Abstract
Objectives:
The study aims to evaluate the efficacy and safety of thoracic radiotherapy in epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor (TKI)-treated patients with stage IV non-small-cell lung cancer (NSCLC).
Methods:
Patients with non-oligometastatic NSCLC harboring EGFR mutations were recruited. All patients received the first-generation TKI treatment with or without radiotherapy. The irradiated sites included primary and/or metastatic lesions. Of all the patients who underwent thoracic radiotherapy, some received radiotherapy before EGFR-TKI resistance, others received radiotherapy after progressive disease.
Results:
No statistically significant difference was observed in progression-free survival (PFS) (median 14.7 versus 11.2 months, p = 0.075) or overall survival (OS) (median 29.6 versus 40.6 months, p = 0.116) between patients treated with EGFR-TKIs alone and those with additional radiotherapy to any sites. However, EGFR inhibitors with thoracic radiation significantly improved OS (median 47.0 versus 31.0 months, p < 0.001) but not PFS (median 13.9 versus 11.9 months, p = 0.124). Moreover, longer PFS (median 18.3 versus 8.5 months, p < 0.001) was achieved in the preemptive thoracic radiation cohort than in the delayed thoracic radiation cohort. However, OS was similar between the two cohorts (median 40.6 versus 52.6 months, p = 0.124). The lower incidence rate of grade 1–2 pneumonitis occurred in preemptive radiation cohort (29.8% versus 75.8%, p < 0.001).
Conclusion:
Non-oligometastatic NSCLC patients with EGFR mutations benefited from thoracic radiotherapy while using EGFR inhibitors. Preemptive thoracic radiotherapy could be a competitive first-line therapeutic option due to superior PFS and favorable safety.
Keywords
Introduction
Over half of the patients with non-small-cell lung cancer (NSCLC) present with metastatic disease at initial diagnosis. 1 The identification of multiple driver gene mutations in NSCLC makes it possible for personalized molecular-targeted therapy. Especially, epidermal growth factor receptor (EGFR)-sensitizing mutations, mainly comprising deletions in exon 19 and the point mutation in exon 21 Leu858Arg, are critical therapeutic targets for NSCLC patients. 2 Previous evidence has shown that EGFR-mutant patients receiving EGFR tyrosine kinase inhibitors (TKIs), such as gefitinib, icotinib, and erlotinib, have a higher overall response rate than those receiving conventional chemotherapy.3,4 Thus, EGFR-TKI monotherapy has been established to be the standard first-line treatment for metastatic NSCLC patients with EGFR mutations.5,6
The cancer cell population of the tumor is heterogeneous, containing tumor cells with activating mutation and those without. 7 Despite the high response of EGFR-TKIs, patients harboring EGFR-mutant experience different time of response due to inconsistent abundance of EGFR mutations 8 and nearly all NSCLC patients unavoidably develop progressive disease (PD) after 9.7–13.1 months of the first-generation EGFR-TKI monotherapy.3,9–11 Efforts have been devoted to exploring mechanisms of acquired resistance to anti-EGFR therapy, but the benefit of TKIs monotherapy in improving survival for patients with advanced lung cancer is limited. 12
Previous studies have indicated that EGFR-TKIs improve radiosensitivity, while radiotherapy reduces EGFR-TKI resistance.13,14 Moreover, it has been reported that EGFR-TKIs plus radiation therapy for metastatic lesions improve local control in stage IV EGFR-mutant NSCLC. 15 Evidence from retrospective studies has revealed that local consolidative radiotherapy improved the progression-free survival (PFS) and overall survival (OS) in oligometastatic NSCLC without increasing the rates of serious adverse events.16–19 Most patients diagnosed with stage IV NSCLC, however, present with commonly widespread metastases. Therefore, more attention should be paid to investigating the role of local treatment in non-oligometastatic NSCLC.
