Abstract
Rectal cancer recurrence remains a major therapeutic challenge, particularly in patients unresponsive to conventional regimens. Tumors with mismatch-repair-proficient (pMMR) or microsatellite-stable (MSS) status represent the majority of colorectal cancers and are characteristically resistant to immune checkpoint inhibitors. We report a 50-year-old woman with recurrent metastatic KRAS-G12D mutant MSS rectal adenocarcinoma refractory to and with complications to standard chemotherapy regimens who achieved a complete and durable molecular response following initiation of the RIN protocol, a combination of regorafenib, ipilimumab, and nivolumab. A sustained decline in biomarker levels was observed, with both circulating tumor DNA and carcinoembryonic antigen becoming undetectable within 6 months, consistent with a complete molecular response accompanied by a marked interval decrease in FDG-avid disease and sustained radiologic and pathologic remission. This case illustrates the potential for multimodal immune modulation to overcome intrinsic resistance in pMMR/MSS in non-liver metastatic (NLM) colorectal cancer. While previous studies have demonstrated limited benefit of immunotherapy in this tumor subtype, the present findings suggest an emerging therapeutic opportunity that warrants prospective evaluation to confirm efficacy, explore the mechanistic basis, and identify biomarkers predictive of durable response beyond the absence of liver metastases. More effective combinatorial regimens like zanzalintinib and atezolizumb (STELLAR-303) trial, as well as newer generation of CTLA-4 inhibitors like botensilimab, vilastobart, and muzastotug are showing more promise for patients with MSS colorectal cancers in particular who do not have liver metastases (NLM).
Plain language summary
Rectal cancer is usually treated with surgery, chemotherapy, and radiation. However, in some people, the cancer can come back and stop responding to standard treatments. When this happens, there are limited treatment options. This report describes the case of a 50-year-old woman whose rectal cancer returned after surgery, chemotherapy, and radiation. Her cancer continued to grow despite further treatment and complications stemming from it. She was then given a combination of three medicines: regorafenib and two immune-based drugs called ipilimumab and nivolumab. These medicines are designed to help the body’s immune system recognize and attack cancer cells. After starting this treatment, tests showed no signs of active cancer in her blood, and her symptoms improved. She tolerated the treatment well without serious side effects. Although this is only a single patient’s experience in the real-world, this case builds upon the literature that combining immune therapy with targeted therapy may be a promising option for some patients with difficult-to-treat metastatic rectal cancer who do not have liver metastases (NLM). More research is needed to understand who may benefit most from this approach.
Keywords
Introduction
Over the past decade, the treatment landscape for locally advanced rectal cancer (LARC) has evolved substantially, with increasing emphasis on personalized and multimodal therapeutic strategies. Standard management typically involved neoadjuvant chemoradiotherapy (CRT) followed by surgical resection; however, recurrence and resistance to therapy remain significant challenges. Among molecular subtypes, proficient mismatch repair (pMMR) or microsatellite-stable (MSS) tumors account for the majority of rectal cancers and are notoriously resistant to immune checkpoint inhibitors (ICIs), 1 limiting the role of immunotherapy in this population.
Recent studies have explored combination strategies to overcome this resistance, including immune checkpoint blockade with tyrosine kinase inhibitors (TKIs) and re-irradiation approaches, showing early promise in small cohorts. 2 These regimens aim to modulate the tumor microenvironment and enhance immune responsiveness. 3
Here, we present a case of recurrent pMMR/MSS rectal cancer with pelvic and nodal metastases but no liver metastases (NLM) that demonstrated a remarkable clinical and radiologic response to a novel combination therapy comprising regorafenib, ipilimumab, and nivolumab (RIN protocol) alongside palliative hypo-fractionated intensity-modulated re-irradiation. This case highlights a potential therapeutic avenue for a subgroup of patients with NLM-colorectal cancer who are traditionally considered refractory to immunotherapy.
