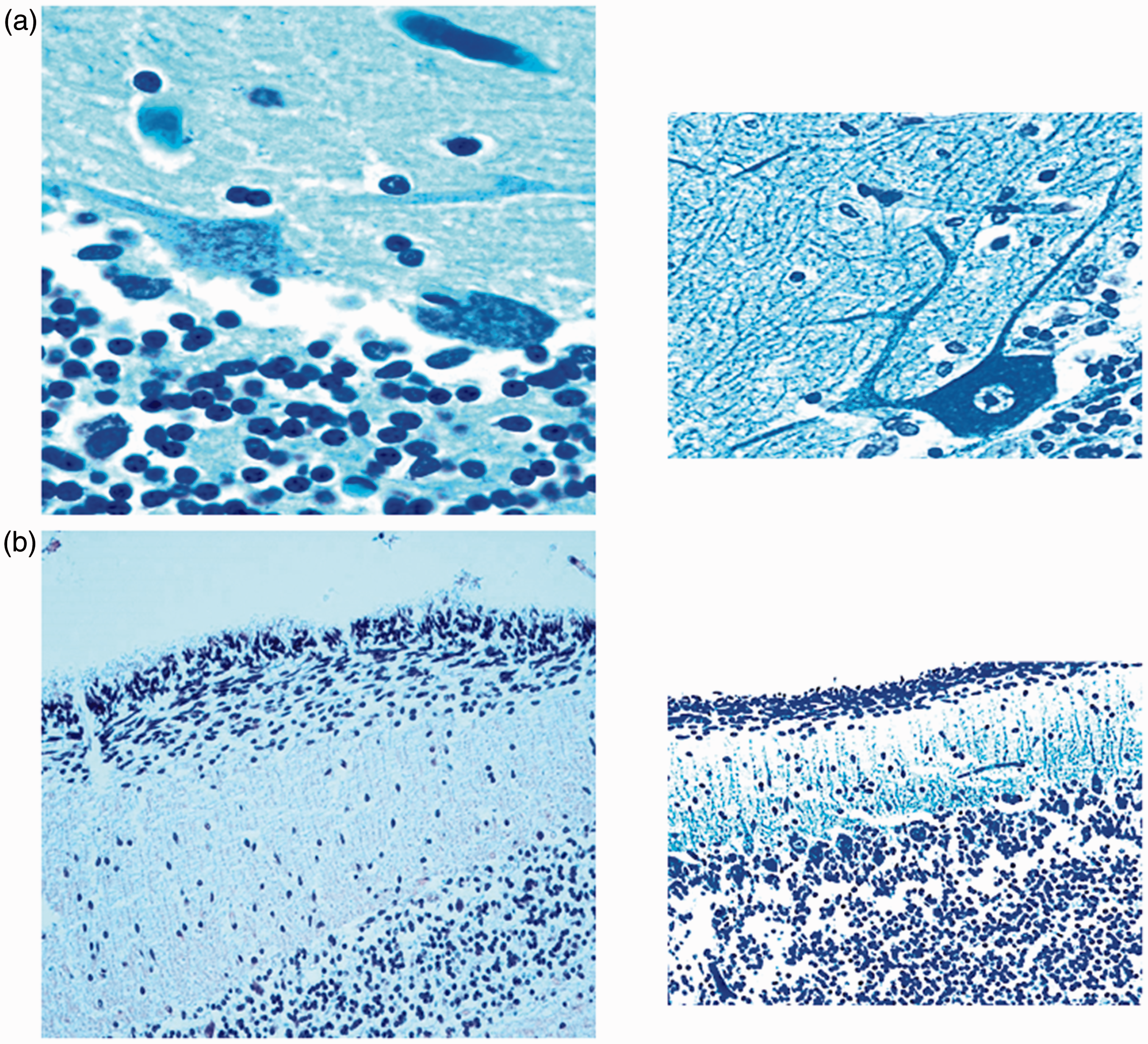
When compared with the normal features of the same developmental stages that were established earlier by us (Lavezzi, Ottaviani, Terni, et al., 2006), abnormal patterns of maturation were observed in 15 SIDS and 12 SIUDS, especially concerning the higher EGL thickness than that expected in relation to age (Figure 1). Alterations of the PCL were also detected in several of these cases. More precisely, the PCs of five SIUDS and seven SIDS were classified as “damaged cells,” as they did not exhibit an evident nucleus and showed shrunken morphology with intensive dark staining of the cytoplasm and rare arborizations (Figure 2(a)). PC loss was observed in extensive stretches of the CC in a further six cases (four SIUDS and two SIDS; Figure 2(b)). The persistence of a thick EGL was only detected in two infants in the control group.
