Abstract
Background:
The LITESPARK-005 trial demonstrated the efficacy and safety of belzutifan in patients with previously treated clear cell renal cell carcinoma (ccRCC). This study aims to evaluate the cost-effectiveness of belzutifan compared to everolimus in treating patients with advanced ccRCC who have received at least one systemic therapy from the perspective of the Chinese healthcare system and the US payers.
Objectives:
To provide previously treated ccRCC patients with the option of belzutifan and to offer recommendations regarding in China.
Design:
The cost-effectiveness analysis.
Methods:
A partitioned survival model was constructed based on data from the LITESPARK-005 trial. Patients transitioned through three mutually exclusive health states: progression-free survival (PFS), progressive disease, and death. The model cycle length was set at 28 days, with a lifetime horizon. Direct medical costs and utility values were obtained from published literature and real-world healthcare data. The model estimated total costs, quality-adjusted life years (QALYs), and incremental cost-effectiveness ratios (ICERs). Price simulations, sensitivity analyses, and scenario analyses were conducted to assess model robustness.
Results:
The base-case analysis showed that belzutifan in China generated 2.038 QALYs at a total cost of $102,561.26 and an ICER of $54,430.16/QALY, which exceeded the willingness to pay (WTP) threshold ($39,076.44/QALY). Belzutifan in the United States generated 2.280 QALYs at a total cost of $796,227.28 with an ICER of $270,864.46/QALY, which significantly exceeded the WTP threshold ($150,000/QALY). The PFS utility value and the drug cost of belzutifan were the main factors affecting the change in ICER, whether in China or the United States. At current pricing, belzutifan was unlikely to be cost-effective. Price simulations indicated the belzutifan would be cost-effective when the price of belzutifan remained below $5.524/mg in the United States and $0.779/mg in China.
Conclusion:
Compared to everolimus, belzutifan is not cost-effective at its current price for treating previously treated RCC in the United States. In China, the belzutifan group was cost-effective when the price of belzutifan was less than $0.779/mg. This study suggests that reducing the price could substantially improve the economic viability of belzutifan.
Keywords
Introduction
Renal cell carcinoma (RCC) is a common and lethal urologic malignancy, accounting for 2%–3% of all cancers globally. Its incidence has been increasing over the past decade.1 –3 In 2022, China reported 73,656 new RCC cases and 23,991 RCC-related deaths. 4 RCC is one of the top 10 most prevalent cancers in the United States. 5 In 2025, around 80,980 new cases of RCC are expected, accounting for 4.0% of all new cancer diagnoses and 2.3% of all cancer-associated deaths. Clear cell renal cell carcinoma (ccRCC) is the most prevalent histological subtype, accounting for 60%–85% of all RCC cases. 6
Anti-angiogenic therapies and immune checkpoint inhibitors (ICIs) have been established as first-line treatments for advanced ccRCC, improving patient outcomes.7 –11 Over 90% of ccRCC cases exhibit mutations, deletions, or methylation of the Von Hippel-Lindau (VHL) gene, which leads to the accumulation of hypoxia-inducible factor-2α (HIF-2α) and disruption of the hypoxia signaling pathway, promoting tumorigenesis, angiogenesis, and metastasis. HIF-2α is a transcription factor that upregulates the expression of hundreds of genes, including those encoding vascular endothelial growth factor (VEGF), platelet-derived growth factor subunit B, transforming growth factor alpha, CXC chemokine receptor 4, erythropoietin, and cytosolic protein D1. All of these play known or potential roles in the progression of ccRCC.
Belzutifan, an innovative small-molecule HIF-2α inhibitor, is the first and currently only FDA-approved (Food and Drug Administration) drug in its class. Recently, the phase III LITESPARK-005 trial, a multicenter, randomized, open-label, active-controlled study across six regions, evaluated belzutifan versus everolimus in patients with advanced ccRCC that had progressed following prior ICIs and anti-angiogenic therapies. In the first interim analysis (median follow-up: 18.4 months), both treatment groups demonstrated a median progression-free survival (PFS) of 5.6 months. At 18 months, 24.0% of patients in the belzutifan group and 8.3% in the everolimus group remained progression-free (p = 0.002). The objective response rate was 21.9% for belzutifan versus 3.5% for everolimus (p < 0.001). The second interim analysis (median follow-up: 25.7 months) reported a median overall survival (OS) of 21.4 months (belzutifan) versus 18.1 months (everolimus), with a hazard ratio for death of 0.88 (p = 0.20). Grade ⩾3 treatment-emergent adverse events (TEAEs) occurred in 61.8% of patients treated with belzutifan and 62.5% of those receiving everolimus; treatment discontinuation due to TEAEs occurred in 5.9% and 14.7%, respectively.
Although belzutifan has shown efficacy in treating previously treated ccRCC and is approved for marketing in the United States, it remains costly for patients who have undergone prior therapy. Currently priced at approximately $6.531 per 1 mg in the United States, a cycle at the recommended daily dose of 120 mg costs over $20,000 and would need to be taken until the disease progresses or an unacceptable toxicity occurs, which would undoubtedly be a sustained and costly financial outlay for patients. Currently, belzutifan is approved by China’s National Medicinal Products Administration (NMPA) for the treatment of VHL-associated RCC. However, the indication of belzutifan for the treatment of ccRCC is still under approval by the NMPA and is expected to be available in China within 1–2 years. The price of belzutifan in China has not yet been determined. In contrast, the per capita disposable income in 2024 was only $5620.26, highlighting the financial burden on patients and the strain on the healthcare system.
