Abstract
Background:
The evolution of nasopharyngeal carcinoma (NPC) involves potential transitions among states of recurrence, metastasis, and/or remission. Understanding the dynamics of NPC progression following radical radiotherapy may help optimize surveillance protocols and patient management.
Objectives:
To characterize the progression dynamics of NPC following radical radiotherapy.
Design:
A retrospective study.
Methods:
The cohort comprised NPC patients who underwent radical intensity-modulated radiotherapy between 2016 and 2022. A multi-state Markov model was employed to estimate transition intensities and probabilities across various disease states, including failure-free, single failure event (recurrence or metastasis, R/M), multi-event progression (recurrence and metastasis, R&M), and death. A Shiny-based web tool was developed to facilitate the clinical translation of the model.
Results:
A total of 4800 NPC patients were included, yielding 18,641 disease-state assessments, with a median follow-up of 3.3 years. The transition intensity from metastasis to death (0.025, 95% confidence interval (CI), 0.021–0.029) was 2.3 times greater than that from recurrence to death (0.011, 95% CI, 0.009–0.014). The estimated probabilities of remaining failure-free, experiencing R/M, and deteriorating to death were 89.9%, 7.7%, and 2.1% at 2 years, respectively. Among patients with recurrence, the probabilities of progression to metastasis and death were 3.8% and 24.5%; among those with metastasis, the probabilities of progression to recurrence and death were 2.1% and 45.0%, respectively. Increasing age, male sex, advanced TNM stage, higher pre-treatment Epstein-Barr virus (EBV) DNA, and detectable post-treatment EBV DNA were associated with treatment failure. A web tool was developed to predict NPC disease-state transitions at 2 years.
Conclusion:
These findings provide initial evidence on the dynamic progression of NPC, which may inform clinical management and patient counseling. Risk-stratified screening and targeted interventions could help mitigate disease progression.
Plain language summary
The risk of failure after therapy is not constant in nasopharyngeal carcinoma (NPC) and the probability of disease progression evolves over time. Static survival estimates are insufficient to address the clinical needs of long-term patient management.
The researchers studied 4,800 NPC patients in China who received radical radiotherapy between 2016 and 2022. They used a special model (Markov model) to track how the disease evolved through different stages-from being cancer-free, to developing recurrence and/or metastasis (including cases where both occurred), and ultimately to death. They also created an online tool to help doctors predict disease progression.
The study included a total of 4,800 NPC patients, yielding 18,641 disease state assessments and found that most NPC patients (89.9%) remained disease-free within two years after treatment. The risk of death was higher for patients with metastasis (45%) compared to those with recurrence (24.5%). Furthermore, older age, male sex, advanced cancer stage, and high EBV DNA levels before treatment were linked to worse outcomes.
This study helps doctors understand how NPC progresses after radiotherapy. The results suggest that personalized follow-up plans and targeted treatments could improve survival, especially for high-risk patients. The online tool developed in this study may assist doctors in predicting disease progression and making better treatment decisions.
Keywords
Introduction
Nasopharyngeal carcinoma (NPC) exhibits a distinct geographical distribution, with the highest incidence observed in southern China and Southeast Asia. 1 While the widespread adoption of intensity-modulated radiotherapy (IMRT) has improved outcomes for non-metastatic NPC, approximately 20%–30% of patients still experience disease recurrence or metastasis (R/M)—the primary causes of treatment failure.2,3 Over the past few decades, extensive research has been conducted to investigate the patterns and risk factors associated with treatment failure in NPC. 4 However, conventional survival estimates provide only static snapshots of prognosis and fail to capture the dynamic nature of NPC progression.
The risk of NPC progression evolves over time, and patients transition through distinct health states.5–7 Understanding these dynamics following radical radiotherapy could help optimize surveillance strategies and personalize patient management. Nevertheless, studying the natural history of the disease is challenging due to variability in disease severity at study entry and differences in follow-up durations. 8 Multi-state Markov (MSM) models offer a powerful framework to address these challenges by quantifying transitions between disease states over time,9,10 yet such approaches remain limited in NPC research.
In this study, we analyzed data from a longitudinal cohort of Chinese NPC patients using an MSM model to elucidate the progression dynamics, estimate transition probabilities between treatment failure events, assess the influence of covariates on these transitions, and compute mean sojourn times in transient states. Furthermore, we developed a web-based tool using the Shiny framework to facilitate the clinical translation and practical use of the model.
