Abstract
Background:
The COMPASSION-15 trial showed that cadonilimab plus chemotherapy has significant clinical advantages in patients with HER2-negative advanced gastric or gastroesophageal junction (G/GEJ) adenocarcinoma compared to chemotherapy. This study evaluated the cost-effectiveness for patients in China and the United States.
Objective:
To provide advice for patients on the use of cadonilimab.
Design:
The cost-effectiveness analysis.
Methods:
A partitioned survival model was conducted from perspective of the Chinese and U.S. healthcare systems over a lifetime horizon. Key parameters of the model were derived from COMPASSION-15 trial and published literature. In this study, costs, quality-adjusted life-years (QALYs), and incremental cost-effectiveness ratios (ICERs) were taken as main outcomes. Sensitivity analyses, price simulations, and programmed death ligand 1 combined positive score (PD-L1 CPS) subgroup analyses were conducted to test robustness.
Results:
In base-case analysis, cadonilimab plus chemotherapy group achieved an ICER of US dollars (USD) 32,630.84/QALY in China and USD 109,996.43/QALY in the United States, falling within the established willingness to pay (WTP) thresholds in both cases. At the current negotiated Chinese price (USD 208.94/100 mg), cadonilimab was cost-effective; in the United States, it remained cost-effective when priced below USD 437.87 (USD 100,000/QALY threshold) or USD 870.23 (USD 150,000/QALY threshold) per 100 mg. Subgroup analyses demonstrated that patients with PD-L1 CPS ⩾5 had 100% (China) and >94% (U.S.) probability of cost-effectiveness, whereas those with CPS <5 had reduced economic favorability. Sensitivity analyses identified progression-free survival utility, drug price, and body weight as key drivers of ICERs.
Conclusion:
As a first-line strategy for patients with HER2-negative advanced G/GEJ adenocarcinoma, cadonilimab combined with chemotherapy represents a cost-effective option in both China and the United States. Its economic advantage is most pronounced in patients with high PD-L1 expression and at lower drug prices. These findings provide quantitative evidence supporting reimbursement negotiations and future pricing strategies for cadonilimab in global markets.
Plain language summary
Keywords
Introduction
In the 21st century, cancer is expected to become the primary contributor to early deaths, transcending cardiovascular diseases and posing a significant barrier to improving population life expectancy. 1 Adenocarcinoma represents the vast majority of gastric malignancies, comprising more than 90% of diagnosed cases and thereby constituting the most prevalent histological form of gastric cancer. The 2022 Global Cancer Statistics indicated that gastric cancer accounted for approximately 968,350 newly diagnosed cases worldwide, making it the fifth most prevalent malignancy. Additionally, gastric cancer caused 659,850 deaths, ranking fifth in contributing to cancer-related mortality worldwide. 2 Gastric cancer can be molecularly classified based on the HER2 expression status in tumor tissues. In Asian populations, approximately 85% of gastric cancer cases are HER2-negative. 3 In China, gastric cancer holds the third position regarding both incidence and mortality throughout all cancer categories.4,5 Approximately 50% of individuals are already found to have either advanced-stage or metastatic disease at diagnosis, yielding a 5-year overall survival (OS) below 10%. Despite global declines in cancer incidence and mortality rates, individuals diagnosed with gastric or gastroesophageal junction (G/GEJ) adenocarcinoma continue to face a poor prognosis and substantial disease burden in China and the United States.
Multiple clinical trials have established that the combination treatment of programmed death-1 (PD-1) inhibitors and chemotherapy yields survival benefits in certain subgroups of patients with G/GEJ adenocarcinoma.6–8 Currently, this regimen has been widely adopted as a standard treatment option. However, their clinical efficacy remains constrained by patients’ low PD-L1 expression levels, and current immunotherapy has shown limited benefit to those patients. For individuals exhibiting HER2-negative profiles along with low PD-L1 expression, survival advantages remain minimal, underscoring the unmet need for novel therapeutic approaches.
Cadonilimab, a dual-targeting bispecific antibody, has received regulatory approval for use in treating cervical and gastric cancer in China. In the United States, cadonilimab was granted an orphan drug by the Food and Drug Administration (FDA). A phase III trial (COMPASSION-15) that enrolled patients from 75 hospitals in China demonstrated that cadonilimab plus chemotherapy substantially extended patients’ survival. 9 Across the entire cohort, patients administered cadonilimab achieved an OS of 14.1 months, whereas OS was 11.1 months in the placebo arm. As for progression-free survival (PFS), it reached 7.0 and 5.3 months, respectively. Although outcomes varied across subgroups with different PD-L1 expression levels, combined treatment conferred a statistically significant survival advantage, indicating a previously unachieved level of treatment effectiveness within a patient population traditionally considered difficult to manage.