Most pharmacoeconomic studies on RCC have focused on advanced RCC treatments such as sunitinib, axitinib, and nivolumab, with limited research evaluating the economic value of belzutifan in ccRCC.12 –14 Given its promising efficacy, a cost-effectiveness analysis of belzutifan is urgently needed. The lack of accurate pricing information poses a challenge in assessing the cost-effectiveness of this new treatment. Everolimus, an mammalian target of rapamycin (mTOR) inhibitor, has been used as monotherapy in specific clinical contexts. Therefore, this study aims to evaluate the cost-effectiveness of belzutifan versus everolimus in previously treated ccRCC based on the data from the LITESPARK-005 trial from the perspective of the Chinese healthcare system and the US payers and provide evidence for decision-making in treatment adoption.
Material and methods
Patients and interventions
This study was conducted in accordance with the Consolidated Health Economic Evaluation Reporting Standards (CHEERS) guidelines (Table S1). 15 The target population and treatment strategies were based on the LITESPARK-005 trial, a randomized, open-label, active-controlled, multicenter phase III clinical study.
Eligible patients were aged ⩾18 years with stage IV ccRCC, a Karnofsky Performance Status score of ⩾70, and at least one measurable lesion per Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1 (available at: https://recist.eortc.org/recist-1-1-2/). Patients had experienced disease progression following prior treatment with a programmed death-1 (PD-1) or programmed death-ligand 1 (PD-L1) inhibitor and a VEGF receptor tyrosine kinase inhibitor (TKI), either sequentially or in combination. Participants had received no more than three prior systemic therapies, and radiographic disease progression had occurred during or after the most recent regimen. Prior treatment with everolimus was not allowed.
Patients were randomized 1:1 to receive either belzutifan 120 mg or everolimus 10 mg orally once daily until disease progression, unacceptable toxicity, or withdrawal from treatment. No crossover was planned between groups. To manage adverse events, the belzutifan dose could be reduced to 80 mg and then to 40 mg per day, with treatment discontinued if further reduction was required. Dose adjustments for everolimus were allowed in accordance with its prescribing information.
Model construction
A partitioned survival model with three mutually exclusive health states, including PFS, progressive disease (PD), and death, was constructed to simulate the clinical and economic outcomes of the two treatment strategies. All patients were assumed to enter the model in the PFS state and transition unidirectionally along the “PFS—PD—death” sequence, with no possibility of returning to a previous state (Figure 1). For example, patients in the PD state could either remain in PD or transition to death but not return to PFS. The model cycle length was set at 4 weeks, consistent with the treatment schedule in the LITESPARK-005 trial. The time horizon was set as a lifetime horizon, with 99% of people dying, to capture the long-term survival and economic impacts of ccRCC treatment. 16
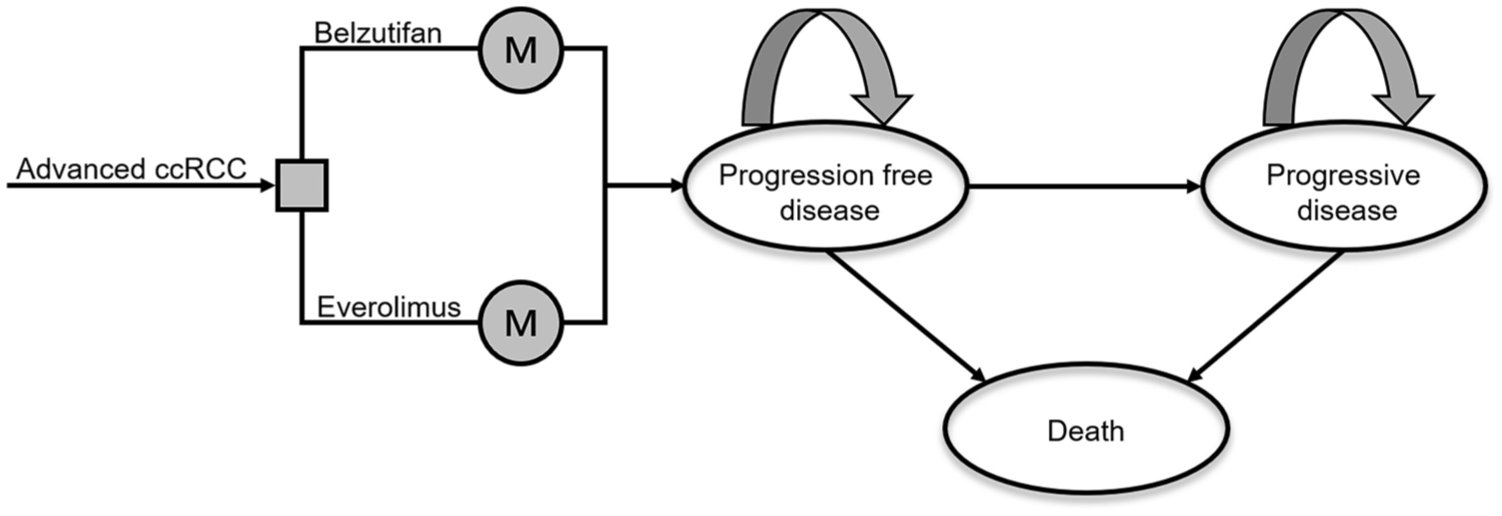
The structure of the partitioned survival model for previously treated ccRCC.