The primary lung lesions are the most common sites of initial failure during EGFR-TKI therapy in patients with advanced NSCLC.12,20 Su et al. reported that aggressive thoracic radiation therapy concurrently combined with chemotherapy may prolonged survival in stage IV NSCLC patients, 21 reminding that the addition of thoracic radiotherapy with EGFR-TKI treatment may also be associated with favorable survival. Currently, limited studies address the efficacy and safety of adding thoracic radiotherapy to EGFR-TKIs as the first-line treatment for metastatic NSCLC patients with EGFR-sensitizing mutations.22,23 In a recent prospective study involving 10 advanced NSCLC patients, Zheng et al. reported that the concurrent EGFR-TKIs combined with thoracic radiotherapy achieve significant control for the primary lung lesions. 22 The data still need to be confirmed in a larger sample study.
Therefore, we implemented a relatively large-scale retrospective study on the efficacy and safety of the dual therapy of EGFR-TKIs and thoracic radiotherapy in patients with non-oligometastatic NSCLC harboring EGFR mutations. The aim of this study is to investigate whether thoracic radiotherapy could prolong the survival of patients with advanced NSCLC during EGFR-TKI treatment.
Methods
Patients selection
It was a relatively large-scale retrospective study. We screened NSCLC patients who visited Cancer Center of Wuhan Union Hospital. Those who were 18 years or older diagnosed as non-oligometastatic NSCLC (more than three metastatic lesions, or pathologically confirmed malignant pleural effusion, pericardial effusion, or peritoneal effusion) with confirmed EGFR mutations, and received the first-generation TKI treatment were included in our study. We enrolled 366 eligible patients. This study was approved by the Institutional Ethics Committee of Wuhan Union Hospital. This study was performed in accordance with the Declaration of Helsinki.
Treatments
All patients received the treatment of first-generation EGFR-TKIs. A portion of patients underwent palliative radiotherapy for primary and/or metastatic lesions. The metastatic sites receiving palliative radiotherapy mainly included bone, brain, lung, and liver. The technologies of radiotherapy included intensity-modulated radiotherapy and stereotactic radiation therapy. Conventional fractionated radiotherapy was given for 15–30 fractions with a daily fractionated dose of 2–5 Gy, while hypofractionated radiotherapy was given for 3–10 fractions with a daily fractionated dose of >5 Gy. The final radiotherapy regimen was designed by a radiotherapist on account of the patient’s age, performance status, tolerance, and willingness.
The enrolled patients were divided into two groups according to whether receiving radiotherapy during ongoing EGFR-TKI treatment was performed. According to different irradiated sites, all patients who had received radiotherapy were further divided into three cohorts, including preemptive thoracic radiotherapy before PD (preemptive thoracic radiation cohort), delayed thoracic radiotherapy after PD (delayed thoracic radiation cohort), and radiotherapy only to metastases. Screening and groups of patients are shown in Figure 1.
