Abstract
Background:
Breast phyllodes tumors (PT) are rare fibroepithelial tumors classified by the World Health Organization into benign, borderline, and malignant subtypes. Although surgery is the primary treatment, local recurrence remains a concern. The role of postoperative radiotherapy (PORT) in improving local control remains unclear.
Objectives:
This study aimed to investigate the effect of PORT in patients with borderline PT (BoPT) and malignant PT (MPT) after R0 resection (complete resection without tumor margins) and to identify prognostic factors related to local recurrence-free survival (LRFS) and overall survival (OS).
Design:
This was a retrospective multicenter study.
Methods:
We retrospectively evaluated patients with BoPT and MPT who underwent R0 resection between January 2002 and October 2023. Propensity score matching was used to balance the covariates between the PORT (n = 37) and non-PORT (n = 83) groups. Kaplan–Meier curves were used to estimate the 5-year LRFS and OS, while Cox regression analyses were used to identify prognostic factors. Subgroup analysis was used to assess the potential benefits of radiotherapy.
Results:
Of 480 patients, 438 (91.25%) underwent surgery alone and 42 (8.75%) received PORT. PORT did not improve the LRFS or OS. After matching, 5-year LRFS (71.42% vs 70.17%, p = 0.58) and OS (82.41% vs 80.02%, p = 0.82) were similar between the groups. Multivariate analysis of matched samples showed that axillary lymph node metastasis was significantly associated with LRFS and OS. Malignant heterologous elements were independent poor prognostic factors for LRFS. No subgroup benefited from radiotherapy.
Conclusion:
In our study, PORT did not significantly improve LRFS or OS in patients with BoPT and MPT. Key prognostic factors may guide treatment decisions in these patients.
Introduction
Breast phyllodes tumors (PT) are rare fibroepithelial neoplasms of stromal origin that account for <1% of all breast tumors. According to pathological features such as mitotic activity, cellular atypia, tumor margin invasion, and stromal composition, the World Health Organization (WHO) classifies PT into benign, borderline, and malignant. 1 The presence of malignant heterologous elements characterizes a tumor as malignant. 2 Surgery is the primary treatment. However, even after R0 resection, patients with borderline PT (BoPT) and malignant PT (MPT) remain at a high risk of local recurrence, with rates of 15%–20% and 25%–30%, respectively.3,4 Moreover, metastases occur almost exclusively in MPT, with a distant metastasis rate of 22%.5,6
Adjuvant radiotherapy has attracted attention because of its potential to improve local control. Given the high recurrence rates of BoPT and MPT, the latest National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology for Breast Cancer recommend postoperative radiotherapy (PORT). However, the effect of PORT after R0 resection remains unclear. Belkacémi et al. 4 indicated that radiotherapy significantly reduced local recurrence rates in BoPT and MPT, but showed no substantial benefit in benign cases. Yu et al. 7 found that this effect was limited to the MPT. Furthermore, the effect of radiotherapy on survival rates in PT remains controversial.8,9 The rarity of PT and conflicting evidence regarding the impact of radiotherapy necessitate further studies. Although prospective trials are difficult to conduct due to the limited number of cases, well-designed retrospective studies can offer valuable insights. We conducted a retrospective study to evaluate the effect of radiotherapy after R0 resection in patients with BoPT and MPT. We also analyzed prognostic factors and identified subgroups to provide a clinical reference for the application of PORT in patients with BoPT and MPT.
Materials and methods
Patient characteristics
We retrospectively reviewed the medical records of patients pathologically diagnosed with BoPT or MPT who underwent surgical intervention at Sun Yat-sen Memorial Hospital of Sun Yat-sen University, Sun Yat-sen University Cancer Center, or the Third Affiliated Hospital of Nanchang University between January 2002 and October 2023. We included patients who were initially diagnosed with BoPT or MPT and underwent R0 resection. R0 resection was defined as complete resection without residual tumor at the surgical margin. The exclusion criteria included patients who (1) underwent initial surgery at other hospitals without accurate baseline data, (2) received radiotherapy before surgery, (3) had a history of or concurrent breast cancer, precancerous lesions in the ipsilateral breast, or contralateral breast cancer, (4) lost to follow-up after treatment, or (5) presenting with initial distant metastasis. Patients were classified into a PORT group, comprising those who received radiotherapy following R0 resection, and a non-PORT group, comprising those who underwent R0 resection alone without subsequent radiotherapy. The Institutional Ethics Review Board approved this study (project ID: SYSKY-2024-1127-01). Written informed consent was waived due to the retrospective nature of the study. A flowchart of this process is shown in Figure S1. This study conformed to the STROBE guidelines (Supplemental File). 10 Our study was registered at ClinicalTrials.gov (NCT06780059).
