Abstract
Background:
Immune checkpoint inhibitors (ICIs) have changed the treatment landscape for non-small cell lung cancer, but the role of race/ethnicity is not well understood.
Design:
This is a retrospective study of non-small cell lung cancer (NSCLC) patients receiving ICI.
Objective:
We evaluated the role of race/ethnicity in ICI response and immune-related adverse events (irAEs) in NSCLC patients.
Methods:
NSCLC patients treated with ICIs from 2014 to 2022 at Los Angeles General Medical Center and Norris Comprehensive Cancer Center were included. Primary endpoints were irAE incidence, time on ICI treatment (TOT), overall survival (OS), and progression-free survival (PFS). TOT, OS, and PFS were evaluated using the Kaplan–Meier method. Landmark analysis was performed of patients receiving >6 month ICIs. Fisher’s exact test was performed for analysis of variables between groups.
Results:
In total, 211 NSCLC patients receiving ICIs were analyzed, including 86 (40.8%) Asian American/Pacific Islander (AAPI), 65 (30.8%) non-Hispanic White/Caucasian (NHW), 37 (17.5%) Hispanic/Latino (HIS), and 23 (10.9%) African American/Black (AA). Among stage IV patients, median OS for AAPI was 23.2 months (95% confidence interval (CI) 16.0–not reached (NR)), 23.6 months (95% CI 6.9–38.8) for NHW, 12.7 months for AA (95% CI 2.2–37.0), and 11.0 months (95% CI 4.8–14.8) for HIS (
Conclusion:
Our study demonstrates that AAPIs and NHW had higher irAE incidence and longer OS. Validation studies evaluating the role of race/ethnicity in ICI response and toxicity are merited.
Plain language summary
Immunotherapy that can help re-signal the immune system to fight tumor cells and has contributed to improving outcomes in non-small cell lung cancer but Hispanics and African Americans have been severely underrepresented in clinical trials making up 2-7% of key lung cancer trials. However, immunotherapy can cause significant side effects such as inflammation of the colon, lung, liver, or thyroid. In our study of NSCLC patients receiving immunotherapy, we found that Asian Pacific Islanders (23.2 months) and Non-Hispanic Whites (23.6 months) had longer median overall survival than 12.7 months in African Americans and 11.0 months in Hispanic patients. Asian Pacific Islander (48.3%) had a higher prevalence of immunotherapy side effects versus 39.4% in Non-Hispanic Whites, 27.3% in African Americans, and 27.0% in Hispanics. Among patients with immunotherapy side effects, Asian Pacific Islanders had a longer median survival of 60.6 months, compared to Non-Hispanic Whites at 48.4 months, African Americans at 35.7 months, and Hispanics at 15.2 months. No significant survival difference was seen in those who did not have immunotherapy side effects. Factoring in other variables, mortality in Hispanics compared to Asian Pacific Islanders was a significant variable towards overall survival. Thus, our study showed that Asian Pacific Islanders had longer median overall survival while having a higher prevalence of immunotherapy side effects while Hispanics and African Americans had a shorter median overall survival while having a lower prevalence of immunotherapy side effects. These findings demonstrate the importance of studying the environment of immune cells in lung cancer factoring in race/ethnicity and to have clinical trials on immunotherapy drugs with greater representation of minorities.
