Abstract
Background:
Nanoliposomal irinotecan with fluorouracil and folinic acid (NFF) is a standard second- or later-line regimen for unresectable or recurrent pancreatic cancer (urPC). However, validated prognostic biomarkers are lacking.
Objectives:
In this study, we aimed to identify clinical predictors of progression-free survival (PFS) in patients receiving NFF and to develop a nomogram for early prediction of therapeutic efficacy.
Design:
This was a pre-planned analysis of a prospective, multicenter observational study conducted across 17 hospitals in Japan.
Methods:
We enrolled 150 patients with urPC who received NFF between 2021 and 2023. Prognostic factors independently associated with PFS were identified using multivariable Cox proportional hazards regression analysis. Based on these factors, we constructed a nomogram to estimate the probabilities of 2-, 4-, and 6-month PFS. Finally, we performed risk stratification according to total nomogram scores and validated this model using an independent retrospective cohort.
Results:
The median overall survival was 7.8 months, and the median PFS was 3.7 months. Multivariable analysis identified a longer duration of previous chemotherapy (hazard ratio (HR), 0.52; 95% confidence interval (CI), 0.38–0.71; p < 0.01) as a favorable prognostic factor; third-line treatment (vs second-line; HR, 1.67; 95% CI, 1.04–2.67, p = 0.03) and higher C-reactive protein/albumin ratio (CAR; HR, 1.14; 95% CI, 1.01–1.29, p = 0.04) were associated with unfavorable PFS. In risk stratification, median PFS in the low-, moderate-, and high-risk groups was 5.9 months (reference), 3.9 months (HR, 1.78; 95% CI, 1.16–2.71; p < 0.01), and 2.2 months (HR, 2.30; 95% CI, 1.51–3.50; p < 0.01), respectively. Significant risk stratification was also confirmed in an independent retrospective cohort.
Conclusion:
The duration of previous chemotherapy, treatment line, and the CAR are useful predictors of PFS in patients with urPC receiving NFF. The proposed nomogram and risk stratification system may facilitate individualized treatment planning and support clinical decision-making.
Keywords
Introduction
Pancreatic cancer (PC) remains a serious global health burden, ranking as the third leading cause of cancer-related mortality in Japan and the sixth worldwide.1,2 At the time of diagnosis, approximately 80% of patients with PC present with unresectable disease due to either locally advanced tumor invasion or distant metastases. 3 Even among patients with resectable disease, most experience disease recurrence within 2 years following surgical resection. 4 Consequently, most patients with PC eventually face unresectable or recurrent disease, for which palliative systemic chemotherapy is the standard of care to prolong survival and preserve quality of life. 5
FOLFIRINOX—a combination of 5-fluorouracil (5-FU), leucovorin, irinotecan, and oxaliplatin—as well as gemcitabine plus nab-paclitaxel, are recognized as standard first-line therapies for PC, supported by findings from prospective randomized phase III clinical trials.6,7 More recently, a combination of nanoliposomal irinotecan (nal-IRI), 5-FU, leucovorin, and oxaliplatin (NALIRIFOX) has emerged as a potential first-line chemotherapy option. 8 In the second-line setting, nal-IRI in combination with 5-FU and folinic acid (NFF) is currently recommended for patients with good performance status, presenting with disease progression after first-line gemcitabine-based chemotherapy.5,9 We previously reported findings from the NAPOLEON-2 observational study, in which we evaluated the efficacy and safety of NFF in a real-world setting.10,11 Although NFF demonstrates clinical utility, outcomes remain poor, with a median overall survival (OS) of 6.1–8.1 months,9–11 and no validated prognostic biomarkers have been established to date. Notably, progression-free survival (PFS) has been recognized as a clinically important endpoint in PC, with several studies suggesting its role as a surrogate for OS.12–14 In clinical practice, even among patients receiving NFF, cases of prolonged PFS are occasionally observed, highlighting the heterogeneity of treatment outcomes. However, objective data on predictors of longer PFS in this setting remain scarce. Identifying patients who are likely to achieve prolonged PFS could support treatment selection and patient counseling. Moreover, the development of simple, clinically applicable prognostic models such as nomograms may help translate real-world data into personalized treatment strategies.
The aim of this study was to identify clinical factors associated with PFS in patients with unresectable or recurrent PC (urPC) treated with NFF, and to develop and validate a simple nomogram to support individualized prognostication in daily clinical practice.