Treatment
Variables and surgery
Clinical and tumor information was obtained from electronic medical records and pathological reports. The clinical data included age at diagnosis, menstrual status, surgical procedures, axillary surgery, and whether radiotherapy or chemotherapy was administered. Tumor characteristics included laterality, histopathological type, tumor size, mitoses per 10 high-power fields (HPF), Ki67 index, the presence of malignant heterologous elements, and axillary lymph node metastases. Histopathological examination confirmed the diagnosis for all patients. The classification of BoPT and MPT was based on the 2019 WHO criteria. 1 BoPT was defined as moderate stromal cellularity, 4–9 mitoses per 10 HPF, mild-to-moderate nuclear atypia, and pushing tumor borders. MPT was characterized by marked stromal overgrowth, ⩾10 mitoses per 10 HPF, severe atypia, infiltrative margins, and/or the presence of malignant heterologous elements. Axillary surgery was categorized as either performed (sentinel lymph node biopsy or axillary lymph node dissection) or not performed. The surgical procedures were classified as breast-conserving surgery (BCS) or mastectomies. BCS refers to local excision of normal breast tissue with clear margins, including wide local excision, lumpectomy, quadrantectomy, and partial mastectomy. Mastectomy involves the complete removal of unilateral breast tissue, which includes simple, radical, and modified radical mastectomies. Only patients who underwent R0 resection, ensuring that there were no residual tumors at the surgical margins, were included in this study. For patients with initially positive surgical margins, reoperation was performed until no residual tumor was detected. The surgical type, date, and pathological information were recorded after the final surgery.
Radiotherapy
PORT was administered at multiple institutions using a linear accelerator system. The clinical target volume, which included the tumor bed and the entire breast, was expanded by 5–10 mm to create the planning target volume. Treatment protocols varied across institutions and time periods, reflecting the evolution of clinical practice. Conventional fractionation involved delivering 50 Gy in 25 fractions or 50.4 Gy in 28 fractions to the breast or chest wall, with optional coverage of the axillary lymph nodes over 5–5.6 weeks. For patients who require a tumor bed boost, an additional 10 or 9.8 Gy was delivered in five fractions to improve local tumor control. Dose escalation techniques, including simultaneous integrated boosts and sequential boosts, were employed based on clinical judgment and available technology. In some cases, hypofractionated regimens were administered, delivering 42.56 Gy in 16 fractions over 3.2 weeks, with a tumor bed boost of 7.98 Gy in 3 fractions, as indicated.
Three-dimensional conformal radiotherapy or intensity-modulated radiotherapy was administered. Computed tomography (CT) simulation and dose optimization prioritized sparing of the lungs, heart, and contralateral breasts.
Outcome
Local recurrence was defined as tumor recurrence within the breast or chest wall on the ipsilateral side, including the ipsilateral axillary, internal mammary, subclavian, or supraclavicular lymph nodes. Distant metastases were defined as recurrences at other sites. Local recurrence-free survival (LRFS), the primary endpoint, was calculated as the time from the date of initial R0 resection to the date of local recurrence or the last follow-up. All recurrences were confirmed by imaging modalities, such as ultrasound, mammography, CT, magnetic resonance imaging, or positron emission tomography-CT, and ultimately validated by postoperative pathology. Overall survival (OS), the secondary endpoint, was calculated as the time from the date of initial R0 resection to all-cause mortality or the last follow-up. Each patient was followed up until death or the final follow-up date, regardless of whether local recurrence occurred, to ensure comprehensive survival data. The date of the last follow-up was determined by either the most recent telephone contact from the follow-up office or the most recently recorded hospital visit.