Although combination therapy of cadonilimab and chemotherapy has demonstrated clinical efficacy and safety and offers a groundbreaking therapeutic option, cadonilimab’s substantial cost thus leaves this therapy’s economic value undetermined. The evaluation of cost-effectiveness is critical to healthcare decision-making. Currently, there is a lack of evidence in published studies concerning economic efficiency of incorporating cadonilimab into chemotherapy for patients with G/GEJ adenocarcinoma, and this research gap is particularly prominent when focusing on subgroups of PD-L1 CPS. To make cost-effectiveness analysis clinically meaningful, we also used published phase III PD-1 trials (KEYNOTE-859, CheckMate-649, RATIONALE-305) to indirectly compare cadonilimab plus chemotherapy with other combination therapies. This research focuses on assessing the economic value for treating gastric cancer, aiming to provide evidence-based support for more efficient allocation of medical resources and informed clinical decision-making in China and the United States.
Materials and methods
Following the Consolidated Health Economic Evaluation Reporting Standards guidelines, this study was designed and undertaken from the viewpoint of Chinese and U.S. health service systems, 10 as detailed in Table S1.
Population and interventions
Since no human subjects were directly involved and the study utilized mathematical modeling exclusively, ethical approval was waived for this analysis. The characteristics of the participants and the interventions included in the model were identical to those in COMPASSION-15 trial. Participants were categorized into two cohorts: a high expression level cohort (PD-L1 CPS ⩾5) and a low expression level cohort (PD-L1 CPS <5).
A total of 610 participants were randomly divided into two separate arms of the same number. All participants’ baseline characteristics demonstrating a balanced distribution between the two arms. In the COMPASSION-15 trial, the duration of cadonilimab administration was capped at 2 years, and according to the results of clinical trials, the duration of treatment with cadonilimab was 8 weeks, and that of oxaliplatin and capecitabine was 6 weeks. 9 The combination treatment period comprised six 21-day cycles. In each 3-week treatment cycle, individuals assigned to the cadonilimab plus chemotherapy arm were administered cadonilimab intravenously (10 mg/kg, Day 1 (D1)), along with oral capecitabine twice daily (1000 mg/m², D1–D14), and an intravenous infusion of oxaliplatin (130 mg/m², D1). Patients in the chemotherapy group received placebo instead of cadonilimab, with other procedures identical to those in the cadonilimab group. Following the combination treatment period, maintenance therapy with cadonilimab or placebo (10 mg/kg, intravenously) was continued until disease progression. Throughout the study period, tumor assessments were conducted at baseline and subsequently every 6 weeks during the initial 54 weeks, followed by evaluations at 9-week intervals. The drug dosage of nivolumab group, pembrolizumab group, and tislelizumab group was consistent with clinical trials. It is assumed that the three groups use the same chemotherapy regimen as the cadonilimab group. After discontinuation of study treatment, subsequent systemic anticancer therapy was given to 186 (61%) patients in cadonilimab group and 283 (93%) patients in chemotherapy group. The proportion of receiving subsequent anticancer therapy in the nivolumab group, pembrolizumab group, and tislelizumab group were 38%, 45%, and 53%, respectively. Subsequent therapies mainly included chemotherapy, and patients were treated with intravenous irinotecan. Other patients received best supportive care (BSC). Presumptions were established for individuals’ physical characteristics: participants’ average weight was 65 kg in China and 75 kg in the United States, mean body surface area was 1.72 and 1.8 m², respectively.11,12
Model structure
We evaluated a partitioned survival model (PSM) to simulate the outcomes of the treatment strategies. The model was designed to operate on a 3-week cycle and extended across a lifetime analytical horizon. A half-cycle adjustment was applied to the estimates of cost and utility, with both subsequently discounted at a yearly rate of 5% in China and 3% in the United States.13–15 The structure of the model comprised three distinct health states, as shown in Figure 1. At the beginning, all subjects were considered to be in the PFS state, with potential transition to either progression disease (PD) or death (D).16,17 The primary outcomes included costs, quality-adjusted life-years (QALYs), and incremental cost-effectiveness ratio (ICER).

Partitioned survival model structure.
We evaluated the risk bias of several clinical trials, and the results of the literature quality assessment are shown in Figure S1. In the clinical trial, OS and PFS were presented for both the overall population and subgroups, this study employed GetData Graph Digitizer (version 2.26, available at: http://www.getdata-graph-digitizer.com/index. php, Moscow, Russia) to retrieve patients’ data by digitizing the Kaplan–Meier curves. The original survival data and simulated reconstruction data of the clinical trials are detailed in Table S2. The subsequent step involved fitting the data to various parametric survival models. The curves with the minimum Akaike Information Criterion and Bayesian Information Criterion values were preferred, 15 supplemented by visual inspection of Kaplan–Meier plot. Lognormal distribution was applied to fit the two survival curves in cadonilimab group. The OS and PFS curves of the chemotherapy group were fitted by loglogistic distribution and lognormal distribution, respectively. The OS and PFS curves of the other three groups were fitted using loglogistic distribution. Table 1 shows the scale and shape parameters of these curve fitting distributions. Survival curve fitting was performed with R 4.2.1 software (available at: http://mirrors.pku.edu.cn/CRAN/). The corresponding parameters of all survival curves and the simulation plots are shown in Table S3 and Figures S2–S25 in Supplemental Materials. We performed a network meta-analyses (NMA) of hazard ratio (HR) for survival curves across four clinical trials to compare which group had the most significant survival advantage. 17
Parameters of the best-fitted distributions.