Methods
Study population
We conducted a retrospective review of 4,942 non-metastatic NPC patients who were initially diagnosed at Fujian Cancer Hospital between January 2016 and December 2022. All patients were treated with radical IMRT and had complete baseline clinical data available. Exclusion criteria included: (1) prior anti-tumor treatment, (2) disease progression during treatment, or (3) loss to follow-up, defined as less than 3 months of post-treatment follow-up. After applying these initial criteria, 4,850 patients remained. Subsequently, 50 patients with indeterminate event order of concurrent recurrence and metastasis (R&M) were excluded, resulting in a final cohort of 4,800 for MSM analysis. The patient selection process is detailed in Figure S1. The characteristics of these excluded patients with R&M are summarized in Table S1. The institutional review board of Fujian Cancer Hospital approved the study (K2025-317-01), and the requirement for informed consent was waived because of the retrospective design. All data collection and analysis for this research were conducted after obtaining ethical approval. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology Statement (Supplemental File). 11
Patient assessment and treatment
At baseline, patients underwent a comprehensive assessment comprising medical history, physical examination, hematological analysis, and nasopharyngoscopy with biopsy. Primary tumor and regional lymph node staging were determined by magnetic resonance imaging of the head and neck. Distant metastasis screening was performed using either combined 18F-fluorodeoxyglucose positron emission tomography with computed tomography or a standard series of examinations comprising computed tomography of the chest, abdominal ultrasonography, and whole-body bone scintigraphy.
Institutional guidelines recommended radical IMRT for patients with stage I NPC, and IMRT combined with platinum-based chemotherapy for stage II to IVa disease. The radiation dose per fraction ranged from 2.0 to 2.12 Gy, delivered 5 times weekly over 6–7 weeks. Cumulative doses exceeding 66 Gy were delivered to primary tumors and involved lymph nodes. The selection and duration of chemotherapy, encompassing induction, concurrent, and adjuvant phases, were determined by the treating clinician, with predominant use of platinum-based agents. Deviations from guidelines occurred in cases of clinical trial participation, patient refusal, advanced age, or organ dysfunction raising concerns about treatment tolerance. Further treatment details have been described previously. 12
Follow-up
After treatment completion, patients were monitored with clinical evaluations every 3 months for the first 2 years, every 6 months for the next 3 years, and annually thereafter. The follow-up duration was calculated from the end of radiotherapy until the date of death or the last documented clinical or telephone contact. Tumor recurrence was defined as the first identification of a new, pathologically confirmed lesion at least 6 months after radiotherapy, verified by biopsy of the primary site or fine-needle aspiration of a metastatic lesion. For cases in which biopsy was not feasible, recurrence was diagnosed clinically based on the presence of at least two distinct radiological features consistent with disease progression. Salvage treatments were provided when feasible for patients with relapse or metastatic disease.
Covariates
Demographic, tumor, and treatment data were systematically collected. Demographic variables included age and sex. Tumor characteristics encompassed TNM stage and pre‑ and post‑treatment plasma Epstein‑Barr virus (EBV) DNA levels. Treatment modality was also recorded. TNM staging was reassessed by two oncologists with radiological reviews, according to the 8th edition of the American Joint Committee on Cancer staging system.
MSM models for disease states
Disease progression was classified into four distinct states: (1) failure-free, (2) single failure event (R/M), (3) multiple failure events (R&M), and (4) death. According to the Markov assumption, transition rates depend only on the current state. Patients could either remain in their current state, transition to an adjacent state, or progress directly to death from any state. The transition intensity matrix, which determines these transitions, is denoted as Q and is presented below:
The elements qrs (for r ≠ s) represent the instantaneous rate of transition from state r to state s, and are non-negative. Diagonal elements qrr are defined by
We further analyzed progression through two predominant pathways: Pattern A (recurrence as initial failure) and Pattern B (metastasis as initial failure). Patients whose trajectory deviated from the designated sequence were censored at the point of divergence. Univariate and multivariate analyses were developed to evaluate covariate effects on transition risks, with the latter incorporating key clinical predictors to estimate adjusted transition probabilities and state-specific sojourn times.
Sensitivity analysis
To evaluate the time-homogeneity assumption of the Markov model, we performed an annual transition probability analysis. This approach quantified the temporal stability of disease progression by estimating state-transition probabilities for discrete follow-up intervals (years 0–5). The stability of each transition pathway was assessed using the coefficient of variation (CV = standard deviation/mean), with CV < 0.5 indicating relative temporal stability.
Statistical analysis
Participants were followed from enrollment until death, loss to follow-up, or July 20, 2024, whichever occurred first. Overall survival (OS) was defined as death from any cause; disease-free survival (DFS) as relapse, metastasis, or death from any cause; and locoregional control (LRC) and distant control (DC) were defined accordingly. All time-to-event endpoints were measured from the completion of radiotherapy and estimated using the Kaplan–Meier method. The MSM model in continuous time was implemented using the “msm” R package. We developed a web-based application using the “shiny” R package to enable the prediction of a patient’s disease state transition probabilities at 2 years based on specific covariates. All analyses were performed using R (version 4.0.0; R Foundation for Statistical Computing, Vienna, Austria). A two-tailed p < 0.05 was considered statistically significant.