Statistical analyses
The significance level was 0.05 for all hypotheses, and statistical analyses were performed using R version 4.5.2. All variables were categorical and analyzed using the Chi-square test. Kaplan–Meier curves were generated to estimate LRFS and OS. A stratified log-rank test was used to compare survival between the groups. Univariate Cox proportional hazard models were used to assess the association between potential risk factors and clinical outcomes. Multivariate Cox proportional hazard models were used to investigate the independent effects of radiotherapy and other factors with p < 0.10 in the univariate models. The final models satisfied the proportional hazards assumption (p > 0.05). To explore the differential effects of radiotherapy on baseline characteristics, stratified Cox models were constructed and visualized using forest plots. Interaction tests were used to assess potential effect modifications. To further explore the potential interaction between treatment modalities, the patients were stratified into three groups based on adjuvant therapy: no radiotherapy (No RT), radiotherapy only (RT), and combined chemoradiotherapy (CRT). LRFS and OS were analyzed using Kaplan–Meier curves and multivariate Cox models. The analysis was performed on the overall cohort. The baseline characteristics and outcomes are summarized in Table S1 and Figure S1. Given the unbalanced covariates between the PORT and non-PORT groups, we estimated the propensity scores (PS) for each patient using a logistic regression model, including age, menstrual status, laterality, histopathology, tumor size, mitoses per 10 HPF, Ki67, malignant heterologous elements, lymph nodes, axillary surgery, surgery procedure, and chemotherapy. A 1:3 nearest neighbor matching method with a 0.1 caliper was used to create the matched cohort. The balance of all baseline covariates was assessed using the standardized mean difference (SMD) after matching.
Results
Patient characteristics
The baseline demographic, clinicopathological, and treatment-level characteristics are shown in Table 1. A total of 480 patients were included, of whom 438 (91.25%) underwent surgery alone and 42 (8.75%) received PORT. Regarding surgical procedures, 168 patients (35.0%) underwent mastectomy and 312 (65.0%) underwent BCS. Among those who underwent mastectomy, six patients (3.6%) underwent subcutaneous mastectomy with implant-based reconstruction. Among the 42 patients who received radiotherapy after R0 resection, 8 (19.0%) underwent three-dimensional conformal radiotherapy and 34 (81.0%) received intensity-modulated radiotherapy. Of these, 36 patients (85.7%) received conventional fractionation, while the remaining 6 (14.3%) underwent hypofractionated radiotherapy. Most patients received radiotherapy targeting the breast or chest wall, with only four (9.5%) receiving combined chest wall and regional lymph node radiotherapy. All patients successfully completed the prescribed radiotherapy doses.
Comparisons of baseline characteristics between the PORT and non-PORT groups before and after PSM.

Values are n (%).
BCS, breast-conserving surgery; HPF, high-power fields; PORT, postoperative radiotherapy; PSM, propensity score matching; SMD, standardized mean difference.
The median age of the entire cohort was 43 years (interquartile range: 36–50 years). Compared with patients in the non-PORT group, those in the PORT group were more frequently diagnosed with MPT (90.5% vs 29.2%, p < 0.001) and had larger tumor sizes (66.7% vs 41.3%, p = 0.003). They also exhibited more aggressive pathological features, including higher mitotic counts and elevated Ki67 levels (p < 0.05).
Notably, patients undergoing PORT were significantly more likely to receive axillary surgery (p < 0.001) and mastectomy (p < 0.001). In addition, a higher proportion of these patients received chemotherapy (p < 0.001). Given the significant baseline differences, PS matching (PSM) was used to balance the distribution of most of the demographic and clinical characteristics between the two groups. After 1:3 nearest-neighbor matching, the PORT and non-PORT groups were well-balanced, with no statistically significant differences observed across all included covariates. All SMDs for baseline characteristics were reduced, and the majority fell well below 0.1, indicating successful covariate balance between the two groups.