OS, overall survival; PD-L1 CPS, programmed death ligand 1 combined positive score; PFS, progression-free survival.
Utility and cost inputs
To represent patients’ health conditions, utility values were applied, ranging from 0 to 1 (0 indicates death and 1 signifies optimal health). 16 Since patient-reported quality of life (QOL) outcomes were not reported in the clinical trial, utility estimates were derived from existing literature (0.797 for PFS and 0.577 for PD). Only those serious adverse events (AEs) classified as grade 3 or more and reported in ⩾5% of cases were analyzed, with their disutility values obtained from the published literature, too.17–21 All AEs in both treatment arms were presumed to occur within the initial cycle of therapy. The details are shown in Table 2.
Key model inputs.
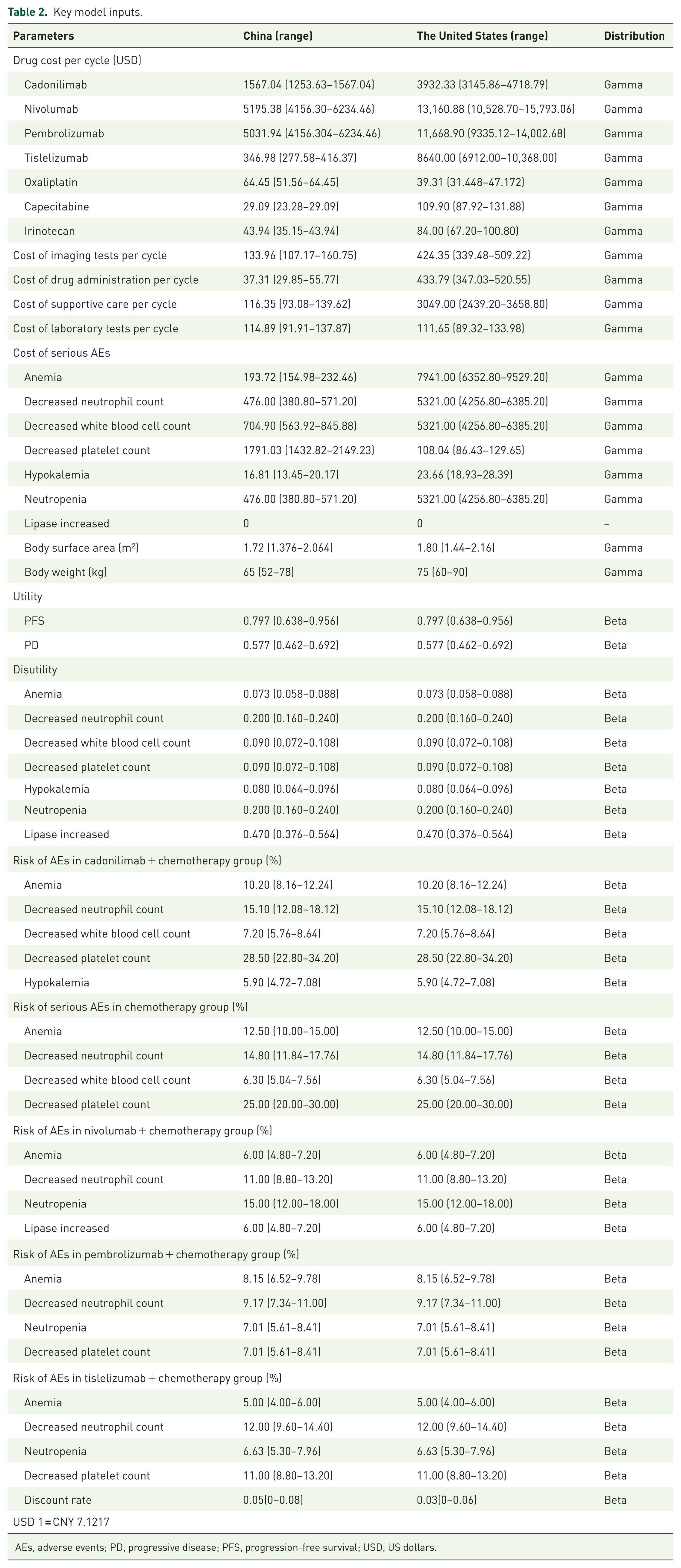
AEs, adverse events; PD, progressive disease; PFS, progression-free survival; USD, US dollars.