Introduction
Lung cancer remains the leading cause of cancer death among men and women in the United States. In 2022, an estimated 130,180 deaths in the United States were attributed to lung cancer. 1 However, there has been significant improvement in recent years, with annual decreases in mortality of 2.4% between 2005 and 2014 and 4.9% from 2014 to 2019. 1 This drop in mortality has been attributed to the advancement of systemic therapy options, including immune checkpoint inhibitors (ICIs) and targeted therapies. 2 Non-small cell lung cancer (NSCLC) consists of 85% of the cases with adenocarcinoma histology being the common histology, while small cell lung cancer (SCLC) consists of about 10%–15% of lung cancer cases. Adenocarcinoma is also the most prevalent subtype in women and never-smokers. 3 Global trends of incidence and mortality of lung cancer have correlated closely with smoking incidence, as there has been a large decrease in developed countries secondary to decreases in smoking prevalence, while low-middle-income countries, in general, have had lower rates of incidence and mortality. In the United States, there have been decreases in lung cancer in both men and women correlating with decreases in smoking, but the decrease has been substantially more in men. 4
ICIs block immune-inhibitory checkpoint signals (programmed cell death 1 (PD-1), programmed cell death ligand 1 (PD-L1), and cytotoxic T-lymphocyte-associated protein (CTLA-4)) expressed on malignant cells as well as lymphocytes and other hematopoietic cells, thus activating the immune system against malignancy. 5 In the treatment of both NSCLC and SCLC, ICIs have led to improved clinical outcomes for patients, both as monotherapy and in combination with other therapies.6–13 However, due to the break on immune-tolerance, the use of ICIs has been associated with the risk of immune-related adverse events (irAEs) in various organs. Rates of irAE have varied with low-grade toxicity ranging from 20% to 50% and severe-grade 3–4 reactions occurring in 10%–70% of patients, depending on the regimen used, type of cancer, and target organ.6,7,9,14–17 Common irAEs include dermatologic events, which are the most common and include rashes, pruritic, and vitiligo, gastrointestinal including diarrhea, colitis, enterocolitis, endocrine including thyroid dysfunction, hepatotoxicity including hepatitis, pulmonary including pneumonitis, and musculoskeletal including arthritis. The severity and treatment of irAEs varies as minor irAEs can be treated outpatient with hydrocortisone topical for rash or levothyroxine for acquired hypothyroidism, while more serious irAEs require hospitalization and can be life-threatening requiring high-dose steroids and biologics such as mycophenolate mofetil and infliximab. 18
Compellingly, multiple studies have shown that the development of irAE may be associated with improved overall survival (OS) and progression-free survival (PFS) in NSCLC patients receiving ICIs; it has been theorized that the development of irAE may indicate a more robust immune response to treatment and greater therapeutic effect.19–22 Although controversial, this association has been shown for other tumor types, various types of irAEs, and regimens.23–26 Despite this, there is still limited understanding of how ICIs and irAE affect diverse populations, including those of varying ethnic and racial backgrounds; a recent study showed that only 3.4% Hispanic/Latinos and 3.4% African Americans were enrolled in NSCLC clinical trials from 2010 to 2019.
27
Growing evidence suggests that the efficacy of ICIs along with the frequency of irAE among NSCLC patients differs across racial/ethnic groups.28–30 It is, therefore, important to ensure that conclusions drawn regarding the efficacy and safety profile of ICIs and how race/ethnicity could impact both efficacy and safety. Furthermore, there are incidence and mortality differences in lung cancer by race/ethnicity. In the United States, data indicate that non-Hispanic Whites and Blacks have the highest incidence and mortality rates of lung cancer, which has been thought to reflect smoking patterns, but what is unclear is how this relates to treatment response. In addition, there are genomic differences in NSCLC by race/ethnicity as we have seen that Hispanics at our institution have greater prevalence and
Materials and methods
Data selection
A multicenter, retrospective analysis of patients diagnosed with NSCLC treated with ICI therapy from 2014 to 2022 at the Los Angeles General Medical Center (LAG) and Norris Comprehensive Cancer Center (Norris) was conducted. This study was approved by the Institutional Review Board (IRB) of the University of Southern California (USC), which covers the study for both LAG and Norris. Because of the retrospective nature of the study, the study received exempt status from the IRB for HS-19-00304 Examining Outcomes to Immunotherapy at USC. The reporting of the results adheres to the STROBE guidelines. 32
Inclusion criteria
Eligible patients for analysis included any patient who received at least one dose of ICI, as monotherapy or combination therapy, at any point in treatment course, and for whom at least one follow-up visit was available within the medical records.
Variables/data sources
Clinical data, including patient demographics (age, gender, race/ethnicity), cancer-related variables (including stage, histology subtype, driver mutation analysis, and PD-L1 expression), cancer treatment history, time of death and last follow-up, and irAEs, were collected from the electronic health record. irAEs were graded as per CTCAE version 5.0 and were evaluated by the authors from medical documentation from clinic notes.
Study size
From 2014 to 2022, a total of 1584 cancer patients were identified who had been treated with an ICI. Of those 1584, 226 patients with a diagnosis of NSCLC were identified and 211 with reported race/ethnicity were included for analysis. A total of 172 stage IV NSCLC patients were further included in survival analysis.