The primary outcomes included total costs, quality-adjusted life years (QALYs), and incremental cost-effectiveness ratio (ICER). A half-cycle correction was applied to costs and outcomes, and both were discounted by 3% for the United States 17 and 5% for China annually. 18 A willingness-to-pay (WTP) threshold was set as $39,076.44 (three times Chinese gross domestic product (GDP) per capita) per QALY gained for China and $100,000–$150,000 for the United States. 19
Clinical data input
Due to the unavailability of individual patient data and limited follow-up, survival curves from the LITESPARK-005 trial were digitized using GetData Graph Digitizer (version 2.26, available at: http://www.getdata-graph-digitizer.com/index.php, Moscow, Russia) to extract PFS and OS data. The extracted curves are shown in Figures S1 and S2. Individual patient-level data were reconstructed in R (version 4.2.2, available at: https://mirrors.pku.edu.cn/CRAN/), and parametric survival models were fitted. 20 Based on visual inspection and statistical fit using the Akaike information criterion (AIC) and Bayesian information criterion (BIC), the best-fit distributions were selected from exponential, gamma, Weibull, log-logistic, log-normal, and Gompertz models. 21
As shown in Table S2, the log-normal distribution provided the best fit for both PFS curves and the OS curve for the everolimus arm, while the log-logistic distribution was optimal for the OS curve in the belzutifan arm. Finally, we built survival functions to obtain the proportion of patients in each state in each cycle. The fitting results are shown in Figures S3 to S6. The proportion of patients in the PFS state was directly obtained from the PFS curve; the deaths were calculated as 100% minus the OS curve, and the PD proportion was derived by subtracting the PFS curve from the OS curve.
Cost and utility input
Only direct medical costs were involved, including the cost of medicines, subsequent treatment, laboratory and imaging tests, hospitalization, routine follow-up visits, and treatment of TEAEs. All these costs were reported in US dollars at a conversion rate of $1 = ¥7.3509 (March 2025). Only grade ⩾3 with an incidence of ⩾5%, including anemia, hypoxia, hypertriglyceridemia, and hyperglycemia, were considered in this study. Drug and adverse event costs were obtained from public databases, local charges, and clinical expert consultations. TEAE incidence rates were derived from the LITESPARK-005 trial, while other direct medical costs were based on public databases and real-world data from local charges.
Utility values represent patient preferences for specific health states, where 0 equals death, 1 equals perfect health. The LITESPARK-005 trial did not collect sufficient information about patients’ quality of life; therefore, utility values for PFS and PD states (0.78 and 0.66, respectively) were obtained from published literature.22 –24 These values were derived from studies using the EuroQol Five Dimensions Questionnaire (EQ-5D) scale and the European Organisation for Research and Treatment of Cancer (EORTC QLQ-C30) scale, both of which are well-established tools for measuring health utility in oncology. The EQ-5D is widely used as a generic health-related quality of life measure, while the EORTC QLQ-C30 is a core scale in cancer patient quality of life assessment. Disutility values for TEAEs (grade ⩾3, incidence ⩾5%) were also sourced from the literature.12,13,25 –27 Key parameters are summarized in Tables 1 and 2.
Clinical and health parameters.

AEs, adverse events; OS, overall survival; PD, progressive disease; PFS, progression-free survival.
Cost parameters and the range of the sensitivity analysis.
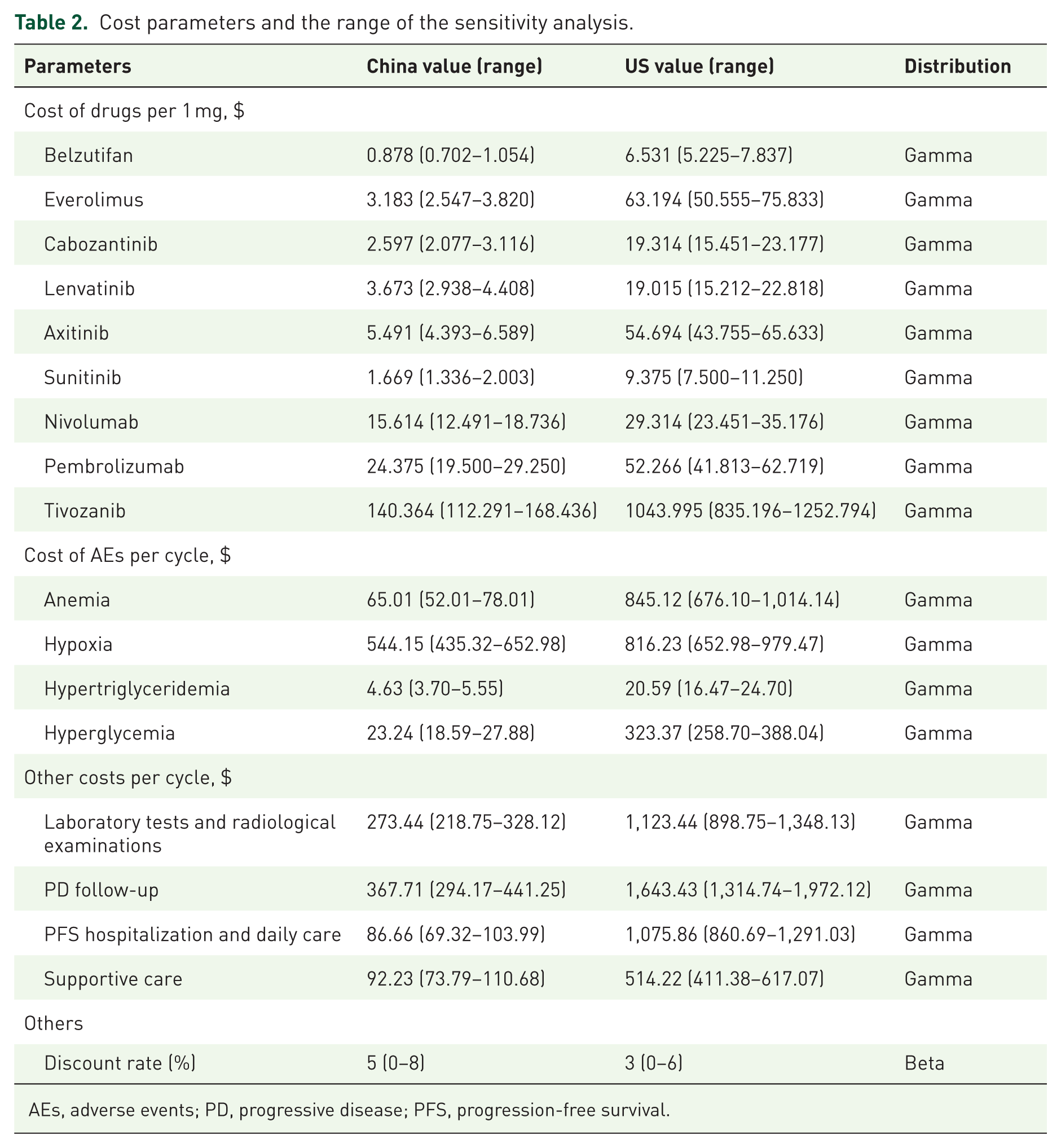
AEs, adverse events; PD, progressive disease; PFS, progression-free survival.
Price simulation