Survival
The median follow-up duration was 65 months (35–105 months) for the entire cohort, 46 months (23–69.5 months) in the PORT group, and 67 months (37.25–105.75 months) in the non-PORT group. The median survival was not reached. For the entire cohort, the non-PORT group exhibited a slightly higher 5-year LRFS; however, this difference was not statistically significant (77.79% vs 72.7%, log-rank χ2 = 0.033, p = 0.86; Figure 1(a)). The non-PORT group showed a higher 5-year OS (94.21% vs 79.51%, log-rank χ2 = 6.74, p = 0.0094; Figure 1(b)).

LRFS and OS of patients with borderline and malignant phyllodes tumors are displayed as Kaplan–Meier curves stratified by radiotherapy. (a) LRFS curves for non-PORT versus PORT groups before PSM. (b) OS curves for non-PORT versus PORT groups before PSM. (c) LRFS curves for non-PORT versus PORT groups after PSM. (d) OS curves for non-PORT versus PORT groups after PSM.
After matching, the 5-year LRFS was 71.42% (95% confidence interval (CI): 61.22%–83.33%) for the non-PORT group and 70.17% (95% CI: 52.96%–93.7%) for the PORT group. This adjustment narrowed the difference, and the log-rank test indicated no significant difference (log-rank χ2 = 0.034, p = 0.85; Figure 1(c)). Similarly, the 5-year OS rates after matching were 82.41% for the non-PORT group and 80.02% for the PORT group, with no significant difference observed (log-rank χ2 = 0.019, p = 0.89; Figure 1(d)).
Before matching, univariate and multivariate Cox regression models (Table 2) revealed that malignant heterologous elements (LRFS: hazard ratio (HR) = 2.74, 95% CI: 1.28–5.83, p = 0.009; OS: HR = 4.95, 95% CI: 1.76–13.92, p = 0.002) and positive lymph nodes (LRFS: HR = 4.34, 95% CI: 1.32–14.26, p = 0.016; OS: HR = 19.01, 95% CI: 2.92–123.63, p = 0.002) were independent risk factors for both LRFS and OS. Notably, MPT was also associated with shorter OS compared with BoPT (HR = 7.72, 95% CI: 2.09–28.47, p = 0.002).
Univariate and multivariate Cox proportional hazards model analyses of LRFS and OS before PSM.

Values are n (%).
BCS, breast-conserving surgery; CI, confidence interval; HPF, high-power fields; HR, hazard ratio; LRFS, local recurrence-free survival; OS, overall survival; PSM, propensity score matching.
In the matched cohort, univariate Cox regression analysis (Table 3) identified positive lymph node status (HR = 16.94, 95% CI: 4.58–62.7, p < 0.001) and malignant heterologous elements (HR = 3.83, 95% CI: 1.56–9.43, p = 0.004) as significant predictors of poorer LRFS. For OS, univariate analysis also indicated potential associations with age (HR = 3.22, 95% CI: 1.07–9.73, p = 0.038), malignant heterologous elements (HR = 3.88, 95% CI: 1.27–11.79, p = 0.017), and positive lymph node status (HR = 20.34, 95% CI: 4.06–101.93, p < 0.001). After adjustment in multivariate Cox analysis, positive axillary lymph node status (HR = 24.93, 95% CI: 5.95–104.52, p < 0.001) and malignant heterologous elements (HR = 4.31, 95% CI: 1.71–10.84, p = 0.002) remained independent predictors of poor LRFS. For OS, only positive lymph node status remained statistically significant (HR = 67.95, 95% CI: 8.81–524.13, p < 0.001), while no other variables reached significance in the multivariate model. No significant effect of adjuvant radiotherapy on LRFS or OS was observed in the matched cohort.
Univariate and multivariate Cox proportional hazards model analyses of LRFS and OS after PSM.