Methods
Study design and patients
This study was performed as a pre-planned analysis of the prospective cohort in the NAPOLEON-2 study, a multicenter observational study comprising a retrospective portion and a prospective portion, with the aim to investigate the efficacy and safety of NFF in patients with urPC.10,11 The study was conducted and reported in accordance with the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) Statement 15 (Supplemental File 1). The inclusion criteria of the prospective study were as follows: patients with urPC who received NFF after at least one regimen; submitted informed consent; aged ⩾20 years; had adequate organ functions; and Eastern Cooperative Oncology Group (ECOG) performance status (PS) of 0–2. The exclusion criteria were as follows: neuroendocrine tumors; active double cancers; and contraindications for NFF, such as allergies, ileus, uncontrollable infection, or the physician’s discretion. Finally, 150 patients were enrolled from 17 centers (Supplemental File 2) in Japan between June 1, 2021 and October 26, 2023, with follow-up until May 26, 2024. The primary objective of this analysis was to identify prognostic factors associated with PFS and to subsequently develop a nomogram to predict the 2-, 4-, and 6-month probabilities of PFS (Figure 1).

Flow diagram for developing a nomogram to predict PFS in the NAPOLEON-2 prospective study.
This study was approved by the institutional review board of each participating institution (Supplemental File 2) and was conducted according to the Declaration of Helsinki. The study was registered in the University Hospital Medical Information Network (UMIN ID: 000043939), a site for registering clinical studies in Japan that fulfills the standards of the International Committee of Medical Journal Editors.
Treatment
NFF was administered as follows: a 90-min intravenous (i.v.) infusion of nal-IRI (70 mg/m2), 2-h i.v. infusion of leucovorin (200 mg/m2), and 46-h continuous i.v. infusion of 5-FU (2400 mg/m2) every 2 weeks. At the discretion of the physician in charge, dose reduction at initiation and dose modification during treatment owing to toxicities were allowed. Treatment was discontinued when disease progression or unacceptable AEs occurred, even with dose adjustment, or at the patient’s request.
Assessments
OS was calculated from the date of administration of NFF to the date of death from any cause or was censored at the final follow-up examination. PFS was calculated from the date of administration of NFF to the date of progression or death from any cause, whichever was earlier, or was censored at the final follow-up examination.
Statistical analyses
Missing data were imputed using the method of multiple imputation with predictive mean matching. 16 The imputation model included all variables listed in Table 1. OS and PFS were estimated using the Kaplan–Meier method, and the 95% confidence interval (CI) was calculated using the method of Brookmeyer and Crowley. Loss to follow-up was accounted for by censoring data at the last follow-up date for each patient. Comparisons of survival curves were performed using the log-rank test. To identify prognostic factors independently associated with PFS and OS, multivariable Cox proportional hazards regression analyses were performed for all clinically meaningful variables, regardless of their significance in univariate analysis. 17 Continuous variables were log-transformed to normalize their distributions and minimize the impact of outliers in this analysis. Variables with a p value < 0.05 were selected as significant prognostic factors. Prognostic nomograms were then developed based on the final Cox models to predict survival probabilities for both PFS and OS. These nomograms were internally validated using the concordance index (C-index) and a calibration plot using the bootstrapping method (200 resamplings). We subsequently developed risk stratification systems for both PFS and OS according to tertiles of their respective total nomogram points and compared the survival times among risk groups. The risk stratification for the PFS nomogram was validated using the independent retrospective cohort in the NAPOLEON-2 study. 10 Statistical significance was set to p < 0.05. All statistical analyses were performed using R version 4.4.0 (The R Foundation for Statistical Computing, Vienna, Austria).
Patients’ baseline characteristics (N = 150).

ALT, alanine aminotransferase; CA19-9, carbohydrate antigen 19-9; CAR, C-reactive protein/albumin ratio; ECOG PS, Eastern Cooperative Oncology Group performance status; NFF, nanoliposomal irinotecan with fluorouracil and folinic acid; NLR, neutrophil-to-lymphocyte ratio; UGT1A1, uridine diphosphate glucuronosyltransferase family 1 member A1.
Results
Patient characteristics
Baseline characteristics of the 150 included patients are summarized in Table 1. The median age was 72 years (range: 45–85 years), and 81 patients (54%) were men. Most patients had a good performance status, with 93% having an ECOG PS of 0 or 1. The most common site of metastasis was the liver (51%), followed by the peritoneum (31%) and lung (29%). The median duration of previous chemotherapy was 8.7 months (range: 2.1–42.8 months). NFF was administered as second-, third-, and fourth- or later-line therapy in 87 (58%), 53 (35%), and 10 (7%) patients, respectively.
Survival outcomes and prognostic factors for PFS
The median follow-up duration was 7.2 months (95% CI: 6.3–8.8). The median OS was 7.8 months (95% CI: 6.6–9.2), and the median PFS was 3.7 months (95% CI: 2.8–4.9). 11 Results of the univariate and multivariable Cox proportional hazards regression analyses for PFS are listed in Table 2. Univariate analysis identified longer duration of previous chemotherapy (log-transformed; hazard ratio (HR), 0.68; 95% CI: 0.55–0.85, p < 0.01) and higher C-reactive protein/albumin ratio (CAR; log-transformed; HR, 1.12; 95% CI: 1.00–1.24, p = 0.04) as significant prognostic factors. Multivariable analysis identified longer duration of previous chemotherapy (log-transformed; HR, 0.52; 95% CI: 0.38–0.71, p < 0.01) as an independent prognostic factor associated with favorable PFS; third-line treatment (vs second-line; HR, 1.67; 95% CI: 1.04–2.67, p = 0.03) and higher CAR (log-transformed; HR, 1.14; 95% CI: 1.01–1.29, p = 0.04) were associated with unfavorable PFS.
