Abstract
Background:
Concurrent chemoradiotherapy (cCRT) is essential for stage III unresectable non-small cell lung cancer (NSCLC) but increases the risk of radiation pneumonitis (RP), especially with immunotherapy (IO). Induction chemotherapy (ICT) controls tumor progression but damages lung tissue, raising RP risk. Research on the RP risk from ICT, cCRT, and IO consolidation is poorly investigated.
Objectives:
We evaluated whether ICT increases RP incidence and affects survival in the IO era.
Design:
This was a retrospective analysis conducted at a single cancer center.
Methods:
Retrospective data from stage III unresectable NSCLC patients treated with cCRT and IO between 2018 and 2024 were analyzed. Patients were divided into cCRT without ICT (N = 55) and ICT before cCRT groups (N = 103). The cumulative incidence of RP was evaluated using competing risk analysis (Gray’s test and Fine-Gray models), with death as a competing risk. Multivariable Cox regression was used to analyze overall survival, progression-free survival, and RP risk among ICT regimen subgroups.
Results:
The ICT before cCRT group showed a higher incidence of ⩾Grade 2 RP compared to the cCRT without ICT group (Gray’s test, SHR = 1.964, 95% confidence interval (CI): 1.137–3.394, p = 0.013), with no survival benefit. Subgroup analysis based on ICT regimens, compared with the Etoposide + Platinum regimen, Pemetrexed + Platinum regimen (hazard ratio (HR): 0.277, 95% CI: 0.090–0.854, p = 0.025), Paclitaxel + Platinum regimen (HR: 0.294, 95% CI: 0.094–0.926, p = 0.037), and Docetaxel + Platinum regimen (HR: 0.059, 95% CI: 0.010–0.356, p = 0.002) were associated with a lower incidence of ⩾Grade 2 RP.
Conclusion:
ICT followed by cCRT showed a higher incidence of ⩾Grade 2 RP compared to cCRT alone in patients with unresectable stage III NSCLC receiving consolidation immunotherapy.
Keywords
Introduction
Lung cancer remains the primary cause of both cancer incidence and mortality worldwide,1,2 with non-small cell lung cancer (NSCLC) as the predominant subtype, representing 85%–90% of all cases.2,3 Approximately 30% of NSCLC patients are diagnosed at stage III, often missing the critical window for surgical intervention. The 2024 National Comprehensive Cancer Network and American Society of Clinical Oncology guidelines recommend that unresectable stage III NSCLC patients with a performance status (PS) score of 0–1 receive consolidation immunotherapy (IO) following concurrent chemoradiotherapy (cCRT).4,5 Despite the PACIFIC trial’s recent 5-year overall survival (OS) rate of 42.9%, 6 over half of patients did not benefit from durvalumab treatment, 7 highlighting the need for improved treatment strategies and further exploration of more effective approaches.
In clinical practice, tumor downsizing is often required when tumors are too large or when patients have poor PS or comorbidities that prevent immediate cCRT.8,9 In such cases, induction chemotherapy (ICT) is used to enhance tumor radiosensitivity, improve local control, reduce lymph node volume, and control micrometastases.10,11 The CALGB 39801 and CALGB 9130 studies demonstrate that ICT followed by cCRT significantly increases the incidence of radiation pneumonitis (RP), although the median survival is marginally extended, with the difference not reaching statistical significance.12–14 IO has reshaped the treatment landscape, but the comparative impact of ICT followed by cCRT versus cCRT alone on survival benefits and pulmonary toxicity in patients with unresectable stage III NSCLC remains unexplored.
This study aims to evaluate the effects of ICT combined with cCRT on the incidence of RP, as well as its impact on OS and progression-free survival (PFS) in the context of IO consolidation. This study may provide valuable insights for future therapeutic strategies combining chemotherapy and radiotherapy in the context of IO consolidation, aiming to improve patient outcomes and minimize treatment-related pulmonary toxicity.
Methods
Patient selection
We retrospectively analyzed data from 158 patients with unresectable stage III NSCLC who received cCRT followed by consolidation IO at Shandong Cancer Hospital and Institute between January 2018 and February 2024. Patients were divided into cCRT without ICT (N = 55) and ICT before cCRT groups (N = 103). Refer to Supplemental Table 1 for detailed inclusion and exclusion criteria for patients. This study received approval from the institutional review board of Shandong Cancer Hospital (approval number: SDTHEC202409042). The study was conducted in accordance with the Declaration of Helsinki.