Although belzutifan has been approved for marketing by the FDA and NMPA, we did not have access to its price in China, so we estimated the price of belzutifan using a comparative approach in our base-case analysis. Since belzutifan was the first HIF-2α inhibitor approved for marketing, there were no drugs in its class for price comparison, so we selected six commonly used backline therapies for ccRCC as alternatives. Everolimus, lenvatinib, axitinib, sunitinib, nivolumab, and pembrolizumab are available in both China and the United States. We calculated a drug index from the prices of these drugs (Table S3). We obtained a price of $6.531 per mg for belzutifan, which was initially marketed in the United States, and converted the Chinese price of belzutifan through the drug index. Ultimately, we estimated the price of belzutifan to be $0.878 per mg.14,28 –34
Sensitivity analyses
To examine the robustness of the model, both deterministic sensitivity analysis (DSA) and probabilistic sensitivity analysis (PSA) were performed. Since standard deviations, standard errors, and 95% confidence intervals were not available for most parameters, we varied parameter values within a plausible range for DSA. Most parameters were varied by ±20% from their baseline values, while a wider range of ±30% was applied to parameters associated with higher uncertainty.17,18 The annual discount rate varied from 0% to 6% for the United States and from 0% to 8% for China. 18 The results were presented as a tornado diagram.
For the PSA, appropriate distributions were assigned to each input parameter: gamma distribution for cost parameters and beta distribution for utilities and probabilities. 35 In each simulation, all parameters were randomly sampled from their respective distributions. A total of 10,000 Monte Carlo simulations were performed to assess the uncertainty in model outputs across a range of plausible values. Results were displayed using a scatter plot and a cost-effectiveness acceptability curve (CEAC). Given the substantial uncertainty in the price of belzutifan in China, in the analysis, the price of belzutifan varied between $0.550/mg and $0.900/mg to explore its potential cost-effectiveness.
Scenario analysis
In this study, we simulated two scenarios as follows.
Scenario analysis 1: To improve the accessibility and affordability of novel anti-tumor therapies, China’s National Reimbursement Drug List (NRDL) has undergone dynamic updates to include such treatments. Although belzutifan is not currently listed in the NRDL, future price negotiations are anticipated. Therefore, ICERs were calculated for every 5% price reduction in belzutifan to explore the impact of price changes on cost-effectiveness.
Scenario analysis 2: Substantial economic disparities and differences in access to healthcare resources exist not only between China and the United States, but also within each country. In China, economic development and medical infrastructure vary considerably across provinces and municipalities; in the United States, similar disparities are observed among different states. These internal regional imbalances can influence both the affordability and accessibility of innovative therapies such as belzutifan. To better reflect real-world differences in economic context, we selected several representative provinces in China and states in the United States and applied three times their respective GDP per capita as the WTP thresholds. This approach allowed us to simulate the regional economic feasibility of belzutifan use and enhance the relevance and generalizability of our analysis.
Scenario analysis 3: To address the concern regarding structural uncertainty in survival modeling, we conducted a scenario analysis using the log-normal distribution to fit OS curves. Among all tested parametric models, the log-normal distribution yielded the highest AIC and BIC values, indicating the worst fit to the observed trial data. By replacing the base-case survival distribution with the log-normal function, we re-estimated total costs, QALYs, and the ICER.
