Abstract
HER2-positive acantholytic squamous cell carcinoma (ASCC) of the breast is exceptionally rare, and its clinicopathologic features are poorly understood. The impact of neoadjuvant therapy on HER2-positive breast ASCC is unclear. Here we report on a 58-year-old woman who was diagnosed with HER2-positive ASCC of the right breast, who underwent neoadjuvant treatment with albumin-paclitaxel, carboplatin, and trastuzumab, and surgery. Neoadjuvant therapy was effective, with no recurrence or metastasis after 1.5 years of postoperative follow-up.
Keywords
Introduction
Squamous cell carcinoma (SCC) of the breast is an uncommon malignant tumor, characterized by more than 90% of malignant cells, independent tumors covering the skin and nipple, and exclusion of primary SCC of other sites. SCC is believed to represent less than 0.1% (0.04%–0.075%) of all malignant breast cancers. 1 SCC comprises several histopathological variants, including warts, spindle-shaped, basaloid, adenosquamous, adenoid, and undifferentiated types.2,3 Acantholytic SCC (ASCC) was first defined as a specific type of SCC in 1947, as a malignant epithelial tumor with a strong glandular pattern extending into the dermis, referred to as adenoacanthoma of the sweat gland.4–6 It was also noted as a rare variant of SCC, with distinctive histological features compared with classic SCC, but its aggressiveness has been interpreted differently by several authors.6–9 The clinical and radiological features of ASCC are nonspecific, the tumors are resistant to therapy, and the prognosis is poor. Treatment of HER2-positive breast cancer also presents a challenge and it has become a focus of global research. In this study, we report on the successful treatment of a patient with HER2-positive ASCC, with important implications for the comprehensive therapy of this tumor.
Case report
Pre-treatment examination findings
In November 2020, a female patient in her late 50s was confirmed with a right breast lump, accompanied by skin swelling and ulceration (Figure 1). She had no family history of breast cancer and there were no abnormalities in other parts of her body. We diagnosed locally advanced HER2-positive breast ASCC (TMN classification: T4N1M0) based on the test results (Figure 2).

Pre-treatment examination findings. (a, b) Physical examination revealed a right breast mass measuring approximately 8 cm × 7 cm. The overlying skin was dark red and the central area was approximately 2 cm × 2 cm. Pus and blood flowed from the mass, but the right axillary lymph nodes were not swollen. (c) Breast ultrasonography showed a 72-mm hypoechoic, heterogeneous solid mass in the right breast. (d) Magnetic resonance imaging confirmed the right breast mass with necrosis and invasion of the surrounding tissue, adjacent skin, and breast fascia

Pre-treatment pathological findings. (a) A cyst in the upper quadrant of the right breast showed squamous cell acantholysis with pseudoadenoid structure (×20, hematoxylin and eosin staining). (b) Cytokeratin 5/6 was overexpressed in cancer cells (×20, immunohistochemistry). (c) HER2 3+ immunoreactivity in tumor cell membrane (×20, immunohistochemistry)
Neoadjuvant chemotherapy treatment
The patient completed six cycles (21 days per cycle) of neoadjuvant treatment with albumin-paclitaxel (260 mg/m2), carboplatin (area under the curve = 5), and trastuzumab (8 mg/kg for the first cycle, 6 mg/kg for the remaining cycles). There were no side effects and the size of the tumor was considerably reduced after chemotherapy (Figure 3).