Four-layered structure of the cerebellar cortex. (a) SIDS case 4 month old; (b) age-matched control case. From left to right: external granular layer (EGL), molecular layer (ML), Purkinje cell layer (PCL), and internal granular layer (IGL). In (a) higher thickness of the EGL than that expected in relation to age. Klüver-Barrera stain—Magnification 20×. Morphological alterations of the PC layer. (a) To the left: shrunken PC morphology without a discernable nucleus in a SIDS case (2 month old); to the right: normal morphology of a Purkinje cell in an age-matched control case. (b) To the left: extensive loss of PCs in a SIUDS case (40 gestational weeks); to the right: normal PC layer in an age-matched fetus of the control group.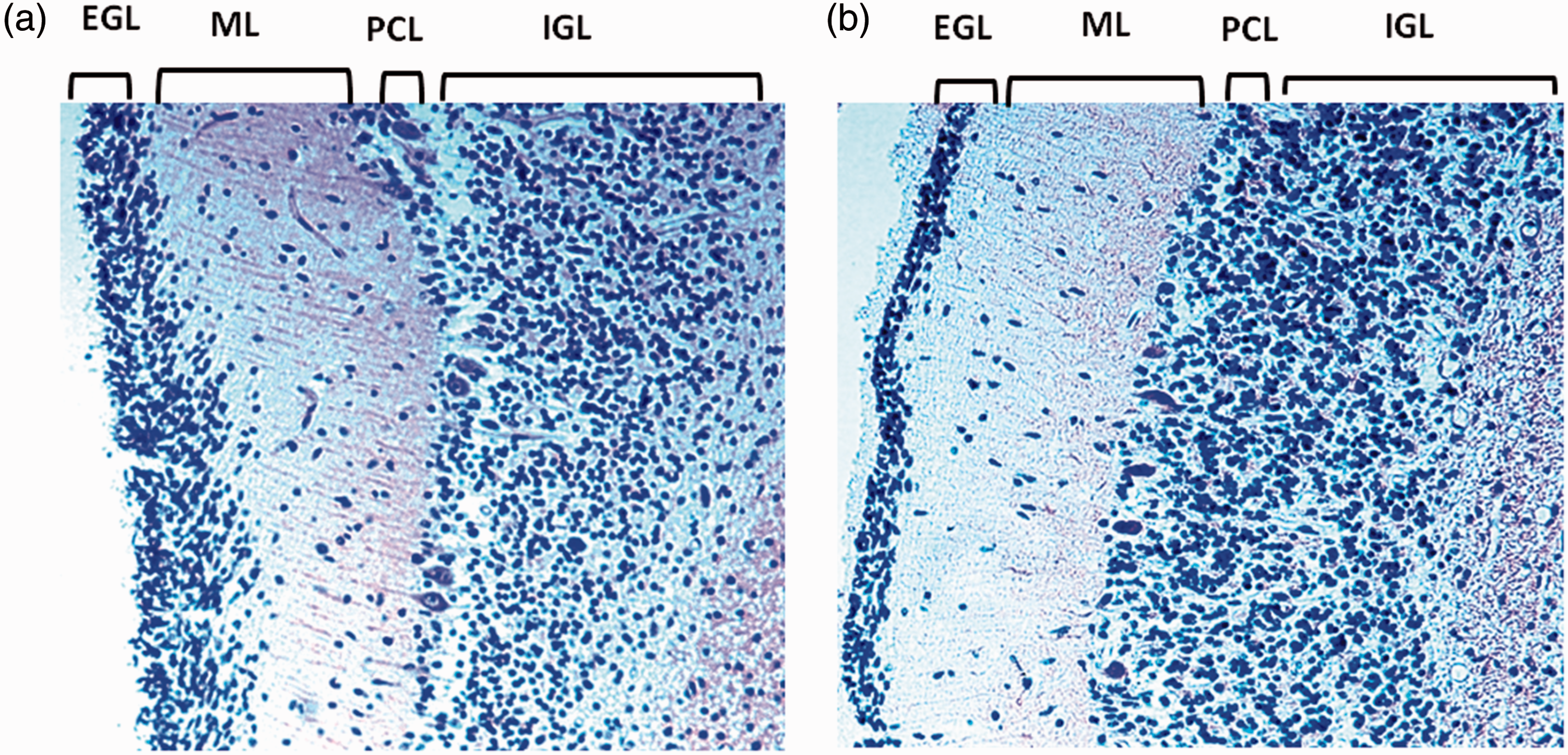
Immunohistochemical Expression of α7-nAChR in CC
Granule cells of the external and IGLs of all the cerebellar lobules showed α7-nAChR immunoreactivity in 8 SIUDS, 8 SIDS, and 16 controls. The percentage of dark-stained neurons, evaluated in the IGL, due to its high cellularity, ranged from 