Statistical analysis
Statistical analysis was conducted with Prism Graphpad 10. (Graphpad Software, Inc., San Diego, California, USA) Primary endpoints included time on ICI treatment (TOT) and OS. Patients with stage III disease were excluded from survival analysis. TOT and OS were plotted with Kaplan–Meier curves and comparisons performed with log-rank testing. To minimize immortal time bias, we also performed survival analysis in which we excluded all patients who discontinued ICI prior to use of ICI for 6 months. Chi-square analysis and Fisher’s exact test were performed for analysis of demographic and cancer variables between ethnic groups. Hazard ratios were calculated using Cox multivariate regression. A
Data availability statement
The data generated in this study are available upon reasonable request from the corresponding author.
Results
Patient characteristics
From 2014 to 2022, a total of 1585 cancer patients were identified who had been treated with an ICI. Of those 1585, 226 patients with a diagnosis of NSCLC were identified. Of the 226 patients, 86 (38.1%) were identified as Asian American/Pacific Islander (AAPI), 65 (28.8%) non-Hispanic White/Caucasian (NHW), 37 (16.4%) Hispanic/Latino (HIS), 23 (10.2%) African American/Black (AA), and 15 patients (6.6%) were identified as “other/unknown” (Figure 1). The “other/unknown” group of 15 patients was subsequently excluded from all between race comparisons, and thus, 211 patients were included in analysis.

CONSORT diagram of patients who received ICIs.
Descriptive information for the included 211 patients with a specific racial/ethnic group designation is displayed in Table 1 and CONSORT diagram in Figure 1. In total, 128 (60.7%) patients were male with an average age of 65.7 years and average BMI 24.5. Thirty-nine patients (18.5%) were diabetic, and 66 patients (31.3%) reported never smoking history.
Demographic and tumor information.