Treatment strategy
All patients received intensity-modulated radiotherapy to a total dose of 40–70 Gy, combined with at least two cycles of platinum-based doublet chemotherapy, either concurrently or as induction. Patients who did not experience severe adverse events during induction therapy and cCRT were eligible to continue consolidation IO for up to 1 year. Immune checkpoint inhibitor (ICI) therapy included either anti-programmed cell death protein 1 (anti-PD-1) or anti-programmed death-ligand 1 (anti-PD-L1).
Assessment of pneumonitis
The diagnosis of RP was established by a multidisciplinary oncology team, which included at least one pulmonologist, pathologist, and radiologist. Diagnosis was based on CT scans and clinical symptoms, excluding other causes like infections or immune checkpoint inhibitor-associated pneumonia (Supplemental Table 2). RP severity was graded according to the Common Terminology Criteria for Adverse Events (CTCAE v.5.0). 15 The representative images are displayed in Figure 1.
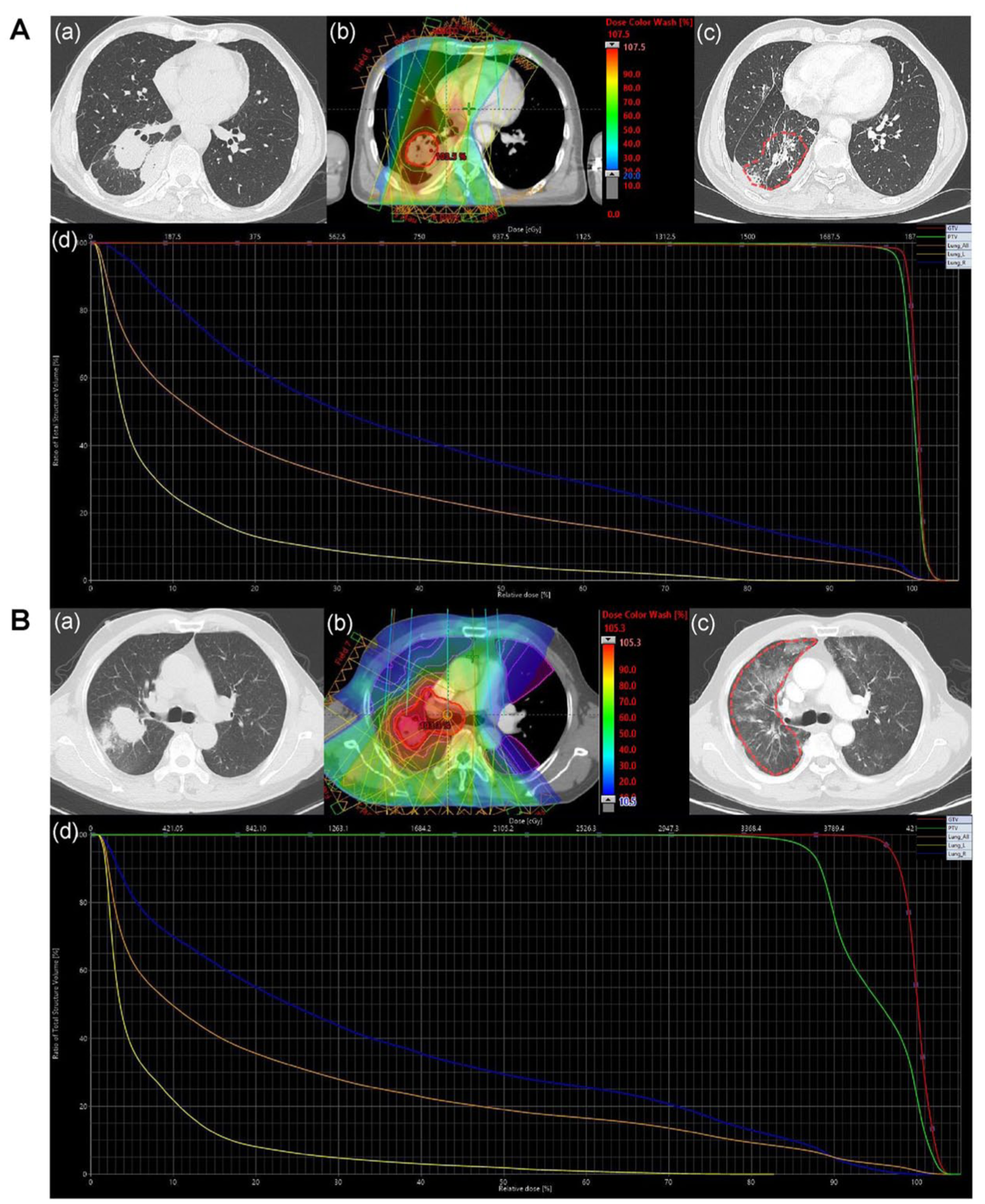
Typical clinical cases. (A) cCRT without ICT. (B) ICT before cCRT. (a) CT image before RT. (b) Isodose curve of the treatment plan. (c) CT image after 3 months of RT. (d) Dose distribution histograms for the total lung, right lung, and left lung. The CT image showing RP is outlined by a red dotted line.
