Abstract
The treatment of patients with renal cell carcinoma (RCC) is evolving rapidly, with promising new regimens being developed and approved for patients with advanced disease, particularly the combination of tyrosine kinase inhibitors with immune checkpoint inhibitors. Within the last 6 months, favorable first-line setting results for patients with clear cell RCC have been reported for the combination of cabozantinib plus nivolumab in the phase III CheckMate 9ER study, leading to its regulatory approval, and lenvatinib plus pembrolizumab in the phase III CLEAR study. Additional systemic first-line treatments for clear cell RCC include axitinib plus pembrolizumab, pazopanib, and sunitinib for favorable-risk patients and ipilimumab plus nivolumab, axitinib plus pembrolizumab, axitinib plus avelumab, and cabozantinib for intermediate- or poor-risk patients. In this review of novel approaches for first-line treatment of advanced RCC, we present an overview of current treatment strategies, the basis behind emerging treatment approaches, a summary of key results from the pivotal studies using tyrosine kinase inhibitor and immune checkpoint inhibitor combination therapy, novel treatments and strategies under development, and efforts for identifying biomarkers to guide treatment decisions.
Keywords
Introduction
Worldwide, approximately 400,000 new cases of kidney cancer occurred in 2018, with 175,000 deaths associated with this disease. 1 Around 85% of kidney cancers are renal cell carcinoma (RCC), of which 70% have a clear cell histology (ccRCC). 2 For patients with localized disease, the 5-year relative survival rate is 93%, which drops to 13% for patients with distant metastasis. 3
ccRCC is a highly vascularized tumor in which levels of angiogenic factors, including vascular endothelial growth factor (VEGF), are correlated with patient prognosis. 4 Upregulation of RCC-related genes, including those coding for VEGF, platelet-derived growth factor (PDGF), MET, AXL, and mTOR, are associated with mutations in the von Hippel–Lindau (VHL) tumor suppressor gene. The VHL tumor suppressor is a ubiquitin ligase that regulates levels of the hypoxia-inducible factor 2α (HIF-2α) transcription factor.5–7 A majority of patients with ccRCC have a loss of heterozygosity of the VHL tumor suppressor gene. 8
ccRCC is also a highly immunogenic tumor characterized by an abundance of immune cells that exhibit a suppressed phenotype.9,10 VEGF, AXL, MET, and other RCC-upregulated growth factors have immunosuppressive effects on the tumor microenvironment.11–13 Consequently, many current and emerging therapeutic regimens incorporate agents that target angiogenic or immunosuppressive pathways, or both. These include VEGF receptor (VEGFR) tyrosine kinase inhibitors (TKIs; cabozantinib, sunitinib, pazopanib, axitinib, lenvatinib), the anti-VEGF antibody bevacizumab, and immune checkpoint inhibitors [ICIs; nivolumab (anti–PD-1), pembrolizumab (anti–PD-1), avelumab (anti–PD-L1), atezolizumab (anti–PD-L1), ipilimumab (anti–CTLA-4)].
In this review, we discuss the rationale for current and emerging first-line treatment strategies in advanced ccRCC; summarize key data from pivotal studies of VEGFR inhibitors and ICIs as combination therapy; and discuss novel therapeutic strategies under development and approaches to identifying biomarkers.
Currently approved therapies
Treatment decisions for patients with advanced ccRCC are based on risk stratification models, including multiple independent prognostic factors – performance status, time from diagnosis to treatment of metastatic disease, elevated calcium, platelet and neutrophil counts, and anemia.14,15 Other factors influencing treatment decisions include age; comorbidities; disease-related symptoms; histology; tumor burden; and for ICI-based regimens, history of autoimmune disease and use of immunosuppressive therapy.14–18 Models incorporating these factors include the more commonly used International Metastatic Renal Cell Carcinoma Database Consortium (IMDC) model and the Memorial Sloan Kettering Cancer Center (MSKCC) model.14,15,17 These models classify patients as having favorable, intermediate, or poor risk and share common features mentioned above.
For patients with favorable risk, defined as patients having no poor prognostic features, common treatment management approaches are deferred therapy, metastasectomy, or systemic therapy. A variety of systemic treatments are available. Preferred systemic regimens include axitinib plus pembrolizumab, cabozantinib plus nivolumab, lenvatinib plus pembrolizumab, or single-agent therapy with pazopanib or sunitinib.2,16,19,20 For those classified as having intermediate or poor risk, characterized as ⩾1 poor prognostic factor, preferred first-line treatments are ipilimumab plus nivolumab, axitinib plus pembrolizumab, cabozantinib plus nivolumab, or lenvatinib plus pembrolizumab. Axitinib plus avelumab represents another combination option, with cabozantinib the preferred single-agent TKI therapy for these patients.2,16
Monotherapies
TKIs
VEGFR-TKIs inhibit angiogenic processes such as endothelial cell survival and vascular permeability (Figure 1).21,22 Inhibition of VEGFR leads to vascular normalization, resulting in increased T-cell infiltration, and promotes T-cell function, maturation of dendritic cells, and reduction in immunosuppressive cells such as regulatory T cells and myeloid-derived suppressor cells.22,23

Impact on the tumor microenvironment of VEGF-targeted monotherapy (a) and VEGF-targeted therapy combined with ICI (b).
