Abstract
Background:
Nanoliposomal irinotecan with fluorouracil and folinic acid (NFF) is a standard regimen for patients with unresectable or recurrent pancreatic cancer (urPC) after gemcitabine-based chemotherapy. However, little is known about the clinical impact of previous chemotherapy on the effects of NFF in the real world.
Objectives:
We retrospectively evaluated whether the efficacy and safety of NFF differed depending on the history of prior chemotherapy among urPC patients receiving NFF.
Design:
This study was conducted using the real-world data of Japanese patients with urPC who received NFF in the multicenter NAPOLEON-2 study.
Methods:
We compared the efficacy and adverse events (AEs) after NFF initiation between previous irinotecan users and non-users, fluorouracil users and non-users, and platinum users and non-users. We also investigated whether the treatment duration of gemcitabine plus nab-paclitaxel (GnP) influenced the efficacy of NFF among patients treated with NFF as a second-line therapy after GnP.
Results:
Overall, 161 patients were enrolled, all of whom had previously received gemcitabine. In the efficacy analysis between irinotecan users and non-users, the median overall survival (OS) was 9.2 and 8.0 months, respectively (hazard ratio (HR) 0.88; p = 0.66). For fluorouracil, the median OS was 9.1 months for users and 7.6 months for non-users (HR 0.98; p = 0.93). For platinum, the median OS was 9.1 months for users and 8.0 months for non-users (HR 0.88; p = 0.67). There was no clinical difference in AEs between irinotecan users and non-users, which affected ⩾10% of patients in both groups. The median OS in the group with previous GnP for ⩾7.8 months was 9.5 months, while it was 4.8 months in the group with GnP for <7.8 months (HR 0.50; p < 0.01).
Conclusion:
NFF may be effective and tolerable, even in patients previously treated with irinotecan-, fluorouracil-, or platinum-based chemotherapy, in the real world.
Plain language summary
Nanoliposomal irinotecan with fluorouracil (chemotherapy drugs) with folinic acid (used to decrease the toxic effects of cancer drugs), collectively referred to as NFF therapy, is a common treatment for patients with pancreatic cancer that cannot be removed by surgery or that has come back after gemcitabine-based chemotherapy. However, our understanding of how previous chemotherapy influences the effects of NFF is limited. This study looked at whether the effects of NFF varied based on the patients’ history of prior chemotherapy. The study used data from Japanese patients with pancreatic cancer who received NFF. The researchers compared the effectiveness and side effects of NFF between patients with and without previous use of chemotherapy (irinotecan, fluorouracil, or platinum-based chemotherapy). They also assessed whether the length of treatment with gemcitabine plus nab-paclitaxel (GnP), another chemotherapy regimen, influenced the effectiveness of subsequent NFF after GnP. A total of 161 patients were included in the study. The results showed that the average overall survival time was similar between irinotecan users and non-users (9.2 vs. 8.0 months), fluorouracil users and non-users (9.1 vs. 7.6 months), and platinum users and non-users (9.1 vs. 8.0 months), suggesting that previous chemotherapy did not influence the length of overall survival in patients treated with NFF. Furthermore, there were no differences in the side effects of NFF between irinotecan users and non-users. However, patients who received previous GnP for 7.2 months or longer had a longer average overall survival time (9.2 months) than those who received GnP for less than 7.2 months (4.8 months). These observations suggest that NFF appears to be effective and safe for patients with pancreatic cancer that cannot be removed by surgery, regardless of their previous chemotherapy treatments. Therefore, NFF may expand the number of treatment options available to this population.
Keywords
Introduction
Pancreatic cancer is one of the most difficult cancers to treat. It is the seventh leading cause of cancer mortality worldwide and the fourth leading cause in Japan.1,2 Surgical resection is the only curative treatment for pancreatic cancer, but more than 80% of patients are unresectable at initial diagnosis because they have either distant metastases or local invasion. 3 Also, there is no established screening method for pancreatic cancer. 4 Even in resectable cases, most patients tend to experience a relapse within 2 years after surgery. 5 Therefore, systemic chemotherapy plays an important role in the treatment of patients with unresectable or recurrent pancreatic cancer (urPC).
Gemcitabine improves clinical symptoms and prolongs the survival of patients with advanced pancreatic cancer, and it has been established as the standard of care. 6 Subsequently, FOLFIRINOX (combination of fluorouracil, leucovorin, irinotecan, and oxaliplatin) and gemcitabine plus nab-paclitaxel (GnP) demonstrated significantly prolonged survival compared with gemcitabine monotherapy in phase III trials,7,8 and both regimens are recommended as first-line chemotherapy strategies for advanced pancreatic cancer. The outcomes of chemotherapy for advanced pancreatic cancer have improved, but chemotherapy is frequently discontinued owing to disease progression or adverse events (AEs). Thus, establishing effective and safe treatments is essential for improving the prognosis of patients with pancreatic cancer.