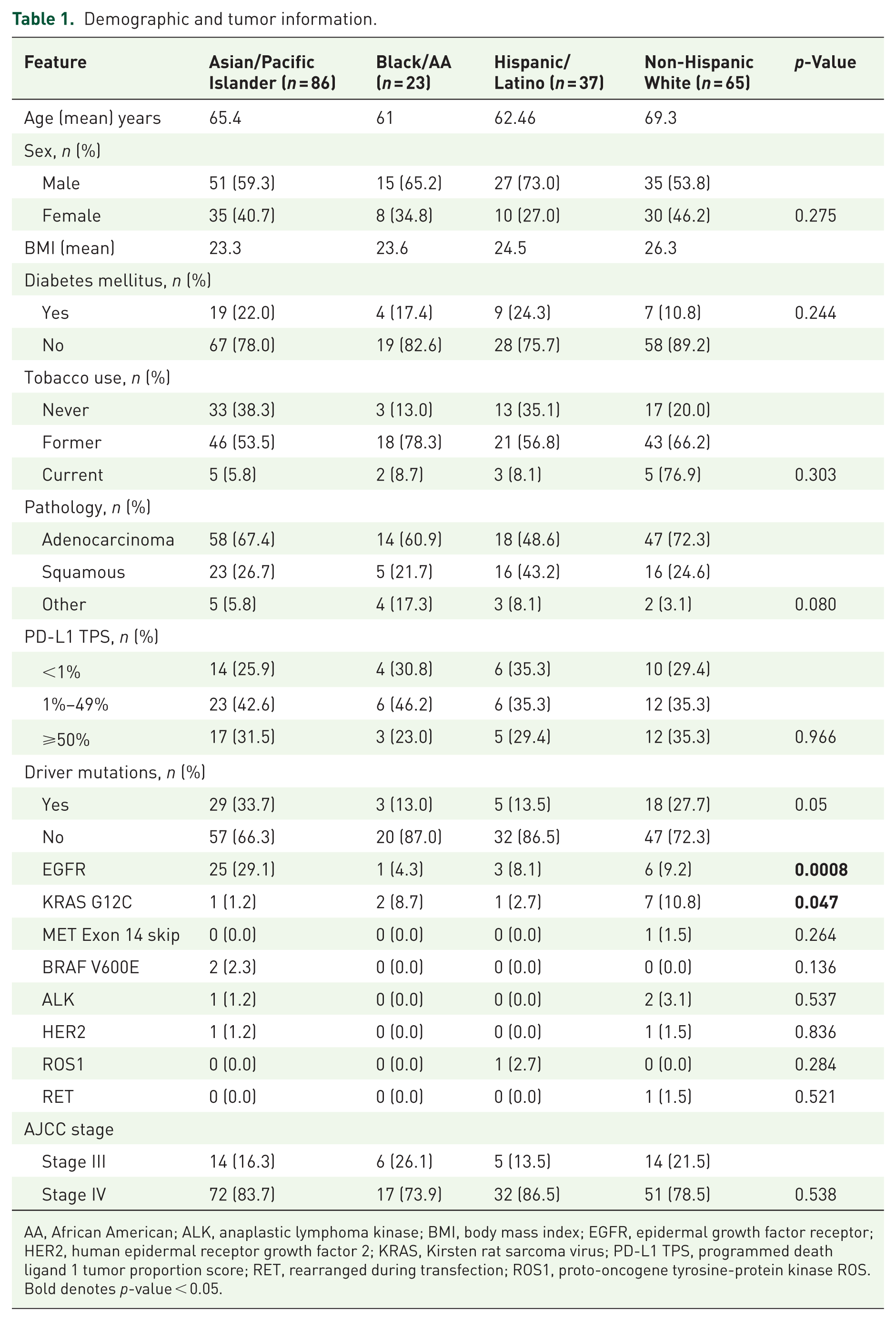
AA, African American; ALK, anaplastic lymphoma kinase; BMI, body mass index; EGFR, epidermal growth factor receptor; HER2, human epidermal receptor growth factor 2; KRAS, Kirsten rat sarcoma virus; PD-L1 TPS, programmed death ligand 1 tumor proportion score; RET, rearranged during transfection; ROS1, proto-oncogene tyrosine-protein kinase ROS. Bold denotes
Tumor characteristics
Tumor characteristics are displayed in Table 1. Per AJCC 8th edition staging system, most (
Treatment outcomes
Most patients received ICI monotherapy (
Treatment and irAE.

AA, Black/African American; ALK, anaplastic lymphoma kinase; CTLA4, cytotoxic T-lymphocyte-associated protein; EGFR, epidermal growth factor receptor; irAE, immune-related adverse event; PD-1/PD-L1, programmed death-1/programmed death ligand 1; TKIs, tyrosine kinase inhibitors. Bold denotes

Survival curves for all patients (

Survival curves by race/ethnicity (AAPI
Immune-related adverse events
Among all patients, (
For the entire cohort, patients who experienced irAE had significantly improved TOT (7.3 months (95% CI 3.8–9.1) vs 4.0 months (95% CI 2.3–5.6),

Survival curves for all patients stratified by irAEs (irAE

Survival curves stratified by irAE by race/ethnicity (AAPI
Among patients who did not experience irAE by race/ethnicity, median TOT was 2.4 months for AAPI (95% CI 1.4–7.4), 4.0 months for NHW (95% CI 1.9–6.3), 5.3 months for AA (95% CI 1.4–9.4), and 4.4 months (95% CI 1.9–7.2) for HIS (

Survival curves for patients without irAE by race/ethnicity (AAPI
Multivariate Cox regression for OS in stage IV patients showed that when considering race/ethnicity, age at diagnosis, sex, smoking history, family history of cancer when compared to AAPI, Hispanic ethnicity had worse OS (hazard ratio (HR) 2.27 (95% CI 1.30–3.94,
Multivariate Cox regression analysis for OS in Stage IV patients.