Statistical analysis
Descriptive statistics summarized the clinical characteristics of the enrolled patients. Differences in baseline characteristics between groups were compared using t tests for continuous variables and χ2 tests for categorical variables. 16 Event outcomes, including pneumonitis and OS, were calculated from the start of radiotherapy. The association between ICT prior to radiotherapy and RP occurrence and severity in unresectable stage III NSCLC patients was analyzed, utilizing the competing risk model to analyze the cumulative incidence of RP. 17 OS was assessed from the start of diagnosis of lung cancer to death. PFS was assessed from the start of diagnosis of lung cancer to the first recorded disease progression or death without progression. Patients who were lost to follow-up were censored at the date of their last known contact. Missing data were handled by complete case analysis, where patients with any missing covariate data were excluded from multivariable regression models. Survival data were analyzed using the Cox proportional hazards regression model, with hazard ratios (HRs) and 95% confidence intervals (CIs) reported for comparisons between groups. 7 A p-value <0.05 was considered statistically significant. Analyses were performed using SPSS (version 27.0.1), with data visualization conducted using R (version 4.4.2).
Reporting guideline
The reporting of this study conforms to Strengthening the Reporting of Observational Studies in Epidemiology (STROBE: Supplemental Table 4). 18
Results
Baseline characteristics
A total of 158 patients were enrolled in this study. The median follow-up time is 37 months (95% CI: 33–40 months). Clinical and subgroup patient demographics are summarized in Table 1. The median age was 61 years (interquartile range (IQR), 56, 67), with 138 male patients (87.3%). Histologically, 94 patients (59.5%) had squamous carcinoma, while 64 (40.5%) had adenocarcinoma. Furthermore, 58 patients (36.7%) were stage IIIA, 79 (50%) were stage IIIB, and 21 (13.3%) were stage IIIC. Seventy-three (46.2%) developed ⩾Grade 2 RP, and 15 (9.5%) developed ⩾Grade 3 RP. Among the patients, 55 (34.8%) received cCRT without ICT, whereas 103 (65.2%) received ICT before cCRT. The median fractionated dose was 2 Gy (IQR, 2, 2), and the median total dose was 60 Gy (IQR, 56, 60). Moreover, 61.4% received PD-1 inhibitors (including tislelizumab, nivolumab, sintilimab, pembrolizumab, camrelizumab, toripalimab, penpulimab, and serplulimab), whereas 38.6% received PD-L1 inhibitors (including durvalumab, sugemalimab, and atezolizumab).
Clinical and subgroup patient demographics.

BMI, body mass index; ECOG, Eastern Cooperative Oncology Group performance status; ICI, immune checkpoint inhibitors; ICT, induction chemotherapy; IQR, interquartile range; MLD, mean lung dose; NRS2002, Nutritional Risk Screening 2002; PD-1, programmed death 1; PD-L1, programmed death-ligand 1; RP, radiation pneumonitis; SD, standard deviation; TNM, tumor node metastasis classification.