This analysis focused solely on direct healthcare expenditures, encompassing expenses related to pharmaceuticals, laboratory testing, imaging examination, medication administration, BSC, subsequent treatment, and AEs management. For the Chinese perspective, the cost of drugs, laboratory tests, imaging tests, and AEs was derived from public databases (https://db.yaozh.com/), local hospital pricing system, and expert consultations. And other costs were obtained from published literature.15–17 For the U.S. perspective, all costs were taken from the published literature.22–24 The currency unit used in this study is US dollars (USD), and the exchange rate used for conversion is “USD 1 = CNY 7.1217.” The key parameters of this study are presented in Table 2.
Price simulation
After being listed in the National Reimbursement Drug List (NRDL), cadonilimab has been priced at around USD 208.94/100 mg in China. Since cadonilimab is not yet available in the United States, we used a hypothetical price as the price of cadonilimab in the base-case analysis. We selected several drugs commonly used for the treatment of HER2-negative G/GEJ adenocarcinoma, calculated the ratio of China/the United States prices, and took several values to form a price index, so as to calculate the hypothetical price of cadonilimab (USD 524.31/100 mg).24–26
In the base-case analysis of China, the price of cadonilimab varied from USD 0–208.94/100 mg, studying the impact of different prices on willingness to pay (WTP). In the United States, we investigated the effect on the WTP threshold when the price of cadonilimab increased by USD 100/100 mg between USD 0 and USD 2000/100 mg.
Sensitivity analysis
To evaluate the stability of the model outcomes, sensitivity analyses were conducted. The deterministic sensitivity analysis (DSA) was employed to explore how alterations in individual parameters influenced the ICER results. The discount rate was allowed to fluctuate 0–0.08 in China and 0–0.06 in the United States. 25 Whereas remaining parameters were set at ±20%. Tornado diagrams were utilized to visually illustrate the results of DSA. The probabilistic sensitivity analysis (PSA) incorporated Monte Carlo simulations with 1000 iterations to assess the acceptability rates of treatment regimens across varying WTP thresholds. To illustrate the outcomes of PSA, we utilized cost-effectiveness acceptability curves (CEACs) along with scatter diagrams.
Results
Meta-analysis
We used a fixed-effect NMA to compare HR for survival outcomes across four therapies. As presented in Figures S26 and S27, the cadonilimab group obtained the best survival benefits, both in terms of PFS and OS.
Base-case analysis
The cadonilimab plus chemotherapy group demonstrated an incremental cost of USD 13,481.71 and an incremental effectiveness of 0.41 QALYs in China. In the United States, cadonilimab group exhibited an incremental cost of USD 48,054.95 and an incremental utility of 0.44 QALYs. The WTP threshold in China is proposed to be set at three times the Chinese gross domestic per capita (GDP). 22 For the year 2024, Chinese GDP per capita was estimated at around USD 13,445, and the WTP threshold applied in this analysis was set at USD 40,335/QALY in China. The WTP in the United States was set at USD 100,000–150,000/QALY. 27 The ICER was USD 32,630.84/QALY, which is below the WTP in China, and USD 112,997.16/QALY, which falls between the WTP range of USD 100,000–150,000/QALY in the United States. More details about basic case analysis and subgroup analysis are shown in Tables 3 and 4. We also analyzed the total cost and utility of nivolumab, pembrolizumab, and tislelizumab from the perspectives of China and the United States, which can be found in Table S4.
Results of base-case analysis.

ICER, incremental cost-effectiveness ratio; QALY, quality-adjusted life year; USD, US dollars.
Results of subgroup analysis.