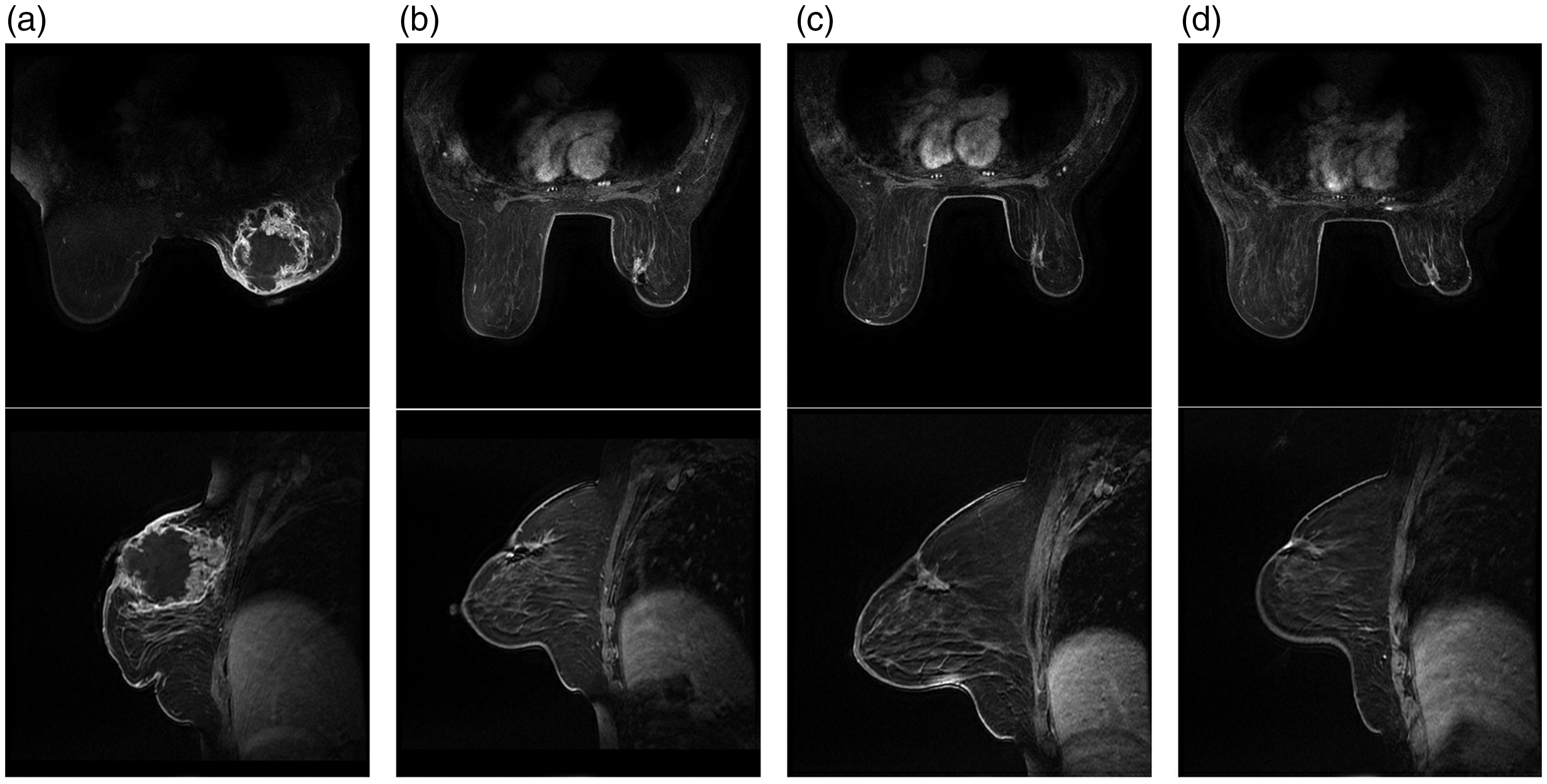
Magnetic resonance imaging (MRI). (a) MRI at the start of neoadjuvant chemotherapy. (b, c, d) MRI showing changes in tumor size after 2nd, 4th, and 6th cycle, respectively, with size reduction from 8 cm × 6.5 cm to 2.4 cm × 1.5 cm
Postoperative test findings
The patient underwent a right mastectomy and level III axillary lymph node dissection. Pathologic tests showed a TMN classification of ypTisN0cM0 with no metastasis in 37 axillary lymph nodes (Figure 4).

Post-treatment examination findings. (a) Physical examination after right mastectomy and grade III axillary lymph node dissection. (b) Pathological findings showed interstitial fibrosis, histiocytic and multinucleated giant cell reaction, local hemorrhage, hemosiderin cell deposition, noninvasive cancer, and some residual ductal carcinoma in situ
Immunohistochemical (IHC) staining results were as follows: catheter in situ: estrogen receptor (ER, (30% moderate), progesterone receptor (PR, 5% moderate), HER2 (3+), Ki-67 (approximately 30% negative), and cytokeratin 5/6 (−); myoepithelium: P63 (+) and calponin (+); and multinucleate giant cell infiltration area: AE1/AE3 (−) and CD68 (histiocyte−). The invasive lesion was determined to have achieved complete remission according to the “Residual Cancer Burden Scoring System” recommended by the “International Breast Collaborative Group” for assessing the effectiveness of neoadjuvant therapy.