Results
Patient characteristics and survival outcomes
A total of 4,800 patients were ultimately included in the analysis, with 72.4% being male and a median age of 49 years (interquartile range (IQR), 41–57 years). The majority of patients (83.8%) presented with locally advanced disease. The median pretreatment plasma EBV DNA level was 1,550 copies/mL (IQR, 201–9,920 copies/mL). Treatment modalities varied, with 6.2% of patients receiving IMRT alone, 9.9% undergoing concurrent chemoradiotherapy (CCRT), and the majority receiving induction chemotherapy (IC) followed by either CCRT (IC + CCRT, 51.1%) or IMRT (32.7%). The median follow-up duration was 3.3 years (IQR, 1.8–4.9 years). The 2- and 5-year OS rates were 96.2% (95% confidence interval (CI), 95.6%–96.8%) and 87.8% (86.5%–89.1%), respectively. Corresponding DFS rates were 87.1% (86.1%–88.1%) and 77.5% (76.1%–79.0%), LRC rates were 95.1% (94.4%–95.7%) and 90.1% (89.1%–91.2%), and DC rates were 93.3% (92.6%–94.1%) and 90.9% (90.0%–91.9%), respectively. Baseline demographic and clinical characteristics are summarized in Table S2.
Transitions between disease states in NPC
Follow-up ended at death; each patient contributed at least two state assessments (including the time of radiotherapy completion (T0) and the last follow-up), yielding a total of 18,641 state-assessment records. The transitions observed from the current disease state to the subsequent state at each follow-up are detailed in Table S3. Among the 4,800 included NPC patients, the cumulative number of patients who experienced a first failure event of R/M was 663 (13.8%), comprising 335 (7.0%) with locoregional recurrence and 328 (6.8%) with distant metastasis. Of the 663 patients who entered the R/M state, 36 (5.4%) subsequently progressed to develop R&M. Ultimately, 398 patients (8.3%) died from any cause during the study period (Figure 1). At the last follow-up, the distribution of patients across disease states was as follows: 3962 patients (82.5%) remained failure-free, 418 (8.7%) were alive with R/M, and 22 (0.5%) were alive with R&M.
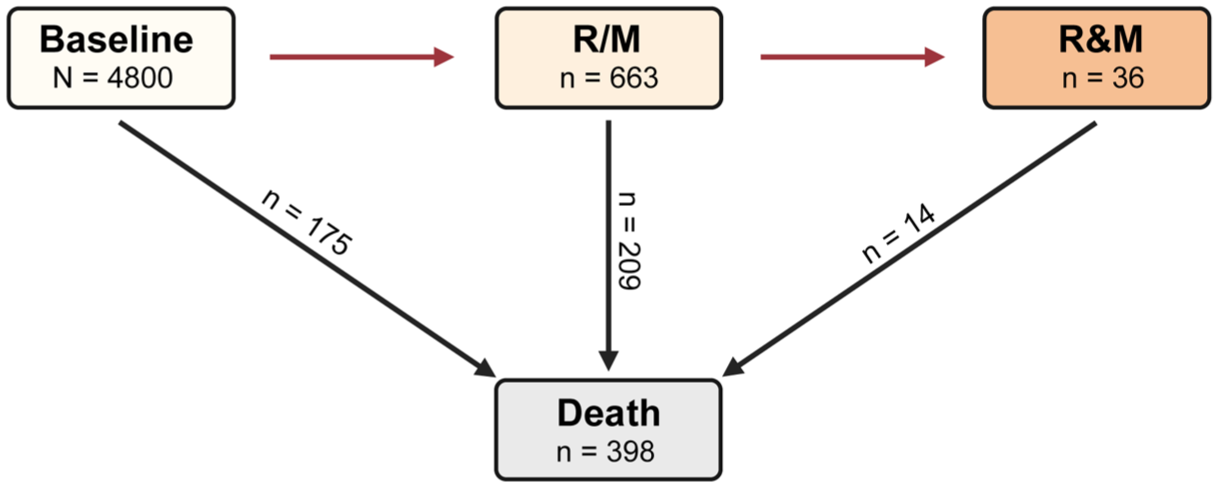
Cumulative number of patients transitioning to each disease state from baseline.
In the pathway-specific analysis, among patients whose first event was recurrence (Pattern A), 75 (22.4%) died, and 23 (6.9%) developed subsequent metastasis; of the latter, 9 (39.1%) later died. Among those whose first event was metastasis (Pattern B), 134 (40.9%) died, and 13 (4.0%) developed subsequent recurrence; of these, 5 (38.5%) later died. Notably, patients with metastasis as the first event were significantly more likely to die during follow-up compared to those with recurrence as the first event (40.9% vs. 22.4%).
