Abstract
Background:
Systemic therapy is standard treatment for postoperative recurrent or metastatic pancreatic adenocarcinoma, yet survival remains poor. While recurrence/metastasis-directed therapy (RDT/MDT) has shown benefits in other cancers, its role in pancreatic adenocarcinoma remains controversial.
Objectives:
To evaluate whether adding RDT/MDT can enhance the efficacy of systemic therapy in patients with postoperative local recurrence or metastasis in pancreatic adenocarcinoma.
Methods:
Patients with recurrence or metastasis of pancreatic adenocarcinoma following R0 resection between 2018 and 2024 at West China Hospital and Shang Jin Hospital were retrospectively enrolled. Patients were categorized into the RDT/MDT plus systemic therapy group versus the systemic therapy alone group. Propensity score matching (PSM; 1:1) was used to minimize selection bias. Survival outcomes were analyzed using Cox regression, with subgroup analyses based on postoperative recurrence/metastasis status. Clinical features and overall survival (OS) of patients receiving radiotherapy as RDT/MDT were also evaluated.
Results:
A total of 203 eligible patients were enrolled. After 1:1 PSM, 76 patients from each group were analyzed for comparative survival outcomes. The median OS was 19.9 months in the RDT/MDT group versus 12.2 months in the systemic therapy group (hazard ratio 0.6, 95% confidence interval (CI): 0.4–0.8, p < 0.05). Subgroup analyses revealed significant survival advantages with RDT/MDT in patients with locoregional recurrence and widespread metastasis. In addition, a single-arm analysis of 64 patients who received radiotherapy as RDT/MDT (from the pre-PSM cohort) demonstrated a median OS of 20.1 months (95% CI: 17.5–28.5), with superior outcomes observed when radiotherapy was administered as first-line treatment.
Conclusion:
This study suggested that adding RDT/MDT to systemic therapy may improve OS in pancreatic adenocarcinoma with postoperative recurrence and metastasis, with radiotherapy as a feasible option. Further randomized controlled trials are warranted.
Design:
A multicenter, retrospective cohort study using PSM.
Plain language summary
Pancreatic cancer often comes back or spreads after surgery. Local therapy that target the recurrence or metastasis directly, combined with standard medicines, may help patients live longer. We studied 203 patients from two hospitals and found that those who received both targeted local therapy and systemic therapy lived longer than those who had only systemic therapy. Radiation therapy, used as a local therapy, was especially effective and safe. Our study suggests adding local therapy that target the recurrence or metastasis may improve survival.
Keywords
Introduction
Pancreatic adenocarcinoma is a lethal malignancy, remaining the fourth leading cause of cancer-related death in the United States and projected to become the second leading cause by 2030. 1 Surgical resection is the only potentially curative option, yet over 80% of patients experience local recurrence or metastasis following curative surgical resection.2–4 The 5-year survival of patients who underwent curative resection is estimated to be 27%, underscoring that local recurrence or metastasis following curative surgical resection remains the primary cause of mortality. 5 Despite these discouraging data, research focused on these patients remains limited.6,7
Systemic therapy is the cornerstone of therapy for local recurrence or metastasis following curative surgical resection in pancreatic adenocarcinoma.8,9 Although recent advancements have led to modest survival improvements, achieving meaningful survival gains remains a major challenge.4,9 In other solid tumors, local therapies directed at local recurrence or metastasis (RDT/MDT), such as radiotherapy and surgery, have been shown to enhance the efficacy of systemic therapy.10–16 However, the role of RDT/MDT in pancreatic adenocarcinoma remains controversial.17–19 Some studies suggest that combining chemotherapy with RDT/MDT may improve outcomes for patients with pancreatic adenocarcinoma.3,20 However, these studies are limited by small sample sizes and often focus on isolated or locoregional recurrences. Most of these studies are retrospective studies with small sample sizes.
The recently published EXTEND study has drawn our attention. 21 Patients who received MDT in this study demonstrated improved survival outcomes along with tolerable toxicity. Moreover, the study showed that MDT induced highly activated (CD25+) T cells and natural killer T cells (CD56+), increased the diversity of the T-cell receptor repertoire, and enhanced the richness of T-cell receptor-dominant motifs, which indicated MDT potentially induces systemic immunity.