The use of ICT drugs and the incidence of RP
As shown in Figure 2(a), compared to the cCRT without ICT group, the incidence of ⩾Grade 2 RP was higher in the ICT before cCRT group (Gray’s test, SHR = 1.964, 95% CI: 1.137–3.394, p = 0.013), with a similar trend for ⩾Grade 3 RP between the groups (Gray’s test, SHR = 4.074, 95% CI: 0.935–17.757, p = 0.046; Figure 2(b)). Cox regression analysis (Figure 3) further showed that ICT before cCRT group had a significantly higher risk of ⩾Grade 2 RP (HR: 1.970, 95% CI: 1.048–3.705, p = 0.035). Cox regression for ICT regimens with ⩾Grade 2 RP (Table 2). Variables with a p-value <0.2 in the univariate analysis were included in the multivariate analysis. Compared with the EP regimen (Etoposide + Platinum), PP regimen (Pemetrexed + Platinum, HR: 0.277, 95% CI: 0.090–0.854, p = 0.025), TP regimen (Paclitaxel + Platinum, HR: 0.294, 95% CI: 0.094–0.926, p = 0.037), and DP regimen (Docetaxel + Platinum, HR: 0.059, 95% CI: 0.010–0.356, p = 0.002) were associated with a lower incidence of ⩾Grade 2 RP.

Cumulative hazard curve of RP between the cCRT without ICT and the ICT before cCRT groups. (a) ⩾Grade 2 RP occurred. (b) ⩾Grade 3 RP occurred.

Cox regression associated with ⩾Grade 2 RP.
Cox regression for ICT regimens with ⩾Grade 2 RP (N = 103).

BMI, body mass index; CI, confidence intervals; DP, Docetaxel + Platinum; ECOG, Eastern Cooperative Oncology Group performance status; EP, Etoposide + Platinum; HR, hazard ratios; ICI, immune checkpoint inhibitors; ICT, induction chemotherapy; IQR, interquartile range; MLD, mean lung dose; PD-1, programmed death 1; PD-L1, programmed death-ligand 1; PP, pemetrexed + platinum; RP, radiation pneumonitis; SD, standard deviation; TNM, tumor node metastasis classification; TP, Paclitaxel + Platinum.
Furthermore, among the 103 patients in the ICT before cCRT group, a longer interval between ICT and radiotherapy was identified as an independent risk factor in the logistic regression model (OR: 1.500, 95% CI: 1.079–2.085, p = 0.016; Supplemental Figure 1).
Impact of ICT on survival outcomes
Among patients with ⩾Grade 2 RP, the ICT before cCRT group was not associated with a significant difference in either OS (HR: 1.020, 95% CI: 0.580–1.780, p = 0.947) or PFS (HR: 1.390, 95% CI: 0.800–2.410, p = 0.242) compared to the cCRT without ICT group (Figure 4). The median OS was comparable between the ICT before cCRT group (42 months) and the cCRT without ICT group (44 months). Similarly, the median PFS was 38 versus 43 months. These results are consistent with previous studies. 14
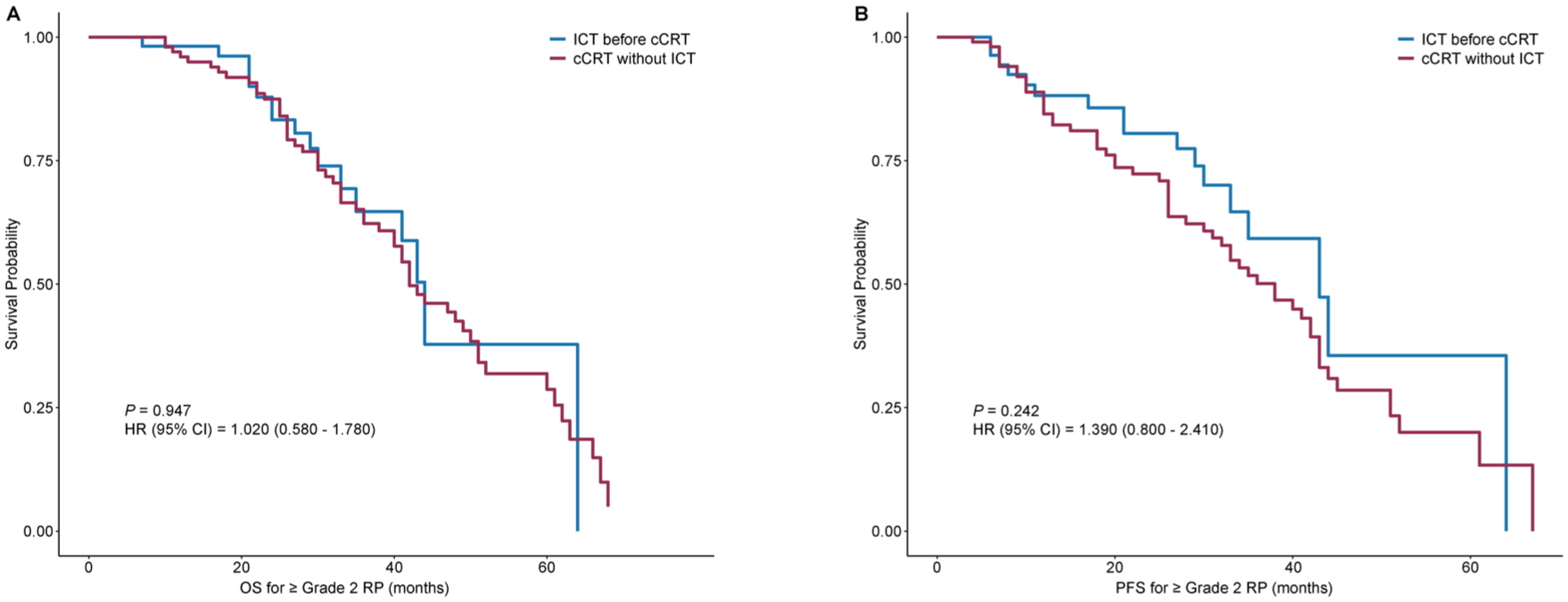
OS and PFS of RP between the cCRT without ICT group and the ICT before cCRT group. (a) OS for ⩾Grade 2 RP. (b) PFS for ⩾Grade 2 RP.
Distribution of ICT-RT time intervals in patients developing ⩾Grade 2RP