Recently, nanoliposomal irinotecan (Nal-IRI) with fluorouracil and folinic acid (NFF) has demonstrated a significant survival benefit compared with fluorouracil and folinic acid after disease progression in patients undergoing gemcitabine-based chemotherapy according to the results of the randomized phase III NAPOLI-1 trial. 9 Nal-IRI is a liposomal encapsulated formulation of the topoisomerase-1 inhibitor irinotecan, which is thought to selectively reach tumors by tumor blood vessel immaturity and leakiness compared with Nal-IRI. 10 Some preclinical data have revealed that conventional irinotecan might have antitumor effects via its active metabolite SN-38 and that tumor cells that are resistant to SN-38 might also exhibit resistance to other topoisomerase I inhibitors.11,12 Nal-IRI is a topoisomerase I inhibitor, so there is a possibility that the therapeutic effect of Nal-IRI might be limited in patients who were previously treated with conventional irinotecan. However, these were pre-clinical data, and there are limited reports based on actual clinical practice. Moreover, patients who have used fluorouracil as a previous therapy might tolerate NFF. The NAPOLI-1 study showed the outcomes of NFF after disease progression followed by gemcitabine-based chemotherapy; however, little is known about the efficacy and safety of NFF in the real-world setting, or when used after fluorouracil-based or platinum-based chemotherapy. In this study, we analyzed whether the efficacy and safety of NFF therapy differed depending on the history of prior therapies among patients who received NFF therapy in the real-world setting.
Materials and methods
Study design
This analysis was a pre-planned analysis of a multicenter retrospective study of NFF administered to patients with urPC (NAPOLEON-2 study) by gastroenterology and medical oncology specialists in Japan. 13 The NAPOLEON-2 study was conducted to investigate the efficacy and safety of NFF by reviewing the medical records of patients with urPC who received NFF at any of 20 institutions from June 2020 to May 2021, and were followed up until November 30, 2022. The efficacy of NFF in previous irinotecan users and non-users only in the third-line or later group has been reported previously. 13 In this analysis, we compared the efficacy and AEs after NFF initiation between previous irinotecan users and non-users among all patients (analysis 1), and between fluorouracil users and non-users (analysis 2). Additional analyses included the comparison of efficacy between platinum users and non-users, and between longer previous GnP duration cases and others in first-line GnP followed by second-line NFF cases based on the treatment period cutoff in our previous data. 14
NFF was administered as a 90-min intravenous infusion of Nal-IRI (70 mg/m2), a 2-h intravenous infusion of L-leucovorin (200 mg/m2), and a 46-h continuous intravenous infusion of fluorouracil (2400 mg/m2) every 2 weeks. The treatment cycles were repeated until disease progression, the emergence of unacceptable toxicity, or the patient’s refusal to continue the treatment. Modifications were allowed, such as dose reduction (including at treatment initiation) or dose delay due to toxicity. This study was conducted with the approval of each participating institution’s institutional review board or ethics committee and according to the Declaration of Helsinki. The reporting of this study conforms to the STROBE statement 15 (Supplemental File 1).
Assessments
The primary endpoint of the study was overall survival (OS). The other endpoints included the proportion of patients who achieved an objective response, disease control, progression-free survival (PFS), dose intensity, and AEs. Computed tomography or interaction magnetic resonance imaging was used to evaluate the antitumor response, which was graded according to the Response Evaluation Criteria in Solid Tumors (RECIST), version 1.1, 16 as a complete response (CR), partial response (PR), stable disease (SD), or progressive disease (PD). The objective response rate (ORR) was defined as a CR or PR, and the disease control rate (DCR) was defined as a CR or PR with SD as the best response. OS was calculated as the interval from the date of NFF initiation to the date of death from any cause or the censoring date at the final follow-up examination. PFS was calculated as the interval from the date of NFF initiation to the date of progression or death from any cause, whichever came first, or the date of censoring at the final follow-up examination. Treatment-related AEs were assessed according to the Common Terminology Criteria for Adverse Events (CTCAE), version 5.0. 17
Statistical analyses
Patient characteristics were evaluated at the start of NFF, including age, sex, Eastern Cooperative Oncology Group (ECOG) performance status, history of pancreatectomy, history of biliary drainage, primary tumor site, disease status, metastatic site, past treatment regimen and duration, and carbohydrate antigen 19-9 and UGT1A1 status. The duration of previous chemotherapy was calculated as the interval from the date of all prior palliative chemotherapy to the date of NFF initiation. PFS and OS were estimated using the Kaplan–Meier method, and the probability was compared using the log-rank test and the Cox proportional-hazards model. Hazard ratios (HRs) were expressed with 95% confidence intervals (95% CIs). p < 0.05 was considered statistically significant. The characteristics of the patient groups were compared using standardized mean differences (SMDs) to assess group balance regardless of the number of cases. Data were collected by clinicians with expertise in clinical research under the supervision of the statistician and then centrally managed. These statistical analyses were performed using R, version 4.2.0 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Patient characteristics
During the period from March 2020 to May 2021, a total of 161 patients were enrolled in this study (Figure 1). The median follow-up period was 7.3 months (95% CI 5.6–8.9 months), and the main data have been reported previously. 13 The patient characteristics are shown in Table 1. The median age was 67 years (range 38–85 years), and 88 patients (55%) were male. All patients had received gemcitabine previously. Overall, 104 (65%) and 57 (35%) patients received NFF as second-line or third-line or later treatment, respectively. In analysis 1, there were several clinically meaningful differences in patient characteristics, including age, lung metastasis, NFF treatment line, and duration of previous chemotherapy. Similarly, there were meaningful differences in the history of pancreatectomy, tumor location, NFF treatment line, and duration of previous chemotherapy in analysis 2.