Previous research has also shown that radiotherapy induces cytoreduction and eliminates macroscopic immunosuppressive stroma, thereby promoting systemic immune responses and overcoming treatment resistance in chemotherapy-refractory tumor cells. These mechanisms suggest that radiotherapy may offer survival benefits even in metastatic disease.21–24
Therefore, our study aims to evaluate whether the addition of RDT/MDT can enhance systemic therapy efficacy for local recurrence or metastasis following curative surgical resection in pancreatic adenocarcinoma and whether radiotherapy as RDT/MDT provides a survival benefit, thereby contributing to the development of improved treatment approaches for this disease.
Methods
Patients and study design
This multicenter, retrospective study collected data of patients with local recurrence or metastasis of pancreatic adenocarcinoma following curative surgical resection from West China Hospital and Shang Jin Hospital of Sichuan University between October 2018 and April 2024. A formal data-sharing agreement exists between the two institutions. 25 Data collection was supervised by Chenyan Zhang at West China Hospital and Xiaofen Li at Shang Jin Hospital. Both investigators ensured adherence to the study protocol and data quality standards. Patient records were systematically reviewed according to predefined inclusion and exclusion criteria at both institutions. This retrospective study received ethics approval from the Ethics Committee on Biomedical Research, West China Hospital of Sichuan University (approval No. 2024-2436, dated December 11, 2024). As member institutions of the Chengdu Regional Medical Ethics Alliance, both hospitals operate under a regional ethics mutual recognition framework, whereby Shang Jin Hospital accepts ethics review decisions from West China Hospital as the coordinating institution. 26 Given the retrospective nature of using de-identified medical records, the requirement for informed consent was waived. The study was registered with the Chinese Clinical Trial Registry (ChiCTR2500095418) on January 7, 2025. The reporting conforms to the STROBE guidelines 27 (Supplemental Table 1).
All patients were reviewed according to the inclusion criteria: (1) pathologically confirmed pancreatic adenocarcinoma, (2) recurrence or extracranial metastasis confirmed by imaging after R0 resection, (3) administration of systemic therapy (e.g., chemotherapy, targeted therapy, or immunotherapy) following postoperative recurrence or metastasis (4) age between 18 and 80 years, (5) Eastern Cooperative Oncology Group (ECOG) performance status of 0–2. Exclusion criteria included: (1) pathological subtypes other than pancreatic adenocarcinoma, (2) no evidence of postoperative recurrence or metastasis, (3) no systemic therapy (e.g., chemotherapy, targeted therapy, or immunotherapy) administered for postoperative recurrence or metastasis, (4) presence of cranial metastases, (5) non-R0 resection, (6) concurrent other malignancies, (7) incomplete data. Patients were categorized into two groups based on treatment received after diagnosis of recurrence or metastasis: (1) RDT/MDT group: patients who received RDT/MDT in addition to systemic therapy; (2) Systemic therapy group: patients who received only systemic therapy without any local intervention.
Treatment and evaluation
Following R0 resection, all patients underwent regular surveillance. Initial radiographic assessment was performed using CT or MRI at 2 months post-surgery. Subsequently, patients were followed up every 2 months with cross-sectional imaging (CT or MRI), serum carbohydrate antigen 19-9 (CA19-9) measurement, and clinical assessment. According to the requirements of clinical doctors, PET-CT may be examined if necessary. Recurrence or metastasis was defined as the appearance of new lesions detected on imaging according to Response Evaluation Criteria in Solid Tumors (RECIST) 1.1 criteria. Postoperative recurrence or metastasis was classified into three types: local recurrence, oligometastasis, and widespread metastasis. Recurrence at locoregional sites—areas near the superior mesenteric artery, celiac axis, hepatic artery, surgical bed, and remnant pancreas—was called locoregional recurrence. Oligometastasis meant metastases outside the above sites, limited to two or fewer organs and five or fewer lesions. Widespread metastasis included all other cases, such as both local recurrence and metastasis, or distant metastasis involving more than two organs or five lesions.
Patients received systemic therapy—including chemotherapy, immunotherapy, and targeted therapy—after diagnosis of recurrence or metastasis. Some patients underwent RDT/MDT in addition to systemic therapy. RDT/MDT refers to local interventions directed at recurrence or metastasis sites, including radiotherapy, transarterial chemoembolization, and surgery.