As shown in Supplemental Figure 2, among the 55 patients in the ICT before cCRT group who developed ⩾Grade 2 RP, the median (IQR) interval between ICT and radiotherapy was 29 days (26, 35). The majority of cases were clustered around the 4-week interval, consistent with clinical guidelines to allow for adequate hematologic recovery.14,19 Specifically, 6 patients (10.91%) had an interval of 20 days, 15 (27.27%) at 25 days, 17 (30.91%) at 30 days, 8 (14.55%) at 35 days, 3 (5.45%) at 40 days, 3 (5.45%) at 45 days, 2 (3.64%) at 50 days, and 1 patient (1.82%) at 60 days.
The count of RP by ICT cycle
Supplemental Figure 3 illustrates the count of ⩾Grade 2 RP across ICT cycles. The highest incidence is observed in the second cycle, with 33 patients affected by ⩾Grade 2 RP, compared to 24 patients without. With progression through the ICT cycles, the number of patients experiencing⩾Grade 2 RP decreases, reflecting a reduction in incidence.
Discussion
The advent of “Onco-Respiratory Medicine” has refocused attention on preserving respiratory health during cancer treatment, 20 with an emphasis on minimizing pulmonary damage. To our knowledge, it remains unclear whether adding ICT to cCRT changes the risk of RP in patients with unresectable stage III NSCLC in the IO era. This study reveals a higher incidence of ⩾Grade 2 RP in the ICT before cCRT group, but no significant differences in OS or PFS. These findings highlight that ICT is associated with a higher risk of RP without improving long-term survival, emphasizing the need for a risk-benefit balance in clinical practice.
Our real-world study showed a higher incidence of ⩾Grade 2 RP in patients receiving ICT, with a similar trend for ⩾Grade 3 RP. Chemotherapy-induced lung injury results from cytotoxicity/apoptosis during drug metabolism, followed by an immune response.21,22 Recent studies indicate that chemotherapy drugs have a dual role in immune responses. 23 Cisplatin enhances anti-tumor immunity by upregulating MHC I expression, recruiting immune effector cells, and inhibiting immune-suppressive molecules. 24 Paclitaxel promotes immune stimulation by enhancing M1 macrophages and inducing apoptosis in immune-suppressive cells.25,26 However, some drugs induce PD-L1 expression and promote immune evasion.27–29 Such immune-inflammatory responses release mediators that exacerbate lung injury in a dose-dependent way. Drug–drug and drug–environment interactions further modulate the severity of injury.21,30 Common drugs like docetaxel, paclitaxel, gemcitabine, and vinorelbine are linked to alveolar epithelial damage.31,32 In most cases, lung injury from these drugs is latent and can either be repaired through alveolar epithelial mechanisms or remain subclinical. In most cases, these drugs are associated with subclinical lung injury that is repaired by alveolar epithelial mechanisms or persists without symptoms. However, when radiotherapy follows, the combined damage from radiation and chemotherapy is often linked to RP.