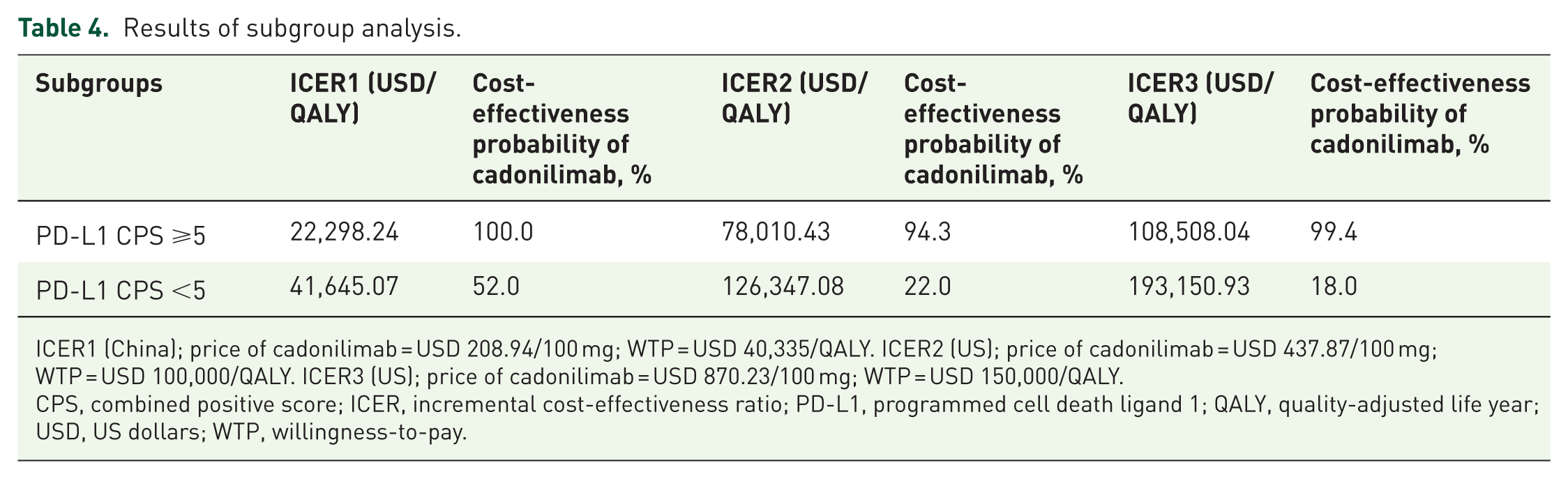
ICER1 (China); price of cadonilimab = USD 208.94/100 mg; WTP = USD 40,335/QALY. ICER2 (US); price of cadonilimab = USD 437.87/100 mg; WTP = USD 100,000/QALY. ICER3 (US); price of cadonilimab = USD 870.23/100 mg; WTP = USD 150,000/QALY.
CPS, combined positive score; ICER, incremental cost-effectiveness ratio; PD-L1, programmed cell death ligand 1; QALY, quality-adjusted life year; USD, US dollars; WTP, willingness-to-pay.
Price simulation
For China, cadonilimab was cost-effective when WTP = USD 40,335/QALY and price was less than USD 272.23/100 mg. The current price of cadonilimab in China is USD 208.94/100 mg, which is more popular in the overall population. For the United States, ICER increased as the price of cadonilimab changed from USD 0/100 mg to USD 2000/100 mg. When the drug price was less than USD 437.87/100mg (WTP = USD 100,000/QALY) and USD 870.23/100 mg (WTP = USD 150,000/QALY), cadonilimab combined with chemotherapy was more cost-effective. Figure 2 shows the results of the price simulation.
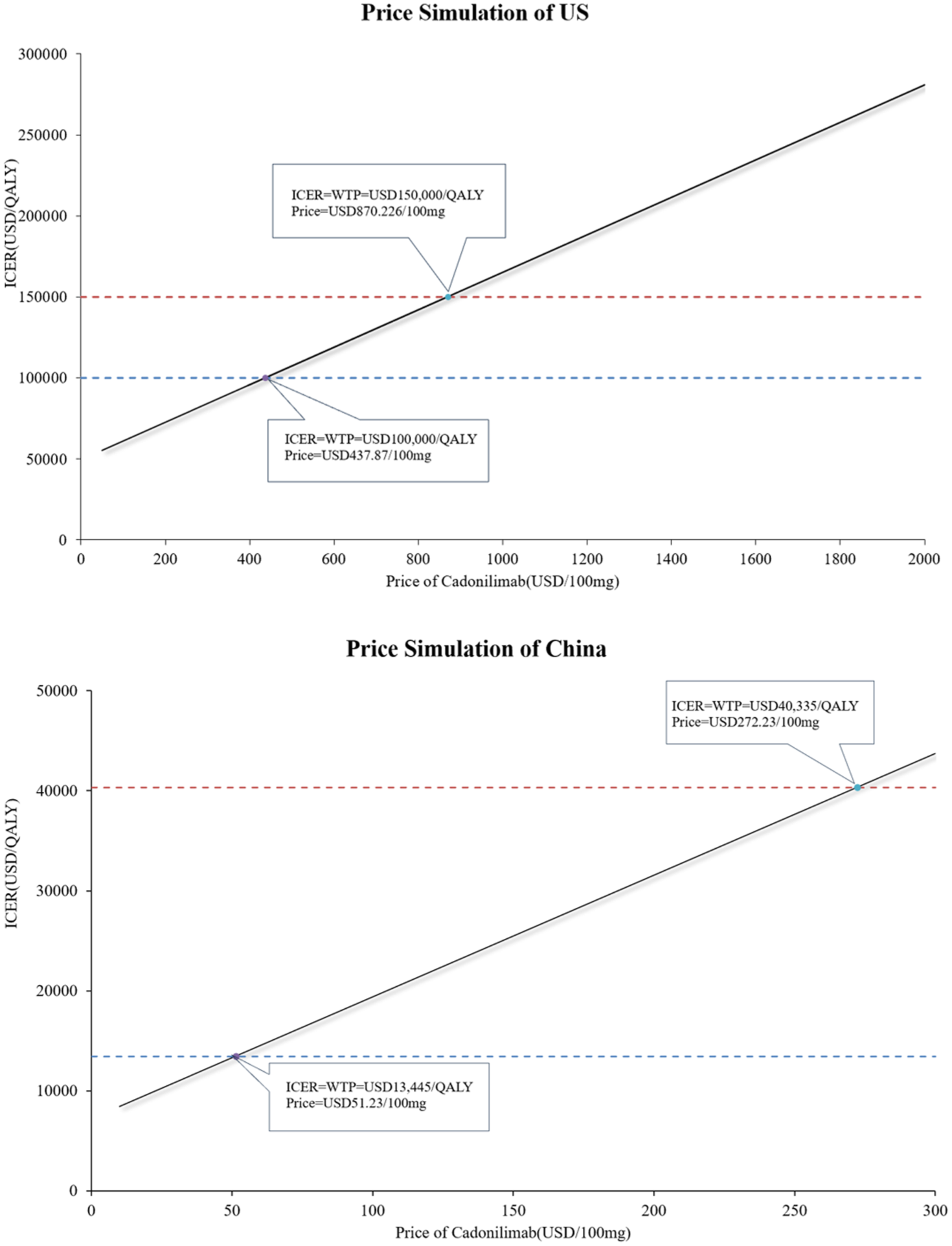
Results of price simulations.
Subgroup analysis
For China, at the current pricing of cadonilimab (USD 208.94/100 mg), ICERs in two subgroups were investigated. The subgroup characterized by PD-L1 CPS ⩾5 had an ICER of USD 22,298.24/QALY, accompanied by a 100% probability of being cost-effective, while the probability of cost-effectiveness for another subgroup stood at 52%. For the United States, when cadonilimab was priced at USD 437.87/100 mg and USD 870.23/100 mg, the probability of cost-effectiveness for the subgroups with good survival advantages was 94.3% and 99.4%, respectively. Table 5 shows the results of subgroup analysis. Monte Carlo scatter plots and CEACs for subgroup analysis are shown in Figures S27–S30 of the Supplemental Material.