Once treatment for postoperative recurrence or metastasis was initiated, disease monitoring was intensified with CT or MRI performed every 2–3 months. Disease progression (PD) was determined radiographically per RECIST 1.1.
Variables
We extracted the baseline characteristics of eligible patients from clinical records. The baseline data included age, gender, ECOG, recurrence or metastasis sites, CA19-9 levels, and local recurrence or metastasis status, as well as surgical and pathological variables (tumor location, stage, and presence of perineural and lymphovascular invasion, prior anti-cancer therapies). The main exposure of interest was receipt of RDT/MDT in addition to systemic therapy. The primary outcome was overall survival (OS), defined as the time from radiographic diagnosis of recurrence or metastasis to death from any cause. Patients alive at the data cutoff date (January 2025) were censored at the date of last follow-up.
Statistical analysis
We categorized patients into two groups: those receiving RDT/MDT in addition to systemic therapy (RDT/MDT plus systemic therapy group) and those receiving systemic therapy alone (systemic therapy group). Descriptive statistics were used to compare baseline variables. The Shapiro–Wilk test assessed normality, and non-normally distributed continuous variables were compared using the Wilcoxon rank-sum test. Categorical variables were analyzed with the chi-squared test, depending on expected frequencies.
To minimize selection bias, propensity score matching (PSM) was conducted using the MatchIt package in R version 4.4.1 (The R Foundation for Statistical Computing, Vienna, Austria). Patients were matched 1:1 using nearest-neighbor matching without replacement, with a caliper width of 0.2 standard deviations of the logit of the propensity score. The propensity score was calculated using logistic regression with the following covariates: gender, tumor location, pathological stage, perineural invasion, lymphovascular invasion, prior chemotherapy, prior radiotherapy, age, ECOG performance status, recurrence/metastasis status, and CA19-9 levels. Balance after matching was assessed using standardized mean differences, with values <0.1 indicating adequate balance.
Kaplan–Meier survival curves were generated and compared with the log-rank test. After matching, we evaluated the impact of RDT/MDT on survival outcomes through univariable and multivariable Cox proportional hazards regression. Variables with p-values <0.1 in univariable analysis were included in the multivariable model. We performed a subgroup analysis based on different recurrence/metastasis statuses. To assess the robustness of PSM, we adjusted the caliper to 0.1 for PSM and repeated univariable and multivariable Cox regression analysis in this cohort as a sensitivity analysis.
For patients receiving radiotherapy as RDT/MDT, we conducted a single-arm survival analysis on the systemic therapy line administered concurrently with radiotherapy. For patients receiving both conventional fractionation and hypofractionation within the same phase, classification was based on the fractionation regimen with the higher total dose to better represent the primary treatment strategy and reduce confounding. In addition, we performed subgroup analyses based on the status of recurrence/metastasis, the number of systemic therapy lines, radiotherapy coverage of all lesions, and fractionation regimen.
All analyses were performed using R version 4.4.1, with statistical significance defined as p < 0.05 (two-sided tests).
Results
Baseline characteristic
This study retrospectively reviewed 411 cases of pancreatic adenocarcinoma diagnosed via pathological assessment of surgical specimens (Figure 1). Of these, 203 (49.4%) received systemic therapy for recurrence or metastasis following curative surgical resection, with sufficient follow-up data available by the cutoff date (Figure 1). Out of 203 patients, 98 (48.3%) received RDT/MDT and systemic therapy. More patients in the RDT/MDT plus systemic group received chemotherapy before recurrence or metastasis (83 vs 68) and had CA19-9 <1000 (54.1% vs 38.1%). These data indicate significant baseline differences between the two groups prior to PSM (Table 1). Following PSM, each group included 76 patients (Figure 1), and no significant differences were observed after PSM (Table 1). The distribution of RDT/MDT modalities among the 76 patients in the post-PSM RDT/MDT group is detailed in Supplemental Table 2.

Flowchart depicting patient enrollment and grouping. Of 411 pathologically confirmed pancreatic adenocarcinoma cases, 203 patients met eligibility criteria and received systemic therapy for postoperative recurrence or metastasis. Patients were divided into RDT/MDT plus systemic therapy group (n = 98) and systemic therapy alone group (n = 105). After 1:1 PSM, 76 matched pairs were included in the comparative analysis (yellow boxes). In addition, 64 patients from the pre-PSM RDT/MDT cohort with complete radiotherapy data were included in a single-arm analysis (blue boxes).