Indeed, a substantial body of evidence indicates that certain chemotherapeutic drugs can promote chronic inflammation and organ fibrosis through the TGF-β and NF-κB signaling pathways.33–36 This provides biological plausibility for our finding: while drugs such as paclitaxel and cisplatin are effective in shrinking tumors, they may simultaneously “prime” or sensitize the pulmonary tissue to subsequent radiotherapy, thereby increasing the risk of injury. 37 Consequently, balancing the anti-tumor benefits of ICT against the potential risk of exacerbated radiotoxicity has emerged as a critical challenge in clinical practice.
ICT regimens commonly used in lung cancer treatment include docetaxel, gemcitabine, pemetrexed, etoposide, paclitaxel, and platinum-based compounds. The incidence of RP varies based on the specific combinations of ICT regimens used.38,39 In the pre-IO era, the previous phase III study on cCRT demonstrated that the EP regimen typically exhibited lower rates of RP compared to TP, but with greater hematologic toxicity.40–42 However, in our study, where all patients received consolidation IO, we observed a partial reversal of this toxicity profile. The EP regimen is associated with significantly higher rates of ⩾Grade 2 RP, suggesting a synergistic, rather than merely additive, adverse effect between etoposide and ICIs. We propose that this is driven by etoposide-induced remodeling of the tumor microenvironment. Through DNA damage pathways like cGAS-STING, etoposide upregulates immune checkpoints such as PD-L1 and CD73 on tumor cells.43,44 This priming effect, when followed by subsequent ICI administration, induces an intensified localized immune response that amplifies inflammation in the irradiated lung, resulting in severe RP. Furthermore, etoposide’s known pro-inflammatory potential, including altered cytokine production, is likely amplified by ICI, exacerbating lung injury, 45 which is an effect unobservable in the non-IO era. Additional studies support our conclusion. 46 Besides, the EP group had a small sample size (n = 6), which raises concerns about potential selection bias; these results should be interpreted with caution. In conclusion, our findings indicate that it is necessary to reassess the toxicity of chemotherapy in the context of IO.
Previous studies have indicated that the toxic effects of ICT are cumulative.47,48 In this study, the number of ICT cycles showed no significant association with the incidence of ⩾Grade 2 RP. The most likely reason is that cumulative toxicity, over the limited number of cycles, did not rise to a level that would be associated with ⩾Grade 2 RP. This study typically used two cycles, in line with clinical practice, in which two ICT cycles are common.
Our findings indicate that a prolonged interval between ICT and radiotherapy initiation is associated with an increased risk of ⩾Grade 2 RP. However, this increased risk is mainly attributed to the toxic effects associated with ICT itself. There is also the possibility of delayed initiation of radiotherapy occurring in patients who experience adverse reactions following ICT. This perspective is indirectly corroborated by the PACIFIC-2 trial, where concurrent durvalumab and cCRT were correlated with a prohibitive incidence of adverse events, preventing nearly 50% of patients from reaching the consolidation phase and resulting in a median OS lower than that of the PACIFIC trial (36.4 vs 47.5 months). 49 This failure suggests that the acute inflammatory stress severely overwhelms the lung’s tolerance capacity. Theoretically, a significantly shortened interval between ICT and radiotherapy may trigger synergistic toxicities (a “1 + 1 > 2” effect). In contrast, an excessively prolonged interval may undermine the therapeutic efficacy of ICT, potentially allowing for tumor repopulation and disease progression. Mechanistically, the risk observed in our study likely originates from the biological priming effect induced by ICT: ICT-induced tumor necrosis releases damage-associated molecular patterns (e.g., HMGB1 and mtDNA), which activate the cGAS-STING or TLR4/NF-κB pathways,50–52 establishing a hyper-inflammatory burden involving IL-6 and TNF-α. 45 Moreover, in clinical practice, a prolonged interval reflects severe ICT-related toxicities or systemic frailty. This represents a primed state of pulmonary vulnerability before radiotherapy begins, which requires a delay in radiotherapy and indicates an extended physiological recovery. Thus, RP risk is driven by both the foundational ICT toxicity and the evolving inflammatory microenvironment. Optimizing the interval between ICT and radiotherapy is crucial for balancing efficacy and the risk of RP.