Case
This report adheres to the CARE (Case Report) guideline as outlined by the EQUATOR Network 4 (see Supplemental File 1).
A 50-year-old woman of Middle Eastern descent presented with advanced, recurrent metastatic rectal adenocarcinoma. A review of her past medical history revealed no significant comorbidities, and her family history was negative for malignancy. At the time of her initial diagnosis in 2021, the patient was found to have a moderately differentiated adenocarcinoma of the rectum, located approximately 5 cm from the anal verge. Comprehensive staging imaging, including contrast-enhanced CT of the chest, abdomen, and pelvis, confirmed locally advanced disease without distant metastases, consistent with clinical stage IIIC (cT4bN2M0) according to the AJCC 8th edition. The tumor invaded the mesorectal fascia and adjacent pelvic structures. Histopathological evaluation revealed moderate glandular differentiation, and immunohistochemistry confirmed pMMR and microsatellite stability (MSS). RAS and BRAF mutation testing were reportedly were both negative. The patient subsequently received neoadjuvant chemoradiation (25 Gy pelvic radiotherapy in five fractions) followed by six cycles of neoadjuvant XELOX (capecitabine and oxaliplatin). After completion of therapy, she underwent laparoscopic ultra-low anterior resection with coloanal anastomosis, involving resection of approximately 15 cm of distal colon and rectum. The procedure achieved an R0 resection, with clear circumferential and distal margins confirmed on histopathologic examination, and no residual tumor identified at the margins. She achieved complete remission for 1 year. Fourteen months postoperatively, surveillance CT imaging revealed a recurrent left mesorectal mass measuring 9 × 7.3 cm, invading the cervix, vagina, pelvic musculature, peritoneal reflection, and adjacent ileal loops. She was denied repeat surgery in her home country and subsequently traveled to the United States for further management. The patient’s case was reviewed in a multidisciplinary tumor board following the confirmation of disease recurrence. This review informed the decision to proceed with an immunotherapy-based treatment approach. In addition, the patient underwent a nutritional assessment and was managed by a nutrition specialist during subsequent therapy to facilitate treatment tolerance and holistic clinical care. 5 Chemotherapy with FOLFOXIRI (5-fluorouracil, leucovorin, oxaliplatin, and irinotecan) was initiated; however, treatment was complicated by necrotizing fasciitis of the perineum and lower extremity, necessitating extensive debridement and long road to recovery after months of intensive care unit, hospital stays, and multiple debridements. Following recovery, the patient was referred to our practice, where treatment with the RIN protocol—comprising regorafenib in combination with low-dose ipilimumab (1 mg/kg) and nivolumab (3 mg/kg), commonly referred to as IPI-1/NIVO-3 was initiated off-label since she was not deemed a suitable candidate for further chemotherapy regimens. The patient tolerated the regimen well and experienced no treatment-related adverse events. In addition, five fractions of intensity-modulated radiation therapy (IMRT) were administered for local symptom control, including pain and bleeding. A concise summary of the clinical timeline is presented in Figure 1.

A timeline of all the events and treatments the patient has received over 3 years.
Response to therapy was assessed by measuring circulating tumor DNA (ctDNA) and carcinoembryonic antigen (CEA) levels, which became undetectable after 10 months of continued treatment, as shown in Figures 2 and 3.

Graphical representation of the change in CEA levels during treatment. At treatment initiation in June 2023, the CEA level was 7.3 ng/mL. It dropped to 6.6 ng/mL by October 2023 (Cycle 2 of IPI-1/NIVO-3). On the day of Cycle 3 in November 2023, CEA became undetectable and remained so during subsequent follow-up visits. Together with ctDNA negativity, this finding indicates a mCR.

Graphical representation of the ctDNA (%) levels of the individual clinical biomarker levels identified in this patient throughout the treatment duration. This patient tested positive for KRAS, TP53, APC, and SMAD4 mutations and MSS by NGS testing. As seen above, as the treatment progressed, ctDNA (%) levels declined to negligible levels, making a compelling case for complete eradication of micro-metastatic disease and a complete immunological response.