Flow diagram of the NAPOLEON-2 study.
Patient characteristics.

CA 19-9, carbohydrate antigen 19–9; ECOG, Eastern Cooperative Oncology Group; NA, not applicable; NFF, nanoliposomal irinotecan and fluorouracil with folinic acid; SMD, standardized mean difference; UGT1A1, uridine diphosphate glucuronosyltransferase family 1 member A1.
Analysis between previous irinotecan users and non-users (analysis 1)
We analyzed the efficacy of NFF in previous irinotecan users (n = 18) and non-users (n = 143) among all patients (Figure 1). All of the previous irinotecan users received FOLFIRINOX prior to NFF. The median OS of irinotecan users was 9.2 months compared with 8.0 months for non-users (HR 0.88, 95% CI 0.49–1.57; p = 0.66; Figure 2(a)). The median PFS of irinotecan users was 2.8 months compared with 3.6 months for non-users (HR 1.23, 95% CI 0.74–2.05; p = 0.42; Figure 2(b)). There were no significant differences between the two groups in OS and PFS. The ORR was 6% and 5% for irinotecan users and non-users, respectively, with no clinically significant difference in the rate among the two groups (SMD = 0.03). The DCR was 44% and 53% for irinotecan users and non-users, respectively, with no clinically significant difference in the rate among the two groups (SMD = 0.18; Table 2). The median number of treatment cycles was 6 (range 1–38) for irinotecan users and 5 (1–31) for non-users. The median relative dose intensity (RDI) for Nal-IRI was 87.1% for irinotecan users and 81.1% for non-users (SMD = 0.33). The median RDI for fluorouracil was 93.5% for irinotecan users and 89.2% for non-users (SMD = 0.18; Supplemental Table 1). Treatment-related AEs affecting ⩾10% of the patients are summarized in Supplemental Table 2(A). There was no clinical difference between the two groups. We assumed that confounding factors were present in the two groups; we therefore performed additional Cox regression analysis for OS, adjusting the following covariates: ECOG performance status, liver metastases, lung metastases, peritoneal metastases, and NFF treatment line. We observed no significant difference in the adjusted analysis (adjusted HR 0.89, 95% CI 0.41–1.92; p = 0.76).