Scenario analysis 4: To assess the impact of uncertainty in utility values on model outcomes, a scenario analysis was conducted using alternative utility values from the literature (PFS = 0.76; PD = 0.68). 22 After replacing the utility values in the base-case analysis with these alternatives, we recalculated the total costs, QALYs, and ICER.
Results
Base-case analysis results
The base-case results are shown in Table 3. The total cost of the belzutifan treatment group was higher than that of the everolimus treatment group in both the United States and China, with costs of $796,227.28 versus $682,483.47 in the United States and $102,561.26 versus $85,613.25 in China. In terms of health outcomes, the belzutifan treatment group also performed better in both the United States and China, with results of 2.280 QALYs versus 1.860 QALYs in the United States and 2.038 QALYs versus 1.726 QALYs in China. The incremental cost and QALY for the belzutifan treatment group in the United States were $113,743.81 and 0.420 QALYs, respectively, while in China, the incremental cost and QALY were $16,948.01 and 0.311 QALYs, respectively. As a result, the ICER for belzutifan in the United States was $270,864.46/QALY, which exceeded the WTP threshold of $150,000/QALY, and in China, the ICER was $54,430.16/QALY, exceeding the WTP threshold of $39,076.44/QALY.
Results of base-case analysis.

ICER, incremental cost-effectiveness ratio; QALY, quality-adjusted life year.
Price simulation results
The results of the price simulations are illustrated in Figure 2. In the United States, when the ICER reached the WTP thresholds of $100,000/QALY and $150,000/QALY, the corresponding belzutifan prices were $5.107/mg and $5.524/mg, respectively. This indicated that the belzutifan treatment group would be cost-effective when the drug price fell below these specific values. In China, where the WTP threshold was $39,076.44/QALY, belzutifan was cost-effective only when the price was below $0.779/mg.
Price simulations of belzutifan compared to everolimus in the United States (a) and China (b), respectively.
Sensitivity analyses results
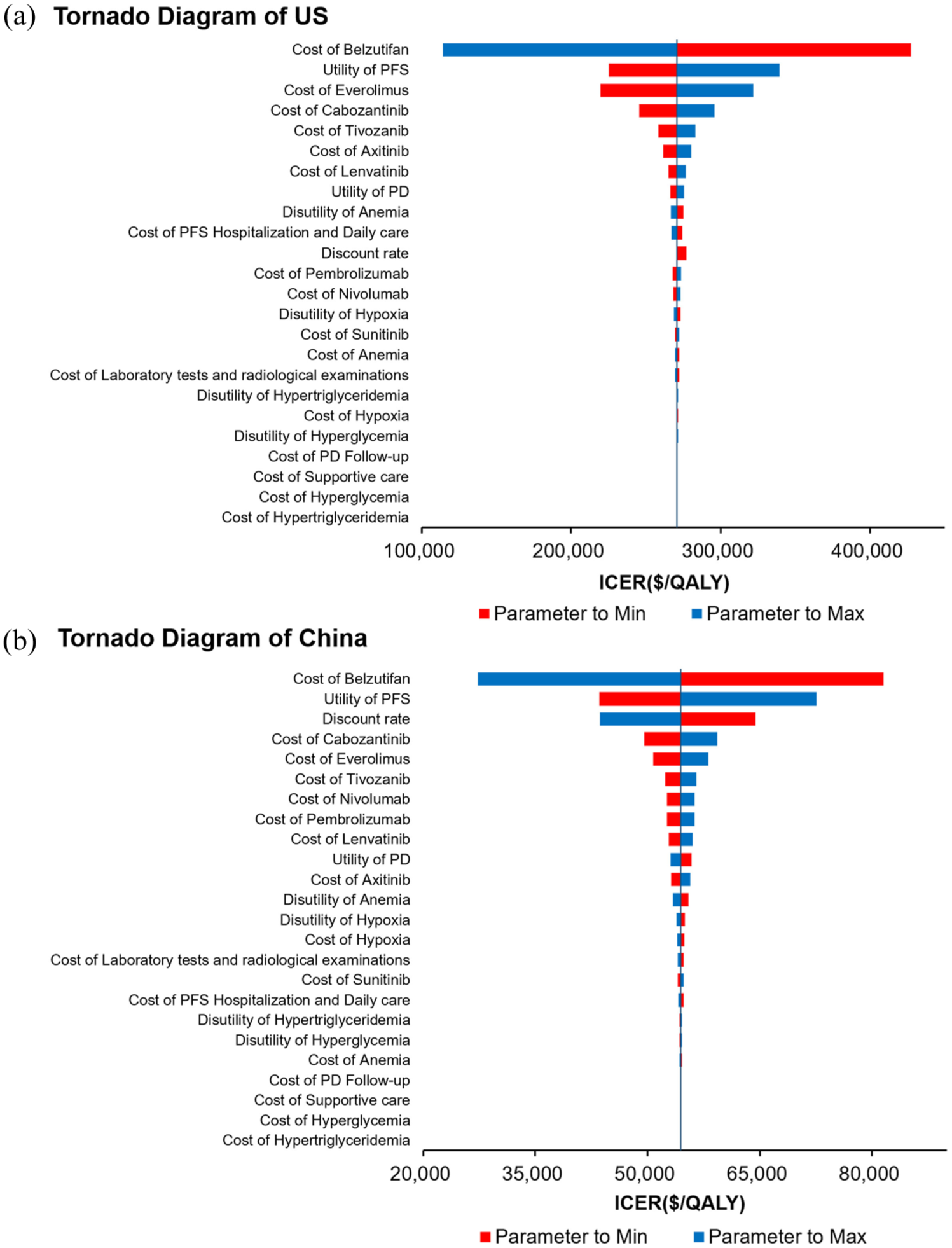
The DSA results are illustrated in the tornado diagram (Figure 3). In China, among all model inputs, the utility of the PFS state, the cost of belzutifan, and the discount rate had the most significant impact on the ICER. Other factors with moderate influence included the utility of the PD state, the cost of everolimus, and the cost of subsequent treatments. The cost of managing adverse events and supportive care had minimal impact (Figure 3(b)). In the United States, among all model inputs, the utility of the PFS state, the cost of belzutifan, and the cost of everolimus had the most significant impact on the ICER. Other factors with moderate influence included the utility of the PD state, the discount rate, and the cost of subsequent treatments. The cost of managing adverse events and supportive care had minimal impact (Figure 3(a)). Across all plausible ranges of these parameters, the ICER remained above the WTP threshold, confirming the robustness of the model.