Univariate and multivariable Cox regression analysis for progression-free survival.
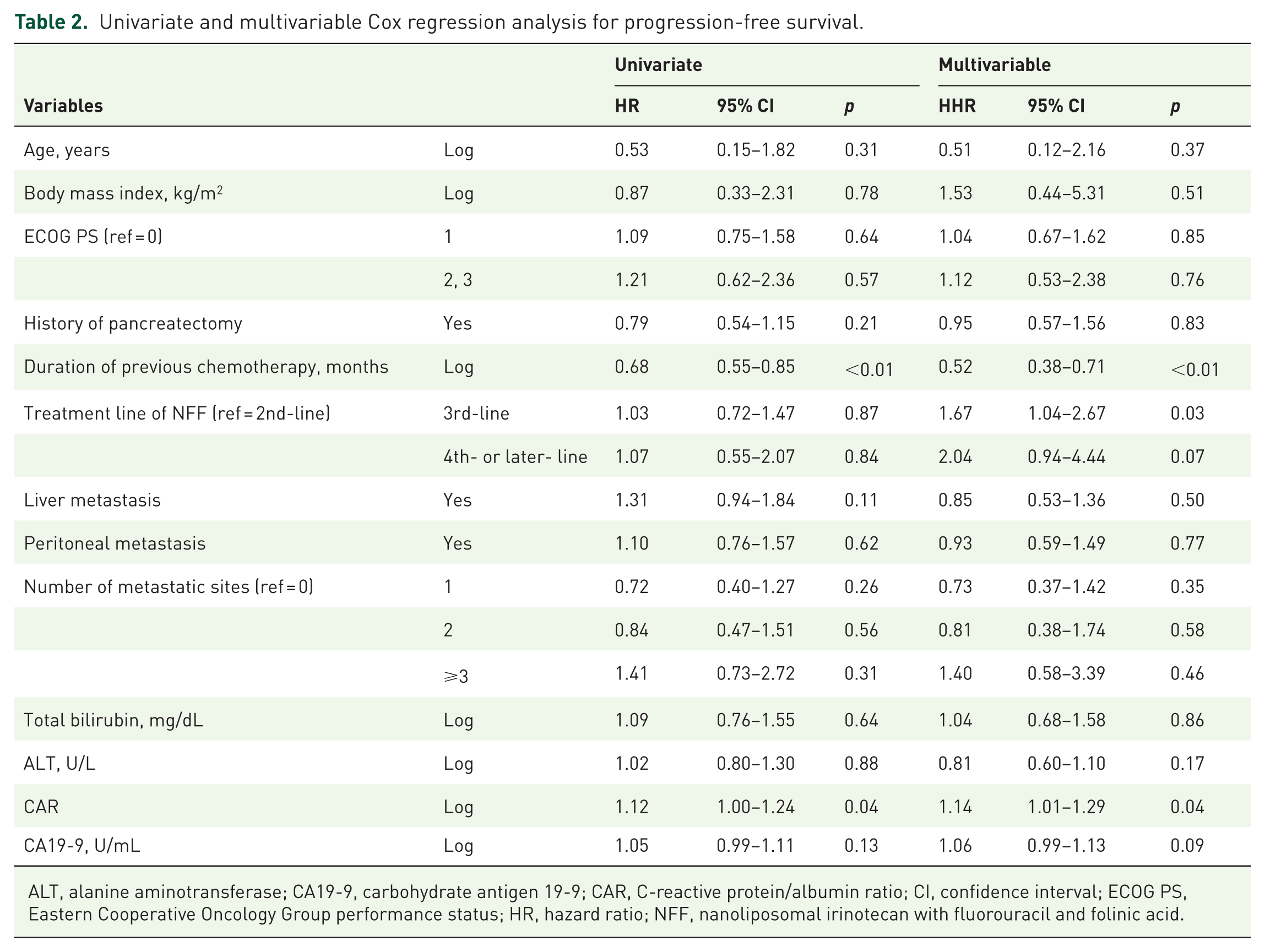
ALT, alanine aminotransferase; CA19-9, carbohydrate antigen 19-9; CAR, C-reactive protein/albumin ratio; CI, confidence interval; ECOG PS, Eastern Cooperative Oncology Group performance status; HR, hazard ratio; NFF, nanoliposomal irinotecan with fluorouracil and folinic acid.