Transition intensity analysis further revealed that the intensity from metastasis to death (0.025 (95% CI: 0.021–0.029)) was 2.3 times higher than that from recurrence to death (0.011 (0.009–0.014); Table 1). Additionally, the transition intensity for deteriorating to death from R&M (0.031 (0.019–0.051)) was 1.9 times greater than that of deteriorating to death from R/M (0.016 (0.014–0.018)). Furthermore, the transition intensity of recurrence following radical treatment (0.002 (0.002–0.002)) was comparable to that of metastasis (0.002 (0.002–0.003)), as was the transition strength for developing another failure event (from recurrence to metastasis: 0.003 (0.002–0.004); from metastasis to recurrence: 0.002 (0.001–0.003)).
Transition intensity between different disease states.
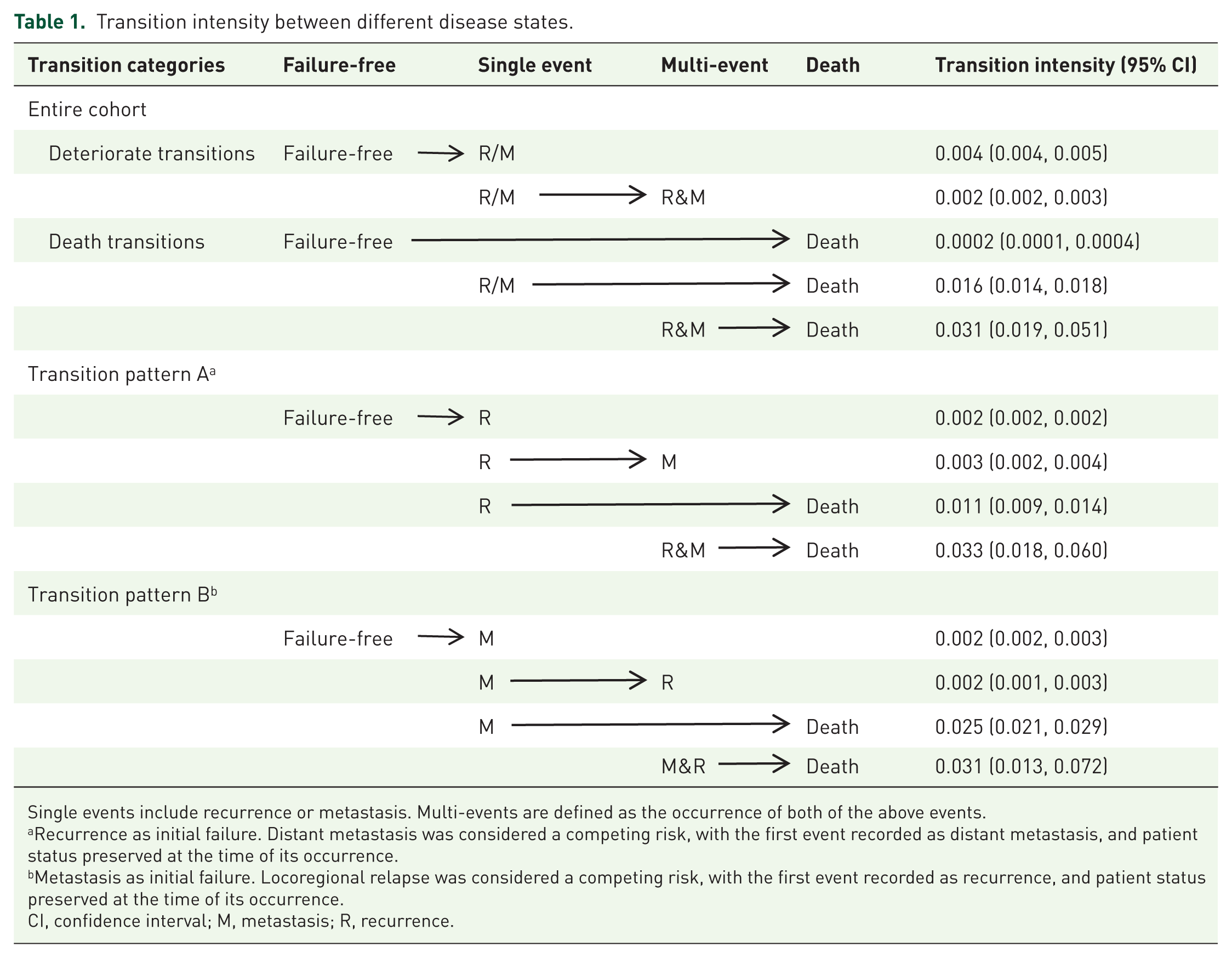
Single events include recurrence or metastasis. Multi-events are defined as the occurrence of both of the above events.
Recurrence as initial failure. Distant metastasis was considered a competing risk, with the first event recorded as distant metastasis, and patient status preserved at the time of its occurrence.