Summary of PSA results.

PSA, probabilistic sensitivity analysis; QALY, quality-adjusted life year; USD, US dollars; WTP, willingness to pay.
Sensitivity analysis
In the base-case analysis, the tornado diagrams illustrated that the utility value of PFS exerted the most significant influence on ICER in both China and the United States. The utility value of PFS fluctuated between 0.5384 and 0.8076, followed by ICER in China at USD 27,558.65/QALY–USD 39,991.27/QALY, and ICER in the United States at USD 93,146.35/QALY–USD 134,289.22/QALY. For China, the subsequent parameters that had a great impact on the model ICER were the price of cadonilimab, patient’s weight, discount rate, and utility value of PD. For the United States, several other parameters that were more sensitive to model ICER were patient’s weight, price of cadonilimab, cost of BSC, and discount rate. Figure 3 presents the results of DSA.

Results of deterministic sensitivity analysis.
For China, the CEACs showed that the likelihood of cadonilimab group being considered cost-effective was 78.0% in overall population. With the price of cadonilimab increased, the WTP for achieving a 50% likelihood of cost-effectiveness in the United States had increased, too. The results of PSA, the CEACs for cadonilimab pricing at USD 437.87/100 mg and USD 870.23/100 mg in the United States, and the results of Monte Carlo simulations are shown in Table 5, Figures 4 and 5, respectively.

Cost-effectiveness acceptability curve.

The Monte Carlo sensitivity analyses for cadonilimab plus chemotherapy compared to chemotherapy.