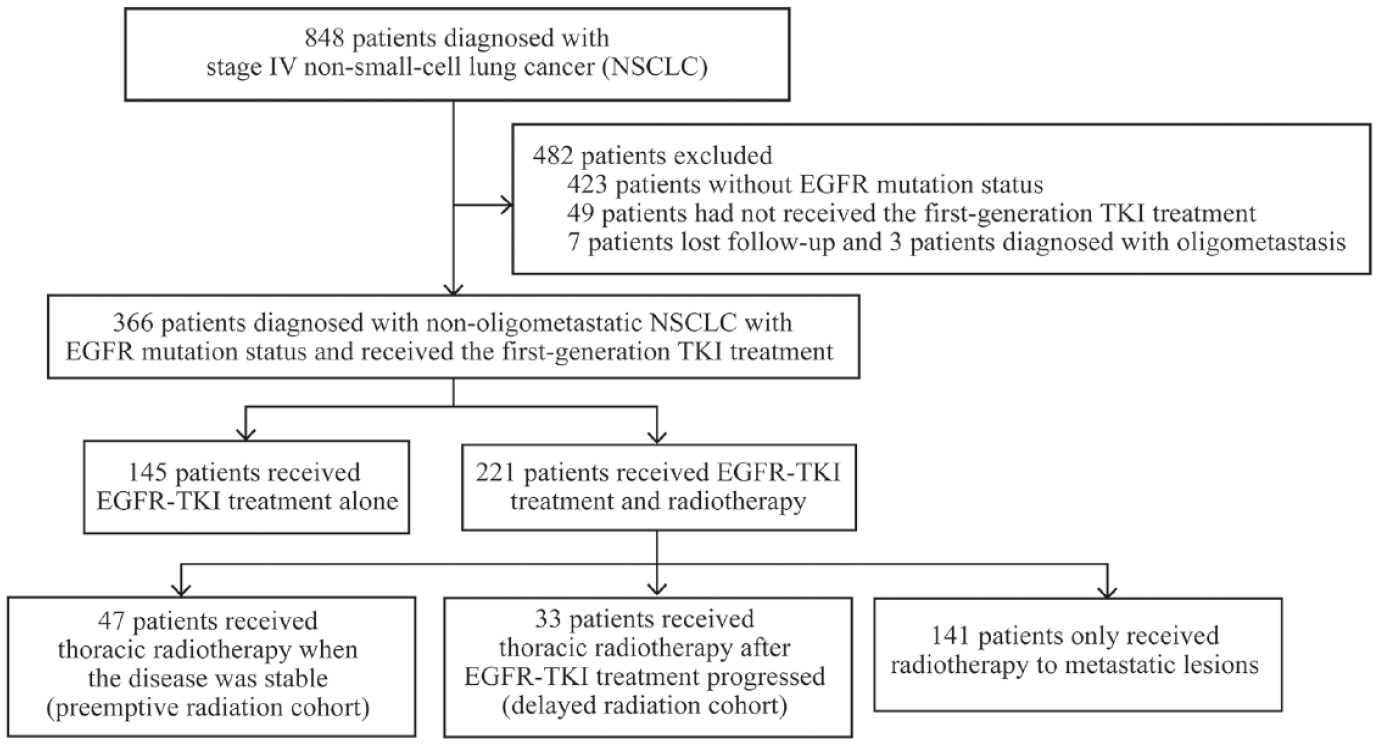
Flow chart of screening and groups of patients.
Follow-up
Patients were followed up at 6–8 weeks, receiving tests, such as thoracic contrast-enhanced computed tomography (CT), imaging examinations of metastases, and laboratory tests. Follow-up was conducted through face-to-face consultation and telephone contact. Adverse events were evaluated based on the patient’s symptoms, laboratory tests including complete blood cell counts and/or biochemical examinations, and imaging of CT. The Common Terminology Criteria for Adverse Events v5.0 was used to estimate treatment-related toxicities.
Statistical analysis
In this study, averages and medians were used to report continuous variables in all outcomes and categorical variables were described by rates. OS was a definition from initiating the first-generation EGFR-TKI treatment to death with any cause or last follow-up if alive. In addition, PFS was defined as the duration from the first date of receiving the first-generation EGFR-TKI treatment to the date of first documented disease progression, the date of death, or the last follow-up. Survival curves were estimated with Kaplan–Meier analysis and log-rank test was used to evaluate the heterogeneity of survival analysis. Chi-square test was used to examine the heterogeneity of rates. Cox proportional hazards model was used to assess for significant related factors of survival and calculate the hazard ratios (HRs). All prospective clinical factors were included as covariates in univariate and multivariate analyses with Cox proportion hazards model. Statistical analyses were conducted with IBM SPSS, version 25.0 software. Survival curves were plotted with Medcalc software. p < 0.05 was confirmed statistically significant.
Results
Patients’ characteristics
A total of 366 patients were eligible for this study. The median age was 59 years. Male cases accounted for 46.2% (n = 169). Of these, 22.4% (n = 82) patients had smoking history. All patients were diagnosed as non-oligometastatic NSCLC with EGFR mutations and received first-generation TKI treatment including erlotinib, gefitinib, or icotinib. Among them, 194 (53.0%) patients were found exon 19 deletion, 172 (47.0%) patients had exon 21 mutation. The common metastatic sites included bone (n = 206, 56.3%), lung (n = 133, 36.3%), brain (n = 129, 35.2%), and liver (n = 31, 8.5%). 50 (13.6%) patients had metastases of other sites. The median follow-up time was 24.4 months. Only 19 patients were lost during follow-up and 219 (59.8%) patients were alive during the follow-up period. Characteristics of the patients are shown in Table 1
Characteristics of patients.

EGFR, epidermal growth factor receptor.