Metastasis as initial failure. Locoregional relapse was considered a competing risk, with the first event recorded as recurrence, and patient status preserved at the time of its occurrence.
CI, confidence interval; M, metastasis; R, recurrence.
Probability of disease state transitions
Transition probabilities between disease states over 2- and 5-year observation periods were estimated for patients across distinct clinical conditions (Figure 2). Among patients receiving radical IMRT, the estimated probabilities of remaining failure-free, experiencing R/M, and transitioning to death were 89.9%, 7.7%, and 2.1% at 2 years, respectively, shifting to 76.7%, 13.2%, and 9.5% at 5 years (Figure 2(a) and (b)). For patients with recurrence, the estimated probabilities of progression to metastasis and death were 3.8% and 24.5% at 2 years, respectively, increasing to 4.3% and 52.2% at 5 years (Figure 2(c) and (d)). Among patients with metastasis, the probabilities of transitioning to recurrence and death were 2.1% and 45.0% at 2 years, respectively, with corresponding values of 1.9% and 77.8% at 5 years (Figure 2(e) and (f)).

Transition probabilities from the unadjusted multi-state model. Probabilities are shown over 2- and 5-year observation intervals for the entire cohort (a, b), patients with transition pattern A (c, d), and patients with transition pattern B (e, f). Arrows represent transitions between states: blue, remaining in the current state; red, progression to R or M; black, progression to death.
Model validation
To evaluate the time‑homogeneity assumption of the Markov model, we performed an annual transition probability analysis as a sensitivity analysis. Key clinical pathways, failure-free → R/M and R/M → death, showed excellent temporal stability (CV < 0.25, Table 2), supporting time homogeneity in primary progression steps. The terminal transition (R&M → death) was fully deterministic, consistent with the clinical course. In contrast, the failure-free → death pathway exhibited moderate temporal variation (CV = 0.578), suggesting that follow-up time may influence direct mortality risk. The transition from R/M to R&M, a rare event, exhibited estimation instability (CV = 0.734), reflecting the complexity of disease progression patterns in clinical practice. Comprehensive annual transition probabilities are provided in Table S4.
Temporal stability analysis of annual transition probabilities.

CV, coefficient of variation; M, metastasis; R, recurrence; SD, standard deviation.
Covariate effects on disease state transitions
The influence of covariates on transitions between distinct disease states is summarized in Table 3. Increasing age (hazard ratio (HR): 1.010 (1.003–1.017)), male sex (HR: 1.460 (1.213–1.757)), advanced T category (HR: 1.556 (1.316–1.839)), advanced N category (HR: 1.783 (1.523–2.088)), higher pretreatment EBV DNA levels (HR: 1.073 (1.054–1.092)), and detectable post-treatment EBV DNA levels (HR: 1.556 (1.276–1.897)) were all associated with an increased hazard of initial treatment failure (transition from a failure-free state). Furthermore, increasing age was a significant risk factor for death, both from the failure-free state (HR: 1.220 (1.170-1.271)) and from the R/M state (HR: 1.041 (1.029-1.054)). A multivariate MSM model refined these associations (Table S5). While the effects of most covariates on the transition to R/M remained consistent with univariate results, the association between IC and treatment failure was not retained in the multivariate model, and the association of male sex with the transition from the R/M state to death was attenuated and no longer significant. Supplementary Kaplan–Meier analyses for OS and DFS confirmed that the evaluated covariates—age, sex, T category, N category, and pre- and post-treatment EBV DNA levels—were significantly associated with survival outcomes (log-rank p < 0.05, Figure S2). The concordance between these conventional survival curves and our Markov-based transition probabilities reinforces the validity of the identified risk factors and highlights the capacity of the MSM framework to delineate state-specific transition risks.
Effects of the covariates on transitions among NPC disease states, shown as HR (95% CI).

Pretreatment EBV DNA levels were log2-transformed (log2 (EBV DNA + 1)) to normalize the distribution before inclusion in the prognostic model. Boldfaced data indicate statistical significance (p < 0.05).
CI, confidence interval; EBV, Epstein–Barr virus; EBV DNApost, post-treatment EBV DNA; EBV DNApre, pretreatment EBV DNA; HR, hazard ratio; NPC, nasopharyngeal carcinoma; R/M, recurrence or metastasis; R&M, recurrence and metastasis.
State-transition probabilities and sojourn times across subgroups
Pretreatment EBV DNA levels were dichotomized at 4,000 copies/mL, and the probabilities of state transitions were subsequently estimated for each subgroup (Figure S3). Across all subgroups, the likelihood of disease progression generally increased with longer observation time, except for transitions from metastasis to recurrence. Notably, probabilities of recurrence, post-recurrence metastasis, and post-metastasis recurrence were similar between the low- and high-EBV DNA groups, whereas the high-EBV DNA group had a significantly higher probability of metastasis. Patients in the high-EBV DNA group were more likely to die following recurrence and after metastasis, and showed an elevated probability of death among those who progressed from recurrence to metastasis. Interestingly, as EBV DNA levels increased, the probability of transitioning from recurrence to death decreased in patients with metastasis.