Our clinical data indicate that the addition of ICT did not significantly improve OS or PFS in patients who received consolidation IO after cCRT. However, it was linked to a higher incidence of ⩾Grade 2 RP. The absence of differences in OS and PFS might be attributable to the small sample size and insufficient follow-up duration. Moreover, the therapeutic benefit of ICT in systemic tumor control may have been offset by the increased risk of RP, thereby masking any potential OS benefit. Similarly, in the pre-IO era, studies showed that ICT did not provide significant survival benefits but was linked to higher toxicity.37,48,53,54 Future research should focus on optimizing treatment sequencing and identifying patient subgroups that may benefit from ICT, aiming to minimize adverse events and maximize survival outcomes.
This study is the first to explore the pulmonary toxicity of ICT within the context of consolidation IO, revealing its effects on survival outcomes. These findings offer new insights into reducing treatment-related toxicities in unresectable stage III NSCLC. However, we acknowledge that our study has several limitations. First, as a retrospective study, it is subject to selection bias and confounding factors. Second, the limited sample size may have reduced statistical power, particularly when analyzing events such as ⩾Grade 3 RP and in subgroup analyses of the ICT regimens. Future multicenter, prospective randomized trials are needed to further explore the link between ICT and RP, providing evidence to optimize treatment strategies.
Conclusion
ICT followed by cCRT showed a higher incidence of ⩾Grade 2 RP compared to cCRT alone in patients with unresectable stage III NSCLC receiving IO consolidation. If ICT is used, the EP regimen should be used with caution. This study suggests that greater attention should be paid to pulmonary safety when using ICT and provides valuable insights for optimizing treatment timing and patient selection.
Supplemental Material
sj-docx-1-tam-10.1177_17588359261421552 – Supplemental material for Induction chemotherapy administration increases the risk of radiation pneumonitis in unresectable stage III NSCLC patients undergoing concurrent chemoradiotherapy with immunotherapy
Supplemental material, sj-docx-1-tam-10.1177_17588359261421552 for Induction chemotherapy administration increases the risk of radiation pneumonitis in unresectable stage III NSCLC patients undergoing concurrent chemoradiotherapy with immunotherapy by Pengwei Li, Yiwei Qin, Xinyi Liang, You Mo and Dawei Chen in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359261421552 – Supplemental material for Induction chemotherapy administration increases the risk of radiation pneumonitis in unresectable stage III NSCLC patients undergoing concurrent chemoradiotherapy with immunotherapy
Supplemental material, sj-docx-2-tam-10.1177_17588359261421552 for Induction chemotherapy administration increases the risk of radiation pneumonitis in unresectable stage III NSCLC patients undergoing concurrent chemoradiotherapy with immunotherapy by Pengwei Li, Yiwei Qin, Xinyi Liang, You Mo and Dawei Chen in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359261421552 – Supplemental material for Induction chemotherapy administration increases the risk of radiation pneumonitis in unresectable stage III NSCLC patients undergoing concurrent chemoradiotherapy with immunotherapy
Supplemental material, sj-docx-3-tam-10.1177_17588359261421552 for Induction chemotherapy administration increases the risk of radiation pneumonitis in unresectable stage III NSCLC patients undergoing concurrent chemoradiotherapy with immunotherapy by Pengwei Li, Yiwei Qin, Xinyi Liang, You Mo and Dawei Chen in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-4-tam-10.1177_17588359261421552 – Supplemental material for Induction chemotherapy administration increases the risk of radiation pneumonitis in unresectable stage III NSCLC patients undergoing concurrent chemoradiotherapy with immunotherapy
Supplemental material, sj-docx-4-tam-10.1177_17588359261421552 for Induction chemotherapy administration increases the risk of radiation pneumonitis in unresectable stage III NSCLC patients undergoing concurrent chemoradiotherapy with immunotherapy by Pengwei Li, Yiwei Qin, Xinyi Liang, You Mo and Dawei Chen in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We would like to express our gratitude to our colleagues at Shandong Cancer Hospital for their invaluable support and guidance throughout this study. We also extend our appreciation to the patients who participated in this research, as their contribution was essential to our work.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