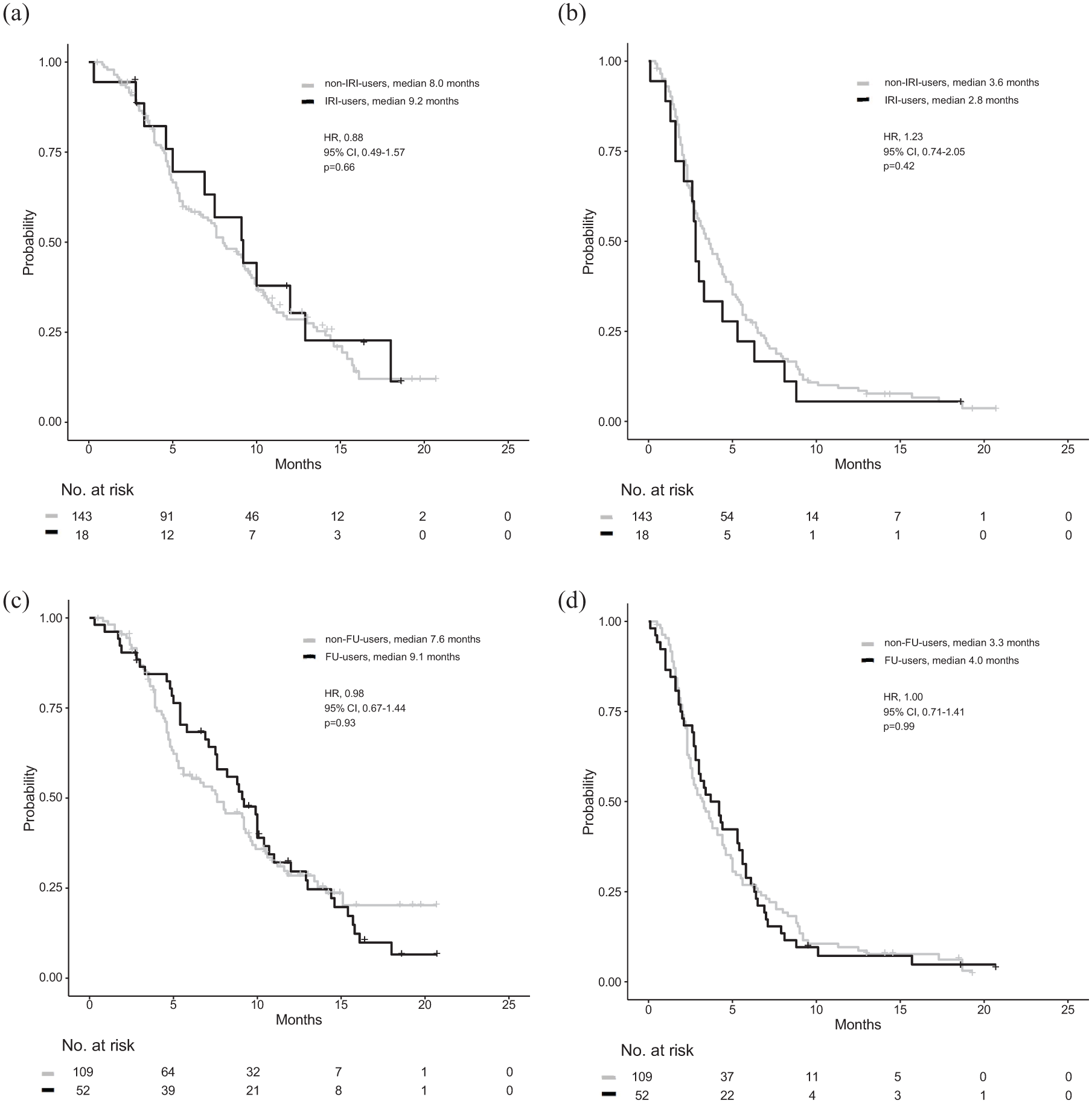
Kaplan–Meier survival curves for nanoliposomal irinotecan and fluorouracil with leucovorin. (a) Comparison of overall survival between irinotecan users and non-users. (b) Comparison of progression-free survival between irinotecan users and non-users. (c) Comparison of overall survival between fluorouracil users and non-users. (d) Comparison of progression-free survival between fluorouracil users and non-users.
Response to NFF among irinotecan users and non-users and among fluorouracil users and non-users.

CR, complete response; DCR, disease control rate; NE, not evaluable; NFF, nanoliposomal irinotecan and fluorouracil with folinic acid; ORR, overall response rate; PD, progressive disease; PR, partial response; SD, stable disease; SMD, standardized mean difference.
The median duration of previous irinotecan treatment was 6.9 months, and nine patients were administered irinotecan just before NFF. There was no significant difference in OS between those with previous irinotecan treatment for a duration of <6.9 months and those with a duration of ⩾6.9 months (median 7.5 vs 10.0 months; HR 0.68, 95% CI 0.22–2.20; p = 0.53; Supplemental Figure 1(A)). Also, there was no significant difference in OS between patients who were administered irinotecan just before NFF and those who were not (median 9.2 vs 9.1 months; HR 0.61, 95% CI 0.18–2.06; p = 0.43; Supplemental Figure 1(B)). All previous irinotecan users had received FOLFIRINOX prior to NFF; seven patients received FOLFIRINOX as first-line therapy, another seven as second-line therapy, and four patients as third-line therapy (Supplemental Table 3). Nine patients had received FOLFIRINOX just prior to NFF, and the remaining nine had not. The intervals from discontinuation of FOLFIRINOX to NFF were short in the group receiving treatment just prior (within approximately 1 month). Supplemental Table 4 shows the comparison of antitumor responses to NFF between the PR cases and the SD + PD cases with irinotecan. There was no significant difference in OS between the PR cases and the SD + PD cases with irinotecan (median 9.2 vs 7.5 months; HR 0.89, 95% CI 0.29–2.76; p = 0.85; Supplemental Figure 1(C)). Moreover, among all previous irinotecan users, 14 discontinued NFF owing to PD, and 4 discontinued NFF owing to AE; the best response was SD in 3 patients, and the outcome was NE in 1 patient. As shown in Supplemental Table 3, the OS of three patients who stopped FOLFIRINOX owing to AE was more than 10 months; however, four patients had an OS of more than 10 months.