We estimated the mean sojourn times in each transient state for the overall cohort and for subgroups (Table S6). In the overall population, mean sojourn times were 18.9 years for the failure-free state, 4.5 years for the R/M state, and 2.7 years for the R&M state. Among patients with recurrence as the first event, mean sojourn times were 6.0 years in the recurrence state and 2.6 years in the subsequent metastasis state. Conversely, for patients with metastasis as the initial event, mean sojourn times were 3.1 years in the metastasis state and 2.7 years in the subsequent recurrence state.
Web-based application development
To facilitate the practical implementation of our MSM model, we developed an interactive web-based tool using the R “Shiny” framework. This publicly accessible platform (https://huangshiny.shinyapps.io/NPC_MSM/) allows clinicians to input individual patient parameters—including age, disease stage (T/N category), EBV DNA dynamics, and treatment history—to generate personalized estimates of 2-year disease transition probabilities. An illustrative case is presented in Figure 3, featuring a 60-year-old male with locally advanced disease, a pretreatment EBV DNA load of 4,000 copies/mL, and persistent post-treatment EBV DNA positivity. For this patient, the tool serves as an aid in patient communication. Translating a patient’s prognosis into individualized probabilities (e.g., “You have a 94% chance of remaining disease-free in the next two years, but we will closely monitor the 5% risk of recurrence”) moves beyond vague qualitative descriptors, enabling more precise risk counseling and informed shared decision-making.

Illustration for the Shiny-web-based application (https://huangshiny.shinyapps.io/NPC_MSM/). The left side is the panel to input the participant’s information, and the right side shows the result, namely the transition probability with 95% CI at 2 years. After entering the pretreatment EBV DNA value, the application will be subject to log2 (EBV DNA + 1) transformation to normalize its distribution for subsequent model integration.
Discussion
This large-scale comprehensive study elucidates dynamic transitions among disease progression states and investigates the impact of risk factors on state transitions of NPC in a Chinese cohort—an aspect previously unexplored in the literature. Our findings reveal that the transition intensity from metastasis to death was 2.3 times greater than that from recurrence to death. Within a 2-year observation period, the probabilities of remaining failure-free, developing R/M, and progressing to death were estimated at 89.9%, 7.7%, and 2.1%, respectively. Factors including age, sex, TNM stage, pre- and post-treatment EBV DNA were identified as significant determinants of progression transitions. To support clinical translation, we developed a web-based tool that provides individualized estimates of disease-state transitions. These insights could help enhance clinical management strategies and patient counseling, alleviating disease-related anxiety and reduce unnecessary financial burdens.
Our results demonstrate that 89.9% of patients achieved failure-free survival at the 2-year follow-up, which aligns with the findings of Liu et al., 14 who reported an 82% probability of being alive and tumor-free for patients surviving 2 years after treatment—a critical period associated with the highest risk of recurrence. The relatively low incidence of disease progression (13.8% for R/M) observed in our cohort further underscores the high probability of durable remission following radical radiotherapy for NPC. These data provide clinicians and patients with a clearer understanding of the long-term benefits of current treatment strategies. Nevertheless, outcomes remain poor once progression occurs. The relatively high mortality rates and shorter sojourn times observed in patients who develop R/M highlight the persistent challenge of managing advanced disease. In recent years, significant efforts have been focused on integrating systemic chemotherapy with radiotherapy to improve survival in advanced-stage NPC. 15 The use of CCRT in combination with IC or adjuvant chemotherapy has yielded significant clinical improvements. 16 However, rates of local and especially distant failure remain considerable, and the prognosis for patients with R/M remains suboptimal. 2 Recent advances in immunotherapy offer new promise; for example, adding toripalimab to chemotherapy has been established as a first-line treatment for recurrent or metastatic NPC and has extended median progression-free survival by 13.2 months compared with placebo. 17 Despite these advances, the management of recurrence and metastasis in high-risk groups remains a formidable challenge, calling for continued exploration of novel therapeutic strategies and prognostic biomarkers to improve outcomes in advanced NPC.