Clinical characteristics of the cohort before and after matching.

CA 19-9, carbohydrate antigen 19-9; ECOG, Eastern Cooperative Oncology Group; RDT/MDT, recurrence/metastasis-directed therapy.
Survival analysis according to whether receive RDT/MDT after PSM
As of January 2025, the median follow-up time for surviving patients was 15.0 months (95% CI: 12.8–17.5), with 127 deaths recorded in the post-PSM cohort. The median OS remained 19.9 months (95% CI: 16.3–24.1) for the RDT/MDT group and 12.2 months (95% CI: 11.1–13.1) for the systemic therapy group, with corresponding 1- and 2-year OS rates of 75.0% and 39.0%, versus 52.4% and 20.7% (p < 0.05, Figure 2). RDT/MDT conferred a survival advantage (post-PSM: hazard ratio (HR) 0.6, 95% CI: 0.4–0.8, p < 0.05).
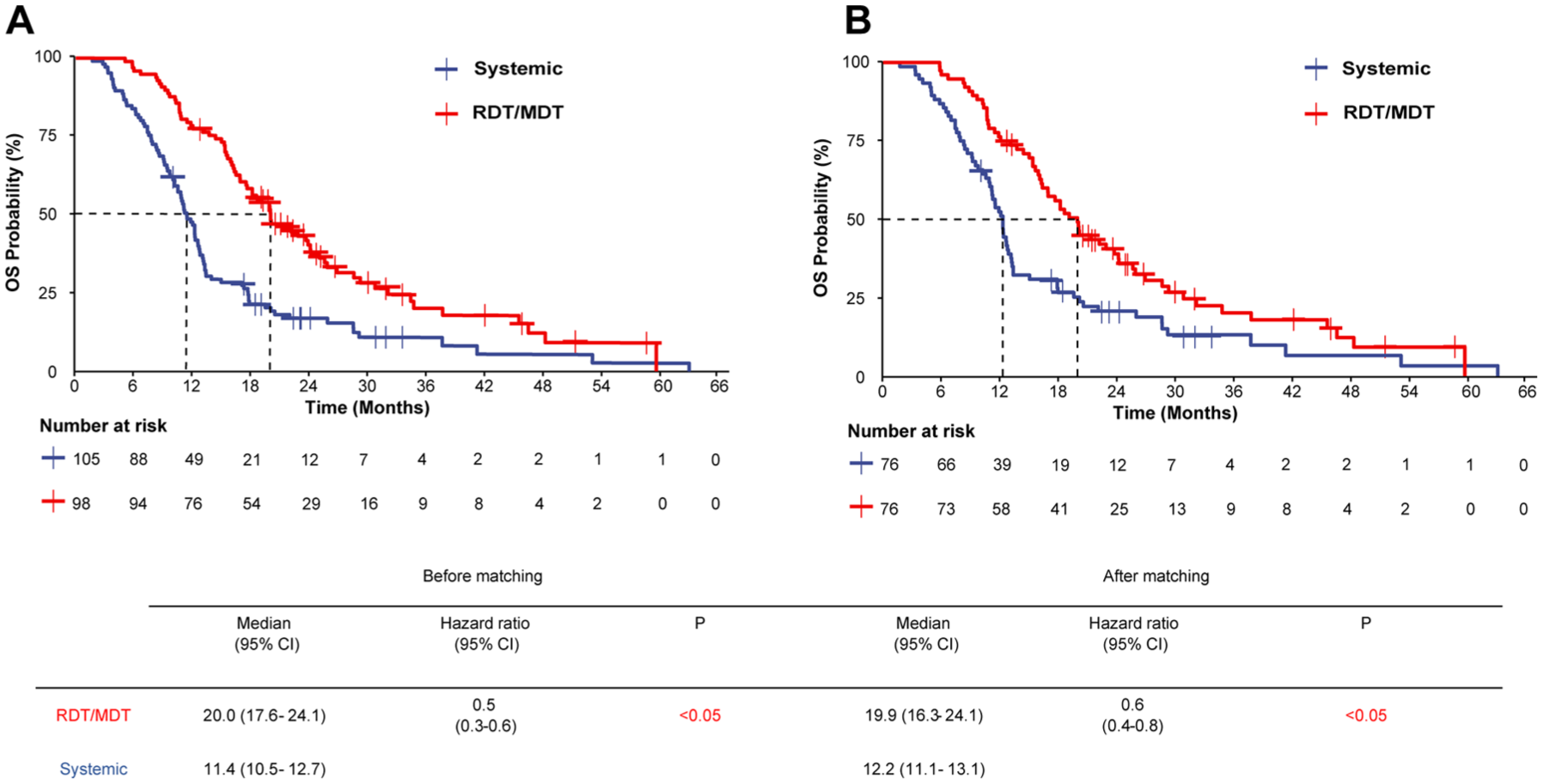
Kaplan–Meier survival analysis demonstrating the survival benefit of adding RDT/MDT to systemic therapy in patients with postoperative recurrent or metastatic pancreatic adenocarcinoma. (a) Pre-PSM cohort analysis (n = 203): The RDT/MDT plus systemic therapy group (red curve, n = 98) demonstrated significantly longer OS compared to the systemic therapy alone group (blue curve, n = 105), with median OS of 20.0 months (95% CI: 17.6–24.1) versus 11.4 months (95% CI: 10.5–12.7), respectively (HR 0.5, 95% CI: 0.3–0.6, p < 0.05). (b) Post-PSM matched cohort analysis (n = 