The deterministic sensitivity analyses for belzutifan compared to everolimus in the United States (a) and China (b), respectively.
Results of the Monte Carlo simulations and CEAC are presented in Figure 4, Figure S7, Figure 5, Figure S8, Table 4, respectively. The PSA showed that at the US WTP threshold, belzutifan was not cost-effective (Figure 4a). The CEAC showed that the probability of cost-effectiveness of belzutifan began to rise only when the WTP threshold exceeded $204,000/QALY in the United States. At a WTP of $270,000/QALY, the probability of being cost-effective reached 50%, and at a WTP of $315,000/QALY, the probability of being cost-effective rose to 90% (Figure 5(a)).

The Monte Carlo sensitivity analyses for belzutifan compared to everolimus in the United States (a) and China (b), respectively.

The cost-effectiveness acceptability curves of belzutifan compared to everolimus in the United States (a) and China (b), respectively.
Summary of PSA results.

With a probability of 50% to be cost-effective.
PSA, probabilistic sensitivity analysis; QALY, quality-adjusted life year; WTP, willingness to pay ratio.
Similarly, in the United States, when belzutifan was priced at 0.878/mg, the PSA for China showed that all points were above the line of China’s triple GDP per capita ($39,076.44/QALY), which suggested that belzutifan was not cost-effective at China’s WTP threshold (Figure S7). The CEAC showed that the probability of belzutifan being cost-effective began to rise only when the WTP threshold exceeded $42,000/QALY in China. At a WTP of $54,000/QALY, the probability of being cost-effective reached 50%, and at a WTP of $65,000/QALY, the probability of being cost-effective rose to 90% (Figure S8). The scatter plot and CEAC curves corresponding to the price change of belzutifan from $0.700/mg to $0.900/mg are shown in Figures 4(b) and 5(b). As the price of the drug changes every $0.050/mg, all the points change from being all above the WTP line (at $0.900/mg) to being almost all at the WTP line ($0.700/mg). Additional details are specified in the figure. In addition, we also did a PSA for the case where the belzutifan price is a specific price when ICER was equal to the China and the US WTP thresholds, and the results are shown in Figure S9.
Scenario analysis results
Results of scenario analysis 1 are summarized in Figure S10. In this scenario, only the total cost of the belzutifan arm changed, while QALYs remained constant for both arms. The ICER decreased significantly as the price of belzutifan was reduced. A 10% price reduction led to an ICER of $40,889.93/QALY; and a 20% reduction resulted in $27,349.70/QALY, which was in the range of 1–3 times the GDP per capita, making belzutifan cost-effective. Subsequently, we conducted more precise calculations. We fitted the price reduction ratio of belzutifan with the ICER and found that the two were inversely proportional. When the price of belzutifan was reduced by 11.34%, that is, when the price was $0.779 per 1 mg, the ICER was equal to the WTP threshold.
In scenario analysis 2, the total costs and benefits of the two treatment strategies remained unchanged, with the only difference being the WTP threshold. The results of scenario analysis 2 are shown in Figure S11. There are significant differences in per capita GDP across cities. In China, we selected Shanghai, which has the highest per capita GDP ($28,194.13), followed by Beijing, which has the second highest GDP per capita ($26,839.17), and then Anhui Province ($21,191.67), which is slightly below the national average, and finally Gansu Province, which has the lowest per capita GDP ($6698.68). 36 Similarly, in the United States, we selected Massachusetts ($86,840.00) and Washington ($85,748.00) as the two states with the highest GDP per capita, as well as Oregon ($66,612.00), which represents a moderate level, and Mississippi ($48,048.00), which has the lowest per capita GDP. 37 According to the results, in Mississippi and Oregon, where the per capita GDPs are lower, the probability of belzutifan being cost-effective was 0%. However, in the higher GDP regions of Massachusetts and Washington, the probabilities of belzutifan being cost-effective were 37.48% and 32.9%, respectively. Similarly, in China, belzutifan had a 0% probability of being cost-effective in Gansu and Anhui, which have lower per capita GDPs, but in the higher GDP cities of Beijing and Shanghai, the probability was close to 100% at three times the per capita GDP.