Our analysis shows that the transition intensity for recurrence after radical treatment is comparable to that for metastasis. However, patients with metastatic disease demonstrated markedly higher mortality during follow-up, a finding consistent with reports from other cancer centers.3,18 For instance, Wang et al. 19 analyzed 695 NPC patients treated with IMRT and found that distant metastasis accounted for 95 of 133 recorded deaths. Hematogenous dissemination in NPC commonly involves critical organs such as the lungs, liver, and bones. 20 Comparative analyses of primary tumors and distant metastases suggest that systemic spread can occur rapidly following malignant transformation. 21 Prognosis in metastatic NPC is strongly influenced by the anatomical site and extent of metastatic involvement, as reflected in the ninth edition of the NPC staging system, which subclassifies M1 disease based on the number of affected organs and metastatic lesions. 22 Furthermore, Du et al. 23 identified liver involvement as a particularly strong predictor of poor outcomes in NPC. Accumulated evidence indicates that systemic dissemination may begin 2–4 years before primary tumor detection, and adjuvant therapies targeting micrometastatic disease can delay progression. 24 However, treatment-induced remodeling of metastatic clones may select for drug-resistant mutations, promoting outgrowth of more aggressive, therapy-resistant subpopulations. 25
The prognosis of NPC is influenced by a complex interplay of clinical, molecular, and therapeutic factors. 26 Previous studies have delineated a spectrum of risk factors, including sex, age, TNM stage, EBV DNA levels, tobacco use, and excessive alcohol consumption, all of which have been implicated as predictive covariates for NPC progression. 27 However, these investigations have predominantly focused either on the impact of risk factors on progression or the outcomes of patients with R/M. 28 While numerous risk factors have been identified, the fragmented nature of these analyses—often limited to a single disease state—has constrained comprehensive comparisons of how these factors affect transitions across different stages of progression. Our findings indicate that the association of age with mortality was most pronounced for the direct transition from a failure-free state to death (HR: 1.220 per year). This elevated estimate likely reflects the profile of the small (3.6%), elderly-enriched subgroup (61.7% ⩾60 years) in which this event occurred, where all-cause mortality is conflated with a higher burden of frailty and comorbidities. Thus, this estimate is inflated and should therefore be interpreted with caution. Additionally, older age was associated with a stronger impact on the transition from the R/M state to death. The heightened vulnerability of older individuals, coupled with accumulated genetic and epigenetic alterations over time, may predispose them to more aggressive tumor phenotypes even following initial treatment success. 29 These factors may partially account for the elevated mortality rates observed among older NPC patients.
Our analysis identifies advanced T and N categories as significant predictors of recurrence and metastasis, aligning with the established prognostic significance of tumor stage. 22 It is important to note that our study used the AJCC 8th edition staging system. While the updated 9th edition introduces refinements for risk stratification, we believe the fundamental prognostic relationship between T/N stage and outcomes captured by our model is likely to remain valid. Future validation in cohorts staged according to the AJCC 9th edition would be a valuable step toward its broader clinical application. We also note that limited event numbers for certain transitions result in wide CIs, for example, the direct transition to death in T3–4 disease yielded a HR of 2.695 (95% CI: 0.306–23.738). These estimates were imprecise and should be interpreted with caution. Additionally, our findings align with prior studies indicating that female patients with NPC generally exhibit a more favorable prognosis compared with males. 27 This appears to be driven by consistently lower transition risk between key disease states in female patients. However, we found no evidence that specific treatment modalities independently influence disease progression trajectories. The initial association between IC and treatment failure likely reflects its administration in higher-risk patients rather than a causative effect, as it was nullified in multivariate analysis.
Higher EBV DNA levels are frequently correlated with advanced disease and a more aggressive clinical trajectory. 30 In our study, the association between higher pretreatment EBV DNA levels or detectable post-treatment EBV DNA and an increased risk of R/M further underscores the potential utility of viral load as a robust biomarker for tumor aggressiveness and metastatic potential.31,32 However, caution is warranted when interpreting the impact of EBV DNA on disease progression based solely on the level during treatment, as subsequent fluctuations after disease relapse are not captured. Regrettably, due to limitations in our study design, we were unable to systematically collect comprehensive follow-up data on EBV DNA levels, particularly after disease progression, which precluded further analysis of how EBV DNA dynamics influence transitions between states during follow-up. Future longitudinal studies that correlate post-treatment EBV DNA trajectories with clinical outcomes may provide valuable insights into the temporal dynamics of EBV reactivation and its role in driving disease progression. 33
This study introduces, to our knowledge, the first application of an MSM model to quantify disease progression dynamics in NPC patients following radical radiotherapy. Unlike traditional survival endpoints or conditional survival analyses, which provide static or interval-specific estimates, the MSM framework probabilistically models transitions between multiple disease states.10,14 This offers a more dynamic representation of cancer progression and enables estimation of clinically actionable metrics, including state-specific transition probabilities and mean sojourn times.34,35 To support translational application, we developed an interactive web tool that generates individualized risk projections, thereby facilitating a more intuitive understanding of prognosis for patients and clinicians. By replacing ambiguous qualitative statements with transparent, data-driven risk profiles, this approach aligns with growing evidence that quantitative risk communication enhances patients’ cognitive controllability, allowing them to better calibrate expectations and engage in anticipatory coping.36–39 Consequently, this approach holds promise for mitigating anxiety, improving adherence to surveillance protocols through clarified personal risk-benefit understanding, and strengthening the therapeutic alliance—fundamental components of patient-centered oncologic care.40,41 However, the generalizability of our model—developed in an endemic Chinese population—to non-endemic regions requires careful consideration. Geographical variations in NPC incidence, etiology (e.g., differential EBV strain distribution), environmental factors, and treatment protocols may influence disease behavior and transition patterns.26,42,43 The performance and calibration of our prediction tool should therefore be validated in diverse, multi-institutional cohorts before broader clinical implementation. Future collaboration with international centers will be essential to establish the universal applicability of these findings and to refine the model for different patient populations. Furthermore, a key consideration in interpreting our results is the foundational Markov assumption that transition hazards depend only on the current state. While this assumption confers mathematical tractability and yields robust state-specific estimates, it may not fully capture clinical complexity. We observed departures from temporal homogeneity in specific pathways, such as the time-dependent risk of direct death, which may reflect underlying population heterogeneity, age-related comorbidities, or late treatment-related complications.44,45 Furthermore, prognosis may also depend on time already spent in a given state; for example, early recurrence was associated with poorer survival. 7 Future methodological work employing semi-Markov or time-dependent models could further refine our understanding by incorporating sojourn time.