Analysis between previous fluorouracil users and non-users (analysis 2)
We analyzed the OS of patients after dividing them into two groups based on previous fluorouracil use (Figure 1). The previous fluorouracil regimens were as follows: S-1 (n = 29), FOLFIRINOX (n = 11), both FOLFIRINOX and S-1 (n = 7), gemcitabine + S-1 (n = 4), and both FOLFOX (combination of fluorouracil, leucovorin, and oxaliplatin) and S-1 (n = 1). The median OS of fluorouracil users (n = 52) was 9.1 months compared with 7.6 months for fluorouracil non-users (n = 109; HR 0.98, 95% CI 0.67–1.44; p = 0.93; Figure 2(c)). The median PFS of fluorouracil users was 4.0 months compared with 3.3 months for fluorouracil non-users (HR 1.00, 95% CI 0.71–1.41; p = 0.99; Figure 2(d)). There were no significant differences between the two groups in OS and PFS. The ORR was 6% and 5% for fluorouracil users and non-users, respectively, with no clinically significant difference in the rate between the two groups (SMD = 0.05). The DCR was 56% and 50% for fluorouracil users and non-users, respectively, with no clinically significant difference in the rate between the two groups (SMD = 0.11; Table 2). The median number of treatment cycles was 7 (range 1–31) for fluorouracil users and 5 (range 1–38) for fluorouracil non-users. The median RDI for Nal-IRI was 79.4% for fluorouracil users and 82.9% for fluorouracil non-users (SMD = 0.09). The median RDI for fluorouracil was 85.3% for fluorouracil users and 92.9% for fluorouracil non-users (SMD = 0.34; Supplemental Table 1). There was no significant difference in AEs between the two groups (Supplemental Table 2(B)).
Clinical impact of previous platinum therapy or GnP on NFF (additional analyses)
Of the 161 patients, 20 (12%) previously received a platinum-based regimen. The previous platinum-based regimens were as follows: FOLFIRINOX (n = 18), FOLFOX (n = 1), and gemcitabine + cisplatin (n = 1). The median OS of platinum users was 9.1 months compared with 8.0 months for non-users (HR 0.88, 95% CI 0.50–1.55; p = 0.67; Supplemental Figure 2). Supplemental Figure 3 shows a flow diagram of the patients who received GnP and NFF as first-line and second-line chemotherapy, respectively, and their characteristics are shown in Table 3. The median OS of those with a previous GnP duration of ⩾7.2 months was 9.2 months compared with 4.8 months for those with a GnP duration of <7.2 months (HR 0.51, 95% CI 0.31–0.81; p < 0.01; Figure 3). In addition, in our previous report, we conducted a clinical comparison of NFF and FOLFIRINOX when used as second-line treatment after GnP failure 18 ; we found no clinical differences in OS and PFS between NFF and FOLFIRINOX after first-line GnP.
Characteristics of the patients who had received GnP as first-line therapy and NFF as second-line chemotherapy.

CA 19-9, carbohydrate antigen 19–9; ECOG, Eastern Cooperative Oncology Group; GnP, gemcitabine and nab-paclitaxel; NFF, nanoliposomal irinotecan and fluorouracil with folinic acid; SMD, standardized mean difference; UGT1A1, uridine diphosphate glucuronosyltransferase family 1 member A1.