Up until 2017, the multikinase inhibitors sunitinib [which targets VEGFR, PDGF receptor (PDGFR), FLT-3, and c-KIT] and pazopanib (which targets VEGFR, PDGFR, and c-KIT) formed the standard of care for first-line treatment of ccRCC, based on superiority over interferon-alpha and placebo, respectively.24–26 Studies in first-line RCC have generally used sunitinib as the comparator.
Pazopanib was compared with sunitinib in the randomized phase III COMPARZ noninferiority study of 1110 patients with metastatic, untreated ccRCC, of which 25% had favorable risk and 55% had intermediate risk.
27
Median progression-free survival (PFS) was similar between pazopanib and sunitinib [median 8.4
The oral multikinase inhibitor cabozantinib, which targets VEGFR, MET, and the TAM family of kinases (TYRO3, AXL, MER), was compared with sunitinib in the randomized phase II study CABOSUN, which included 157 patients with metastatic, untreated ccRCC who had either intermediate (81%) or poor risk.28,29 PFS was significantly improved with cabozantinib
Although the VEGFR TKIs all have distinct safety profiles, there is considerable overlap in types of adverse events (AEs), with hypertension, fatigue, diarrhea, and palmar–plantar erythrodysesthesia being the most common grade 3/4 AEs.27,28 In COMPARZ, grade 3 and 4 treatment-emergent AEs occurred in 59% and 15% of patients with pazopanib, respectively, and 57% and 17% of patients with sunitinib, respectively; discontinuation due to an AE occurred in 24% and 20% of patients receiving pazopanib and sunitinib, respectively. 27 In CABOSUN, 68% of patients had a grade 3/4 AE with cabozantinib compared with 65% with sunitinib; and discontinuation due to an AE occurred in 21% and 22% of patients, respectively. 28
Immunotherapies
Checkpoint proteins such as PD-1 and CTLA-4 are cell surface receptors expressed on immune cells, and their activation leads to immune inhibition. 30 PD-L1 and PD-L2 are both ligands for PD-1, with PD-L1 the most widely expressed within tumors. 30 ICIs disrupting the PD-1/PD-L1 axis restore the function of effector T cells and suppress regulatory T-cell function, thereby promoting an antitumor immune response.30,31 However, the ability of the immune system to mount an effective antitumor response depends on a variety of other factors, including tumor intrinsic factors, the tumor microenvironment, the expression and activation of other checkpoint proteins, host genetics, and microbial diversity. 32
ICI monotherapy has demonstrated clinical activity in the first-line setting.33–36 In a randomized phase II study, atezolizumab monotherapy was compared with sunitinib, with a median PFS of 6.1