The result of scenario analysis 3 is shown in Table S4. The total cost of the belzutifan treatment group was higher than that of the everolimus treatment group in both the United States and China, with costs of $803,181.52 versus $680,842.08 in the United States and $104,499.39 versus $85,513.03 in China. In terms of health outcomes, the belzutifan treatment group also performed better in both the United States and China, with results of 2.303 QALYs versus 1.855 QALYs in the United States and 2.081 QALYs versus 1.725 QALYs in China. Ultimately, we arrived at ICERs of $273,173.58/QALY and $53,288.26/QALY for the United States and China, respectively, which are similar to the base-case analysis results.
The result of scenario analysis 4 is shown in Table S5. The total cost of the belzutifan treatment group was higher than that of the everolimus treatment group in both the United States and China, with costs of $796,227.28 versus $682,483.47 in the United States and $102,561.26 versus $85,613.25 in China. In terms of health outcomes, the belzutifan treatment group also performed better in both the United States and China, with results of 2.300 QALYs versus 1.890 QALYs in the United States and 2.053 QALYs versus 1.753 QALYs in China. Ultimately, we arrived at ICERs of $277,362.76/QALY and $56,458.62/QALY for the United States and China, respectively, which are similar to the base-case analysis results.
Discussion
This study used phase III data from the LITESPARK-005 trial to evaluate the cost-effectiveness of belzutifan in patients with previously treated RCC. By constructing a partitioned survival model, we estimated total costs, QALYs, and the ICER for belzutifan compared with everolimus. The base-case analysis yielded an ICER of $54,430.16/QALY for belzutifan, which exceeded the WTP threshold of $39,076.44/QALY, indicating that belzutifan was not cost-effective under current pricing in China. Similarly, the ICER for belzutifan in the United States was $270,864.46/QALY, which exceeded the WTP threshold of $150,000/QALY, indicating that belzutifan was not cost-effective under current pricing in the United States. Sensitivity analyses, including both DSA and PSA, further confirmed the robustness of these findings. DSA revealed that the utility value of the PFS state and the drug cost of belzutifan were the most influential factors. PSA showed that the probability of belzutifan being cost-effective under the current threshold was 0% in the United States and China. The probability rose to 50% at a WTP of $54,000/QALY and 90% at $65,000/QALY in China. The probability rose to 50% when the WTP was $270,000/QALY and to 90% when the WTP was $315,000/QALY in the United States.
The approved first-line treatment options for RCC in China are sunitinib, axitinib combined with toripalimab, nivolumab combined with cabozantinib, and atezolizumab combined with bevacizumab.12 –15,38 In previous pharmacoeconomic studies, whether from the perspective of the Chinese healthcare system and the US payers, none of these four treatment options has been found to be cost-effective. For example, the ICERs for toripalimab plus axitinib versus sunitinib in the treatment of advanced RCC were $64,337.49/QALY and $923,962/QALY in China and the United States, respectively, both of which were higher than the ICER calculated in this study.14,39 Moreover, all of these drugs are either TKIs or PD-L1 monoclonal antibodies. Belzutifan, as an innovative small-molecule HIF-2α inhibitor, holds certain advantages. HIF-2α is a key factor in RCC, particularly in VHL gene-mutated RCC, where it plays a central role. 40 Small-molecule HIF-2α inhibitors inhibit tumor angiogenesis by reducing the expression of angiogenesis-related factors, such as VEGF, thereby limiting the formation of blood vessels that supply the tumor with oxygen and nutrients, which in turn slows tumor growth. Some RCC patients exhibit resistance to traditional targeted therapies (such as VEGF inhibitors) or immune therapies. As a new treatment strategy, HIF-2α inhibitors may improve resistance to existing therapies through different mechanisms of action, offering new treatment options. However, no cost-effectiveness analysis of belzutifan has been conducted, and our study fills this gap. Although belzutifan was not found to be cost-effective at its current price in either China or the United States, its clinical benefits remain notable. In the LITESPARK-005 trial, belzutifan demonstrated superior objective response rates, a more favorable safety profile, and improved PFS compared to everolimus. These health gains are particularly relevant for patients resistant to prior VEGF or ICI therapies. Belzutifan shows potential for greater economic efficiency if priced appropriately.
In the results of DSA, the high price of belzutifan significantly impacted ICER. Currently, belzutifan has just been approved for marketing in China but has not yet been included in the NRDL, and its potential for future price adjustments remains uncertain. For example, after NRDL negotiations in 2023, the price of axitinib was reduced from approximately $189.16 per 5 mg to $26.75 per 5 mg, and the out-of-pocket cost for patients dropped to just a few hundred RMB after insurance reimbursement. Considering belzutifan’s favorable efficacy, if successful negotiations with the National Healthcare Security Administration are achieved, its cost-effectiveness could be significantly improved. In 2024, the average price reduction of negotiated drugs reached 63%, and our scenario analysis suggests that when the price of belzutifan is reduced by more than 11.34%, this therapy will become an economical option. Our findings also offer valuable insights for drug pricing strategies in other countries. Given that China is one of the most populous countries in the world, the National Healthcare Security Administration has adopted a volume-based procurement model during drug price negotiations, leading to substantial reductions in drug prices. Consequently, drug prices in China are generally lower than in many other countries. Therefore, other countries may consider using this scenario analysis to inform potential price adjustments.