As shown in Figures 4 and 5, the patient’s PET scan revealed a marked radiologic response after therapy. This case is distinctive as a pMMR/MSS tumor, which is generally considered refractory to immunotherapy. Chen et al. 1 reported on a series that responded favorably to an immunotherapy and anti-VEGF combination regimen.

PET scan images of the patient before and after treatment. (a) and (c) show the pelvis’s transverse section before treatment initiation in October 2023. Enlarged hypermetabolic retroperitoneal lymph nodes can be visualized. (b) and (d) represent imaging performed in January 2024, showing a decrease in size and FDG avidity of the retroperitoneal lymph nodes following treatment.
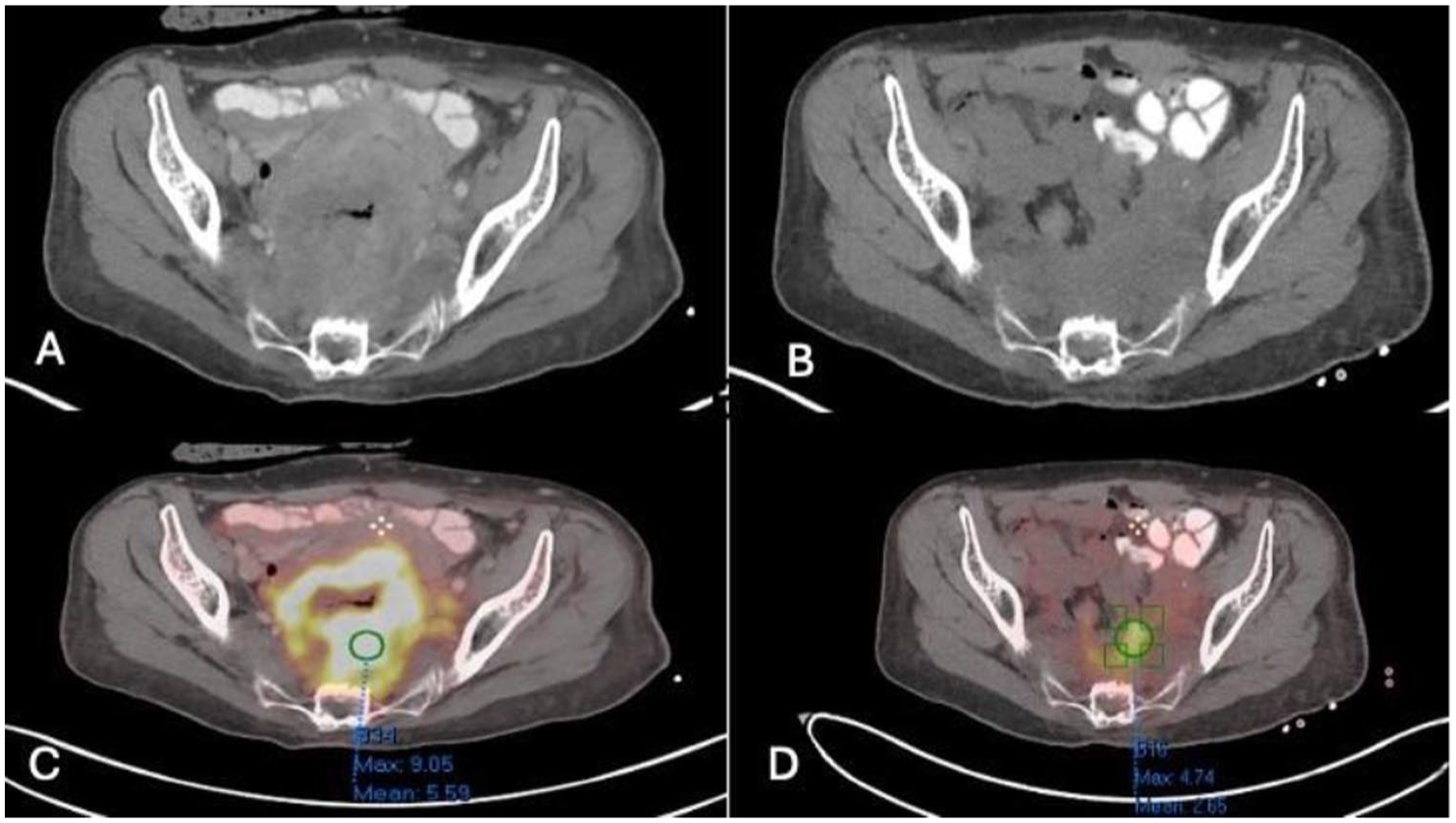
(a) and (c) Represent the pre-treatment images. A large FDG-avid rectal mass with SUV max 9 can be visualized. (b) and (d) represent post-treatment images demonstrating a marked reduction in size and decreased FDG avidity of the rectal mass.
