Abstract
Background:
Relationships between cancer onset, progression, and inflammation have been reported; yet, the significance of inflammatory markers before and after treatment with nanoliposomal irinotecan and fluorouracil with folic acid (NFF) is unclear.
Objectives:
We aimed to (1) investigate whether worsening inflammation is associated with disease progression and whether it can be used as a biomarker by comparing inflammatory markers before and after treatment with NFF and (2) verify which is a superior biomarker.
Design:
This was a preplanned analysis of a retrospective cohort of the NAPOLEON-2 study, a multicenter observational study. Patients from 20 institutions were enrolled. Those with unresectable pancreatic cancer who received NFF as a second-line or later-line treatment between June 1, 2020, and May 31, 2021, were included.
Methods:
The primary endpoint was overall survival, and the main secondary endpoint was progression-free survival. The following inflammatory markers were assessed: neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio, Prognostic Nutrition Index (PNI), C-reactive protein/albumin ratio (CAR), Glasgow prognostic score (GPS), Prognostic Index (PI), and Komori score.
Results:
A total of 161 patients were enrolled. The CAR, NLR, GPS (1 vs 2), PI (1 vs 2), PNI, and Komori score were useful pre-NFF biomarkers (hazard ratios (HRs): 2.59, 2.28, 2.76/3.35, 2.16/18.89, 2.13, and 3.39, respectively). The CAR, NLR, GPS (compared with a score of 1/2), and PI (compared with a score of 1/2) were useful post-NFF biomarkers (HRs: 2.63, 1.88, 2.54/3.09, 2.30/5.39, respectively). In terms of the receiver operating characteristic curve and Akaike information criterion, the CAR, GPS, and PI showed favorable trends, and overall, CAR was a useful biomarker.
Conclusion:
Inflammatory markers can be used as biomarkers both before and after NFF treatment, and CAR may be useful. We hope these findings will contribute to the selection of appropriate treatment options.
Keywords
Introduction
Pancreatic cancer has a very poor prognosis. In Japan, the 5-year relative survival rate of patients with unresectable pancreatic cancer is about 1%. 1 For the treatment of pancreatic cancer, FOLFIRINOX and gemcitabine + nab-paclitaxel (GEM) have been reported to be superior to gemcitabine (GnP).2,3 The efficacies of nanoliposomal irinotecan and fluorouracil with folic acid (NFF) as second-line therapies have also been reported.4,5 Furthermore, the superiority of NALIRIFOX, a regimen that uses nanoliposomal irinotecan (nal-IRI), over GnP has been reported. 6 Although NALIRIFOX prolongs overall survival (OS), the prognosis of pancreatic cancer remains poor. Owing to advances in treatment, an increasing number of people are living longer despite having a poor prognosis. Thus, markers are needed to predict which patients will have a favorable outcome.
Various reports have indicated that inflammation is involved in cancer development and progression.7,8 Various inflammatory markers, including the neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), Prognostic Nutrition Index (PNI), C-reactive protein/albumin ratio (CAR), Glasgow prognostic score (GPS), and Prognostic Index (PI), have been reported as potential prognostic biomarkers.9–14 We have reported the utility of a combined score of albumin and tumor markers, which can reflect inflammation. 15 Cancer is not a constant condition, and worsening inflammatory markers are predicted with disease progression. However, most previous studies have classified patients using inflammatory markers only before the start of treatment. Furthermore, a biomarker for post-nal-IRI has not yet been established, and the significance of inflammatory markers before and after NFF is unclear.
Therefore, we aimed to examine whether inflammatory markers before and after NFF could be biomarkers for a more acute understanding of the disease status and which of the aforementioned markers is a superior biomarker.
Materials and methods
Ethics statements
The institutional review board of each participating institution approved the study protocol (2021-08), and this study was conducted in accordance with the Declaration of Helsinki guidelines. Detailed information on the names of the Institutional Review Boards and approval numbers/IDs and/or dates are provided in Supplemental File 1. Additional written or verbal consent was not required from the study participants because the NAPOLEON-2 study used only pre-existing data. However, the opportunity for patients or their representatives to refuse to become study participants was ensured by posting information about the study on the websites of the participating facilities.
Study design and patients
This was a pre-planned analysis of a retrospective cohort from the NAPOLEON-2 study, a multicenter observational study. 16 Patients from 20 institutions were enrolled in the NAPOLEON-2 trial. Those aged ⩾20 years with locally advanced, metastatic, or recurrent pancreatic cancer who underwent NFF as second-line or later chemotherapy between June 1, 2020, and May 31, 2021, and were followed up until November 30, 2022, were included. Patients with multiple primary cancers were excluded, except those with intraepithelial or intramucosal cancers that were deemed curable with local treatment. Patients who received NFF based on a clinical diagnosis, which consisted only of biochemical and/or radiological characteristics, were included in this study, although those with a diagnosis of neuroendocrine tumors were excluded. The reporting of this study conforms to the STROBE statement 17 (Supplemental File 2).