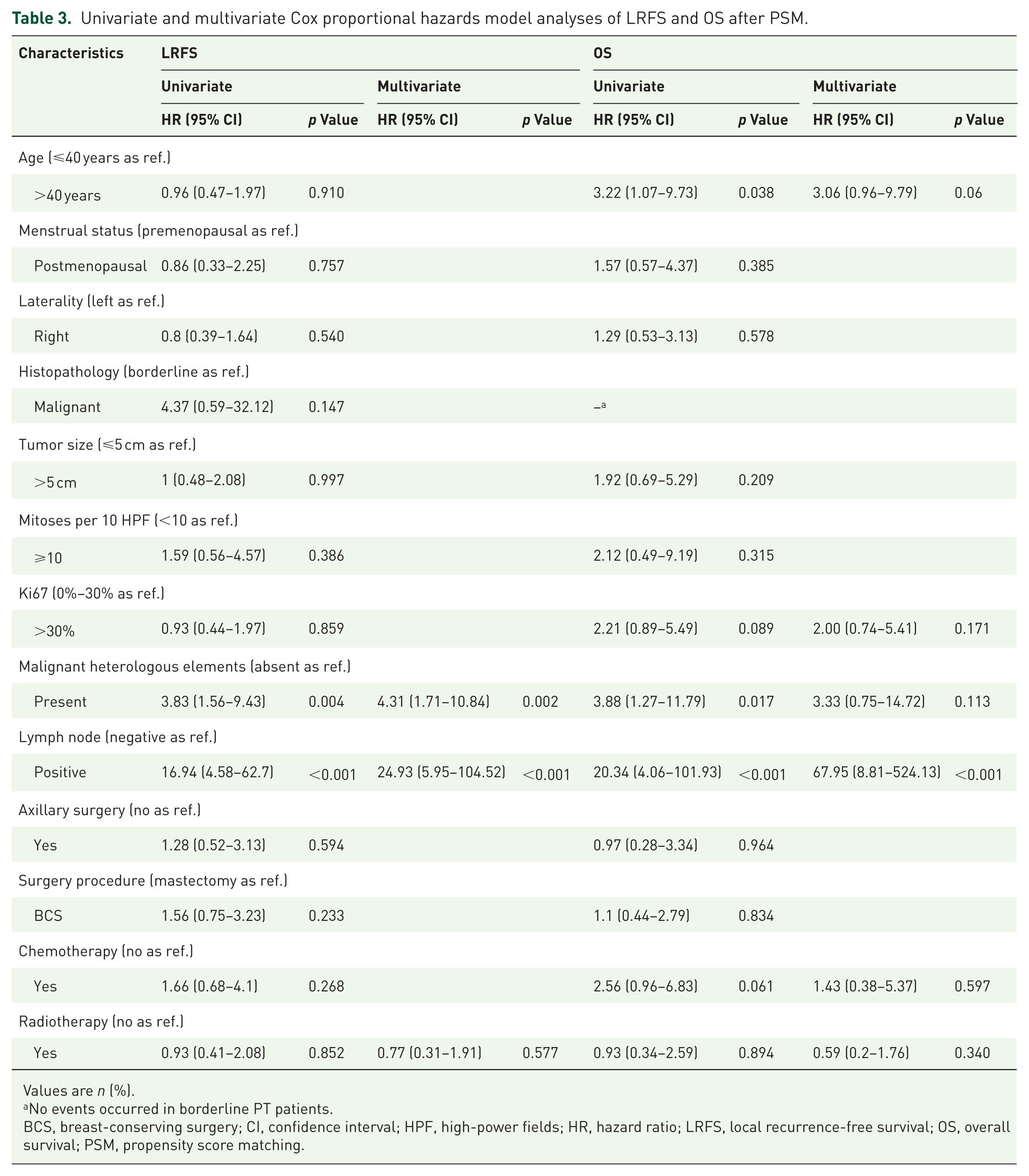
Values are n (%).
No events occurred in borderline PT patients.
BCS, breast-conserving surgery; CI, confidence interval; HPF, high-power fields; HR, hazard ratio; LRFS, local recurrence-free survival; OS, overall survival; PSM, propensity score matching.
Subgroup
Subgroup analyses were conducted to further investigate the effect of PORT on prognosis across various subgroups stratified by clinicopathological factors. The results showed that none of the subgroups benefited from PORT before or after matching (Figures S2–S5).
Recurrence/progression pattern
After treatment, 106 recurrences (22.1%) occurred (Table 4). In the PORT group, 42 patients were treated, of whom 8 (19.0%) developed local failure and 7 (16.7%) experienced distant failure. In the non-PORT group, 98 patients had local recurrence, and 17 patients had distant metastasis. Local recurrence was the predominant failure pattern in both groups, accounting for 19.0% and 22.4%, respectively. The lungs (n = 19) were the most frequent sites of metastasis, comprising 79.2% of all distant metastases, followed by bones (n = 7), liver (n = 2), adrenal glands (n = 2), and soft tissues (n = 2). Rare metastases were observed in the brain (n = 1) and the heart (n = 1).