Prognostic nomogram and risk stratification for PFS
The prognostic nomogram integrating the significant independent predictors of PFS identified in the multivariable Cox analysis is shown in Figure 2. The formula of total risk points was as follows: total points = 61.563–1.368 × (duration of previous chemotherapy) + 18.182 × (CAR) + treatment line score (0 for 2nd-line, 10.396 for 3rd-line, and 19.412 for 4th- or later-line). C-index values (bootstrapping 95% CI) for the nomogram in predicting the probabilities for PFS at 2, 4, and 6 months were 0.631 (0.547–0.712), 0.679 (0.612–0.742), and 0.676 (0.595–0.749), respectively (Supplemental Figure 1). The calibration plots for the PFS probability at 2, 4, and 6 months demonstrated good agreement between the nomogram-predicted probabilities and the actual observations. Notably, the bias-corrected curves generated via bootstrapping with 200 resamples generally tracked the ideal diagonal line, confirming the model’s acceptable calibration (Supplemental Figure 2). Patients were stratified using tertiles of the total risk points (Figure 3(a)). Median PFS in the groups with low (total nomogram points <52.1), moderate (total nomogram points 52.1–58.4), and high (total nomogram points ⩾58.4) risk was 5.9 months (reference), 3.9 months (HR, 1.78; 95% CI: 1.16–2.71, p < 0.01), and 2.2 months (HR, 2.30; 95% CI: 1.51–3.50, p < 0.01), respectively (Figure 3(b)). We validated this nomogram using the independent retrospective cohort in the NAPOLEON-2 study. The validation cohort was stratified using tertiles of total risk points (Figure 4(a)). The median PFS in the low-, moderate-, and high-risk groups was 5.1 months (reference), 3.7 months (HR, 1.68; 95% CI: 1.06–2.67, p = 0.03), and 2.4 months (HR, 2.28; 95% CI: 1.58–3.30, p < 0.01), respectively (Figure 4(b)).

Prognostic nomogram for predicting 2-, 4-, and 6-month progression-free survival.

Risk stratification using tertiles of total risk points. (a) Patients were stratified using tertiles of total risk points. (b) Median progression-free survival times in the low-, moderate-, and high-risk groups were 5.9 months (reference), 3.9 months (HR, 1.78, 95% CI: 1.16–2.71, p < 0.01), and 2.2 months (HR, 2.30, 95% CI: 1.51–3.50, p < 0.01), respectively.
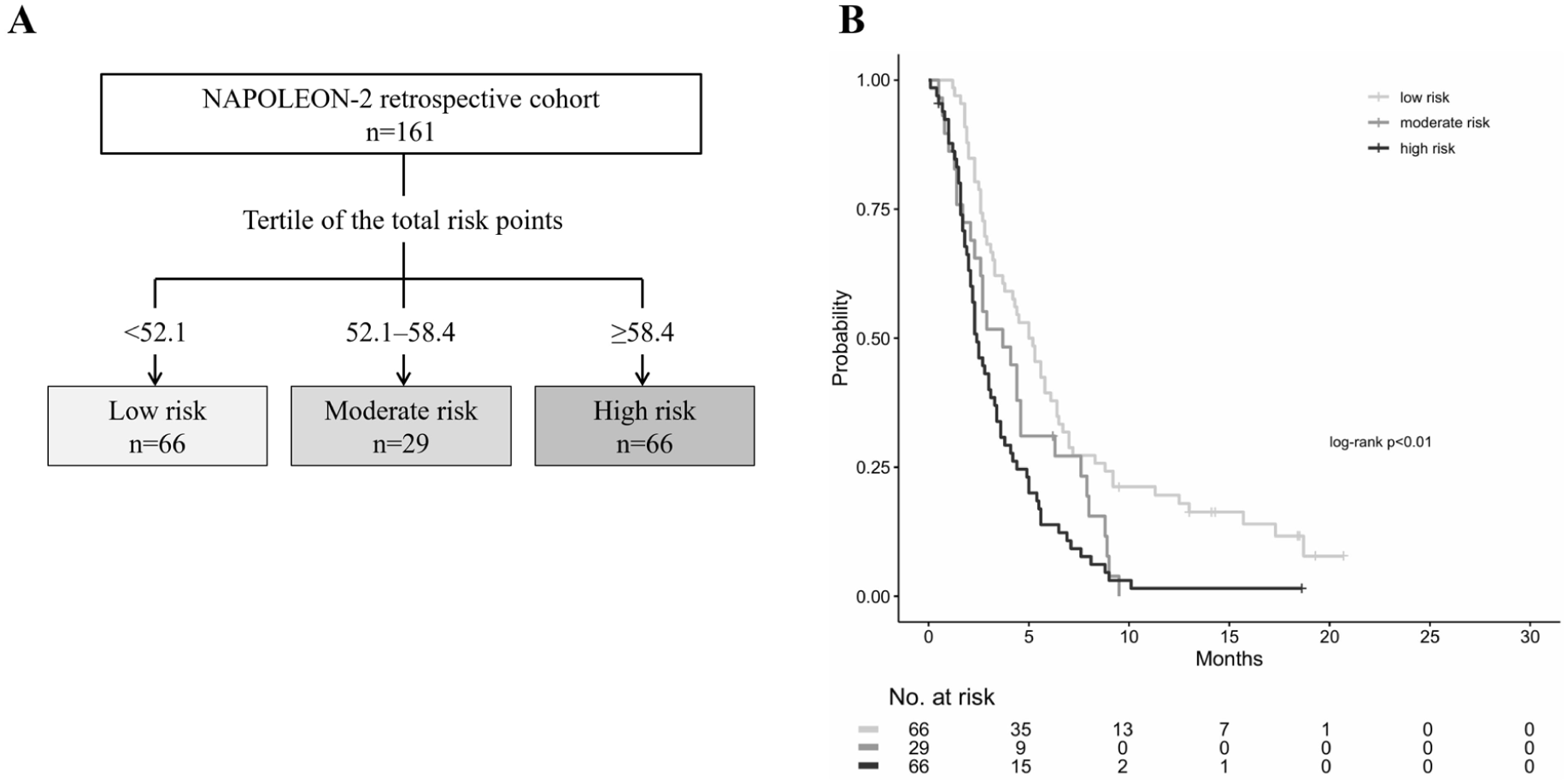
Validation of risk stratification. (a) The NAPOLEON-2 retrospective cohort was stratified using tertiles of total risk points. (b) Median progression-free survival times in the low-, moderate-, and high-risk groups were 5.1 months (reference), 3.7 months (HR, 1.68, 95% CI: 1.06–2.67, p = 0.03), and 2.4 months (HR, 2.28, 95% CI: 1.58–3.30, p < 0.01), respectively.
Association between PFS risk stratification and OS
The risk stratification model also demonstrated prognostic value for OS. In the prospective cohort, the median OS was 11.0 months (reference), 8.5 months (HR, 1.84; 95% CI: 1.16–2.93, p < 0.01), and 3.8 months (HR, 3.25; 95% CI: 2.07–5.13, p < 0.01), respectively (Supplemental Figure 3). This stratification was validated in the independent retrospective cohort, where the high-risk group showed significantly shorter OS (median 4.8 months; HR, 2.73; 95% CI: 1.81–4.12, p < 0.01) compared with the low-risk group (median 10.0 months); however, the difference between the low- and moderate-risk (9.5 months) groups was not significant (Supplemental Figure 4).