Outcomes
The primary endpoint was OS, and the secondary endpoints were progression-free survival (PFS), overall response rate, disease control rate, adverse events, and relative dose intensity. The period from the initial NFF administration until patient death was designated as OS. The period from the initial NFF administration until the earliest instance of disease progression or death from any cause was designated as PFS. In the OS and PFS analyses, patients lost to follow-up were considered censored observations. The reference doses were as follows for every 2 weeks: nal-IRI, 70 mg/m2; 5-fluorouracil (5-FU), 2400 mg/m2; and="left"leucovorin, 200 mg/m2. Inflammatory markers at the beginning and inflammatory markers at the end of NFF administration were defined as pre-NFF and post-NFF, respectively. In addition, post-discontinuation survival (PDS) was defined as the survival time after NFF treatment ended. The score reported by Komori et al. 15 was referred to as the Komori score. The NLR was calculated as the neutrophil count (/μL) divided by the lymphocyte count (/μL), the PLR was calculated as the platelet count (/μL) divided by the lymphocyte count (/μL); PNI was calculated as 10 × albumin (g/dL) + 0.005 × lymphocyte count (/μL); and CAR was calculated as C-reactive protein (CRP; mg/dL) divided by albumin (g/dL). The GPS was defined as 0 for CRP level ⩽1.0 and albumin level ⩾3.5 (g/dL), 2 for CRP level >1.0 and albumin level <3.5, or 1 otherwise. The PI was defined as 0 for CRP level ⩽1 and white blood count (WBC; cells/μL) ⩽11,000, 2 when CRP level >1 and WBC count >11,000, or 1 otherwise. The Komori score was calculated by adding 1 point for albumin level <3.5 and 1 point for carbohydrate antigen 19-9 (CA19-9) >1000 (U/mL), with a maximum score of 2 points. Based on previous reports, we determined the cutoff values for the NLR, PLR, PNI, and CAR as follows: ⩽5 or >5, ⩽150 or >150, <47 or ⩾47, and <0.54 or ⩾0.54, respectively.9,10,13,15
Statistical analysis
OS, PFS, and PDS were estimated using the Kaplan–Meier method and compared using the log-rank test. Hazard ratios (HRs) were estimated using Cox proportional hazards regression analysis with 95% confidence intervals (95% CIs). Missing values were obtained using the multiple assignment method for WBC, neutrophil count, lymphocyte count, platelet count, albumin, CRP, and CA19-9, which are used to calculate inflammatory markers. 18 Multivariate Cox regression analyses were used for assessing OS, PFS, and PDS by age (⩾75/<75); presence or absence of ascites; performance status ⩾1/0; history of resection; and each inflammatory marker. They were selected according to the factors that clinicians considered important and consensus statements on mandatory measurements in pancreatic cancer trials, except for inflammatory markers. 19 Time-dependent receiver operating characteristic (ROC) and Akaike information criterion (AIC) analyses were performed for each inflammatory marker.20,21 All statistical analyses were performed using EZR ver. 1.63 (Saitama Medical Center, Jichi Medical University, Saitama, Japan). 22
Results
Patient characteristics
In all, 161 patients were enrolled in this study. Twelve patients (7.4%) remained on NFF therapy and one patient relocated, while 149 patients (92.5%) stopped because of disease progression (n = 133, 89.3%), adverse events (n = 12, 8.1%), patient refusal (n = 2, 1.3%), or other reasons (n = 2, 1.3%). Four patients were excluded because they lacked the WBC, neutrophil, lymphocyte, and platelet counts and albumin, CRP, and CA19-9 levels required to calculate the respective inflammatory markers, leaving 144 patients for post-NFF analysis (Figure 1). The median follow-up duration was 7.3 months (range, 0.3–20.7 months). The patient background characteristics pre-NFF and post-NFF are shown in Table 1. The CAR was ⩾0.54 in 33 patients (20%) pre-NFF and 64 (44%) post-NFF. PLR ⩾150 was observed in 115 patients (71%) pre-NFF and 104 (72%) post-NFF; NLR ⩾5 was observed in 35 (22%) pre-NFF and 47 (33%) post-NFF; and PNI ⩾47 was observed in 43 (27%) pre-NFF and 15 (10%) post-NFF. GPS 0/1/2 was observed in 80/49/32 patients (50/30/20%) pre-NFF and 48/34/62 (33/24/43%) post-NFF. The Komori scores were 53/74/34 (33/46/21%) in 0/1/2 patients pre-NFF and 13/28/10/93 (9/19/7/65%) in 0/1/2/best supportive care (BSC) post-NFF. In the pre-NFF group, there were 117 OS events, including 116 cancer deaths, and there were 150 and 115 PFS and PDS events, respectively.

Flow diagram.
Patient background characteristics, N=161.