Discussion
In both males and females, rectal cancer is responsible for 3.9% of all cancer diagnoses and 3.2% of cancer-related deaths. 6 Rectal cancers account for approximately 30%–35% of all colorectal cancers, and approximately half of them are diagnosed at a locally advanced stage.
Improving the prognosis of patients with LARC and metastatic CRC (mCRC) continues to be a significant and difficult issue. The prognosis for patients with mCRC is poor, with a 5-year survival rate of less than 15%. 7 For approximately two decades, the standard of care (SOC) for patients with LARC has been neoadjuvant long-course chemoradiotherapy or short-course radiotherapy, followed by surgery—total mesorectal excision with or without adjuvant chemotherapy. Nevertheless, this conventional method has several drawbacks, including a low pathological complete response (pCR) rate (10%–25%), a high metastasis rate (30%–35%), and highly inconsistent compliance with adjuvant chemotherapy (25%–75%). 8 More recently, total neoadjuvant therapy (TNT) began to gain momentum as it attempted to administer systemic chemotherapy and neoadjuvant CRT prior to surgery. TNT was linked to improved compliance, decreased toxicity, and a reduced need for surgery (watch-and-wait), as indicated by numerous studies. The TNT group also exhibited a higher pCR rate than the adjuvant chemotherapy group (36% vs 21%). 9 However, there is still room to improve the efficacy and safety of TNT with multiple trials building upon this backbone. Fluorouracil in combination with platinum, irinotecan, or both is the SOC frontline doublet or triplet chemotherapy regimen with or without a biologic where appropriate for a patient with mCRC. The recommended first-line therapies for a small subset of patients (3%–5%) with mCRC whose tumors are mismatch repair-deficient/microsatellite instability-high (dMMR/MSI-High) include ICIs with anti-programmed death-1 inhibitors with or without anti-CTLA-4 inhibitors. These medications have demonstrated exceptional efficacy, with response rates exceeding 65% and median progression-free survival (PFS) exceeding 16.5 months, including prolonged durable controls and even cures. Nevertheless, the majority of patients (>95%) with mCRC have tumors that are (pMMR/MSS), which are resistant to ICIs due to aspects of the tumor microenvironment that are not associated with cytotoxic anti-tumor immune responses. Salvage therapy options for patients with pMMR/MSS tumors are limited to regorafenib (multi-TKI), tipiracil/trifluridine (TAS-102) with or without bevacizumab, and most recently fruquintinib, which have suboptimal efficacy and toxicity. 10
Therefore, there is a significant clinical need to advance new treatment approaches, particularly those targeting the tumor microenvironment to induce cytotoxic immune responses against tumors, aiming to enhance the effectiveness of ICIs in metastatic colorectal cancer (mCRC) patients with pMMR/MSS status.