Discussion
The effects and adverse reactions of traditional chemotherapy are unsatisfactory. Over the past few years, immunotherapy has achieved remarkable breakthroughs for managing advanced gastric cancer.28,29 One of the standard first-line treatment options recommended for patients with HER2-negative advanced gastric cancer involves PD-1 inhibitors in conjunction with chemotherapy. Several phase Ⅲ trials showed better efficacy for patients than chemotherapy alone, and these drugs have been approved for the first-line treatment of gastric cancer in China. Nivolumab, pembrolizumab, tislelizumab have been marketed in China and the United States. In China, nivolumab and pembrolizumab have not yet been approved for insurance coverage for this indication, with prices of USD 1298.84/100 mg and USD 2515.97/100 mg, respectively. In this study, patients in the cadonilimab group achieved the highest utility of 1.23 QALYs. The nivolumab group had the highest cost and the lowest utility, resulting in a low probability of nivolumab being favored. This is consistent with the results of previously published literature. A study showed that the ICER of the nivolumab group was USD 430,185.04/QALY, which has no economic benefits in China. 30 Another study showed that pembrolizumab was more economical than nivolumab in China, but neither therapy had an overall advantage compared to chemotherapy alone. 31 Compared with chemotherapy alone, the ICER of pembrolizumab combined with chemotherapy was much higher than WTP threshold, indicating that pembrolizumab combined with chemotherapy for the treatment of HER2-negative gastric cancer was not economical in China.32,33 The cost and utility of the pembrolizumab group were 59,372.53 USD and 0.92 QALYs, which were not dominant compared with the cadonilimab group in this study. Tislelizumab has been included in medical insurance in China, with drug cost of USD 176.02/100 mg. In line with published evidence, tislelizumab plus chemotherapy was cost-effective compared with chemotherapy alone in China, which is concordant with the results of our study. 34 Cadonilimab group’s cost was higher than tislelizumab group, while yielding superior health utility. The ICER of cadonilimab versus tislelizumab was found to be below the WTP threshold in China. From the perspective of the United States, the results of this study show that none of the three-drug combination therapy is cost-effective compared with chemotherapy. According to the available evidence, nivolumab was more economical than pembrolizumab, and the ICER of two combination therapies far exceeded the WTP threshold, and neither drug will be prioritized in the United States.31–33 Tislelizumab was also non-cost-effective in the United States, with an ICER of USD 502,786.22/QALY, which was much higher than the WTP in the United States. 34 Against the global momentum of biopharmaceutical innovation, novel immunotherapeutic agents, particularly bispecific antibodies, have demonstrated significant potential in clinical studies.35,36 This development marks a new stage of cancer immunotherapy characterized by multi-target immune modulation. Cadonilimab, a representative bispecific antibody, has received approval for use alongside with chemotherapy as a first-line regimen. This application aims to tackle the limited efficacy of conventional PD-1/PD-L1 inhibitors among individuals exhibiting low or negative expression of PD-L1. This combination provides a more comprehensive and effective immunotherapeutic option for patients, representing an important milestone in the field. Following a median follow-up of 33.87 months, the final analysis of the COMPASSION-15 trial confirmed that cadonilimab treatment arm continued to provide survival benefits in both OS and PFS. Notably, the survival advantage was further enhanced in patients with PD-L1 CPS ⩾5, where HR of OS was 0.58. In another subgroup, the benefit was maintained, suggesting consistent clinical value across different levels of PD-L1 expression. Our study further demonstrated that although cadonilimab plus chemotherapy improved survival outcomes in all patients with HER2-negative advanced G/GEJ adenocarcinoma irrespective of PD-L1 status, the magnitude of economic benefit varied by subgroup. In China, the PD-L1 CPS ⩾5 population showed optimal cost-effectiveness, with a certainty of 100% regarding its cost-effectiveness, while this probability decreased to 52% in patients with PD-L1 expression level. A similar pattern was observed in the United States: high expressors maintained favorable cost-effectiveness (94.3%–99.4%), whereas low expressors showed markedly reduced economic benefit (18%–22%). These findings highlight a critical insight for domestic innovative drugs entering global markets—the importance of achieving both clinical benefit and economic value to enhance global competitiveness. The COMPASSION-15 trial also reported the HR of OS in both subgroups when the CPS cutoff value was set to 10. For HER2-negative patients, a higher CPS cutoff may also identify a subgroup with stronger PD-L1 expression and thus greater benefit from combination therapy. For the subgroup of PD-L1 CPS <10, OS was longer in the cadonilimab group than in the placebo group, with a 28% reduction in the risk of death (HR 0.72, 95% confidence interval (CI): 0.56–0.93). For the subgroup with PD-L1 CPS of 10 or more, the risk of death was reduced by 46% in the cadonilimab group compared with the chemotherapy alone group (HR 0.54, 95% CI: 0.35–0.83). The HR of both subgroups was smaller than that of the CPS cut-off value of 5. The QALY obtained in both subgroups became higher, and the probability of cost-effectiveness increased.