Treatment details
Regarding therapeutics, 60.4% (221/366) of patients received first-generation EGFR-TKI treatment combined with radiotherapy. On the other hand, 39.6% (145/366) of patients received the TKIs alone. Of the patients with thoracic radiotherapy, 12.8% (47/366) of patients received radiation therapy during the treatment of TKIs (preemptive radiation cohort) and 9.0% (33/366) of patients after PD (delayed radiation cohort). The median time from initiating EGFR-TKI treatment to receiving thoracic radiation in the preemptive radiation cohort was 5.8 months. 38.5% (141/366) of patients only received radiotherapy for metastatic lesions.
The clinical characteristics of the cohorts with or without thoracic radiotherapy and the cohorts of comparison between preemptive and delayed radiation were presented in Tables 2 and 3, respectively. Clinical characteristics were well-balanced among the cohorts, except for the smoking factor, which differed in the thoracic radiotherapy group. In addition, a part of radiotherapy information of patients receiving thoracic irradiation are shown in Appendix 1.
Clinical characteristics of patients in thoracic radiotherapy group.

EGFR, epidermal growth factor receptor.
Clinical characteristics of patients in preemptive and delayed radiation cohorts.
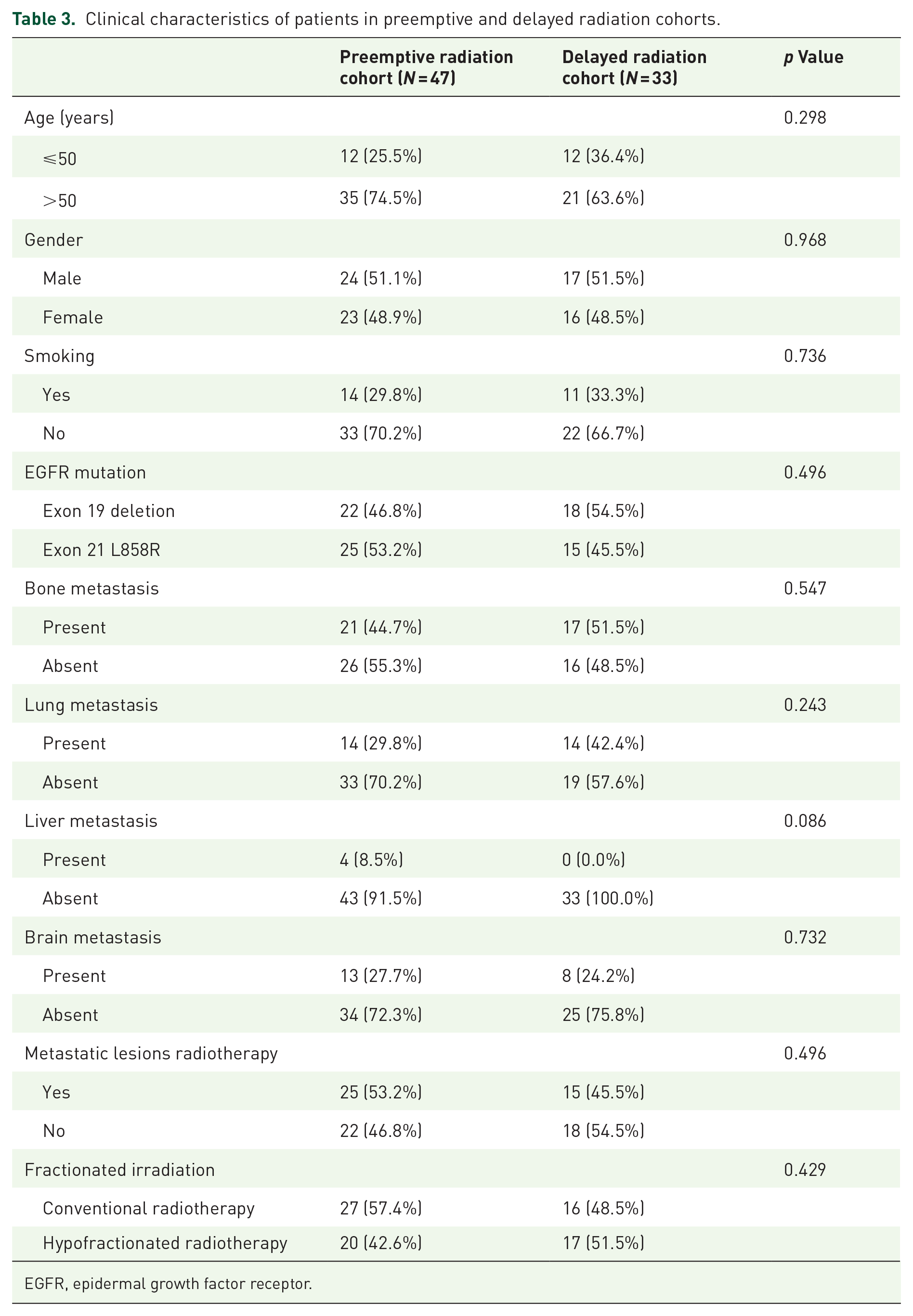
EGFR, epidermal growth factor receptor.