ICI–ICI combinations
Preclinical studies of PD-1 plus CTLA-4 ICIs demonstrated synergistic activity with enhanced effector T-cell expansion, supporting the evaluation of this combination in clinical trials.37,38 Based on positive outcomes from the phase III CheckMate-214 study, which compared nivolumab plus ipilimumab with sunitinib, this combination was approved for first-line use in patients with intermediate-/poor-risk RCC. 39
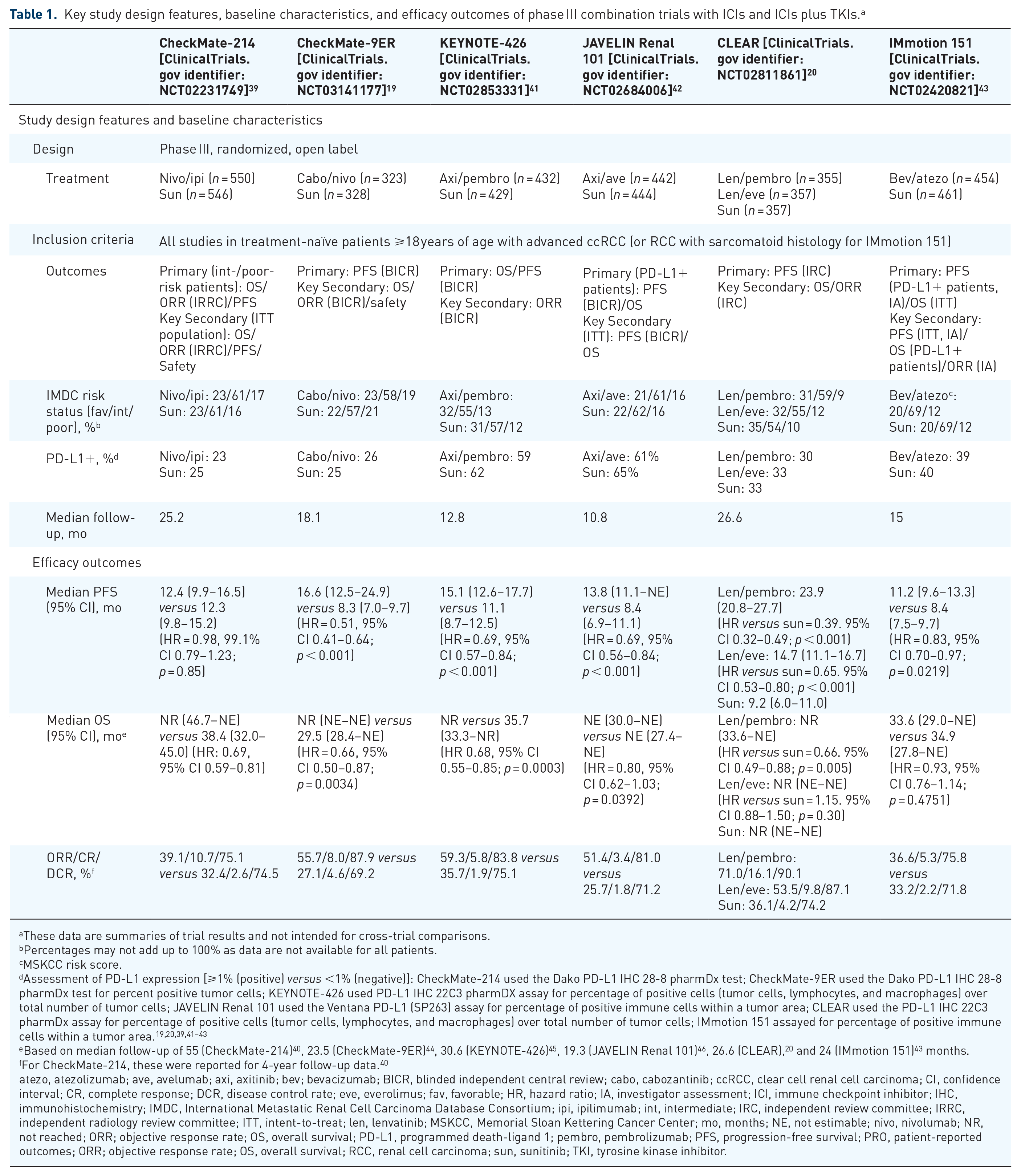
In CheckMate-214, the majority of patients were intermediate risk (61%) and 24% were PD-L1 positive (Table 1).
39
PFS, OS, and ORR (co-primary endpoints in immediate-/poor-risk patients) all favored nivolumab plus ipilimumab over sunitinib in the intermediate-/poor-risk population (Tables 1 and 2).39,40 For the intermediate-/poor-risk population, median PFS was 11.6
Key study design features, baseline characteristics, and efficacy outcomes of phase III combination trials with ICIs and ICIs plus TKIs. a
These data are summaries of trial results and not intended for cross-trial comparisons.
Percentages may not add up to 100% as data are not available for all patients.
MSKCC risk score.
Assessment of PD-L1 expression [⩾1% (positive)
Based on median follow-up of 55 (CheckMate-214) 40 , 23.5 (CheckMate-9ER) 44 , 30.6 (KEYNOTE-426) 45 , 19.3 (JAVELIN Renal 101) 46 , 26.6 (CLEAR), 20 and 24 (IMmotion 151) 43 months.
For CheckMate-214, these were reported for 4-year follow-up data. 40
atezo, atezolizumab; ave, avelumab; axi, axitinib; bev; bevacizumab; BICR, blinded independent central review; cabo, cabozantinib; ccRCC, clear cell renal cell carcinoma; CI, confidence interval; CR, complete response; DCR, disease control rate; eve, everolimus; fav, favorable; HR, hazard ratio; IA, investigator assessment; ICI, immune checkpoint inhibitor; IHC, immunohistochemistry; IMDC, International Metastatic Renal Cell Carcinoma Database Consortium; ipi, ipilimumab; int, intermediate; IRC, independent review committee; IRRC, independent radiology review committee; ITT, intent-to-treat; len, lenvatinib; MSKCC, Memorial Sloan Kettering Cancer Center; mo, months; NE, not estimable; nivo, nivolumab; NR, not reached; ORR; objective response rate; OS, overall survival; PD-L1, programmed death-ligand 1; pembro, pembrolizumab; PFS, progression-free survival; PRO, patient-reported outcomes; ORR; objective response rate; OS, overall survival; RCC, renal cell carcinoma; sun, sunitinib; TKI, tyrosine kinase inhibitor.