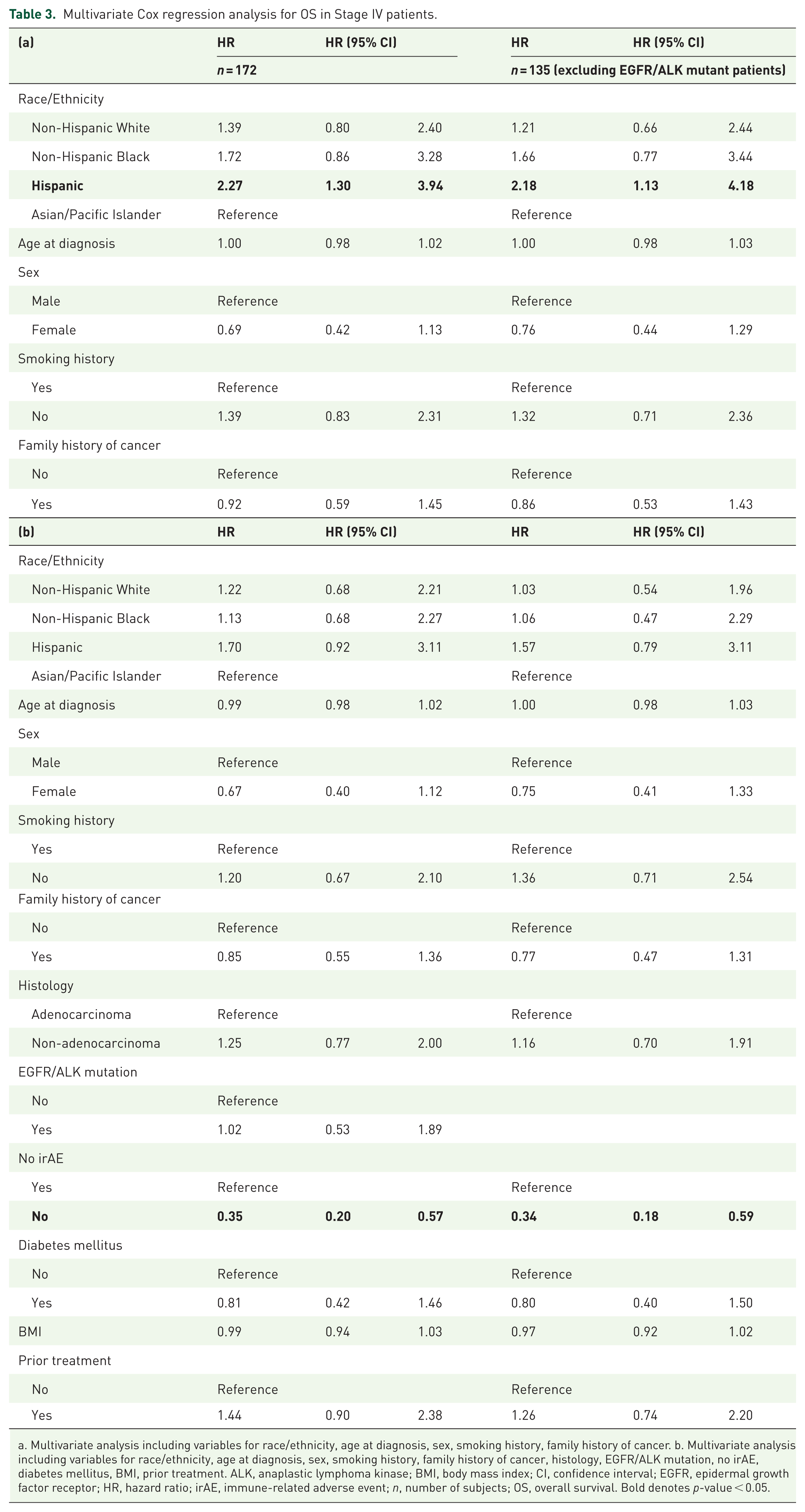
a. Multivariate analysis including variables for race/ethnicity, age at diagnosis, sex, smoking history, family history of cancer. b. Multivariate analysis including variables for race/ethnicity, age at diagnosis, sex, smoking history, family history of cancer, histology, EGFR/ALK mutation, no irAE, diabetes mellitus, BMI, prior treatment. ALK, anaplastic lymphoma kinase; BMI, body mass index; CI, confidence interval; EGFR, epidermal growth factor receptor; HR, hazard ratio; irAE, immune-related adverse event;
Landmark analysis
Excluding patients who had received ICI for less than 6 months, median TOT was 25.5 months for AAPI (95% CI 12.1–NR), 15.7 months for NHW (95% CI 7.9–31.8), 9.1 months for AA (95% CI 6.3–NR), and 11.3 months for HIS (95% CI 7.2–15.0;

Survival curves for patients who were on ICI greater than 6 months by Race/Ethnicity (AAPI
Survival based on line of systemic therapy and type of systemic therapy
In evaluating OS based on line of therapy, there was superior median OS in AAPI patients (median NR) receiving ICI as first-line systemic therapy versus NHW (38.0 months, 95% CI 6.9–NR), AA (8.2 months, 95% CI 1.9–NR), and HIS (11.7 months, 95% CI 0.7–14.8;
Discussion
Our study demonstrates the potential important role of irAEs in both incidence and survival in race/ethnicities. We saw greater prevalence of irAEs in AAPI and NHW compared to AA and HIS. We then saw a superior OS in AAPI and NHW as well, but this pattern was specifically seen in median OS in patients who experienced irAEs. Landmark analysis also showed that AAPI who had irAEs having longer median OS than other race/ethnicities. These results suggest that race/ethnicity could play a role in immune response efficacy with ICI particularly in those who experience irAEs. When comparing to other studies, there have been mixed results as shown in Table 4 in evaluating differences in survival by race/ethnicity.30,33–39 AAPIs have consistently demonstrated to have longer median OS when compared to other race/ethnicities across many of the studies.30,33,34,36 In our diverse cohort, we did not find improved OS when comparing AAPI with NHW. Other studies such as Qian et al. 34 have shown a persistent difference in OS even when adjusting for other variables such as smoking, driver mutations, PD-L1 expression, and TMB. 34 While there has been more consistent data comparing AAPI and NHW, the data evaluating AA are mixed (Table 4). There have been studies by Ayres et al. 36 and Gupta et al. 39 where AA had improved outcomes with ICI compared to NHW, while other studies such as Khozin et al. 33 and Olateju et al. 30 did not show any difference in survival. In our study, we showed that AA had worse OS than NHW. We also found worse OS in Hispanics, which is in line with another study led by Raez et al. showing that among patients with locally advanced NSCLC receiving durvalumab after chemoradiation, Hispanics had inferior OS and PFS versus NHW. However, our findings contradict another study led by Saravia et al., 37 which demonstrated no difference in median OS and PFS in between Hispanics and NHW among advanced NSCLC patients receiving ICI. 35 What stands out specifically in our study is the differentiation of race/ethnicity outcomes in those who experience irAEs.