35% to 80% (corresponding to “Class 2” and “Class 3” of nAChR-index; Figure 3(a)). On the contrary, in the remaining 13 SIUDS, 18 SIDS, and 2 controls, regardless of developmental status, the percentage of immunostained neurons in the same layers was very low or totally absent (“Class 1” and “Class 0” of nAChR-index; Figure 3(b)). Overall, SIUDS or SIDS cases had a significantly higher incidence of α7-nAChR defective expression in the cerebellar granule cell layers, when compared with age-matched controls (p < .01). Immunoreactivity was negative in the PCL in all of the cases under study.
Immunohistochemistry for α7-nicotinic receptor subunit in the granule cells of the IGL of the cerebellar cortex. (a) High immunopositivity (“Class 3” of nAChR-index) of a control case (3 month old). (b) Immunonegativity (“Class 0” of nAChR-Index) in a SIUDS case (39 gestational weeks). Magnification 100×. Evidence for significant correlation between SIUDS and SIDS, no or weak nAChR immunoexpression and maternal smoking. Histogram shows the comparison of numbers of cases belonging to the different nAChR-index classes related to maternal smoking. Interestingly, SIUDS and SIDS display a significantly increased number of cases included in Classes 0 and 1 with a smoking mother, compared with controls (p < .01).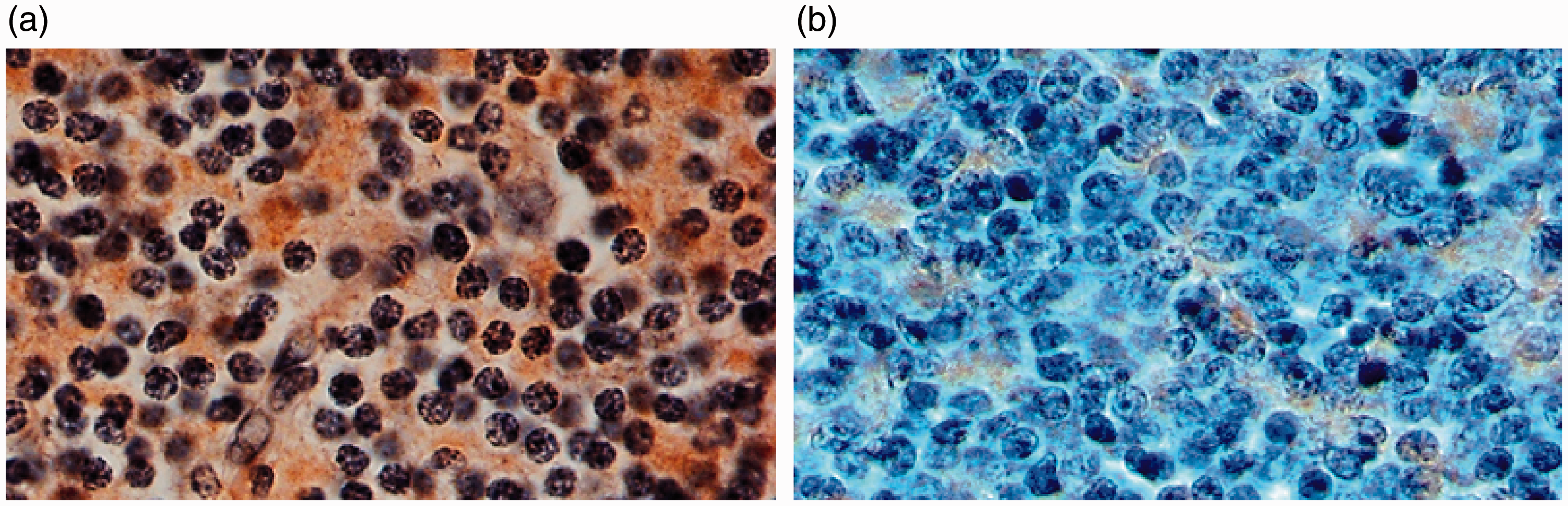
Correlations of the Results With Cigarette Smoke
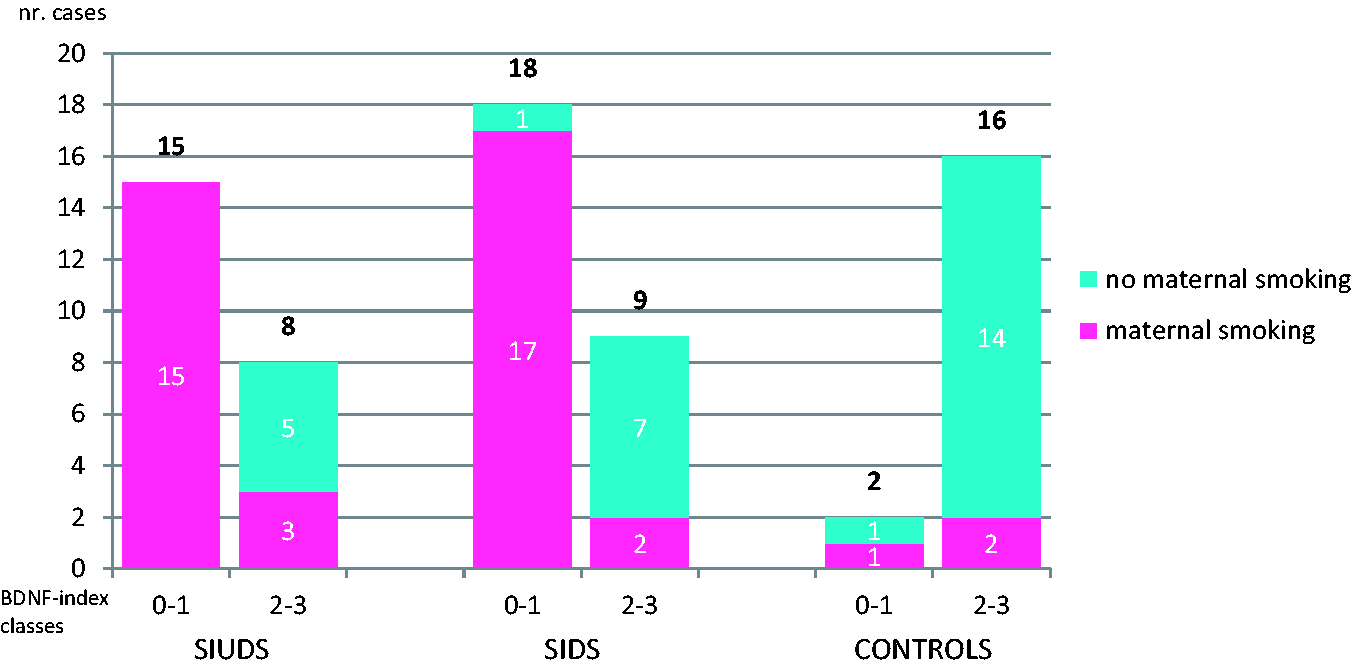
Strong correlation was observed between the immunoexpression of α7 receptors in CC and maternal smoking, validated in all cases by carrying out qualitative cotinine tests on the victims’ hair (p < .01). Moreover, we were able to apply the quantitative test on seven SIUDS and five SIDS, obtaining cotinine values ranging between 0.74 ng/mg and 1.0 ng/mg. Overall, all of the 13 SIUDS, 17 of the 18 SIDS and 1 control case with no or light α7-immunostaining had smoking mothers. Contrariwise, among the 31 cases with consistent α7 nAChR immunopositivity (8 SIUDS, 9 SIDS, and 16 controls), maternal smoking was only documented in three SIUDS, two SIDS, and two victims of the control group.