152): After 1:1 PSM, the survival advantage persisted in the RDT/MDT group (red curve, n = 76) compared to the systemic therapy group (blue curve, n = 76), with median OS of 19.9 months (95% CI: 16.3–24.1) versus 12.2 months (95% CI: 11.1–13.1), respectively (HR 0.6, 95% CI: 0.4–0.8, p < 0.05). The consistent survival benefit observed both before and after PSM supports the robustness of the findings.
Univariate analysis identified pancreatic tail lesions, lymphovascular invasion, and RDT/MDT use as significant predictors of improved OS, while extensive metastasis was associated with worse survival. In multivariate analysis, only RDT/MDT addition remained an independent prognostic factor (HR 0.57, 95% CI: 0.39–0.84, p < 0.01, Table 2).
Univariable and multivariable Cox proportional hazards models predicting OS in 152 propensity score-matched patients.
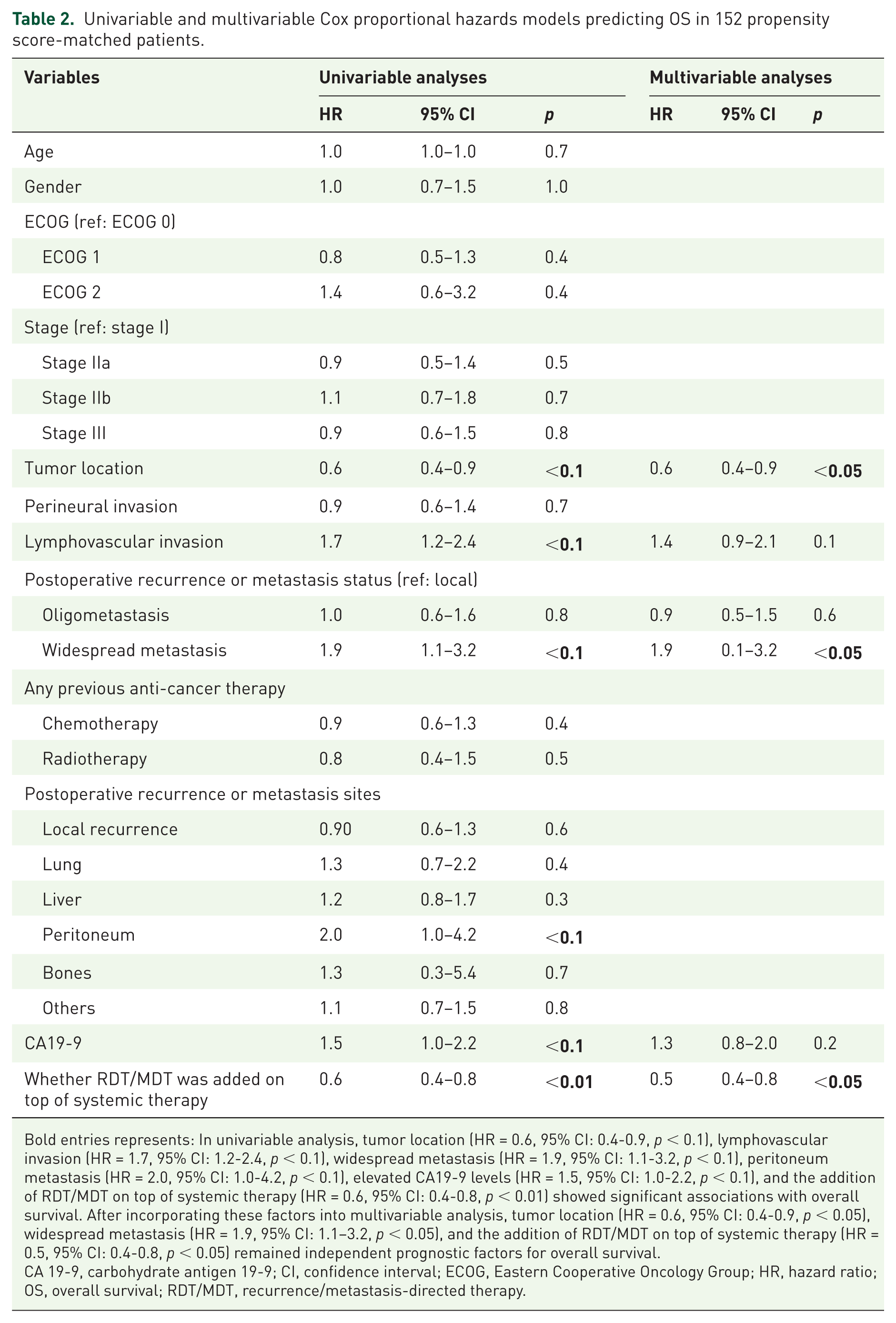
Bold entries represents: In univariable analysis, tumor location (HR = 0.6, 95% CI: 0.4-0.9, p < 0.1), lymphovascular invasion (HR = 1.7, 95% CI: 1.2-2.4, p < 0.1), widespread metastasis (HR = 1.9, 95% CI: 1.1-3.2, p < 0.1), peritoneum metastasis (HR = 2.0, 95% CI: 1.0-4.2, p < 0.1), elevated CA19-9 levels (HR = 1.5, 95% CI: 1.0-2.2, p < 0.1), and the addition of RDT/MDT on top of systemic therapy (HR = 0.6, 95% CI: 0.4-0.8, p < 0.01) showed significant associations with overall