Numerous initial-stage trials were unsuccessful in identifying an effective combination of ICIs for metastatic pMMR/MSS colorectal cancer.11–14 These findings are highly indicative of the fact that the immunomodulating properties of the tumor microenvironment are generally not enhanced by the specific inhibition of a single pathway. Consequently, the manifold layers of immune evasion necessitate the inhibition of multiple signaling pathways.
The Imblaze370 trial, which compared cobimetinib, a MEK inhibitor, plus atezolizumab to regorafenib in refractory pMMR/MSS mCRC, was one of the largest and earliest trials to evaluate an ICI plus TKI combination. 15 The study did not observe any survival benefit or superiority of cobimetinib plus atezolizumab over regorafenib.
The REGONIVO trial’s initial phase I/II, also referred to as EPOC1603, was designed to evaluate the efficacy of nivolumab plus regorafenib in patients with metastatic pMMR/MSS CRC. Despite the initial trials’ promising results, the success was not replicated in subsequent studies. The primary endpoint of phase II of the trial was not achieved. The mPFS of 11.9 months in patients without liver involvement (NLM) and 1.8 months in those with hepatic metastases 2 confirmed the lack of effect of this combination in patients with liver metastases. Patients with liver metastases did not exhibit any responses, while those liver metastases (NLM) without had an overall response rate (ORR) of 21.7%. 16
Another study, the REGOMUNE trial, evaluated the efficacy of regorafenib in combination with avelumab in a single-arm phase II trial. Nevertheless, it demonstrated only modest efficacy, with objective response rates of less than 1%. 17
MAYA study published in 2021 proved the immune-sensitizing role of temozolomide in MSS or O6-methylguanine DNA methyltransferase silenced mCRC. The trial was successful in achieving its primary endpoint and proved to be an innovative treatment strategy worthy of further development. 18
Recently, in a phase III trial, the lenvatinib plus pembrolizumab (LEAP-017) study, which combined ICI and TKI regimens, failed to achieve the primary endpoint of overall survival (OS), indicating that it was not efficacious. 19
Despite the different immunotherapeutic agents used, CAMILLA’s efficacy analyses revealed an ORR of almost 30% and a disease control rate of 86.2% (25/29), with a median PFS of 4.4 months and a median overall survival (mOS) of 9.1 months. The results of the CAMILLA trial were slightly superior. Durvalumab plus cabozantinib was employed as a third-line therapy for patients with pMMR mCRC. The trial was conducted in accordance with preclinical findings, which demonstrated that cabozantinib in combination with anti-PD1 inhibited tumor growth and increased the expression of the CD4+ T cell ligand HLA-DR on the tumor cells. 20 CAMILLA’s efficacy analyses demonstrated a disease control rate of 86.2% (25/29), an ORR of nearly 30%, a median PFS of 4.4 months, and a mOS of 9.1 months. 21
The RIN protocol assessed regorafenib, ipilimumab, and nivolumab (RIN) as a combination therapy for the treatment of chemotherapy-resistant MSS mCRC. In summary, RIN (80/1/240) demonstrated significant activity in MSS non-liver metastases mCRC patients. The data indicate that a regorafenib starting dose of 80 mg is crucial for priming the immune response to RIN (Table 1). 22
A summary of the trials discussed in the paper and a comparison of the treatments and their relative outcomes.

ORR, overall response rate; OS, overall survival; PFS, progression-free- survival; RIN, Regorafenib, ipilimumab, and nivolumab.
Most clinical trials combining immunotherapy and TKI in MSS mCRC have not shown significant results. 23
The RIN protocol has demonstrated promising activity in refractory MSS or pMMR colorectal cancer. In multiple published studies by Fakih and colleagues22,24 the objective response rate was approximately 27%, with a median PFS of 4 months and mOS of 20 months in heavily pretreated patients. The present case builds upon these findings and exhibited a deeper and more durable response, with complete clearance of ctDNA and CEA within 6 months of therapy initiation and sustained molecular remission beyond 12 months. This outcome exceeds the magnitude and duration of responses typically reported in prior RIN cohorts, suggesting exceptional biological sensitivity to this regimen. Collectively, these findings further support the potential of multimodal immune modulation in overcoming immunotherapy resistance in pMMR/MSS colorectal cancer, especially in patients with no liver metastases.