Alb, albumin; BSC, best supportive care; CA19-9, carbohydrate antigen 19-9; CAR, C-reactive protein-to-albumin ratio; CRP, C-reactive protein; ECOG, Eastern Cooperative Oncology Group; FOLFIRINOX, 5-fluorouracil, leucovorin, irinotecan, and oxaliplatin; 5-FU, 5-fluorouracil; GEM + nab-PTX, gemcitabine plus nab-paclitaxel; GPS, Glasgow prognostic score; Ly, lymphocytes; NA, not applicable; nal-IRI, nanoliposomal irinotecan; Ne, neutrophil; NFF, nanoliposomal irinotecan and fluorouracil with folic acid; NLR, neutrophil-to-lymphocyte ratio; PI, Prognostic Index; PLR, platelet-to-lymphocyte ratio; Plt, platelet; PNI, Prognostic Nutrition Index; RDI, relative dose intensity; UGT1A1, uridine diphosphate glucuronosyltransferase 1A1; WBC, white blood cell.
Pre-nanoliposomal irinotecan and fluorouracil with folic acid
OS in the pre-NFF group was significantly stratified by the CAR, NLR, GPS, PI, PNI, and Komori score (Figure 2). OS with a CAR <0.54 (9.24 months; 95% CI, 7.63–10.46) was significantly better than that ⩾0.54 (4.67 months; 95% CI, 3.26–5.56) with an HR of 2.59 (95% CI, 1.66–4.02); OS with NLR ⩽5 (9.24 months; 95% CI, 7.60–10.63) was significantly better than that >5 (5.20 months; 95% CI, 3.16–7.47) with an HR of 2.28 (95% CI, 1.51–3.45). Regarding the GPS, OS with scores 1 (5.23 months; 95% CI, 4.67–7.63; HR = 2.76; 95% CI, 1.80–4.22) and 2 (4.77 months; 95% CI, 3.49–5.56; HR = 3.35; 95% CI, 2.02–5.57) was significantly different from that with a score of 0 (10.72 months; 95% CI, 9.24–13.55). Concerning the PI, OS with scores 1 (5.23 months; 95% CI, 4.61–7.60; HR = 2.16; 95% CI: 1.45–3.21) and 2 (2.99 months; 95% CI, 1.12–not applicable; HR = 18.89; 95% CI, 6.53–54.62) was significantly different from that with a score of 0 (9.87 months; 95% CI, 8.06–11.18). OS with a PNI ⩾47 (11.64 months; 95% CI, 9.87–14.38) was significantly better than that <47 (6.84 months; 95% CI, 5.20–8.03) with an HR of 2.13 (95% CI, 1.38–3.29). There was a significant difference in OS with a Komori score of 2 (3.95 months; 95% CI, 3.49–4.93) compared with that of 0 (10.72 months; 95% CI, 9.08–13.36; HR = 3.39; 95% CI, 2.00–5.75). Using multivariate Cox regression analyses, significant differences in NLR, PNI, CAR, GPS, PI, and Komori score were found (Table S1(A)).

OS analysis for pre-NFF and PDS analysis for post-NFF. Below 1 is the OS analysis for pre-NFF and 2 is the PDS analysis for post-NFF. In addition, below are the analyses for the following: (a) is for CAR, (b) is for PLR, (c) is for NLR, (d) is for GPS, (e) is for PI, (f) is for PNI, and (g) is for Komori score. (a-1) OS categorized by CAR ⩾0.54 or <0.54. CAR <0.54 was significantly better with an HR of 2.59 (95% CI, 1.66–4.02). (a-2) PDS categorized by CAR ⩾0.54 or <0.54. CAR <0.54 was significantly better with an HR of 2.63 (95% CI, 1.80–3.86). (b-1) OS categorized by PLR ⩽150 or >150. There was no significant difference (HR 1.18; 95% CI, 0.79–1.77). (b-2) PDS categorized by PLR ⩽150 or >150. There was no significant difference (HR 1.37; 95% CI, 0.90–2.10). (c-1) OS categorized by NLR ⩽5 or >5. NLR ⩽5 was significantly better with an HR of 2.28 (95% CI 1.51–3.45). (c-2) PDS categorized by NLR ⩽5 or >5. NLR ⩽5 was significantly better with an HR of 1.88 (95% CI 1.26–2.80). (d-1) OS categorized by GPS 0, 1, and 2. Score 1 (HR 2.76; 95% CI 1.80–4.22) and score 2 (HR 3.35; 95% CI 2.02–5.57) were significantly different from score 0. (d-2) PDS categorized by GPS 0, 1, and 2. Score 1 (HR 2.54; 95% CI 1.51–4.29) and score 2 (HR 3.09; 95% CI 1.96–4.87) were significantly different from score 0. (e-1) OS categorized by PI 0, 1, and 2. Score 1 (HR 2.16; 95% CI 1.45–3.21) and score 2 (HR 18.89; 95% CI 6.53–54.62) were significantly different from score 0. (e-2) PDS categorized by PI 0, 1, and 2. Score 1 (HR 2.30; 95% CI 1.54–3.45) and score 2 (HR 5.39; 95% CI 2.85–10.16) were significantly different from score 0. (f-1) OS categorized by PNI <47 or ⩾47. PNI ⩾47 was significantly better with an HR of 2.13 (95% CI 1.38–3.29). (f-2) PDS categorized by PNI <47 or ⩾47. There was no significant difference (HR 1.75; 95% CI, 0.91–3.36). (g-1) OS categorized by Komori score 0, 1, and 2. There was a significant difference for score 2 compared to score 0 (HR 3.39; 95% CI 2.00–5.75). There was no significant difference for score 1 compared to score 0 (HR 1.34; 95% CI 0.87–2.05). (g-2) PDS categorized by Komori score 0, 1, 2, and BSC. There was a significant difference in score BSC compared to score 0 (HR 2.90; 95% CI 1.46–5.76). There was no significant difference for score 2 compared to score 0 (HR 1.95; 95% CI 0.71–5.35). There was no significant difference for score 1 compared to score 0 (HR 1.00; 95% CI 0.47–2.13).