Several limitations also warrant mention. First, the retrospective design inherently carries the risk of selection bias. However, in contrast to clinical trials with stringent inclusion criteria, the data presented here better reflect real-world prognostic estimates, thereby enhancing their applicability to a broader, unselected cohort of NPC patients receiving standard care. While the substantial sample size and recruitment from a high-incidence region enhance the representativeness of our findings for the core affected population in China, the single-center design may limit generalizability to other settings. Second, building the multi-state model required the exclusion of 50 patients (1.0% of the initial cohort) who presented with concurrent recurrence and metastasis, as the temporal sequence of events could not be determined. This subgroup likely represents a more aggressive tumor phenotype. Although methodologically necessary for the current analysis, their exclusion may lead to underestimation of transition intensities into the R&M state and could introduce bias in model estimates. Future studies using statistical approaches that accommodate interval-censored or simultaneous events would be valuable to characterize progression dynamics in this high-risk subgroup. Third, the complexity of the Markov model may pose challenges for clinical implementation. To address this, we developed a web-based tool that allows clinicians to obtain individualized 2-year transition probabilities based on risk factors identified in this study. However, the tool faced challenges with internal validation via bootstrapping for certain transitions due to sparse data and convergence issues. While the tool offers a practical interface for hypothesis generation and exploratory prognosis, its clinical utility for guiding individual patient management requires rigorous external validation. Finally, although disease progression may be influenced by numerous factors, our analysis was confined to demographic and tumor-related covariates because of the retrospective nature of the study. Future investigations that incorporate modifiable variables such as lifestyle, dietary habits, and environmental exposures could provide valuable insights and help inform preventive interventions aimed at delaying NPC progression. 43
Conclusion
Survival outcomes in non-metastatic NPC have improve over the years, and our findings offer valuable insights into the probabilities of state transitions in the context of contemporary therapeutic strategies. These results can assist oncologists and patients in better understanding the likelihood of disease progression, thereby supporting more informed, individualized decisions regarding clinical management and surveillance.
Supplemental Material
sj-docx-1-tam-10.1177_17588359261433798 – Supplemental material for Dynamic disease progression in non-metastatic nasopharyngeal carcinoma post-radical radiotherapy: a Markov model-based analysis in a Chinese cohort
Supplemental material, sj-docx-1-tam-10.1177_17588359261433798 for Dynamic disease progression in non-metastatic nasopharyngeal carcinoma post-radical radiotherapy: a Markov model-based analysis in a Chinese cohort by Ying Li, Zongwei Huang, Zihan Chen, Ting Lin, Jue Wang, Qisi Zhang, Jiajia Zheng, Yuye Lin, Jinghua Lai, Zhiming Zhong and Sufang Qiu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359261433798 – Supplemental material for Dynamic disease progression in non-metastatic nasopharyngeal carcinoma post-radical radiotherapy: a Markov model-based analysis in a Chinese cohort
Supplemental material, sj-docx-2-tam-10.1177_17588359261433798 for Dynamic disease progression in non-metastatic nasopharyngeal carcinoma post-radical radiotherapy: a Markov model-based analysis in a Chinese cohort by Ying Li, Zongwei Huang, Zihan Chen, Ting Lin, Jue Wang, Qisi Zhang, Jiajia Zheng, Yuye Lin, Jinghua Lai, Zhiming Zhong and Sufang Qiu in Therapeutic Advances in Medical Oncology