Figure 4 summarizes the immunohistochemical results obtained for SIUDS, SIDS, and the controls related to maternal smoking.
Neuropathological Examination of the Brainstem
For most of the SIUDS and SIDS cases (14 SIUDS and 16 SIDS), the histological examination of the brainstem showed the hypodevelopment of various nuclei or structures checking the vital functions (hypoplasia or agenesis of one or more nuclei of the raphé system, of the facial/parafacial complex, pre-Bötzinger, Kölliker-Fuse, and arcuate nuclei). Individual victims frequently showed combinations of these alterations. The hypoplasia of the facial/parafacial complex, in terms of decreased neuronal number and complex area, was the most common finding in SIUDS (12/21 cases). More information on the localization of the aforementioned structures and their delayed development can be found in our previous studies (Lavezzi et al., 2004; Matturri et al., 2004; Lavezzi et al., 2008a, 2008b). There was no histological evidence of brainstem abnormalities in any of the control victims, except for hypoplasia of the arcuate nucleus, detected in 22% of cases (4/18).
Positivity for α7-nAChR subunits was generally observed in brainstem nuclei or neuronal complexes that control vital functions, with both normal and delayed maturation. Surprisingly, the cholinergic system of the same subject could be differentially expressed in CC and brainstem nuclei. In fact, we observed at the same time α7-nAChR immunopositivity in hypoplasic brainstem nuclei and α7-nAChR immunonegativity in CC, in eight SIUDS and eight SIDS.
Discussion
To our knowledge, this is the first study carried out on nAChR expression during human CC development and to investigate the adverse effects of nicotine toxicity in this process, in order to gain insight into the pathogenesis of both SIUDS and SIDS.
nAChRs, which are the key molecules that process ACh messages in specific brain regions, are widely expressed in the nervous system, including the cerebellum. The abundance of cholinergic synthetic enzymes during the long cerebellar maturative process (Clos et al., 1989) supports this assertion. More specifically, experimental studies have demonstrated that the dynamic sequence of morphological steps that regularly occur during CC development, mainly consisting in a progressive thinning until the disappearance of the EGL due to cell death or cell migration in the IGL, coincide with the maturation of the cholinergic system (Clos et al., 1989; Court et al., 1995). The activation of the α7-nAChR subtype appears to be particularly involved in the maturation of the granule layers (Caruncho et al., 1997; Opanashuk et al., 2001; Prestori et al., 2013).
Although, as well known, nicotinic receptors can be activated by both the endogenous neurotransmitter ACh and the exogenous ligand nicotine, the effects of nicotine on cholinergic functions are quite complex. In particular, prolonged exposure and absorption of this agonist can incorrectly drive the cholinergic system, ultimately decreasing nAChR responsiveness until desensitization or permanent inactivation (Wang and Sun, 2005; Buccafusco et al., 2009). The consequent inhibition of ACh secretion can be irreversible, with obvious repercussions on many vital functions, especially respiratory activity. This also applies to CC, due to its involvement in breathing control. In fact it is well known that the CC plays a major role in the coordination of muscle activity and functions like a clock to coordinate the time intervals between two successive contractions (Freeman, 1982). This ability is also extended to respiratory muscles, thus giving CC an important role in regulating the respiratory rhythm in normal and adverse conditions, such as in case of hypoxia or hypercapnia (Xu et al., 1994). Therefore, perturbations of cholinergic functions in the CC caused by nicotine exposure can affect breathing patterns with consequent implications for the pathophysiology of neurological disorders involving respiratory control.