PFS in the pre-NFF group differed significantly according to the CAR, NLR, GPS, PI, PNI, and Komori score (Figure S1). PFS with CAR <0.54 (3.83 months; 95% CI, 2.93–4.61) was significantly better than that ⩾0.54 (2.30 months; 95% CI, 1.68–3.26) with an HR of 1.84 (95% CI: 1.23–2.75); PFS with NLR ⩽5 (4.11 months; 95% CI, 2.93–4.87) was significantly better than >5 (2.60 months; 95% CI, 1.64–3.12) with an HR of 1.77 (95% CI, 1.21–2.59). Regarding the GPS, PFS with scores 1 (2.76 months; 95% CI, 2.07–4.14; HR = 1.93; 95% CI: 1.32–2.83) and 2 (2.30 months; 95% CI, 1.68–2.83; HR = 2.65; 95% CI, 1.70–4.13) were significantly different from that with a score of 0 (4.92 months; 95% CI, 3.68–5.76). Concerning the PI, PFS with scores 1 (2.53 months; 95% CI, 2.07–3.13; HR = 1.96; 95% CI 1.36–2.81) and 2 (1.64 months; 95% CI, 0.76–2.34; HR = 6.39; 95% CI: 2.75–14.84) were significantly different from that with a score of 0 (4.34 months; 95% CI, 3.42–5.33). PFS with PNI ⩾47 (5.00 months; 95% CI, 4.38–6.97) was significantly better than that with <47 (2.93 months; 95% CI, 2.53–3.42) with an HR of 1.90 (95% CI: 1.30–2.78). There was a significant difference in PFS with a Komori score of 2 (2.11 months; 95% CI, 1.68–2.73) compared with that of 0 (HR = 2.52; 95% CI, 1.58–4.02). Using multivariate Cox regression analyses, significant differences in NLR, PNI, CAR, GPS, and PI were found (Table S1(B)).
The AIC pre-NFF was the most predominant for the GPS (CAR, 1001.7; PLR, 1016.1; NLR, 1003.6; GPS, 989.65; PI, 991.18; PNI, 1003.9; Komori score, 999.79). The time-dependent ROC curve was favorable for the CAR and GPS (Figure 3).
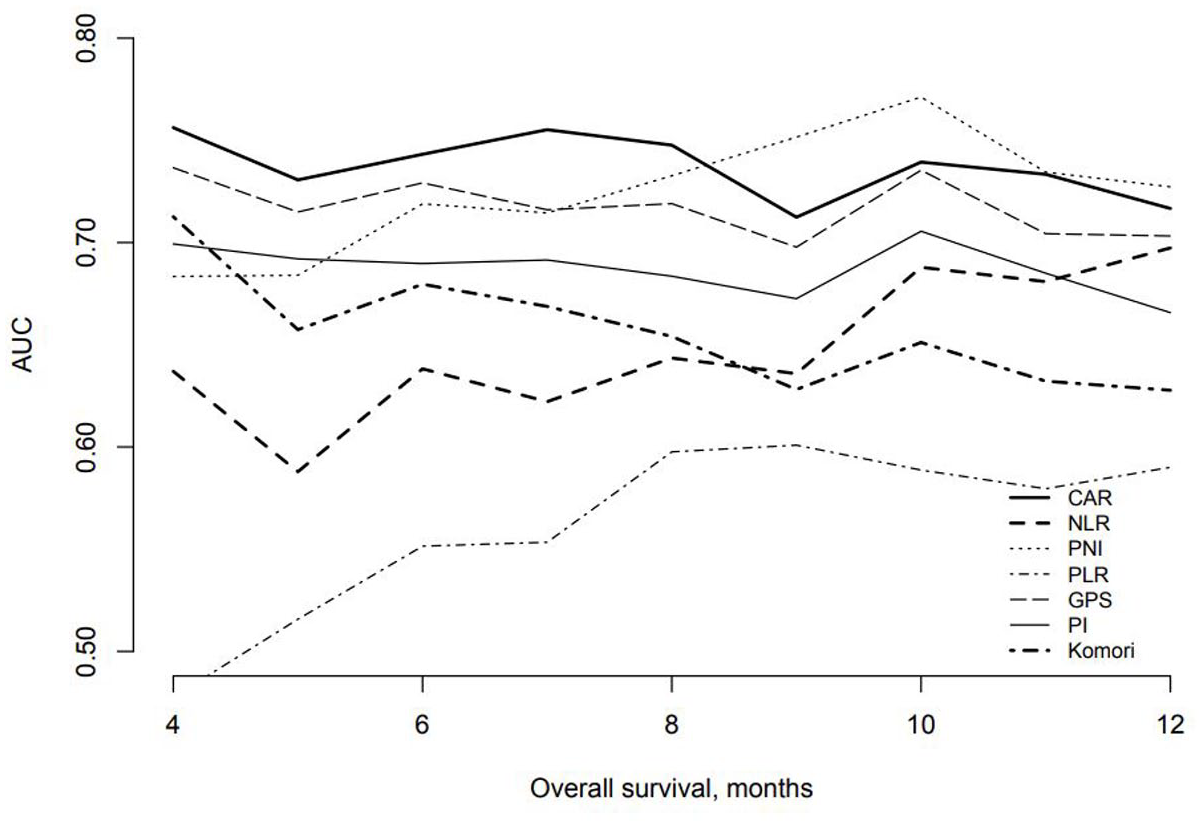
Time-dependent ROC. Time-dependent ROCs were compared for each inflammatory marker. The results were favorable in CAR and GPS.