Survival outcomes and prognostic factors for OS
In the multivariable Cox proportional hazards regression analysis for OS, longer duration of previous chemotherapy (log-transformed; HR, 0.44; 95% CI: 0.31–0.63, p < 0.01) was identified as an independent prognostic factor associated with favorable OS. Conversely, third-line treatment (vs second-line; HR, 1.82; 95% CI: 1.09–3.03, p = 0.02), higher CAR (log-transformed; HR, 1.35; 95% CI: 1.17–1.55, p < 0.01), and higher carbohydrate antigen 19-9 levels (CA19-9; log-transformed; HR, 1.09; 95% CI: 1.02–1.17, p = 0.01) were associated with unfavorable OS. An OS-specific nomogram integrating these four predictors demonstrated high predictive accuracy, with C-indices from 0.710 to 0.746. Based on this model, median OS in the low-, moderate-, and high-risk groups was 11.6 months (reference), 8.1 months (HR, 1.77; 95% CI: 1.11–2.81, p = 0.02), and 3.7 months (HR, 3.28; 95% CI: 2.09–5.14, p < 0.01), respectively (data not shown).
Reasons for discontinuation and subsequent therapy
Among the 134 patients who discontinued NFF therapy, the primary reason was disease progression (including clinical PD), accounting for 87% (n = 116) of cases. Discontinuation due to adverse events occurred in 10 patients (8%; Supplemental Table 1). Following the discontinuation of NFF, 54 patients (40%) received subsequent anti-tumor therapy, most of whom (96%) underwent chemotherapy; 80 patients (60%) transitioned to best supportive care (Supplemental Table 2).
Discussion
In this prospective multicenter study of patients with urPC treated with NFF, we identified the duration of previous chemotherapy, the treatment line, and the CAR as independent predictors of PFS. By integrating these readily available clinical variables, we developed a nomogram capable of estimating the probability of achieving 2-, 4-, and 6-month PFS. To our knowledge, this is the first prospective analysis to generate a clinically applicable nomogram for predicting PFS in patients receiving NFF. The simplicity of the model allows immediate application in daily practice without additional testing costs.
Previous studies have primarily developed prognostic nomograms based on OS in patients receiving first- or second-line chemotherapy for advanced PC.18–21 Although OS remains the gold standard for assessing overall prognosis, it is often confounded by subsequent therapies and supportive care, limiting its ability to directly reflect the efficacy of a specific regimen. By contrast, PFS captures disease control during a defined therapy period and may provide a more immediate measure of the treatment effect. In our analysis, an OS-specific model incorporating CA19-9 achieved a higher predictive accuracy than the PFS model. This discrepancy suggests that although OS is strongly driven by the total tumor burden and biological reserve, PFS is influenced by drug-specific sensitivity, which is more challenging to predict using clinical factors alone. Nevertheless, we prioritized the PFS-based nomogram because clinical decision-making at this stage focuses on whether a patient will derive benefit from NFF specifically. Our model offers clinical utility for this “go/no-go” decision, distinguishing patients who will achieve disease control from those likely to experience rapid progression.