Effect of thoracic radiotherapy on survival
The median PFS (mPFS) and median OS (mOS) of all the patients were 12.2 months [95% confidence interval (CI): 11.1–13.3] and 34.8 months (95% CI: 28.9–40.7), respectively. The mPFS in patients treated with TKIs with or without radiotherapy for any sites exhibited no statistically significant difference (11.2 versus 14.7 months, p = 0.075; Figure 2(a)), neither did mOS (40.6 versus 29.6 months, respectively, p = 0.116: Figure 2(b)). The patients receiving thoracic radiotherapy (both preemptive and delayed thoracic radiotherapy) and TKIs had better mOS than the rest patients (47.0 versus 31.0 months, respectively, p < 0.001; Figure 3(b)), but no significant difference was observed in mPFS between these two cohorts (13.9 versus 11.9 months, respectively, p = 0.124, Figure 3(a)). The mPFS of the preemptive radiation cohort and the delayed radiation cohort were 18.3 months and 8.5 months, respectively, indicating a statistically significant difference (p < 0.001; Figure 4(a)). However, there was no statistical difference in mOS between these two cohorts (40.6 versus 52.6 months, respectively, p = 0.124; Figure 4(b)). Statistical parameters are shown in Table 4.

Kaplan–Meier survival curve for PFS (a) and OS (b) between patients receiving radiotherapy and who did not.

Kaplan–Meier survival curve for PFS (a) and OS (b) between patients receiving thoracic radiotherapy and who did not.

Kaplan–Meier survival curve for PFS (a) and OS (b) between preemptive radiation cohort and delayed radiation cohort.
Statistical parameters of survival.

PFS, progression-free survival; OS, overall survival; 95% CI, 95% confidence interval.
Relevant factors on OS
According to univariate analysis, thoracic radiotherapy and EGFR mutation as related factors were presumably correlation with the OS. Further multivariate analysis was implemented. The results confirmed that receiving thoracic radiotherapy (HR: 0.39, 95% CI: 0.24–0.62, p < 0.001) and exon 19 deletion (HR: 0.60, 95% CI: 0.42–0.87, p = 0.006) were statistically significant contributors of prolonged survival. Univariate and multivariate analyses of factors associated with OS are shown in Table 5.
Univariate and multivariate analyses of factors associated with OS.

EGFR, epidermal growth factor receptor; HR, Hazard ratio; OS, overall survival; 95% CI, 95% confidence interval.
Toxicity
The majority of toxicities were grade 1–2. Grade 3 myelosuppression and grade 3 hepatic insufficiency were observed in 21 and 17 patients during EGFR-TKI therapy. All of them were cured by prompt treatment. No patient discontinued EGFR-TKI treatment because of toxicities. We found that the rate of grade 1–2 radiation-related pneumonitis in the preemptive radiation cohort was lower than that in the delayed radiation cohort (29.8% versus 75.8%, p < 0.001). No one had grade ⩾3 radiation-related pneumonitis in radiotherapy-treated patients. No grade 5 treatment-associated toxicities were recorded in all patients.