In previous reports, each cutoff is classified as a good or poor prognosis.9,10,13,15 One point was assigned to each inflammatory marker classified as poor prognosis. GPS, PI, and Komori score were assigned 1 point for scores of 1 or more. The total score was classified as a maximum of 7 points (Table S2). The total score was further classified into 0, 1–3, 4–6, and 7 points, with the higher total score indicating a poorer prognosis (Figure S2). The number of missing values was as follows: albumin level in two cases, CRP level in two cases, and CA19-9 level in four cases.
Post-nanoliposomal irinotecan and fluorouracil with folic acid
PDS was significantly different according to the CAR, NLR, GPS, PI, and Komori score (Figure 2). PDS with CAR <0.54 (4.84 months; 95% CI, 3.59–5.76) was significantly better than that with ⩾0.54 (1.64 months; 95% CI, 1.18–2.20) with an HR of 2.63 (95% CI, 1.80–2.20); PDS with NLR ⩽5 (4.31 months; 95% CI, 2.83–5.13) was significantly better than that with >5 (1.51 months; 95% CI, 1.15–1.97) with an HR of 1.88 (95% CI, 1.26–2.80). Regarding the GPS, PDS with scores 1 (2.60 months; 95% CI, 1.97–3.88; HR = 2.54; 95% CI, 1.51–4.29) and 2 (1.61 months; 95% CI, 1.18–2.20; HR = 3.09; 95% CI, 1.96–4.87) was significantly different from that with a score of 0 (5.26 months; 95% CI, 4.47–7.14). Concerning the PI, PDS with scores 1 (1.97 months; 95% CI, 1.51–2.70; HR = 2.30; 95% CI: 1.54–3.45) and 2 (1.15 months; 95% CI, 0.36–1.61; HR = 5.39; 95% CI, 2.85–10.16) was significantly different from that with a score of 0 (5.07 months; 95% CI, 4.31–5.79). There was a significant difference in the Komori score for BSC (1.97 months; 95% CI, 1.41–2.53) compared with a score of 0 (6.94 months; 95% CI, 1.97–9.05; HR = 2.90; 95% CI, 1.46–5.76). Using multivariate Cox regression analyses, significant differences in NLR, CAR, GPS, and PI were found (Table S1(C)). The AIC post-NFF was the most predominant for the PI (CAR, 898.72; PLR, 920.54; NLR, 914.08; GPS, 895.84; PI, 893.43; PNI, 919.52; Komori score, 895.14).
The number of missing values was as follows: neutrophil count, two cases; lymphocyte count, two cases; albumin level, five cases; CRP level, two cases; and CA19-9 level, 37 cases. Because the CA19-9 value was not required if the patient was classified into the BSC group by the Komori score, the number of missing CA19-9 values needed to calculate the Komori score was 9.
Discussion
This study’s results suggest that several inflammatory markers may be promising biomarkers, and that the CAR may be a superior biomarker to the others.
There was a statistically significant difference in PFS and OS for the CAR, NLR, GPS, PI, PNI, and Komori score (0 vs 2). Using multivariate Cox regression analysis, the usefulness of these factors for OS and PFS was confirmed, except for the Komori score for PFS. We also observed significant differences in many inflammatory markers that support the relationship between cancer and inflammation. The most promising AUCs were for the CAR and PNI, whereas the most promising AICs were for the CAR, GPS, and PI.
Post-NFF, there was a statistically significant difference in PDS for the CAR, NLR, GPS, PI, and Komori score of 0 (vs the BSC group), although there was no significant difference between the groups in post-treatment cases. However, the Komori score included many BSC cases, and the number of cases in each group was small owing to the further subdivision of the treatment groups, which may have prevented significant differences from being observed. Using multivariate Cox regression analysis, the usefulness of these factors for PDS was confirmed, except for the Komori score.