Recurrence/progression pattern in patients with borderline and malignant phyllodes tumors.

Values are n (%).
PORT, postoperative radiotherapy.
Exploratory analysis of patients receiving both radiotherapy and chemotherapy
Among the entire cohort (n = 480), 10 patients received both radiotherapy and chemotherapy (CRT group), 32 received radiotherapy only (RT group), and 438 underwent surgery without radiotherapy (No RT group), including those who also received chemotherapy. Baseline characteristics differed significantly across groups in histopathology, mitosis per 10 HPF, Ki67, and lymph nodes, as well as other variables (Table S2).
Kaplan–Meier analysis (Figure S6) showed no significant difference in 5-year LRFS among the three groups (log-rank χ2 = 1.127, p = 0.57). However, 5-year OS differed significantly (log-rank χ2 = 7.556, p = 0.03), with the CRT group showing the poorest outcomes.
In multivariable Cox regression, CRT was not independently associated with either LRFS (HR = 0.29, 95% CI: 0.04–2.17, p = 0.228) or OS (HR = 0.32, 95% CI: 0.05–1.97, p = 0.216; Table S3).
Discussion
To explore the adjuvant treatment strategies for PT, it is necessary to briefly review the effects of adjuvant chemotherapy, as it has shown limited efficacy in patients with PT. Existing studies11,12 and our multivariate analysis confirmed no survival benefit in MPT, consistent with our previous findings. 13 Chemotherapy may only be considered for extensive tumors (e.g., chest wall invasion). 14 By contrast, a growing number of patients with PT are receiving adjuvant radiotherapy, 15 yet its role remains controversial. Two meta-analyses indicated that radiotherapy primarily influences local control in MPT.7,16 Another real-world study from the Netherlands reported no significant impact of radiotherapy on local recurrence in patients with PT, which is consistent with our conclusion. 17 Notably, none of the above studies found a correlation between radiotherapy and improved long-term survival. Our study reinforces this hypothesis by identifying local recurrence as the most frequent cause of treatment failure, suggesting that current treatment strategies may be improved to better control local recurrence.
To further investigate the potential interaction between chemotherapy and radiotherapy, we conducted a stratified analysis, categorizing the patients into three groups: No RT, RT, and CRT. Although the Kaplan–Meier analysis showed poorer OS in the CRT group, this result was likely confounded by baseline differences. Patients receiving CRT tended to have more aggressive pathological features, including tumors >5 cm in size, malignant heterologous elements, a high Ki67 index, and lymph node involvement. After adjustment for these factors in the multivariate analysis, CRT was not independently associated with OS or LRFS. Given the small number of CRT patients (n = 10), these results should be interpreted with caution and regarded as exploratory.
Several studies have shown that patients with PTs and positive surgical margins have a higher risk of recurrence. 18 Although the NCCN guidelines recommend wide excision with margins ⩾1 cm for BoPT or MPT, 19 the relationship between the width of negative margins and local recurrence remains controversial.7,20 Even if wide negative margins are achieved, the local recurrence rate after surgery can still reach up to 40%. 21 Consequently, many researchers are exploring the role of radiotherapy in PT with different margin statuses. In our study, owing to the limitations in the pathology reports, the specific width of the negative margins was not consistently available, as most reports only indicated whether the margins were negative or positive. The results indicated that for patients with negative margins, radiotherapy had no significant effect on survival and recurrence between those who underwent mastectomy and BCS. BCS is recommended when sufficient negative margins are achieved to ensure good cosmetic results. In a prospective single-arm study, Barth et al. 22 reported no local recurrence after radiotherapy in 46 patients who underwent BCS. However, prospective randomized evidence is lacking. Although BCS is preferred because of its cosmetic and functional advantages, large tumor sizes often limit its feasibility. Zhang et al. 23 suggested that 44.3% of MPT exceeded 5 cm, making it anatomically difficult to achieve sufficient negative margins while preserving the breast. A similar situation was reported by Rumyantseva et al., 24 where a giant tumor required mastectomy and lymph node assessment. In such cases, large tumors often require aggressive or multimodal treatment strategies, including surgery, radiotherapy, and chemotherapy. However, both our findings and those of Zhang et al. indicate that these intensified approaches do not translate into improved survival outcomes, underscoring the need for individualized risk-adapted treatment in this subgroup.