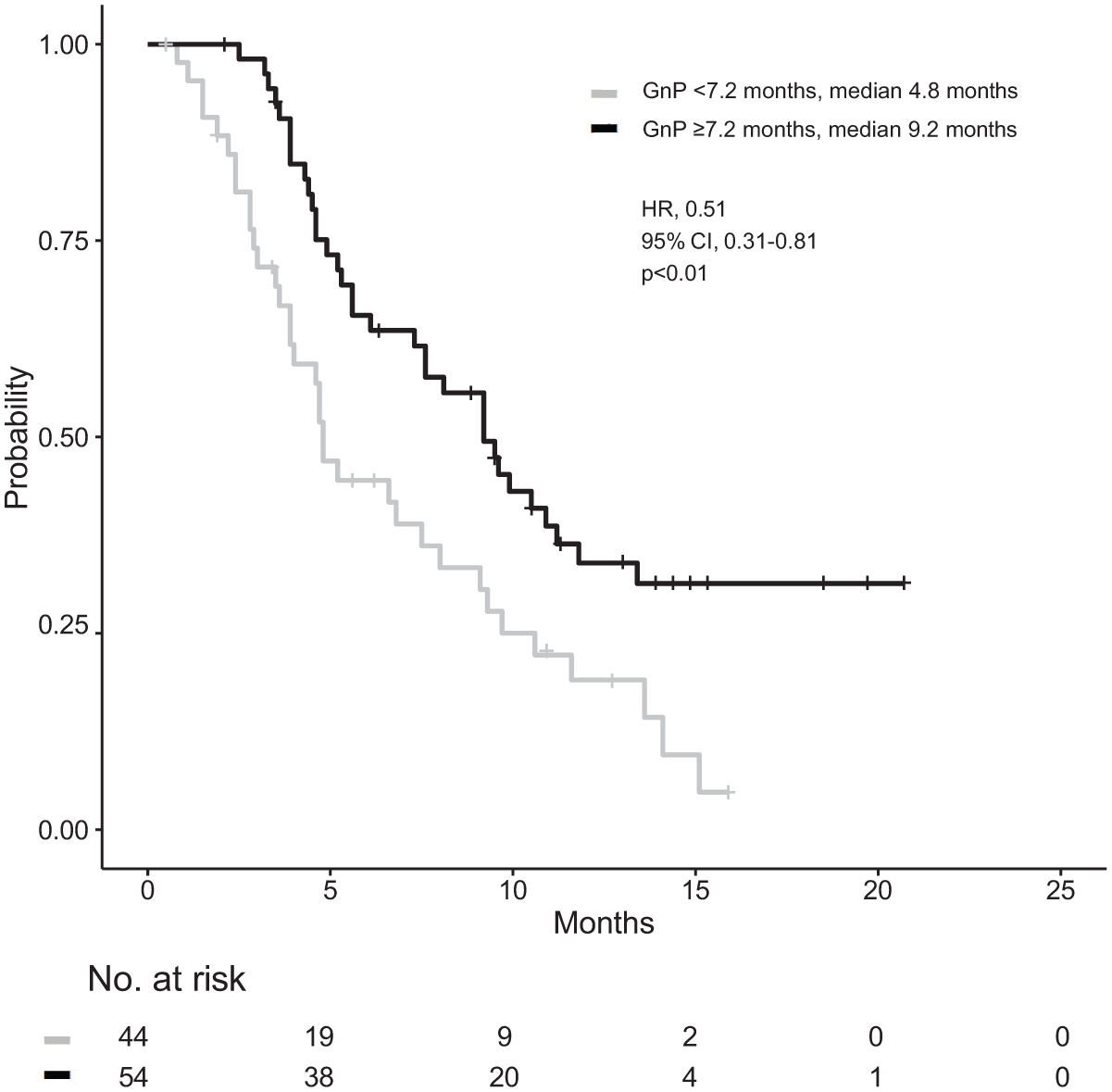
Kaplan–Meier survival curves for nanoliposomal irinotecan and fluorouracil with leucovorin. Comparison of overall survival between those with previous GnP for a duration of ⩾7.2 and <7.2 months.
Discussion
We conducted this study to analyze whether prior chemotherapy had an impact on the efficacy and safety of NFF in patients with urPC in the real-world setting. There are limited clinical data on the influence of prior irinotecan, fluorouracil, and platinum therapy, with only around 100 cases reported in the real-world setting. The median follow-up duration was 7.3 months, which is a little shorter than the median OS of 8.1 months in this study. 13 This might have been influenced by the fact that the median OS for confirmed deaths was 6.5 months, so we considered the median follow-up duration to be reasonable. We used SMDs to compare baseline characteristics because this was a retrospective observational study, and biostatistical calculations such as α, β, and power were not performed, as in a prospective interventional trial. Therefore, p-values were considered estimates, and the interpretation of CIs was an interval estimate. In other words, p-values might show significant differences, but this might not reflect clinical significance. Some analyses in this study did not include a large number of patients, so we considered that the SMD would be useful to evaluate group balance and could be evaluated regardless of the number of patients.
NFF showed similar survival outcomes to those reported in the NAPOLI-1 study and other real-world analyses.9,19 –31 Therefore, NFF might be effective and safe, even in patients with urPC who receive conventional irinotecan as prior therapy, irrespective of its duration; the interval to NFF; and its antitumor efficacy. NFF was similarly safe and effective among patients who had received prior chemotherapy, including fluorouracil and platinum. In addition, NFF was effective in patients who had received prior GnP for a longer duration.