survival. After incorporating these factors into multivariable analysis, tumor location (HR = 0.6, 95% CI: 0.4-0.9, p < 0.05), widespread metastasis (HR = 1.9, 95% CI: 1.1–3.2, p < 0.05), and the addition of RDT/MDT on top of systemic therapy (HR = 0.5, 95% CI: 0.4-0.8, p < 0.05) remained independent prognostic factors for overall survival.
CA 19-9, carbohydrate antigen 19-9; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; HR, hazard ratio; OS, overall survival; RDT/MDT, recurrence/metastasis-directed therapy.
After PSM, 152 patients were classified as having local recurrence (n = 29), oligometastasis (n = 72), or widespread metastases (n = 51). RDT/MDT significantly improved survival in patients with local recurrence and widespread metastases (HR 0.4, 95% CI: 0.1–0.9, p < 0.05; HR 0.4, 95% CI: 0.2–0.7), but no benefit was observed in those with oligometastasis (Supplemental Figure 1).
Sensitivity analysis
In the sensitivity analysis of the cohort obtained after adjusting the caliper to 0.1 for PSM, the RDT/MDT group continued to demonstrate a significant survival advantage (HR 0.6; 95% CI: 0.4–0.9; p = 0.01; Supplemental Figure 2). Furthermore, the addition of RDT/MDT was significantly associated with improved OS in both univariable (HR 0.6; 95% CI: 0.4–0.0; p < 0.1) and multivariable analyses (HR 0.5; 95% CI: 0.3–0.8; p < 0.05; Supplemental Table 3).