The identified predictors are biologically plausible. A longer duration of previous chemotherapy, identified as a favorable independent predictor, likely reflects not only favorable treatment tolerability but also—and more importantly—indolent tumor biology or preserved chemosensitivity. Because chemotherapy is typically continued until disease progression, the duration of therapy serves as a practical surrogate for tumor responsiveness. This is consistent with findings from ex vivo ATP-based chemosensitivity assays in primary PC cells, which showed substantial variability in response to cytotoxic agents, with gemcitabine sensitivity correlating with human equilibrative nucleoside transporter-1 expression and longer time to relapse. 22 Moreover, previous nomograms for second-line chemotherapy have also incorporated the duration of previous treatment as a prognostic factor, highlighting its relevance across different predictive models.20,21,23 In addition, we identified later treatment lines as a negative prognostic factor. Patients receiving NFF in later lines often present with reduced bone marrow reserve due to cumulative toxicity from prior regimens. 24 Furthermore, therapeutic pressure from earlier lines drives clonal evolution and the acquisition of multidrug resistance mechanisms, such as the upregulation of drug efflux pumps or activation of alternative survival pathways, rendering the tumor more refractory.25,26 Regarding inflammatory markers, CAR was adopted as a predictive marker in the present study, whereas the neutrophil-to-lymphocyte ratio has more commonly been incorporated as an inflammatory marker in past studies.19–21 The prognostic value of CAR is supported by the biological link between systemic inflammation and nutritional decline. Cancer-related inflammation plays a key role in tumor progression by promoting angiogenesis, cell proliferation, and inhibition of apoptosis through the upregulation of pro-inflammatory cytokines such as interleukin-6.27,28 Concurrently, serum albumin serves as an index of nutritional status and also reflects cancer cachexia. In addition, serum albumin is closely associated with sarcopenia, which adversely affects survival and treatment tolerance in patients with PC.29–31 Therefore, the CAR serves as a comprehensive biomarker reflecting both cancer-related systemic inflammation and unfavorable host factors, such as cachexia and sarcopenia. In our earlier retrospective analysis of various inflammatory markers—including the neutrophil-to-lymphocyte ratio and CAR—as prognostic factors in patients with urPC, CAR was identified as the most reliable marker. 32 The present study extends these findings by corroborating our earlier observations and providing prospective evidence in patients with urPC receiving NFF. Interestingly, CA19-9 was identified as a significant predictor for OS but not for PFS in the multivariable analysis. This supports the notion that whereas CA19-9 reflects the systemic tumor burden determining life expectancy, it may not necessarily predict the specific therapeutic response to NFF.
The discriminatory ability of our PFS nomogram was moderate, with C-indices of approximately 0.65; however, its clinical utility is best illustrated by its robust risk stratification. In both the prospective and independent retrospective cohorts, the model could successfully distinguish three prognostic groups with widely separated outcomes. Notably, the high-risk group experienced a median PFS of approximately 2 months, compared with 5–6 months in the low-risk group. Furthermore, although constructed for PFS, the risk stratification strongly predicted OS; high-risk patients had a median OS of less than 5 months—approximately one-third to half that of the low-risk group (10–11 months). Clinically, this clear stratification suggests that low-risk patients may derive sustained benefit from NFF, whereas high-risk patients—characterized by rapid progression—might be better served by prioritizing quality-of-life care or exploring clinical trials.
This study has several limitations. First, although it included 150 patients from 17 centers, the sample size is relatively modest for developing a prognostic nomogram, which may limit the statistical power and stability of estimates. Second, the study cohort consisted exclusively of Japanese patients. Given the potential differences in genetic ancestry regarding pharmacokinetics (e.g., UGT1A1 polymorphisms distribution) or tumor biology, the applicability of our findings to populations of different genetic backgrounds remains to be verified. Third, the predictive accuracy of the PFS nomogram was moderate compared with that of the OS model. This suggests that although clinical parameters can be used to stratify risk, the precise prediction of the drug response likely requires the incorporation of molecular or genetic biomarkers. Finally, although validated in the independent retrospective cohort (temporal validation), further validation in a completely independent dataset from different institutions or countries is needed to confirm the nomogram’s generalizability. Nevertheless, the strength of this study lies in its prospective, multi-institutional design, which provides a robust foundation for future validation. Importantly, the prognostic factors identified here may have translational relevance for other liposomal irinotecan-based regimens, such as NALIRIFOX. Although the “duration of previous chemotherapy” is specific to the salvage setting, biological markers such as CAR likely share prognostic utility across treatment lines. Therefore, further accumulation of prospective data, including evaluation in broader populations and in the first-line setting with NALIRIFOX, will be essential to confirm these hypotheses and extend the present findings.