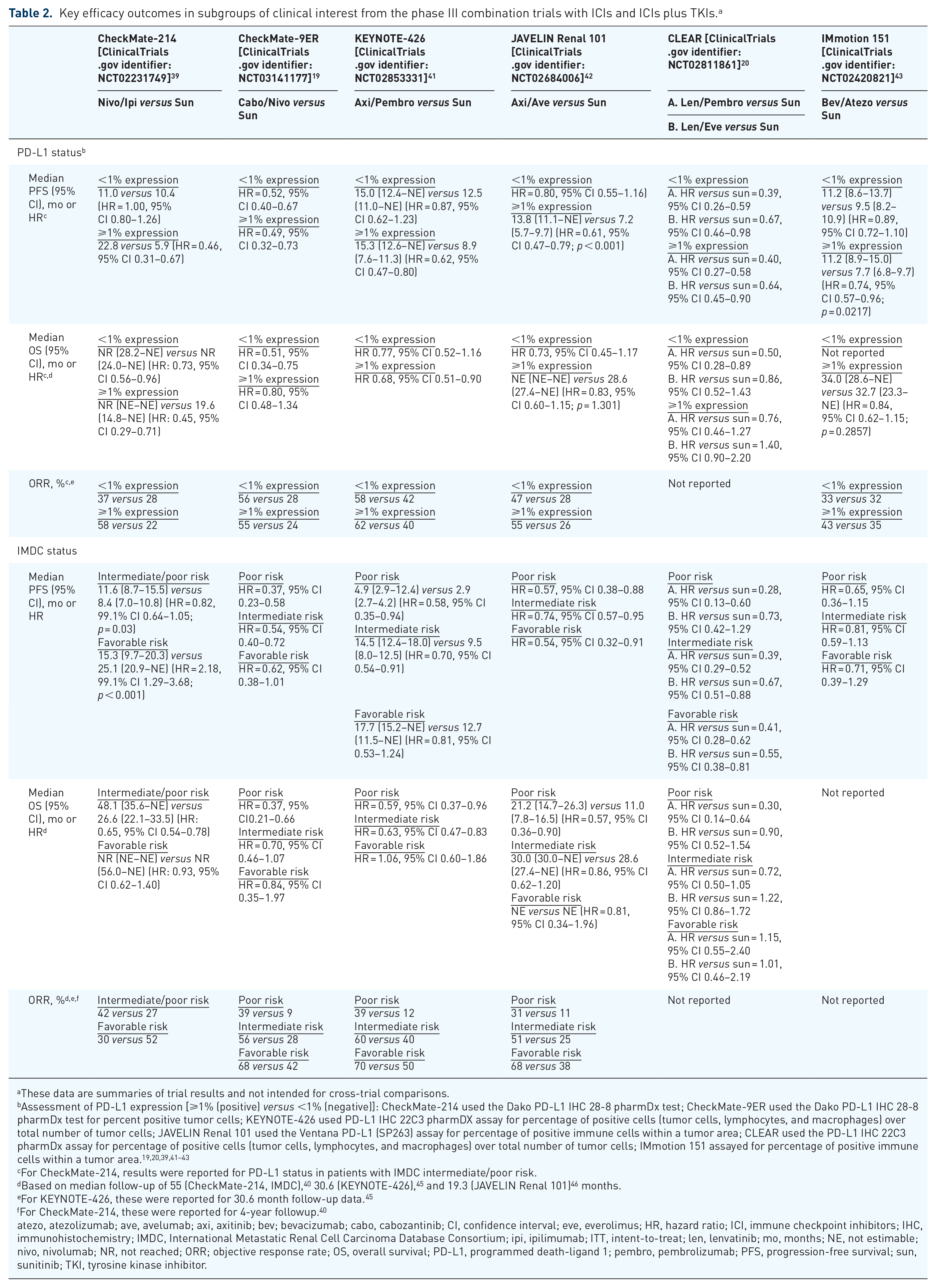
Key efficacy outcomes in subgroups of clinical interest from the phase III combination trials with ICIs and ICIs plus TKIs. a
These data are summaries of trial results and not intended for cross-trial comparisons.
Assessment of PD-L1 expression [⩾1% (positive)
For CheckMate-214, results were reported for PD-L1 status in patients with IMDC intermediate/poor risk.
Based on median follow-up of 55 (CheckMate-214, IMDC), 40 30.6 (KEYNOTE-426), 45 and 19.3 (JAVELIN Renal 101) 46 months.
For KEYNOTE-426, these were reported for 30.6 month follow-up data. 45
For CheckMate-214, these were reported for 4-year followup. 40
atezo, atezolizumab; ave, avelumab; axi, axitinib; bev; bevacizumab; cabo, cabozantinib; CI, confidence interval; eve, everolimus; HR, hazard ratio; ICI, immune checkpoint inhibitors; IHC, immunohistochemistry; IMDC, International Metastatic Renal Cell Carcinoma Database Consortium; ipi, ipilimumab; ITT, intent-to-treat; len, lenvatinib; mo, months; NE, not estimable; nivo, nivolumab; NR, not reached; ORR; objective response rate; OS, overall survival; PD-L1, programmed death-ligand 1; pembro, pembrolizumab; PFS, progression-free survival; sun, sunitinib; TKI, tyrosine kinase inhibitor.