Considering pre-NFF and post-NFF, as well as the AUC and AIC values, the CAR is useful for predicting prognosis at each time point. Comparison of inflammatory markers before and after first-line chemotherapy with FOLFIRINOX or GnP has been reported, with the CAR being particularly useful, and the results replicated this with NFF as a second- or later-line treatment. 23 These results suggest that inflammatory markers may be universally useful in patients with pancreatic cancer who are candidates for chemotherapy, regardless of the treatment line or regimen. The CAR is suggested to be superior to other inflammatory markers for several reasons. The GPS and PI are categorical variables that may have made it difficult to fully reflect the patient’s condition. Only seven patients had a PI score of 2 pre-NFF, making verification difficult. Although cutoff values were used for each inflammatory marker in accordance with previous reports, there were only 15 cases of PNI ⩾47, which may have made it difficult to obtain a significant difference. The Komori score may have been influenced by the small number of patients with a score of 0–2, although most post-NFF patients were in the BSC group, making it difficult to determine a significant difference. Although we judged the CAR to be superior as an overall judgment, based on the ROC curve and AIC, there was no significant difference between the CAR and inflammatory markers. In particular, the differences between the CAR and NLR, GPS, and PI should be confirmed in future studies. The results of the total score showed that higher scores tended to show a worse prognosis; the combination of multiple inflammatory markers should be evaluated in future research.
The usefulness of multiple inflammatory markers reaffirms the relationship between cancer and inflammation. In other words, inflammation can be considered an indicator of not only cancer development but also cancer progression and metastasis in basic research, and this study was replicated in clinical practice.7,8,24–27 In addition, inflammation contributes to tumor progression through mechanisms other than tumorigenesis, by establishing a microenvironment that promotes tumorigenesis through excessive production of inflammatory cytokines and chemokines. 24 Therefore, inflammation requires close monitoring, and considering the findings of this study and previous reports, it was consistent that inflammatory markers are consistently useful in any line of treatment. 23 CRP is a representative indicator of inflammation and has been reported as a prognostic marker for pancreatic cancer. 28 The CAR seemed to be the most dominant ROC curve, whereas the GPS had the most dominant AIC pre-NFF and the PI had the most dominant AIC post-NFF. Therefore, we considered the importance of CRP, which is a common component of the three inflammatory markers. Another important aspect is that the liver, which produces albumin, is involved in the metabolism of 5-FU and nanoliposomal irinotecan. However, since CRP can be altered by infections and other factors besides tumorigenicity, we considered it worthwhile to conduct biomarker studies using a composite inflammatory marker. In addition to cancer, albumin levels tend to be low in patients with cirrhosis, nephrotic syndrome, other chronic debilitating diseases, and active infections. Patients with such underlying diseases are predicted to have a poorer prognosis than others, and the albumin level is an important factor. The results suggest that inflammatory markers may be useful in predicting prognosis during NFF treatment and even after NFF treatment is completed, and it may help determine whether the patient should be switched to the next chemotherapy.
This study has some limitations. First, this was a retrospective study. Based on the results of this study, stating that CAR was superior to other inflammatory biomarkers is difficult. However, CAR apparently is a good result in this study similar to that in a previous report; this result could be remarkable. 22 Second, this study did not include many cases as a multicenter observational study; the stratification based on inflammatory markers was uneven. However, this was a retrospective study conducted approximately 1 year after nanoliposomal irinotecan was approved to be covered by insurance in Japan. Therefore, even though it was a multicenter study, the number of cases was limited to 161. After verifying the results in this retrospective study, we plan to verify reproducibility in prospective studies and ensure recruiting many cases for integrated analysis to produce higher-quality data. The cutoff values of inflammatory markers were based on previously published studies.9,10,13,15 It is possible that a significant difference might have been found if the number of cases was increased; however, the p-value is only an estimate, and a clinically meaningful difference has been detected. Verification is required by accumulating more cases, performing integrated analysis with prospective cohorts, and accumulating further research data. Third, cutoff values for each inflammatory marker were adopted in accordance with previous reports, although this clearly biased the number of cases and made it difficult to interpret cases in which no significant differences were found. However, the appropriateness of these cutoff values requires further validation. Fourth, in Figure 2, the proportion of patients with poor prognosis in terms of the CAR, NLR, GPS, and PNI seems to have increased post-NFF compared with pre-NFF. This is because the proportion of patients with a poor prognosis increased at all time points during NFF treatment. However, this study only evaluated inflammatory markers before and after NFF and did not assess changes in inflammatory markers during NFF. This is worth examining in relation to the changes in inflammatory markers over time and tumor growth during NFF treatment. Fifth, it should be noted that AIC and ROC were not intended to exclude other markers; there is no sufficient evidence to do this. Lastly, it should be noted that the p-values for statistical significance decreased as the number of strata increased. The p-value is an estimate, presented as a complementary indicator to determine whether the difference in median survival time is clinically significant. To resolve these limitations, we examined the association between changes in inflammatory markers over time and imaging in a prospective cohort of the NAPOLEON-2 study. Furthermore, to make the data even more robust, verification with more cases is warranted.