In the present study, there were no significant differences between the irinotecan users and non-users in OS and PFS. Also, there was no clinical difference in the RDI between the two groups. We speculated that irinotecan users could continue NFF therapy for a longer duration because they were significantly younger with better tolerability, and their disease progression might not have been active due to lung metastases or the longer pre-treatment period.32 –34 Moreover, there was no significant difference in OS between the two groups, although all irinotecan users received NFF as third-line or later therapy. Of the irinotecan non-users, around 70% used NFF as a second-line therapy. Bang et al. 35 reported that the prognosis of patients who had previously received irinotecan was poor due to the later-line usage. However, our data suggest that NFF could be indicated for later-line therapy, paying attention to choosing cases with appropriate indications. In the current analysis, there were no clinical differences in OS between irinotecan users and non-users or between fluorouracil users and non-users. As shown in Table 1, most irinotecan users and fluorouracil users received NFF as third- or later-line therapy; their characteristics would indicate those patient populations most likely to benefit from NFF in later-line settings, namely, patients with younger age, a history of pancreatectomy, and lower carbohydrate antigen 19-9 levels. Previous reports have shown that NFF was effective for pre-irinotecan users,26,36 which is consistent with our results. Nal-IRI leaks from tumor vessels into tumor tissues. Nal-IRI concentrates in tumor tissues because the tissue cannot exclude Nal-IRI due to the immature lymph tissue, so Nal-IRI might be effective for irinotecan-resistant cases.37,38 Although a previous report demonstrated that patients with longer intervals between irinotecan and NFF have poorer PFS but better OS than non-irinotecan users, 28 our findings suggested that the OS with NFF might not be influenced by the efficacy or treatment duration of prior irinotecan-containing regimens such as FOLFIRINOX. We should also discuss the presence of resistance to prior irinotecan-containing regimens. Supplemental Table 3 demonstrates that in three patients who stopped FOLFIRINOX owing to AE with prior irinotecan, the OS was more than 10 months; however, four patients had an OS of more than 10 months. Our real-world data were limited; however, there may be potential factors that could influence treatment efficacy with or without the presence of resistance from prior irinotecan-containing regimens.
We also observed no significant differences between fluorouracil users and fluorouracil non-users in OS and PFS. The rate of previous tumor resection was higher for fluorouracil users than non-users (46% vs 15%), probably because adjuvant chemotherapy with S-1 has become a standard of care for resected pancreatic cancer in Japanese patients. 39 Fluorouracil users included more patients with recurrent pancreatic cancer who were supposed to have a better prognosis, as well as fewer patients with liver metastasis who were supposed to have a worse prognosis than fluorouracil non-users (40% vs 62%).40 –42 Taken together, the OS of fluorouracil users, most of whom had received NFF as third-line or later therapy, was similar to that of fluorouracil non-users who had received NFF as second-line therapy.
The OS of patients with longer GnP pre-treatment was significantly better than that of patients with shorter GnP pre-treatment; however, patients with shorter pre-treatment had a significantly higher rate of liver metastasis and carbohydrate antigen 19-9. We speculate that NFF might have a weak impact on patients with shorter GnP pre-treatment because these patients are considered to have strongly progressive tumors.40 –42 As for pre-treatment with platinum, there was no significant difference in OS between platinum users and non-users. Moreover, there were some differences in AEs between users and non-users. Platinum-based agents, including oxaliplatin, are among the main chemotherapy drugs used in pancreatic cancer. However, to the best of our knowledge, there are no reports comparing the efficacy of NFF between platinum users and non-users in the real world; for this reason, we conducted this exploratory analysis to add to the relevant evidence. The clinical importance of this comparison involves the usefulness of NFF in second- or later-line settings when platinum agents are used with fluorouracil.
Overall, the findings of this study could increase the number of regimens available to patients, which could improve their prognosis; however, further analyses are needed as our results are based on a small sample size. This study has some limitations that should be considered. First, as this study was retrospective, our results could have been affected by selection bias. Patients who are eligible for second- or later-line treatment have a relatively better general condition and better prognosis, 43 which might cause inherent selection bias. NFF is reportedly effective regardless of the treatment line, 13 but there is a difference in prognosis between patients receiving post-treatment subsequent to NFF and those receiving best supportive care alone 44 ; this could result in selection bias. In fact, there were some clinical differences in our patients’ characteristics; however, the adjusted analysis showed no clinical significance. Our data should be interpreted with consideration for the presence of selection bias. Also, we are currently conducting a multicenter prospective observational study to verify reproducibility. Second, our study included patients who used NFF chemotherapy, and we analyzed NFF cases irrespective of the therapeutic line at which it was used. Third, the number of patients who used each pre-treatment was small. In particular, the sample size for previous irinotecan users was small, which we consider an important study limitation. Because this study was retrospective, p-values were regarded as estimates, and the interpretation of the CIs was an interval estimate. We assume this to be a considerable limitation that affects the reliability of the survival outcome analysis. However, we conducted comparisons, including in subgroup analysis, as an exploratory analysis because we had limited real-world data, and we consider the results to be clinically meaningful. Indeed, we are currently verifying reproducibility, not only comparing non-irinotecan users and irinotecan users but also prior fluorouracil use. To overcome these limitations, we plan to undertake future prospective studies with different cohorts to validate the repeatability of our observations. In addition, the present study only included a small number of patients who were diagnosed with malignancy based on imaging studies rather than by histological or cytological methods. Some patients who were evaluated during routine clinical practice had no choice other than to undergo systemic chemotherapy in the absence of any histological evidence, such as patients who were diagnosed in an emergency or in anatomically difficult positions. Thus, to build stronger evidence to support the findings of the present study, further studies are warranted.