The role of radiotherapy as MDT/RDT in survival before PSM
Among the 98 patients in the MDT/RDT group in the pre-PSM cohort, 72 underwent radiotherapy for local recurrence or metastases following surgical resection, with 8 patients receiving radiotherapy at external medical institutions and lacking complete radiotherapy-related data. The remaining 64 patients received radiotherapy at our institution with complete radiotherapy-related data. The radiation regimens received by these 64 patients can be found in Supplemental Table 4. The demographic and clinical characteristics of these 64 patients at the initiation of systemic therapy, which is concurrent with radiotherapy, are detailed in Supplemental Table 5. The specifics of the systemic therapy administered during radiotherapy are presented in Supplemental Figure 3.
As of January 8, 2025, 56 of 64 patients experienced PD or intolerable adverse events following radiotherapy combined with systemic therapy. By the same date, 45 patients had died, with a median OS of 20.1 months (95% CI: 17.5–28.5) and a 1-year OS rate of 79.5% (Figure 3). Subgroup analysis was conducted based on the status of recurrence or metastasis, number of systemic therapy lines, radiotherapy coverage of all lesions, and different fractionation regimens. Two patients who received both hypofractionated and conventional fractionated radiotherapy at the same stage were classified under the hypofractionation group. The analysis revealed superior survival outcomes in patients receiving radiotherapy as RDT or MDT during first-line therapy compared to those treated in later lines (Figure 3).

Kaplan–Meier survival analysis of patients who received radiotherapy as RDT/MDT with subgroup analyses. (a) OS of all 64 patients who received radiotherapy as RDT/MDT, showing a median OS of 20.1 months (95% CI: 17.5–28.5). (b) Survival comparison between patients whose radiotherapy covered all lesions (red line, n = 36) versus those with incomplete lesion coverage (blue line, n = 28). No significant difference was observed (HR 0.9, p = 0.8). (c) Survival comparison between conventional fractionation (blue line, n = 44) and hypofractionation (red line, n = 20) radiotherapy regimens. No significant difference was observed (HR 1.1, p = 0.7). (d) Survival comparison based on recurrence/metastasis status: local recurrence (blue line, n = 18), oligometastasis (red line, n = 25), and widespread metastases (green line, n = 21). No significant difference was observed among the three groups (p = 1.0). (e) Survival comparison based on the line of concurrent systemic therapy: first-line (blue line, n = 48), second-line (red line, n = 12), and ⩾third-line (green line, n = 3). Patients receiving radiotherapy during first-line systemic therapy showed significantly better survival outcomes (p < 0.05). Numbers at risk are shown below each survival curve at different time points.
Discussion
This study demonstrates that integrating RDT/MDT with systemic therapy significantly improves survival in patients with postoperative recurrent or metastatic pancreatic adenocarcinoma. Furthermore, compared to previous reports, radiotherapy as RDT/MDT was associated with longer PFS and OS. With 203 patients across different patterns of disease recurrence, this study provides complementary evidence to emerging prospective trials in this field.
The NCCN guidelines have yet to propose an optimal therapy strategy for local recurrence or metastasis following surgical resection, focusing primarily on systemic therapy. 28 Some retrospective and small prospective studies suggest a potential role for RDT/MDT.3,29 However, these studies did not assess outcomes in patients with widespread metastases. In our study, we included all patients with postoperative recurrence or metastasis over a 5-year period and conducted a subgroup analysis based on different recurrence and metastatic statuses. Our findings indicate that RDT/MDT significantly improved survival in patients with local recurrence and widespread metastases. However, no statistically significant OS difference was observed in the oligometastatic subgroup. Beyond the limitation of small sample size, organ-specific metastatic patterns may have caused this result. In pancreatic adenocarcinoma, liver metastases are well-established adverse prognostic factors, whereas lung metastases generally confer a relatively more favorable prognosis.6,30,31 Within the oligometastasis subgroup, the RDT/MDT group demonstrated a higher proportion of liver metastases (71.1%, 27/38) compared to the systemic therapy group (52.9%, 18/34), while exhibiting a lower proportion of lung metastases (5.3%, 2/38 vs 14.7%, 5/34). This unfavorable distribution of metastatic sites in the RDT/MDT group, compounded by the limited sample size (n = 72), may have counterbalanced the potential survival benefit of local therapy, thereby masking the treatment effect in this subgroup. These results suggest that, for patients with postoperative recurrence or metastasis, palliative local therapies may offer survival benefits, regardless of the recurrence or metastatic pattern.