Our study followed the China Guidelines for Pharmacoeconomic Evaluations, which recommend a WTP threshold of 1–3 times per capita GDP.41 –43 However, novel anti-tumor agents tend to be priced significantly higher than conventional drugs. Moreover, as cancer is a life-threatening condition, patients are often more willing to pay for life-extending treatments. Therefore, some researchers have proposed higher thresholds for evaluating cancer therapies. For instance, in the United States, the WTP threshold is typically $50,000–$100,000/QALY for conventional drugs but $100,000–$150,000/QALY for novel anti-tumor agents. 44 As shown in Figure S12, when the WTP threshold in China is adjusted to the US WTP threshold of $100,000/QALY, the probability of belzutifan being cost-effective is 99.9% infinitely closer to 100%.
Additionally, given the possible economic disparities and differences in access to healthcare resources between regions in China and the United States, we selected representative cities and states in both countries to explore changes in belzutifan economics using three times their GDP per capita as the WTP threshold. The results of scenario analysis 2 revealed that the affordability of belzutifan varies significantly across regions, and is mainly influenced by the regional economic level, that is, GDP per capita. In regions with lower GDP per capita, such as Gansu and Anhui provinces in China, and Mississippi and Oregon in the United States, patients’ WTP was lower. In contrast, in cities with higher GDP per capita, such as Shanghai and Beijing, and in Massachusetts and Washington in the United States, where patients’ ability to pay was higher, the cost-effectiveness of belzutifan performed more favorably, especially in the triple GDP per capita scenario. The probability of cost-effectiveness is close to 100%. This analysis reflects the significant variation in patients’ payment ability and WTP for medications in different economic regions, which in turn affects the cost-effectiveness evaluation of drugs. In higher GDP areas, patients are more able to afford higher drug costs, and even innovative medications can be accepted due to their health benefits and treatment outcomes. However, in lower GDP regions, patients’ WTP is constrained, and despite the drug’s significant therapeutic effects, its high price may prevent it from being considered cost-effective. Therefore, pharmaceutical companies and policymakers need to adopt flexible pricing and promotional strategies based on the economic conditions of different regions. In low-GDP areas, it may be necessary to reduce drug prices, implement donation strategies, or offer additional discounts to improve drug acceptance. In higher-GDP areas, more flexible pricing strategies can be employed to ensure cost-effectiveness and encourage widespread use of the drug.
For newly launched innovative drugs in China, it is common to implement a drug donation strategy to promote market penetration and improve patient access to medications. For example, levatinib uses a “(2+2)+(2+X)” donation strategy, whereby a 2-month purchase of the drug results in a 2-month free supply, followed by a 2-month purchase of the paid-for drug and an additional X months of free supply, where X can be up to 10 months. This donation strategy helps reduce the financial burden on patients in the initial stages of use, thereby promoting drug adoption and accessibility. Based on a similar strategy, if belzutifan adopts a comparable discount policy, its price would be significantly reduced, with a price reduction ranging from at least 42.8% to a maximum of 75%. According to the results of scenario analysis 1, this donation strategy indicates that, whether the price is reduced by 42.8% or 75%, belzutifan would be cost-effective in China, enhancing the drug’s competitiveness. From an economic perspective, regardless of whether the price reduction is 42.8% or 75%, belzutifan’s treatment outcomes and cost-effectiveness ratio remain within a reasonable range, ensuring good economic value. Furthermore, the donation strategy effectively reduces the direct cost of medication to patients, thereby increasing patient acceptance of Bezutivan and facilitating its widespread clinical use.
This study has several limitations. First, it used data from the LITESPARK-005 trial to simulate real-world patient characteristics and clinical pathways. The trial had a relatively small sample size (746 patients), not all of whom were from Asian populations. Second, long-term survival data had to be extrapolated due to limited follow-up. Although we used statistical methods to reconstruct individual survival data from Kaplan–Meier curves, the extrapolated long-term outcomes may deviate from real-world results. Third, to simplify the model, only grade ⩾3 TEAEs with an incidence ⩾5% were included, which may underestimate costs. However, DSA showed that adverse event costs had minimal impact on the model outcome. Fourth, indirect costs were not included, and cost data came from multiple sources, which may introduce variability in the analysis. Lastly, the absence of real-world data on belzutifan, especially in the Chinese healthcare setting, limits the external validation of our model. Differences between clinical trial settings and routine clinical practice—such as treatment adherence, dosing modifications, and management of toxicities—may influence both costs and outcomes. Future studies incorporating real-world evidence will be important to enhance the robustness, accuracy, and policy relevance of cost-effectiveness assessments.
Conclusion
In conclusion, from the perspective of the Chinese healthcare system and the US payers, belzutifan is not cost-effective for the treatment of previously treated patients with RCC under current drug pricing with appropriate WTP thresholds, despite the prolonged PFS. Especially in China, where belzutifan is not yet priced. Our modeling results have the potential to guide clinical treatment decisions for patients with ccRCC or to develop appropriate pricing and healthcare reimbursement policies for these drugs.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251379708 – Supplemental material for Cost-effectiveness analysis of belzutifan versus everolimus for previously treated advanced clear cell renal cell carcinoma in the United States and China
Supplemental material, sj-docx-1-tam-10.1177_17588359251379708 for Cost-effectiveness analysis of belzutifan versus everolimus for previously treated advanced clear cell renal cell carcinoma in the United States and China by Baolong Ding, Hongting Yao, Tiantian Tao, Yuyang Sun, Yulu Zhu, Haomin Zhu, Jia Wang, Zhuying Jing, Lihong Gao, Yingtao Lin and Xin Li in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