Comparative studies on survival in NSCLC patients receiving ICI by race/ethnicity.

AA, Black/African American; AAPI, Asian Pacific Islander; CI, confidence interval; HR, hazard ratio; ICI, immune checkpoint inhibitors;
irAEs are increasingly recognized as an important consequence of immunotherapy. We observed a correlation between the incidence of irAEs and survival. Intriguingly, we found fewer incidence of irAEs among Hispanics and AAs and worse survival compared to AAPI and NHW. But more notably, our results showed a widening survival difference between race/ethnicity groups when irAEs were accounted for. Current literature regarding the rate of irAE between racial/ethnic groups is mixed. At least two other studies have also reported that irAEs are more frequent among NHW compared to AA patients, while a study by Florez et al. 28 looking at NSCLC and head and neck cancer patients also reported lower rates of irAE among HIS as well. 40 At the same time, other studies have shown no differences in the occurrence of irAE between NHW and AA patients.41,42 In a single-center study of 293 patients with solid tumors (48% NHW, 42% AA) undergoing PD-1/PD-L1 therapy, there was no difference in OS between races when comparing all patients; however, when comparing patients who experienced irAE, there was noted to be improved median OS among NHW patients with irAE compared to AA with irAE. 40 That being said, our study highlights the increased need for preclinical and prospective work studying race/ethnicity in the immune tumor microenvironment and the effect that ICI has on this immune tumor microenvironment in each race/ethnicity particularly when irAEs occur.
In our study, differences in outcomes by race/ethnicity became apparent among patients who experienced irAE. When evaluating those without irAE, no significant differences in outcomes were seen. This suggests that race/ethnicity may influence the impact of irAE. However, it does not imply that irAE alone explains differences in outcomes between different racial/ethnic groups. These mixed but significantly different outcomes ultimately speak to a multifactorial list of issues that need to be addressed: (1) a better understanding of the biology behind the immune microenvironment by different race/ethnicities, (2) socioeconomic factors that have a role in survival, and (3) the heterogeneity within each race/ethnicity. With regard to immune tumor microenvironment, there is mounting evidence of race/ethnicity difference in tumor microenvironment. Xu et al.
43
compared immune cells in NHW and AA NSCLC patients and showed that NHW had greater cell density of CD4+, CD8+, CD45+ T cells, which helps boost immune response. Another study comparing AA and NHW showed that AA had fewer CD8+ T cells but increased γδ T cells and resting dendritic cells.
44
Meanwhile, there were differences in miRNA expression and M1 and M2 macrophage expression in NHW and AA.
45
We have seen that in other solid tumors such as breast cancer, AAs have greater numbers of immunosuppressive tumor-associated macrophages such as CD163+ and CD206+ cells compared to NHW.
46
Another aspect that should be considered when evaluating immune response is Human Leukocyte Antigens (HLA) type. Studies have shown that specific HLA types such as HLA-C could portend worse outcomes to ICI.