The patient did not receive radiotherapy to either the right chest or the right supraclavicular bone. The postoperative pathologic findings were ER (+) and PR (+). She was therefore treated with one dose of oral anastrozole 1 mg for endocrine treatment, and trastuzumab was continued for 17 cycles. The patient was monitored for 1.5 years and examined every 3 months, with no metastasis or recurrence.
Discussion
ASCC of the breast is a distinct tumor entity that has been classified as a variant of SCC, and as a specific type of metaplastic breast carcinoma according to the most recent “WHO classification of tumors 5th edition (2019)”. The histologic features of ASCC are related to the loss of cohesion between tumor cells with typical pseudoglands and pseudovascular structures. IHC may therefore be required to make a differential diagnosis, to rule out eccrine neoplasia and vascular sarcomas.11–13 Pathologists can identify ASCC by IHC detection of endothelial lineage markers (CD31 and CD34) and cytokeratin (high-molecular-weight cytokeratin) in biopsies.14,15 In the current case, a large cyst was detected in the right breast, and the tumor showed squamous epithelial carcinoma components and pseudoadenoid structures, while IHC was positive for vascular endothelial CD34, high-molecular-weight cytokeratin (CK5/6), and broad-spectrum cytokeratin (AE1/AE3). The tumor was also ER- and PR-negative, with a high Ki-67 index, and myoepithelial P63-positive, essentially consistent with the literature. 15 There was no metastasis in the ipsilateral axillary lymph nodes, but the tumor was HER2-positive, which is rare in ASCC.
The efficacy of targeted therapy for HER2-positive SCC is unclear, particularly regarding neoadjuvant therapy. Although some doctors believe the tumors are drug resistant, Usui et al. reported a case in which dose-dense doxorubicin and cyclophosphamide, followed by pertuzumab and trastuzumab plus docetaxel, resulted in complete pathological remission.16–18 Zhu and Chen suggested that endocrine therapy was necessary for primary breast SCC, 19 but the importance of treating this disease has rarely been reported. The inconsistent results regarding the above treatment methods indicate the need to examine more cases of ASCC.19,20 Given the irreversible nature of the mass in the current case, we administered neoadjuvant therapy with albumin-paclitaxel, carboplatin, and trastuzumab, with excellent results, confirming the efficacy of this treatment for HER2-positive SCC. Postoperative pathology suggested that there was some residual carcinoma in situ in the lesion, ER and PR were highly expressed, and that the addition of anastrozole could reduce the risk of recurrence and metastasis. We aim to follow up the patient to determine the long-term efficacy of the treatment.
Conclusion
HER2-positive breast ASCC is a relatively rare breast cancer with tumor heterogeneity and a variable prognosis, and a better understanding of its pathological features and therapeutic approaches are needed. The current report presents the case of a patient with HER2-positive breast ASCC who was treated with a novel adjuvant treatment approach with good results.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231187936 - Supplemental material for Neoadjuvant therapy for HER2-positive acantholytic squamous cell breast carcinoma: a case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605231187936 for Neoadjuvant therapy for HER2-positive acantholytic squamous cell breast carcinoma: a case report by Jia Cong, HyonSu Jo, Na Zou, LingXin Kong and HyokJu Ri in Journal of International Medical Research
Footnotes
Acknowledgements
We would like to thank the staff and nurses for their kind cooperation. We would also like to thank the patient and her family.
Author contributions
Conceptualization: JC, HR; Data curation: NZ, LK; Funding acquisition: JC; Investigation: HJ, Supervision: JC; Writing - original draft: JC; Writing - review & editing: JC, HJ, NZ, LK.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics statement
The reporting of this study conforms to the CARE guidelines. 10 The patient provided written informed consent for publication of this report and informed consent for surgery. This study was approved by Ethics Committee of Dalian Women and Children's Medical Group.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