The rate of grade ⩾3 treatment-related AEs (TRAEs) was 46% for the combination (Table 3). 39 The most frequently occurring grade ⩾3 TRAE was increased lipase level (10%) in the combination. In the combination arm, 22% of patients discontinued study treatment (both agents) due to a TRAE compared with 12% with sunitinib.
Key safety outcomes from the phase III combination trials with ICIs and ICIs plus TKIs. a

These data are summaries of trial results and are not intended for cross-trial comparisons.
AE, adverse event; ALT, alanine aminotransferase; AST, aspartate aminotransferase; atezo, atezolizumab; ave, avelumab; axi, axitinib; bev; bevacizumab; CA, cardiac arrest; cabo, cabozantinib; eve, everolimus; GIH, gastrointestinal hemorrhage; HTG, hypertriglyceridemia; HTN, hypertension; ICH, intracranial hemorrhage; ICI, immune checkpoint inhibitor; ipi, ipilimumab; irAE, immune-related AE; len, lenvatinib; MI, myocardial infarction; nivo, nivolumab; pembro, pembrolizumab; PNA, pneumonia; PPE, palmar-plantar erythrodysesthesia; sun, sunitinib; TKI, tyrosine kinase inhibitor; TRAE, treatment-related AE; TTP, thrombocytopenia.
TKI/antiangiogenic–ICI combinations
RCC is a highly angiogenic and immunogenic tumor.4,9 Anti-VEGFR TKIs and ICIs target these tumor characteristics in different ways (Figure 1), and preclinical studies suggest a potential synergistic effect of these treatment modalities when combined; however, this effect is yet to be demonstrated in clinical studies.49–51 TKIs targeting VEGFR and other kinase receptors may enhance the immune response with ICIs given that their kinase receptor targets contribute to immune regulation, with VEGFR promoting regulatory T-cell and myeloid-derived suppressor cell recruitment; AXL being implicated in T-cell exclusion, suppressing antigen presentation, and low tumor major histocompatibility complex class I expression; and MET shown to increase PD-L1 expression and tumor presence of immunosuppressive neutrophils.52–56
In the following sections, we review results from five phase III trials evaluating these TKI/ICI combinations, including CheckMate-9ER, KEYNOTE-426, JAVELIN Renal 101, CLEAR, and IMmotion 151 (Tables 1–3). Results for CheckMate-9ER, KEYNOTE-426, and JAVELIN Renal 101 led to approval of the respective combination in the first-line setting, while the CLEAR results are under regulatory review.
CheckMate-9ER
The randomized, open-label, phase III Checkmate-9ER study evaluated cabozantinib in combination with nivolumab
Cabozantinib plus nivolumab improved PFS, OS, and ORR compared with sunitinib.
19
Median PFS (primary endpoint) was 16.6 months for cabozantinib plus nivolumab
For HRQoL (FKSI-19), a significant difference (
TRAEs of grade ⩾ 3 were reported in 61% of patients with the combination (Table 3). 19 The most frequently occurring grade ⩾ 3 TRAE was hypertension (11%) in the combination. TRAEs leading to discontinuation of all study treatment occurred in 3% of patients with the combination (15% with TRAEs leading to discontinuation of either study drug) and 9% of patients with sunitinib.
KEYNOTE-426
The combination of axitinib with pembrolizumab was assessed in patients with ccRCC in the open-label phase III KEYNOTE-426 study. 41 The majority of patients were intermediate risk (56%) and PD-L1 positive (60%) (Table 1).
PFS (co-primary endpoint) and ORR favored axitinib plus pembrolizumab
TRAEs of grade ⩾ 3 were reported in 63% of patients with the combination (Table 3). 41 The most frequently occurring grade ⩾ 3 TRAE was hypertension (21%) in the combination. TRAEs leading to discontinuation of all study treatment occurred in 8% of patients with the combination (with 26% having a TRAE that led to discontinuation of either study drug) and 10% of patients with sunitinib.
JAVELIN Renal 101
In the phase III JAVELIN Renal 101, axitinib was combined with avelumab and compared with sunitinib in patients with advanced RCC.42,46 The primary analysis population consisted of patients with PD-L1–positive status, which accounted for 63% of all patients. The majority of those enrolled in the ITT population were intermediate risk (62%) (Table 1).
In JAVELIN Renal 101, median PFS in the PD-L1–positive population (co-primary endpoint) was 13.8 months with axitinib plus avelumab and 7.2 months with sunitinib (
Rates of grade ⩾ 3 TRAEs were similar between the combination and sunitinib arms, at 57% and 55%, respectively. 42 The most frequently occurring grade ⩾ 3 TRAE in the combination arm was hypertension (24%) (Table 3). In the combination arm, 8% of patients discontinued both study treatments owing to a TRAE, compared with 13% in the sunitinib arm.