Conclusion
Inflammatory markers could be used as biomarkers for both pre-NFF and post-NFF, and the CAR could be particularly useful. We hope that these findings will contribute to appropriate treatment selection in patients with pancreatic cancer.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251320768 – Supplemental material for Comparison of inflammatory markers before and after nanoliposomal irinotecan and fluorouracil with folic acid in patients with pancreatic cancer: results from the NAPOLEON-2 study (NN-2302)
Supplemental material, sj-docx-1-tam-10.1177_17588359251320768 for Comparison of inflammatory markers before and after nanoliposomal irinotecan and fluorouracil with folic acid in patients with pancreatic cancer: results from the NAPOLEON-2 study (NN-2302) by Tomonori Araki, Kohei Hayashi, Mototsugu Shimokawa, Taiga Otsuka, Yuki Sonoda, Takuya Honda, Taro Shibuki, Junichi Nakazawa, Shiho Arima, Keisuke Miwa, Futa Koga, Yujiro Ueda, Yoshihito Kubotsu, Hozumi Shimokawa, Shigeyuki Takeshita, Kazuo Nishikawa, Azusa Komori, Satoshi Otsu, Ayumu Hosokawa, Tatsunori Sakai, Hisanobu Oda, Machiko Kawahira, Shuji Arita, Hiroki Taguchi, Kengo Tsuneyoshi, Toshihiro Fujita, Takahiro Sakae, Yasunori Kawaguchi, Tsuyoshi Shirakawa, Toshihiko Mizuta and Kenji Mitsugi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251320768 – Supplemental material for Comparison of inflammatory markers before and after nanoliposomal irinotecan and fluorouracil with folic acid in patients with pancreatic cancer: results from the NAPOLEON-2 study (NN-2302)
Supplemental material, sj-docx-2-tam-10.1177_17588359251320768 for Comparison of inflammatory markers before and after nanoliposomal irinotecan and fluorouracil with folic acid in patients with pancreatic cancer: results from the NAPOLEON-2 study (NN-2302) by Tomonori Araki, Kohei Hayashi, Mototsugu Shimokawa, Taiga Otsuka, Yuki Sonoda, Takuya Honda, Taro Shibuki, Junichi Nakazawa, Shiho Arima, Keisuke Miwa, Futa Koga, Yujiro Ueda, Yoshihito Kubotsu, Hozumi Shimokawa, Shigeyuki Takeshita, Kazuo Nishikawa, Azusa Komori, Satoshi Otsu, Ayumu Hosokawa, Tatsunori Sakai, Hisanobu Oda, Machiko Kawahira, Shuji Arita, Hiroki Taguchi, Kengo Tsuneyoshi, Toshihiro Fujita, Takahiro Sakae, Yasunori Kawaguchi, Tsuyoshi Shirakawa, Toshihiko Mizuta and Kenji Mitsugi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359251320768 – Supplemental material for Comparison of inflammatory markers before and after nanoliposomal irinotecan and fluorouracil with folic acid in patients with pancreatic cancer: results from the NAPOLEON-2 study (NN-2302)
Supplemental material, sj-docx-3-tam-10.1177_17588359251320768 for Comparison of inflammatory markers before and after nanoliposomal irinotecan and fluorouracil with folic acid in patients with pancreatic cancer: results from the NAPOLEON-2 study (NN-2302) by Tomonori Araki, Kohei Hayashi, Mototsugu Shimokawa, Taiga Otsuka, Yuki Sonoda, Takuya Honda, Taro Shibuki, Junichi Nakazawa, Shiho Arima, Keisuke Miwa, Futa Koga, Yujiro Ueda, Yoshihito Kubotsu, Hozumi Shimokawa, Shigeyuki Takeshita, Kazuo Nishikawa, Azusa Komori, Satoshi Otsu, Ayumu Hosokawa, Tatsunori Sakai, Hisanobu Oda, Machiko Kawahira, Shuji Arita, Hiroki Taguchi, Kengo Tsuneyoshi, Toshihiro Fujita, Takahiro Sakae, Yasunori Kawaguchi, Tsuyoshi Shirakawa, Toshihiko Mizuta and Kenji Mitsugi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-4-tam-10.1177_17588359251320768 – Supplemental material for Comparison of inflammatory markers before and after nanoliposomal irinotecan and fluorouracil with folic acid in patients with pancreatic cancer: results from the NAPOLEON-2 study (NN-2302)