PT predominantly metastasizes through hematogenous routes, with the lungs, bones, and liver being the most common metastatic sites. The risk of distant metastasis can reach up to 22%. 14 This study identified a notable difference in distant metastasis rates between the two groups (p = 0.001); however, this difference disappeared in the MPT-specific analyses after excluding the patients with BoPT (Table S1). Moreover, few studies have reported a correlation between axillary lymph node metastasis and PT. One study indicated that only 5% of patients with PT exhibited axillary lymph node metastasis. 25 Based on the rarity of lymph node metastasis, guidelines recommend omitting axillary surgery. However, our findings showed that positive axillary lymph nodes significantly increased the risk of recurrence and death in multivariate Cox regression analysis, a finding that warrants further investigation. We suggest that fine-needle aspiration biopsy should be considered for patients with clinically enlarged lymph nodes to determine the necessity of axillary dissection. In addition, previous studies have revealed that malignant heterologous elements increase the risk of local recurrence and predict distant metastasis in MPT. 26 Malignant heterologous differentiation, including angiosarcoma, chondrosarcoma, leiomyosarcoma, osteosarcoma, and rhabdomyosarcoma, is uncommon in MPT and has only been described in small retrospective studies and case reports.27,28 In this study, its presence was an independent predictor of local recurrence. Notably, in univariate analysis, malignant heterologous elements were also significantly associated with OS (HR = 3.88, 95% CI: 1.27–11.79, p = 0.017), suggesting a potential association with poorer survival outcomes. To our knowledge, this is the first study to demonstrate the possible association between malignant heterologous differentiation and OS in patients with MPT. Although this association was not statistically significant in multivariate analysis (p = 0.113), the observed trend highlights the need for further research into the potential role of malignant heterologous differentiation in MPT prognosis. The strong association between these elements and local recurrence suggests a potential rationale for sarcoma-type chemotherapy similar to those used in osteosarcoma protocols. 29
This study has several limitations. First, owing to its retrospective design spanning 21 years, the randomization of treatments was impossible. However, we improved the objectivity of the results by using PSM. Second, some information was not reported. Since this study primarily focused on local treatment, we supplemented the details of radiotherapy (e.g., techniques and doses). However, the sequences of chemotherapy and radiotherapy were not standardized, which could introduce a potential bias. Moreover, information on radiotherapy toxicity and detailed margin widths was not fully collected. Third, the rarity of PT and the limited application of radiotherapy resulted in a relatively small sample size. This may have constrained our ability to identify the subgroups that could benefit from radiotherapy, necessitating a cautious interpretation of the subgroup analysis results. Despite this, our study is the first to explore clinicopathological subgroups (tumor size, mitosis, Ki67, and heterologous elements) and identify axillary metastasis and heterologous differentiation as key prognostic biomarkers. Larger prospective trials are required to validate these findings and translate them into personalized strategies.
Conclusion
Based on our results, radiotherapy after R0 resection did not improve LRFS or OS in patients with BoPT or MPT. Further prospective randomized studies are required to confirm our findings.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251387556 – Supplemental material for Effects of radiotherapy in borderline and malignant phyllodes tumors after R0 resection: a propensity score analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359251387556 for Effects of radiotherapy in borderline and malignant phyllodes tumors after R0 resection: a propensity score analysis by Bingbing Ma, Xiaojun Li, Zilin Zhuang, Mengjia Han, Yulu Zhang, Feng Ye, Zhuofei Bi, Wen Deng, Jiahao Zhang, Yaping Yang and Yan Nie in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors acknowledge and express their deepest gratitude to all patients of this study.
Declarations
Availability of data and materials
The data that support the findings of this study are available from the corresponding author, Yan Nie, upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