Conclusion
The duration of previous chemotherapy, treatment line, and the CAR are simple but powerful predictors of PFS in patients with urPC receiving NFF. Our prospective multicenter nomogram provides clinically meaningful risk stratification and an immediately applicable tool for individualized treatment decisions. With further refinement and external validation, this model may help in optimizing therapy selection, improving patient counseling, and advancing personalized care in the second- or later-line setting for urPC, where treatment options remain limited.
Supplemental Material
sj-docx-5-tam-10.1177_17588359261442386 – Supplemental material for Predictors of prolonged progression-free survival in patients with unresectable or recurrent pancreatic cancer treated with nanoliposomal irinotecan with fluorouracil and folinic acid (NAPOLEON-2 study)
Supplemental material, sj-docx-5-tam-10.1177_17588359261442386 for Predictors of prolonged progression-free survival in patients with unresectable or recurrent pancreatic cancer treated with nanoliposomal irinotecan with fluorouracil and folinic acid (NAPOLEON-2 study) by Yasunori Kawaguchi, Kohei Hayashi, Mototsugu Shimokawa, Taiga Otsuka, Junichi Nakazawa, Hozumi Shimokawa, Yudai Shinohara, Futa Koga, Noriko Oza, Hisanobu Oda, Shigeyuki Takeshita, Shiho Arima, Shuji Arita, Kazuo Nishikawa, Satoshi Otsu, Hiroki Taguchi, Kenichi Jikuya, Tatsunori Sakai, Yujiro Ueda, Takahiro Sakae, Norimasa Araki, Hironori Sawase, Yasushi Ide, Machiko Kawahira, Kenta Nio, Tsuyoshi Shirakawa, Toshihiko Mizuta and Kenji Mitsugi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-6-tam-10.1177_17588359261442386 – Supplemental material for Predictors of prolonged progression-free survival in patients with unresectable or recurrent pancreatic cancer treated with nanoliposomal irinotecan with fluorouracil and folinic acid (NAPOLEON-2 study)
Supplemental material, sj-docx-6-tam-10.1177_17588359261442386 for Predictors of prolonged progression-free survival in patients with unresectable or recurrent pancreatic cancer treated with nanoliposomal irinotecan with fluorouracil and folinic acid (NAPOLEON-2 study) by Yasunori Kawaguchi, Kohei Hayashi, Mototsugu Shimokawa, Taiga Otsuka, Junichi Nakazawa, Hozumi Shimokawa, Yudai Shinohara, Futa Koga, Noriko Oza, Hisanobu Oda, Shigeyuki Takeshita, Shiho Arima, Shuji Arita, Kazuo Nishikawa, Satoshi Otsu, Hiroki Taguchi, Kenichi Jikuya, Tatsunori Sakai, Yujiro Ueda, Takahiro Sakae, Norimasa Araki, Hironori Sawase, Yasushi Ide, Machiko Kawahira, Kenta Nio, Tsuyoshi Shirakawa, Toshihiko Mizuta and Kenji Mitsugi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-7-tam-10.1177_17588359261442386 – Supplemental material for Predictors of prolonged progression-free survival in patients with unresectable or recurrent pancreatic cancer treated with nanoliposomal irinotecan with fluorouracil and folinic acid (NAPOLEON-2 study)
Supplemental material, sj-docx-7-tam-10.1177_17588359261442386 for Predictors of prolonged progression-free survival in patients with unresectable or recurrent pancreatic cancer treated with nanoliposomal irinotecan with fluorouracil and folinic acid (NAPOLEON-2 study) by Yasunori Kawaguchi, Kohei Hayashi, Mototsugu Shimokawa, Taiga Otsuka, Junichi Nakazawa, Hozumi Shimokawa, Yudai Shinohara, Futa Koga, Noriko Oza, Hisanobu Oda, Shigeyuki Takeshita, Shiho Arima, Shuji Arita, Kazuo Nishikawa, Satoshi Otsu, Hiroki Taguchi, Kenichi Jikuya, Tatsunori Sakai, Yujiro Ueda, Takahiro Sakae, Norimasa Araki, Hironori Sawase, Yasushi Ide, Machiko Kawahira, Kenta Nio, Tsuyoshi Shirakawa, Toshihiko Mizuta and Kenji Mitsugi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-1-tam-10.1177_17588359261442386 – Supplemental material for Predictors of prolonged progression-free survival in patients with unresectable or recurrent pancreatic cancer treated with nanoliposomal irinotecan with fluorouracil and folinic acid (NAPOLEON-2 study)