CLEAR
In the 3-arm CLEAR study, the multikinase inhibitor lenvatinib was combined with either pembrolizumab or the mTOR inhibitor everolimus and compared with sunitinib. 20 Most patients had intermediate risk (56%), with 32% PD-L1 positive (Table 1).
Both combination arms were favored over sunitinib for PFS (primary endpoint), with medians of 23.9 months for pembrolizumab plus lenvatinib, 14.7 months for lenvatinib plus everolimus, and 9.2 months for sunitinib (
Rates of grade ⩾ 3 TRAEs were higher for either combination with lenvatinib (72%–73%)
IMmotion 151
In the phase III IMmotion151, bevacizumab was combined with atezolizumab and compared with sunitinib in patients with ccRCC or RCC with a sarcomatoid histology (
Rates of grade ⩾ 3 TRAEs were lower for the combination (40%)
Ongoing studies and novel approaches
Several ongoing phase III studies are evaluating TKI-ICI combinations with results expected in the next 12–24 months. The combination of PD-1 inhibitor toripalimab plus axitinib is being evaluated
Studies are also evaluating regimens that include novel agents, such as those targeting the immune checkpoint LAG-3 and HIF-2α, as well as cancer vaccines (Table 4). Ongoing phase III studies that are evaluating novel agents for first-line treatment of RCC include: PIVOT-09, which is assessing the pegylated interleukin-2 molecule bempegaldesleukin (NKTR-214) plus nivolumab
Ongoing studies and novel first-line therapies for RCC.
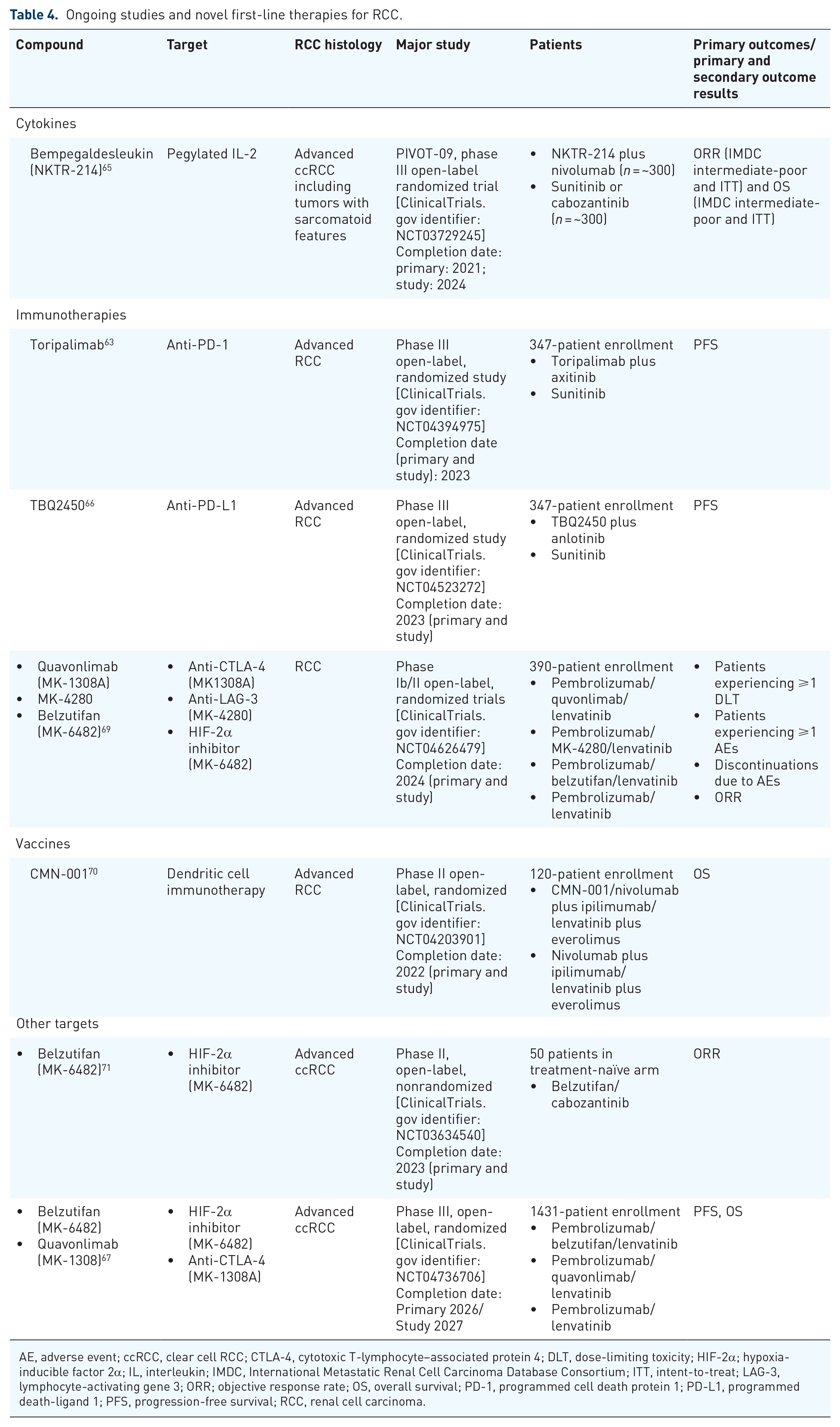
AE, adverse event; ccRCC, clear cell RCC; CTLA-4, cytotoxic T-lymphocyte–associated protein 4; DLT, dose-limiting toxicity; HIF-2α; hypoxia-inducible factor 2α; IL, interleukin; IMDC, International Metastatic Renal Cell Carcinoma Database Consortium; ITT, intent-to-treat; LAG-3, lymphocyte-activating gene 3; ORR; objective response rate; OS, overall survival; PD-1, programmed cell death protein 1; PD-L1, programmed death-ligand 1; PFS, progression-free survival; RCC, renal cell carcinoma.