Conclusion
Based on retrospective data analysis, we demonstrated the efficacy and safety of NFF therapy in those with and without a history of prior therapies among patients with urPC in the real-world setting. NFF might be effective for patients who have received prior irinotecan-based chemotherapy, irrespective of its duration or the interval to NFF; fluorouracil-based, or platinum-based chemotherapy. Therefore, NFF is a possible treatment option after irinotecan-, fluorouracil-, or platinum-based therapies. In the future, it will be important to consider the therapeutic variation in NFF when used as a second-line or later regimen.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251393156 – Supplemental material for Impact of prior chemotherapy on nanoliposomal irinotecan with fluorouracil and folinic acid for unresectable pancreatic cancer: a retrospective analysis of data from the multicenter NAPOLEON-2 study
Supplemental material, sj-docx-1-tam-10.1177_17588359251393156 for Impact of prior chemotherapy on nanoliposomal irinotecan with fluorouracil and folinic acid for unresectable pancreatic cancer: a retrospective analysis of data from the multicenter NAPOLEON-2 study by Tomomi Koike, Yasunori Kawaguchi, Mototsugu Shimokawa, Taiga Otsuka, Kanami Furukawa, Yudai Shinohara, Hozumi Shimokawa, Tomoyasu Yoshihiro, Taro Shibuki, Junichi Nakazawa, Shiho Arima, Keisuke Miwa, Futa Koga, Yujiro Ueda, Yoshihito Kubotsu, Shigeyuki Takeshita, Kazuo Nishikawa, Azusa Komori, Satoshi Otsu, Ayumu Hosokawa, Tatsunori Sakai, Hisanobu Oda, Machiko Kawahira, Shuji Arita, Takuya Honda, Hiroki Taguchi, Kengo Tsuneyoshi, Toshihiro Fujita, Takahiro Sakae, Tsuyoshi Shirakawa, Toshihiko Mizuta and Kenji Mitsugi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251393156 – Supplemental material for Impact of prior chemotherapy on nanoliposomal irinotecan with fluorouracil and folinic acid for unresectable pancreatic cancer: a retrospective analysis of data from the multicenter NAPOLEON-2 study
Supplemental material, sj-docx-2-tam-10.1177_17588359251393156 for Impact of prior chemotherapy on nanoliposomal irinotecan with fluorouracil and folinic acid for unresectable pancreatic cancer: a retrospective analysis of data from the multicenter NAPOLEON-2 study by Tomomi Koike, Yasunori Kawaguchi, Mototsugu Shimokawa, Taiga Otsuka, Kanami Furukawa, Yudai Shinohara, Hozumi Shimokawa, Tomoyasu Yoshihiro, Taro Shibuki, Junichi Nakazawa, Shiho Arima, Keisuke Miwa, Futa Koga, Yujiro Ueda, Yoshihito Kubotsu, Shigeyuki Takeshita, Kazuo Nishikawa, Azusa Komori, Satoshi Otsu, Ayumu Hosokawa, Tatsunori Sakai, Hisanobu Oda, Machiko Kawahira, Shuji Arita, Takuya Honda, Hiroki Taguchi, Kengo Tsuneyoshi, Toshihiro Fujita, Takahiro Sakae, Tsuyoshi Shirakawa, Toshihiko Mizuta and Kenji Mitsugi in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We thank all of the patients and their families, as well as all of the investigators at the 20 institutions that participated in the NAPOLEON-2 study. We would also like to thank the Saga Study Group of Liver Disease (SASLD) for their cooperation. We are indebted to the clinical research coordinators and medical office assistants of the NAPOLEON study group for their assistance in data collection. We thank Emily Woodhouse, PhD, from Edanz (![]() ) for editing a draft of this manuscript.
) for editing a draft of this manuscript.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