Our study further identified that in both China and the United States, the utility value during PFS and the drug price were the two most influential parameters affecting the model outcomes. Among them, the impact of PFS utility on the ICER was the most pronounced, underscoring the central role of QOL in cost-effectiveness analyses. This finding highlights the need for more accurate and patient-centered assessments of QOL during the PFS stage in clinical practice to optimize overall treatment value. Meanwhile, the significant influence of drug price on model outcomes suggests that proactive international pricing strategies are essential. Early planning and rational adjustment of overseas market prices could enable patients with varying PD-L1 expression levels to achieve economic benefits, thereby improving the global accessibility and long-term sustainability of cadonilimab. These findings suggest that the definition of “patient benefit” is expanding beyond clinical efficacy to include accessibility, affordability, and patient-reported outcomes—dimensions increasingly central to assessing the value of innovative therapies. It is noteworthy that in recent years, several PD-1 inhibitors such as nivolumab and pembrolizumab have withdrawn indications for PD-L1 negative populations, while tislelizumab remains under regulatory review by the FDA and EMA, with approvals limited to high PD-L1-expressing subgroups.37,38 Against this backdrop, the COMPASSION-15 trial highlights the potential clinical advantage of cadonilimab in patients with low or negative PD-L1 expression, offering a new direction for global gastric cancer immunotherapy. If cadonilimab plans to enter the U.S. market, updating global clinical data and refining pharmacoeconomic strategies that specifically target cost-effectiveness improvement among patients with PD-L1 CPS <5 may facilitate broader regulatory recognition of this subgroup by the FDA. At its initial launch in China, cadonilimab was priced at approximately USD 1855 per 125 mg vial, resulting in an estimated annual treatment cost of about USD 28,000. Such a high price imposed a substantial financial burden on patients and made the therapy difficult to achieve cost-effectiveness thresholds. In 2024, cadonilimab was included in China’s NRDL through national price negotiations at a significantly reduced price of USD 261.17 per vial—an 85.92% reduction from its launch price. This adjustment dramatically lowered patients’ financial burden and enabled cadonilimab to achieve favorable cost-effectiveness within the Chinese healthcare system. This process exemplifies China’s capacity to balance innovation incentives and patient affordability through a centralized, evidence-based drug negotiation mechanism. This experience also provides valuable policy implications for the U.S. healthcare system. Unlike China’s centralized negotiation framework, the United States maintains a largely market-driven pricing mechanism without a unified national price regulation structure. The case of cadonilimab underscores that a drug’s true clinical value should be reflected through systematic cost-effectiveness evaluation and payer negotiation. For the United States, incorporating pharmacoeconomic evidence—such as value-based pricing models anchored in QALY assessments—into pricing and reimbursement decisions could reduce access barriers to innovative therapies while sustaining incentives for pharmaceutical innovation. 39 Overall, China’s NRDL negotiation model provides a value-oriented policy paradigm for global drug pricing.40,41 By identifying patient subgroups with differential benefits, integrating pharmacoeconomic evidence, and linking prices dynamically to therapeutic value, this approach promotes a balance between patient access, healthcare sustainability, and industrial innovation. Such a framework may serve as a valuable reference for future pricing and access strategies for cadonilimab and other emerging Chinese immunotherapies entering international markets.
There are certain limitations to this research. First, the model relies on extrapolated long-term survival data and uses a simulation method to infer survival outcomes from Kaplan–Meier curves. The uncertainty of long-term efficacy data could undermine how reliable the findings are. Second, this study considered only direct medical costs and did not include indirect costs, resulting in a potentially incomplete cost-effectiveness evaluation. Third, the analysis only included AEs with an incidence ⩾5% and grade ⩾3. It did not account for the long-term impact of AEs, and the limitation may compromise model precision. Fourth, the clinical trial used in this study was conducted in China only, and didn’t include U.S. population. The efficacy and safety of the trial may vary between the Chinese population and the U.S. population. In addition, patient QOL data were not reported in the clinical trial, and utility values were derived from published literature, which may cause discrepancies between model outputs and real-world data. Finally, the WTP threshold was set at the 2024 GDP per capita in China and at USD 100,000–150,000/QALY in the United States. However, substantial economic heterogeneity exists across regions in both countries. Combination therapy may be more acceptable in economically developed regions, whereas less-developed areas may face heavier payment burdens, which could affect the generalizability of our conclusions.
Conclusion
In summary, in China and the United States, findings of this study indicated that combination therapy of cadonilimab and chemotherapy offers a more cost-effective treatment strategy. This therapy may offer a new treatment option and is worthy of recommendation for patients with low PD-L1 expression level particularly. For the United States, it is more likely to be economical to choose cadonilimab when it is priced below USD 437.87/100 mg (WTP = USD 100,000/QALY) and USD 870.23/100 mg (WTP = USD 150,000/QALY). Further price reductions are needed to make people with low PD-L1 expression levels economical. These results provide a reference for the pricing of cadonilimab available in the United States.
Supplemental Material
sj-docx-1-tam-10.1177_17588359261428081 – Supplemental material for Cadonilimab plus chemotherapy for patients with HER2-negative advanced gastric or gastroesophageal junction adenocarcinoma in China and the United States: a cost-effectiveness analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359261428081 for Cadonilimab plus chemotherapy for patients with HER2-negative advanced gastric or gastroesophageal junction adenocarcinoma in China and the United States: a cost-effectiveness analysis by Jia Wang, Baolong Ding, Zhuying Jing, Yulu Zhu, Lihong Gao, Hongting Yao, Tiantian Tao and Xin Li in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