The immunohistochemical findings presented provide valuable insight into the nAChR phenotype expressed in cerebellar granular neurons in human perinatal life, thus we can assume that α7-nAChR subunits are specifically involved in the differentiation of both the EGL and IGL. More precisely, intense immunopositivity of α7-nAChR in granular layers was observed in most of the fetuses and infants in the control group (83%) and several victims of the SIUDS or SIDS groups (38% and 31%, respectively). On the contrary, low levels or total absence of α7-nAChR immunoexpression were detected in a wide subset (over 50% of the cases) of sudden fetal and infant deaths, which was highly related to maternal smoking. These results suggest that the adverse effects of nicotine are not evenly exerted on receptor expression, yet show the ability of nicotine to act differentially during CC development. Therefore, it is reasonable to assume that a subgroup of SIUDS or SIDS is more sensitive to nicotine, with another subset that is also resistant to long-term nicotine exposure. Understanding the mechanisms of how, following intense exposure to cigarette smoke, some nChR become desensitized while others do not, is beyond the scope of this study. Nevertheless, further research is required in order to validate our results and to examine the interaction of nicotine with α7-nAchR subunit in the CC more in depth.
It is interesting to note that α7-nAChR immunonegativity in the granular layers is the only neuropathological alteration in many cases of SIUDS and SIDS. In SIDS cases, this can be explained by the fact that if the respiratory muscles do not contract, due to the desensitization of α7-nAChR in CC, it may prevent newborns from breathing after birth and cause death.
However, this does not apply to fetuses lacking α7-nAChRs immunoexpression. Even if SIDS may be caused by respiratory arrest, this cannot be the case when death occurs in utero, when breathing is not yet a vital condition. In this regard, it is essential to focus on the EGL, the predominant component of the CC in fetal life with the aim of finding a possible explanation. This outer layer, which represents the dorsal surface of the developing cerebellum, is derived from the rostral portion of the rhombic lip, the free margin of the hindbrain that surrounds the dorsal opening of the fourth ventricle (Rivas and Hatten, 1995; Wingate, 2001). In humans, the EGL appears in early embryonic development, before the other cortex layers, as a consistent multistratified structure composed of strips of densely packed, small cells. It then thins gradually from birth and completely disappears around the 12th postnatal month (Lavezzi, Ottaviani, Terni, et al., 2006). The stately presence of EGL during intrauterine life has a specific meaning and indicates its essential role before birth.
Our data show that almost all of the EGL cells in most of the control cases significantly express the α7-nAChR phenotype, thus enabling us to foresee a massive release of Ach. We assume that this neurotransmitter not only promotes the differentiation of the EGL neurons during early cerebellar development but also plays an important role in the synaptogenesis, an essential step of brainstem circuit formation, with consequent strong systemic effects on the vital functions.
For these reasons, it is essential to prevent the intrinsic deleterious effects of maternal smoking not only on the fetal and neonatal growth (Billaud and Lemarie, 2001; Wickström, 2007), but also, more specifically, on the maturation and functionality of the CC, particularly of the EGL in fetuses, which can already have potentially serious consequences on the cholinergic activity in utero. Our previous studies have reported on an array of important side effects of nicotine exposure on the developmental process of the different brainstem centers essential for life (Lavezzi, Ottaviani, Mingrone, et al., 2005; Lavezzi, Ottaviani, Matturri, 2005; Lavezzi et al., 2007; Lavezzi et al., 2013; Lavezzi, Ferrero, et al., 2016). The findings of our study add new insights to this field and highlight the fact that the altered expression of nAChR, also limited to CC, puts fetuses exposed to maternal cigarette smoking in utero, at higher risk of SIDS or SIUDS.
Footnotes
Acknowledgments
The authors would like to thank Dr. Graziella Alfonsi and Dr. Alessia Ricaldi for their excellent technical assistance and Ms. Eleanor M. Fabri for the English revision of the manuscript.
Author Contributions
As this collaboration involved researchers from three institutions, each author’s specific contributions are described later: A. M. L. planned the study, analyzed the data, performed the neuropathological examination, and wrote the manuscript, through extensive discussion with L. M. S. F. participated in the study design, analyzed the results, and collaborated in writing the manuscript. L. R., F. P., and T. P. recruited the subjects of the study and performed the autoptic examination. All authors have read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Italian Health’s Ministry in accordance with the Law 31/2006 “Regulations for Diagnostic Post Mortem Investigation in Victims of Sudden Infant Death Syndrome (SIDS) and Unexpected Fetal Death.”