This case report emphasizes numerous advantages. It describes a profound and enduring clinical and molecular response to the RIN regimen in a patient with pMMR/MSS recurrent rectal cancer that has been heavily pretreated in the real-world. This patient population has a limited number of effective treatment options. The longitudinal assessment of treatment response and the provision of complementary evidence to radiographic evaluation were facilitated by the use of serial biomarkers, such as CEA and ctDNA. It is imperative to recognize significant constraints. Most important, the present paper is a single case report, the findings are limited and cannot determine comparative efficacy or causality. In addition, the relative contribution of each component of the combination regimen cannot be determined, and the absence of a control group restricts interpretation. Regular clinical assessments, serial laboratory monitoring, including CEA and ctDNA, and periodic cross-sectional imaging were implemented to facilitate follow-up. The patient has shown lasting molecular remission with no signs of disease worsening in clinical exams or imaging for over 12 months, supporting the effectiveness of this method in carefully selected patients. There was no evidence of clinical or radiographic disease progression during the follow-up period of over 12 months, corroborating the feasibility of this approach in meticulously selected patients. Patient subsequently completed a total of 2 years of PD-1 blockade and low dose regorafenib back in her home country, and continued to have no evidence of disease till last reported follow-up. Reporting this rare off-label real-world cases are important to report as cumulatively they might help gain insight and provide clinical experience in these challenging situations.
Conclusion
This patient with advanced, recurrent rectal adenocarcinoma demonstrated a remarkable and durable response to combination therapy with the RIN protocol. Within 6 months of treatment initiation, both ctDNA and CEA levels became undetectable, accompanied by a marked interval reduction in FDG-avid disease on PET imaging. The concurrent clearance of ctDNA and CEA supports a molecular complete response, consistent with emerging evidence that biomarker-based remission may precede or occur independently of radiologic resolution.23,25 To our knowledge, such a brisk, deep and lasting ongoing response has not been previously reported in a patient with pMMR/MSS rectal cancer—a subset historically resistant to immunotherapy, or at least builds upon the exceptional responders being reported in these stuides. 26 This case highlights the potential of strategic multimodal immune modulation to restore sensitivity to checkpoint blockade and provides a rationale for exploring similar therapeutic combinations in future clinical and translational studies. Further research is warranted to define biomarkers predictive of response beyond the absence of liver metastases (NLM) and refine patient selection for this promising treatment approach. More effective combinatorial regimens like zanzalintinib and atezolizumb (STELLAR-303) trial, as well as newer generation of CTLA-4 inhibitors like botensilimab, vilastobart, and muzastotug are showing more promise for patients with MSS colorectal cancers in particular who do not have liver metastases (NLM).
Supplemental Material
sj-pdf-1-tam-10.1177_17588359261432049 – Supplemental material for Molecular complete response to the RIN protocol (regorafenib, ipilimumab, and nivolumab) in a patient with advanced recurrent metastatic mismatch repair proficient/microsatellite stable (pMMR/MSS) rectal cancer
Supplemental material, sj-pdf-1-tam-10.1177_17588359261432049 for Molecular complete response to the RIN protocol (regorafenib, ipilimumab, and nivolumab) in a patient with advanced recurrent metastatic mismatch repair proficient/microsatellite stable (pMMR/MSS) rectal cancer by Fatima Qadri, Maaz Khan Afghan, Areeb Lutfi, Sana Javaid, Preethi Guniganti, Higinia Cardenes, Alessio Pigazzi and Pashtoon Murtaza Kasi in Therapeutic Advances in Medical Oncology