47
In addition, specific HLA subtypes may be more prone to irAEs in specific organs; one study on melanoma patients showed that HLA-DR 7 of 10 patients (70%) with type 1 diabetes had DR4; 6 of 12 (50%) with hypothyroidism had DR8; 5 of 8 (63%) with hypophysitis had DR15; 3 of 5 (60%) with pneumonitis had DR1; and 8 of 15 (53%) with hepatitis had DR4.
48
HLA-A*0201 is a frequently studied HLA allele and has been shown to be more frequent in North America and less frequent in Africa and Asia, and in another study, NHW were found to be 1.6 times more likely than AAs to be eligible for these trials.49,50 Finally, it should be noted that 9 of 14 AAPI patients who took an
Regarding patient-related factors, there has been evidence to show that when these factors are accounted for, survival outcomes are more equal. For example, in Gupta et al., in models adjusted for socioeconomic factors factoring in type of facility where patients are receiving treatment (academic vs community), insurance, income, and percent without high school degree by zip code, AAs who actually received ICIs had improved survival compared to NHW.32,46–49 Finally, race/ethnicities are quite heterogeneous. For example, previous studies looking at immunotherapy and irAE outcomes by race/ethnicity involving Hispanics have been predominantly in different locations within the United States. For example, our study in Los Angeles likely consists primarily of those with Mexican heritage while in Miami, Cubans and Puerto Ricans make up the largest Hispanic subgroups.56,57 Similarly, within AAPIs, Chinese make up the largest subgroup in California, but Indians make up the largest subgroup in Texas and Florida. 58 The variability of ethnic background within an aggregated race/ethnicity shows the need for more real-world studies and for more diverse clinical trials that will better reflect the population at large.
Limitations of this study notably include the possibility of sample size particularly when evaluating subgroup data and our study being held at a single institution and the retrospective nature of the study. Many patient outcomes between racial/ethnic groups, such as socioeconomic status, access to care, degree of acculturation, and behavioral risk factors, were self-reported, and availability of some of this information was limited from electronic medical record review. In addition, given that this is an observational retrospective study, there is risk for immortal time bias though our landmark analysis helped demonstrate similar patterns in median OS and showed a trend toward differences in TOT by race/ethnicity among patients who experienced irAEs. Furthermore, it is important to note that this is an observational study, and our findings suggest a need for prospective and preclinical validation. Nevertheless, the strengths of this study include its diverse patient population that includes Hispanic and AAPI subgroups that are not well studied in the literature and the fact that we are looking at both survival and irAEs within the same patient population. Our multivariate Cox regression analysis showing a significant survival difference between AAPI and HIS and irAE incidence as significant variables in OS helps bolster our findings.
Conclusion
In conclusion, we report that survival outcomes to ICI among NSCLC patients may be impacted by the presence of irAE, and when present, race/ethnicity may further influence these differences. Further study into the real-world events of ICI in larger populations is warranted along with translational studies looking into the tumor microenvironment in NSCLC by race/ethnicity. We also hope that this study will bring more emphasis on the absolute need to improve diversity in clinical trials involving immunotherapy to better understand the role of race/ethnicity in both treatment response and toxicities.
Supplemental Material
sj-docx-1-tam-10.1177_17588359261423890 – Supplemental material for Impact of race/ethnicity and the presence of immune-related adverse events on outcomes for non-small cell lung cancer patients treated with immune checkpoint inhibitors
Supplemental material, sj-docx-1-tam-10.1177_17588359261423890 for Impact of race/ethnicity and the presence of immune-related adverse events on outcomes for non-small cell lung cancer patients treated with immune checkpoint inhibitors by Robert Hsu, Karen Resnick, Peter Zang, Travis Larsen, Shirley Ye, April Choi, Shawn Yu, Kevin Brady, Trevor E. Angell, Lily Dara, Fumito Ito, Jorge J. Nieva and Gino K. In in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359261423890 – Supplemental material for Impact of race/ethnicity and the presence of immune-related adverse events on outcomes for non-small cell lung cancer patients treated with immune checkpoint inhibitors
Supplemental material, sj-docx-2-tam-10.1177_17588359261423890 for Impact of race/ethnicity and the presence of immune-related adverse events on outcomes for non-small cell lung cancer patients treated with immune checkpoint inhibitors by Robert Hsu, Karen Resnick, Peter Zang, Travis Larsen, Shirley Ye, April Choi, Shawn Yu, Kevin Brady, Trevor E. Angell, Lily Dara, Fumito Ito, Jorge J. Nieva and Gino K. In in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