With the TKI/ICI combination, there is still limited information on the optimal treatment sequencing strategy. Also, it is unclear if treatment sequencing should be patient-dependent. To address some of these questions, the ongoing phase III PDIGREE study will investigate an adaptive treatment approach, whereby first-line nivolumab plus ipilimumab is followed by either nivolumab monotherapy, nivolumab plus cabozantinib, or cabozantinib monotherapy depending on response to nivolumab plus ipilimumab.
72
It is also unclear if potential improvements with combinations are synergistic or additive and which drug is driving disease control. The phase III CONTACT-03 study will evaluate cabozantinib plus atezolizumab
Although this review focused on ccRCC, there are some recent advancements in non-ccRCC that are worth noting. The phase III SAVOIR study evaluated the MET inhibitor savolitinib
Biomarkers
Data from phase III trials illustrate the potential for long-term, durable benefit in patients who respond to immunotherapy-based regimens, but they also highlight the need for biomarkers of response. Currently, IMDC risk scores are the only validated prognostic markers in clinical use for RCC.19,20,39,41–43 In CheckMate-214, the benefit of nivolumab plus ipilimumab was observed in patients with intermediate-/poor-IMDC risk but not in patients with favorable risk.
39
For the CheckMate-9ER, KEYNOTE-426 studies, and CLEAR studies, the benefit of the ICI-TKI combinations was observed across IMDC risk subgroups with the benefit more pronounced in patients with intermediate/poor risk
Use of additional biomarkers along with IMDC scores could aid in predicting outcomes. In a study evaluating IMDC components as outcome predictors with anti-VEGF therapy, elevated platelet count was associated with worse outcomes in intermediate-IMDC risk patients. 76 Another study identified a high systemic immune-inflammation index based on neutrophil, platelet, and lymphocyte counts, correlating with poorer outcomes for patients receiving a first-line TKI. 77
Histologic characteristics can potentially be utilized as a response predictor. Sarcomatoid features are generally associated with poor outcomes.42,59,61 Although sarcomatoid features are well described as a poor prognostic feature, multiple studies of first-line ICI-TKI show increased response rates with sarcomatoid features as compared with tumors negative of this feature.42,59,61 Given the outcomes of ICI-TKI and ICI-ICI combinations compared with sunitinib, the presence of sarcomatoid features will likely be incorporated into treatment decisions in the near future.
The use of PD-L1 as a biomarker has not been clearly established for RCC. A meta-analysis of recent randomized clinical trials comparing ICIs as monotherapy or in combination with other agents to standard of care suggested that PD-L1 may be predictive of PFS but not OS. 78 In JAVELIN Renal 101, PD-L1 expression on immune cells was not associated with improved PFS with avelumab plus axitinib. 79 Multiple issues have likely confounded PD-L1 biomarker data, including varying definitions of PD-L1 positivity (i.e., immune cells only or tumor cells plus immune cells) and utilizing different antibodies and assay formats.41,42 Potentially, a composite biomarker encompassing key tumor-immune phenotype features and not a single biomarker may be required.