Supplemental material, sj-jpg-1-tam-10.1177_17588359261442386 for Predictors of prolonged progression-free survival in patients with unresectable or recurrent pancreatic cancer treated with nanoliposomal irinotecan with fluorouracil and folinic acid (NAPOLEON-2 study) by Yasunori Kawaguchi, Kohei Hayashi, Mototsugu Shimokawa, Taiga Otsuka, Junichi Nakazawa, Hozumi Shimokawa, Yudai Shinohara, Futa Koga, Noriko Oza, Hisanobu Oda, Shigeyuki Takeshita, Shiho Arima, Shuji Arita, Kazuo Nishikawa, Satoshi Otsu, Hiroki Taguchi, Kenichi Jikuya, Tatsunori Sakai, Yujiro Ueda, Takahiro Sakae, Norimasa Araki, Hironori Sawase, Yasushi Ide, Machiko Kawahira, Kenta Nio, Tsuyoshi Shirakawa, Toshihiko Mizuta and Kenji Mitsugi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-2-tam-10.1177_17588359261442386 – Supplemental material for Predictors of prolonged progression-free survival in patients with unresectable or recurrent pancreatic cancer treated with nanoliposomal irinotecan with fluorouracil and folinic acid (NAPOLEON-2 study)
Supplemental material, sj-jpg-2-tam-10.1177_17588359261442386 for Predictors of prolonged progression-free survival in patients with unresectable or recurrent pancreatic cancer treated with nanoliposomal irinotecan with fluorouracil and folinic acid (NAPOLEON-2 study) by Yasunori Kawaguchi, Kohei Hayashi, Mototsugu Shimokawa, Taiga Otsuka, Junichi Nakazawa, Hozumi Shimokawa, Yudai Shinohara, Futa Koga, Noriko Oza, Hisanobu Oda, Shigeyuki Takeshita, Shiho Arima, Shuji Arita, Kazuo Nishikawa, Satoshi Otsu, Hiroki Taguchi, Kenichi Jikuya, Tatsunori Sakai, Yujiro Ueda, Takahiro Sakae, Norimasa Araki, Hironori Sawase, Yasushi Ide, Machiko Kawahira, Kenta Nio, Tsuyoshi Shirakawa, Toshihiko Mizuta and Kenji Mitsugi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-3-tam-10.1177_17588359261442386 – Supplemental material for Predictors of prolonged progression-free survival in patients with unresectable or recurrent pancreatic cancer treated with nanoliposomal irinotecan with fluorouracil and folinic acid (NAPOLEON-2 study)
Supplemental material, sj-jpg-3-tam-10.1177_17588359261442386 for Predictors of prolonged progression-free survival in patients with unresectable or recurrent pancreatic cancer treated with nanoliposomal irinotecan with fluorouracil and folinic acid (NAPOLEON-2 study) by Yasunori Kawaguchi, Kohei Hayashi, Mototsugu Shimokawa, Taiga Otsuka, Junichi Nakazawa, Hozumi Shimokawa, Yudai Shinohara, Futa Koga, Noriko Oza, Hisanobu Oda, Shigeyuki Takeshita, Shiho Arima, Shuji Arita, Kazuo Nishikawa, Satoshi Otsu, Hiroki Taguchi, Kenichi Jikuya, Tatsunori Sakai, Yujiro Ueda, Takahiro Sakae, Norimasa Araki, Hironori Sawase, Yasushi Ide, Machiko Kawahira, Kenta Nio, Tsuyoshi Shirakawa, Toshihiko Mizuta and Kenji Mitsugi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-4-tam-10.1177_17588359261442386 – Supplemental material for Predictors of prolonged progression-free survival in patients with unresectable or recurrent pancreatic cancer treated with nanoliposomal irinotecan with fluorouracil and folinic acid (NAPOLEON-2 study)
Supplemental material, sj-jpg-4-tam-10.1177_17588359261442386 for Predictors of prolonged progression-free survival in patients with unresectable or recurrent pancreatic cancer treated with nanoliposomal irinotecan with fluorouracil and folinic acid (NAPOLEON-2 study) by Yasunori Kawaguchi, Kohei Hayashi, Mototsugu Shimokawa, Taiga Otsuka, Junichi Nakazawa, Hozumi Shimokawa, Yudai Shinohara, Futa Koga, Noriko Oza, Hisanobu Oda, Shigeyuki Takeshita, Shiho Arima, Shuji Arita, Kazuo Nishikawa, Satoshi Otsu, Hiroki Taguchi, Kenichi Jikuya, Tatsunori Sakai, Yujiro Ueda, Takahiro Sakae, Norimasa Araki, Hironori Sawase, Yasushi Ide, Machiko Kawahira, Kenta Nio, Tsuyoshi Shirakawa, Toshihiko Mizuta and Kenji Mitsugi in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We thank all the patients and their families, and all of the investigators at the 17 institutions that participated in the NAPOLEON-2 study. We would also like to thank the Saga Study Group of Liver Disease (SASLD) for their cooperation. We are also grateful to the clinical research coordinators and medical office assistants of the NAPOLEON study group for their assistance in the data collection. We also thank Analisa Avila, MPH, ELS, of Edanz (![]() ) for providing medical writing support.
) for providing medical writing support.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