Discussion
There is controversy regarding whether EGFR-mutant NSCLC patients during EGFR-TKIs therapy can benefit from thoracic radiation therapy. Cuneo et al. and Zheng et al. reported that NSCLC with EGFR mutation may be associated with radioresistance.24,25 However, other studies suggested that tumors with EGFR mutation, especially ex19del and Leu858Arg, were more radiosensitive than their wild-type counterparts.26,27 Although TKI monotherapy is currently the mainstay treatment for advanced EGFR-mutant NSCLC patients, nearly all patients inevitably progress caused by the development of acquired resistance. Therefore, more attention should be paid to the prevention of acquired resistance. Evidence from the literature suggests that the addition of radiotherapy to TKIs can produce a synergistic effect.13,14 In this study, a total of 366 EGFR-mutant NSCLC patients with the non-oligometastatic disease who received the first-line TKI treatment were included. We found that patients treated with EGFR-TKI and thoracic radiotherapy (both preemptive and delayed thoracic radiotherapy) have longer mOS than all the rest patients. Interestingly, patients who received preemptive thoracic radiotherapy showed a better mPFS than those receiving delayed thoracic radiotherapy.
Several studies have demonstrated the value of local consolidative therapy in oligometastatic NSCLC.16–19 A study by Yu et al. reported that local radiotherapy with EGFR-TKIs improved survival in patients with oligometastatic NSCLC. 17 Recently, other studies by Wu et al. and Gomez et al. concluded that local consolidative therapy offered promising therapeutic perspectives.18,19 Nevertheless, non-oligometastatic NSCLC is common in patients with advanced NSCLC and is closely associated with poor prognosis. Few studies have attempted to explore the value of thoracic radiation therapy in these patients. In this study, we found neither PFS nor OS were different for patients treated with radiation versus non-radiation groups. Thus, we speculate that the site and optimal timing of radiotherapy may be associated with survival rates.
Studies have reported that the lung is the most frequent primary failure site during TKI therapy.28,29 Emerging evidence suggests that radiotherapy for metastatic lesions can improve the outcome in patients with advanced EGFR-mutant NSCLC.15,30,31 Moreover, several studies have examined the activity of thoracic radiation in advanced NSCLC patients harboring TKI-sensitive EGFR mutations.22,23,32,33 Zheng et al. conducted a prospective study to assess the efficacy of thoracic radiotherapy in combination with EGFR-TKIs on stage IV EGFR mutant lung adenocarcinomas. 22 A superior 1-year PFS was observed in the combination therapy group. However, the small sample size, only 10 patients, limits the reliability of the study. Based on the larger sample, our results showed that radiotherapy for primary lesions provided the benefit in OS for non-oligometastatic NSCLC, regardless of the timing of its addition. It enlightened us that local radiotherapy for primary lesions could be an optional regimen to enhance the therapeutic efficacy.
In 2021, Wei et al. reported a retrospective study on 45 EGFR mutation-positive NSCLC patients who were treated with radiotherapy for primary lesions before disease progression. 33 They discovered that patients in the preemptive radiation cohort achieved a significantly superior PFS compared with the delayed radiation group (22.3 versus 12.9 months). Similarly, we also analyzed the differences in mPFS and mOS between the preemptive and delayed thoracic radiotherapy cohorts. As detailed in Figure 1, 33 non-oligometastatic NSCLC patients underwent thoracic radiotherapy after the progression of primary sites. Despite there being no significant difference in OS, the prolonged PFS delayed the advent of further therapy, which may be related to a better quality of life. Notably, the median time from first-line EGFR-TKI treatment to thoracic radiotherapy in the preemptive radiation cohort was 5.8 months, similar to the time of maximal tumor shrinkage reported. 34
In addition, it is noteworthy that the incidence of radiation pneumonitis is relatively lower in patients with concurrent thoracic radiotherapy plus EGFR-TKIs. The rate of grade 1–2 radiation pneumonitis was 29.8% in the preemptive radiation group versus 75.8% in the delayed radiation group. Therefore, preemptive thoracic radiotherapy in the course of TKI therapy could be a safe option for patients with stage IV NSCLC. Our current research cannot conclude that radiotherapy combined TKIs is safe. A previous study explored the incidence of radiation pneumonitis in NSCLC patients treated with concurrent thoracic radiotherapy and erlotinib. Radiation pneumonitis of grade 2 or higher was observed in 37.5% (9/24) of patients. 35 Recently, a single-arm phase II study presented a result that the probabilities of grade 3 or higher radiation-associated pneumonitis reached 20% during concurrent chemoradiotherapy. 22 Therefore, advanced patients receiving TKIs combined with thoracic radiotherapy should be carefully monitored considering radiation pneumonitis.