Supplemental material, sj-docx-4-tam-10.1177_17588359251320768 for Comparison of inflammatory markers before and after nanoliposomal irinotecan and fluorouracil with folic acid in patients with pancreatic cancer: results from the NAPOLEON-2 study (NN-2302) by Tomonori Araki, Kohei Hayashi, Mototsugu Shimokawa, Taiga Otsuka, Yuki Sonoda, Takuya Honda, Taro Shibuki, Junichi Nakazawa, Shiho Arima, Keisuke Miwa, Futa Koga, Yujiro Ueda, Yoshihito Kubotsu, Hozumi Shimokawa, Shigeyuki Takeshita, Kazuo Nishikawa, Azusa Komori, Satoshi Otsu, Ayumu Hosokawa, Tatsunori Sakai, Hisanobu Oda, Machiko Kawahira, Shuji Arita, Hiroki Taguchi, Kengo Tsuneyoshi, Toshihiro Fujita, Takahiro Sakae, Yasunori Kawaguchi, Tsuyoshi Shirakawa, Toshihiko Mizuta and Kenji Mitsugi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-5-tam-10.1177_17588359251320768 – Supplemental material for Comparison of inflammatory markers before and after nanoliposomal irinotecan and fluorouracil with folic acid in patients with pancreatic cancer: results from the NAPOLEON-2 study (NN-2302)
Supplemental material, sj-docx-5-tam-10.1177_17588359251320768 for Comparison of inflammatory markers before and after nanoliposomal irinotecan and fluorouracil with folic acid in patients with pancreatic cancer: results from the NAPOLEON-2 study (NN-2302) by Tomonori Araki, Kohei Hayashi, Mototsugu Shimokawa, Taiga Otsuka, Yuki Sonoda, Takuya Honda, Taro Shibuki, Junichi Nakazawa, Shiho Arima, Keisuke Miwa, Futa Koga, Yujiro Ueda, Yoshihito Kubotsu, Hozumi Shimokawa, Shigeyuki Takeshita, Kazuo Nishikawa, Azusa Komori, Satoshi Otsu, Ayumu Hosokawa, Tatsunori Sakai, Hisanobu Oda, Machiko Kawahira, Shuji Arita, Hiroki Taguchi, Kengo Tsuneyoshi, Toshihiro Fujita, Takahiro Sakae, Yasunori Kawaguchi, Tsuyoshi Shirakawa, Toshihiko Mizuta and Kenji Mitsugi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-6-tam-10.1177_17588359251320768 – Supplemental material for Comparison of inflammatory markers before and after nanoliposomal irinotecan and fluorouracil with folic acid in patients with pancreatic cancer: results from the NAPOLEON-2 study (NN-2302)
Supplemental material, sj-docx-6-tam-10.1177_17588359251320768 for Comparison of inflammatory markers before and after nanoliposomal irinotecan and fluorouracil with folic acid in patients with pancreatic cancer: results from the NAPOLEON-2 study (NN-2302) by Tomonori Araki, Kohei Hayashi, Mototsugu Shimokawa, Taiga Otsuka, Yuki Sonoda, Takuya Honda, Taro Shibuki, Junichi Nakazawa, Shiho Arima, Keisuke Miwa, Futa Koga, Yujiro Ueda, Yoshihito Kubotsu, Hozumi Shimokawa, Shigeyuki Takeshita, Kazuo Nishikawa, Azusa Komori, Satoshi Otsu, Ayumu Hosokawa, Tatsunori Sakai, Hisanobu Oda, Machiko Kawahira, Shuji Arita, Hiroki Taguchi, Kengo Tsuneyoshi, Toshihiro Fujita, Takahiro Sakae, Yasunori Kawaguchi, Tsuyoshi Shirakawa, Toshihiko Mizuta and Kenji Mitsugi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-7-tam-10.1177_17588359251320768 – Supplemental material for Comparison of inflammatory markers before and after nanoliposomal irinotecan and fluorouracil with folic acid in patients with pancreatic cancer: results from the NAPOLEON-2 study (NN-2302)
Supplemental material, sj-docx-7-tam-10.1177_17588359251320768 for Comparison of inflammatory markers before and after nanoliposomal irinotecan and fluorouracil with folic acid in patients with pancreatic cancer: results from the NAPOLEON-2 study (NN-2302) by Tomonori Araki, Kohei Hayashi, Mototsugu Shimokawa, Taiga Otsuka, Yuki Sonoda, Takuya Honda, Taro Shibuki, Junichi Nakazawa, Shiho Arima, Keisuke Miwa, Futa Koga, Yujiro Ueda, Yoshihito Kubotsu, Hozumi Shimokawa, Shigeyuki Takeshita, Kazuo Nishikawa, Azusa Komori, Satoshi Otsu, Ayumu Hosokawa, Tatsunori Sakai, Hisanobu Oda, Machiko Kawahira, Shuji Arita, Hiroki Taguchi, Kengo Tsuneyoshi, Toshihiro Fujita, Takahiro Sakae, Yasunori Kawaguchi, Tsuyoshi Shirakawa, Toshihiko Mizuta and Kenji Mitsugi in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We thank all patients, their families, and all investigators who participated in the NAPOLEON 2 study and the Saga Study Group of Liver Disease for their cooperation. We also thank Editage for English language editing.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