A variety of studies have utilized genomic profiling and transcript profiling to identify biomarkers associated with treatment efficacy and safety. A biomarker analysis of samples from the phase II IMmotion150 study showed that markers of pre-existing immunity (PD-L1 expression, effector T-cell signature) identified patients with improved response to bevacizumab plus atezolizumab, while patients with a high angiogenic signature responded better to sunitinib. 35 Comparable results were obtained from the phase III IMmotion151 study, 80 and the same angiogenic gene expression signature was found to be enriched in favorable-risk patients from the JAVELIN Renal 101. 81
Further exploratory analyses of patients treated with axitinib plus avelumab in JAVELIN Renal 101 have shown the high expression of a 26-gene immune-based signature associated with improved PFS with the combination, 79 and a low neutrophil-to-lymphocyte ratio associated with improved PFS, OS, and ORR. 82 Additionally, a 26-gene angiogenesis-based signature was associated with improved PFS with sunitinib, but not with the combination. 79 However, gene signatures from JAVELIN Renal 101 and IMmotion150 were evaluated in CheckMate 214, and no relationship was observed between immune-related gene signature and efficacy. 83
In the phase II BIONIKK study, patients with four different 35-gene expression mRNA signatures, two each associated with low-and high-sunitinib sensitivity, were assessed.84,85 The sunitinib low-sensitivity groups, with immune-high (
Serum molecules are also being evaluated as response biomarkers. Studies include identifying a cytokine signature associated with nivolumab clearance and the association of elevated interleukin-6 and CD146 levels with TKI sensitivity.86–88
Overall, promising candidate biomarkers are centered around identifying patients with an immunogenic phenotype
Challenges, gaps, and future directions
Although significant progress has been made in development of first-line treatment strategies for RCC, relevant issues still need to be addressed. There is a lack of data for patients with favorable-risk RCC, as the majority of patients in phase III trials have intermediate-/poor-risk RCC, and analyses of favorable-risk subgroups have been exploratory. Additional research is also needed to better characterize the tumor biology by IMDC risk; results from such research may also highlight potentially useful biomarkers.
Better strategies need to be developed for the management of AEs with combination treatments. Toxicity tends to be greater with combination treatments relative to monotherapies, with an increased risk of all-grade and grade 3/4 diarrhea, liver toxicity, and endocrinopathies relative to sunitinib monotherapy.19,41,42 Studies have employed strategies including prophylaxis, dose interruption, and dose reduction (TKIs only) to manage AEs. Treatment sequencing may also aid in the development of strategies to reduce toxicity, but there are limited data of second-line treatments following newly approved first-line regimens.
Measuring response with ICI-based regimens poses challenges, as response kinetics differs from those observed with TKI treatments. Although trials continue to use RECIST v1.1, new measures such as irRECIST have been developed and may become used more widely in the future. 16
Summary
The first-line RCC treatment landscape has evolved rapidly, with immunotherapy-based combinations forming a new standard of care. Optimization of these new therapies will involve greater personalization to increase benefit for individual patients. Strategies to mitigate and minimize added toxicity with combination therapies will be needed to translate these clinical trial results to more diverse real-world patient populations.
Footnotes
Acknowledgements
Medical writing and editorial assistance was provided by Karen O’Leary, PhD, and Alan Saltzman, PhD, Fishawack Communications Inc., part of Fishawack Health, Conshohocken, PA.
Conflict of interest statement
DJG reports personal/financial fees as senior editor of American Association for Cancer Research; consultancy fees from Astellas, AstraZeneca, Bayer H/C Pharmaceuticals, BMS, Constellation Pharmaceuticals, Exelixis, Inc., Flatiron, Janssen Pharmaceuticals, Merck Sharp & Dohme, Michael J Hennessey Associates, Myovant Sciences, Inc., Pfizer, Physician Education Resource LLC, Propella TX (formerly Vizuri), RevHealth, LLC, Sanofi; research funding from Astellas (Inst), AstraZeneca (Inst), BMS (Inst), Calithera (Inst), Exelixis, Inc. (Inst), Janssen Pharmaceuticals (Inst), Novartis (Inst), Pfizer (Inst), Sanofi (Inst); advisory boards for Astellas, AstraZeneca, Capio Biosciences, Modra Pharmaceuticals B.V.; steering committee member with AstraZeneca (CAPI-281), BMS, NCI Genitourinary (Leidos Biomedical Research Inc.), Nektar Therapeutics, Pfizer; independent contractor with Axess Oncology; speaker for Bayer H/C Pharmaceuticals, Exelixis, Inc., Sanofi; honorarium from Bayer H/C Pharmaceuticals, EMD Serono, Exelixis, Inc., Ipsen, Michael J Hennessey Associates, Pfizer, Sanofi, UroGPO, UroToday; travel accommodations from Bayer H/C Pharmaceuticals, Exelixis, Inc., Sanofi, UroToday; Independent Data Monitoring Committee for Janssen Pharmaceuticals; Co-Editor-in-Chief for Millennium Medical Publishing, Clinical Advances in Hematology & Oncology. C-HL reports research funds to the institute from BMS, Calithera, Eisai, Eli Lilly, Exelixis, Merck, and Pfizer; consulting with Amgen, BMS, Exelixis, Eisai, Merck, Pfizer, and EMD Serono; honoraria from ACME, Intellisphere; and research grants to his practice. DH reports consultancies and research funding with Ipsen, Exelixis, BMS, Pfizer, Novartis, Merck, and Eisai.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Medical writing and editorial assistance was funded by Exelixis, Inc. (Alameda, CA).