The role of radiotherapy in pancreatic cancer remains controversial due to its low radiosensitivity and the complex peripancreatic anatomy.32–34 However, palliative radiotherapy has been demonstrated to provide survival benefits for patients with metastatic disease in other malignancies. In 2019, the landmark SABR-COMET trial innovatively investigated the impact of stereotactic ablative radiotherapy (SABR) on survival, oncological outcomes, toxicity, and quality of life (QOL) in patients with controlled primary tumors and oligometastatic lesions. 35 The study demonstrated that SABR significantly improved OS and doubled PFS in patients with oligometastatic disease, albeit with increased toxicity risks. Notably, with extended follow-up, the OS benefit of SABR proved durable, no new toxicities emerged, and SABR demonstrated no adverse impact on QOL. 36 Furthermore, the recent ARREST study explored the safety and feasibility of delivering SABR to all lesions in patients with polymetastatic disease (>10 metastases), demonstrating acceptable toxicity profiles. 37
Building upon these findings in other malignancies, emerging evidence supports the role of radiotherapy in recurrent or metastatic pancreatic cancer. Compared to surgery, radiotherapy offers broader coverage with fewer selection constraints and lower complication risks. 38 Several studies suggest that standard radiotherapy improves survival in patients with isolated recurrence with acceptable side effects.39–41 The recent EXTEND study also showed that radiotherapy helps in metastatic pancreatic cancer. 21 This study found that patients receiving MDT had better survival with tolerable side effects. Moreover, the study showed that MDT induced highly activated (CD25+) T cells and natural killer T cells (CD56+), increased the diversity of the T-cell receptor repertoire, and enhanced the richness of T-cell receptor-dominant motifs, which indicated MDT potentially induces systemic immunity. In addition, radiotherapy can synergize with immunotherapy by activating the immune system, inducing cytoreduction, and eliminating macroscopic immunosuppressive stroma. A phase II RCT showed improved survival with Stereotactic Body Radiation Therapy (SBRT) combined with immunotherapy compared to SBRT combined with gemcitabine (median OS: 14.9 vs 12.8 months, HR 0.69, p = 0.021). 42 In our study, the radiotherapy group’s survival outcomes also exceeded those in previous reports. However, due to the retrospective nature of this study and the presence of numerous confounding factors, we were unable to establish a control group to specifically compare outcomes between patients receiving radiotherapy versus those not receiving radiotherapy as RDT/MDT. This limits our ability to isolate radiotherapy’s independent contribution to the observed survival benefit. Future prospective, multicenter, randomized controlled trials with standardized treatment protocols and predefined patient selection criteria are warranted to definitively establish the therapeutic value in patients with postoperative recurrent or metastatic pancreatic adenocarcinoma.
This study is limited by its retrospective design, small sample size, and inability to perform PSM between patients receiving radiotherapy as RDT/MDT and those receiving only systemic therapy. Further prospective studies are needed to address these limitations.
Conclusion
This multicenter retrospective study provides preliminary evidence suggesting that adding RDT/MDT to systemic therapy may be associated with improved OS in pancreatic adenocarcinoma with postoperative recurrence and metastasis. Despite the limited sample size and retrospective design, these findings support radiotherapy as a feasible treatment option and warrant further prospective RCTs with larger cohorts to confirm these results.
Supplemental Material
sj-docx-1-tam-10.1177_17588359261417639 – Supplemental material for Impact of adding recurrence/metastasis-directed therapy to systemic therapy in postoperative recurrent or metastatic pancreatic adenocarcinoma: a retrospective cohort study
Supplemental material, sj-docx-1-tam-10.1177_17588359261417639 for Impact of adding recurrence/metastasis-directed therapy to systemic therapy in postoperative recurrent or metastatic pancreatic adenocarcinoma: a retrospective cohort study by Chenyan Zhang, Ruizhen Li, Jun Ji, Xiaofen Li and Dan Cao in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We are grateful for the understanding and cooperation of patients.
Declarations
Supplemental material
Supplemental material for this article is available online.
Artificial intelligence policy
This manuscript was prepared without the use of generative artificial intelligence (AI) tools.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