The reasons why thoracic radiotherapy combined with TKIs benefits patients with stage IV NSCLC were founded on several theories. First, resistant cells were selectively predominant while mutant cells were inhibited after EGFR-TKI treatment. 7 Administrating thoracic radiotherapy could help decrease the number of cells with TKI resistant but radiosensitivity. Second, tumor cells could spread constantly from the primary lesions with a high tumor burden, which was correlated positively with cancer progression and poor prognosis. Local radiotherapy for the primary site could reduce the total tumor burden compared with TKIs monotherapy in advanced NSCLC patients. 36 Furthermore, Li et al. reported that patients with sensitizing-EGFR mutations were more prone to benefit from thoracic radiotherapy. 37 They also found that residual tumor burden in the patients with double EGFR mutation was lower in comparison with that of the group with single EGFR mutation. Moreover, previous studies revealed that the existence of primary tumors could suppress the immune system.38–40 Since primary tumor resection is rarely used in patients with advanced NSCLC, local radiotherapy for primary lesions may be a feasible option in EGFR mutant NSCLC.
Our study has some limitations. First, the results were restricted by single-center retrospective analysis. Potential biases were inevitable due to retrospective observational nature. We attempted to alleviate the disturbance of confounders by making the cohorts well-balanced through screening clinical features of patients. Second, radiotherapy regimens varied among different patients according to tumor sizes and sites as well as therapeutic objectives. There is a lack of sufficient exploration on radiotherapy techniques, sites, and doses to reduce the interference on patient outcomes. Finally, our results imply that concurrent EGFR-TKI and thoracic radiotherapy has the possibility to change the natural history of disease progression and prolong the PFS of patients with non-oligometastatic EGFR-mutant NSCLC. These findings need to be confirmed by more large-scale prospective studies to ensure comprehensiveness.
Conclusions
To sum up, we retrospectively analyzed the role of radiotherapy, especially preemptive thoracic radiotherapy, in patients with non-oligometastatic NSCLC harboring EGFR mutations. These data demonstrated that the addition of thoracic radiotherapy could improve the survival in non-oligometastatic NSCLC patients irrespective of the timing of thoracic radiotherapy. Moreover, EGFR-TKI plus preemptive thoracic radiotherapy as the first-line treatment achieved better PFS. Compared with delayed radiotherapy for primary lesions, the addition of preemptive radiation had a lower incidence of radiation pneumonitis. The time of maximal tumor shrinkage after initiation of TKI therapy may be the optimal timing for radiotherapy to the primary site. A further prospective study is needed to verify the role of thoracic radiotherapy in non-oligometastatic advanced NSCLC patients harboring EGFR mutations.
Footnotes
Appendix
Radiotherapy information of patients receiving thoracic radiotherapy.

| Irradiated site | Number of patients | Conventional fractionated radiotherapy | Hypofractionated radiotherapy | ||
|---|---|---|---|---|---|
| Number of patients | Median radiation dose/fractions | Number of patients | Median radiation dose/fractions | ||
| Primary site | 80 | 37 | 60 Gy/25 f | 43 | 50 Gy/5 f |
| Bone metastases | 24 | 20 | 36 Gy/12 f | 4 | 29.5 Gy/3.5 f |
| Brain metastases | 23 | 17 | 30 Gy/10 f | 6 | 27 Gy/3 f |
| Cervical lymph node metastases | 1 | 1 | 60 Gy/20 f | 0 | NA |
| lymph node metastases in the supraclavicular region | 1 | 1 | 60 Gy/25 f | 0 | NA |
| Adrenal metastases | 1 | 0 | NA | 1 | 32 Gy/4 f |
| Tumor in shoulder | 1 | 1 | 45 Gy/18 f | 0 | NA |
Acknowledgements
We would like to take the opportunity to thank the patients, their families, and all of the research members in this study.
