Abstract
Background:
The prognosis of centrally located hepatocellular carcinoma (cl-HCC) was unsatisfactory when treated with surgery alone. Adjuvant radiotherapy (RT) and neoadjuvant radiotherapy have been reported to correlate with better disease-free survival (DFS) and overall survival (OS) in cl-HCC.
Objectives:
To compare the survival outcomes and safety of neoadjuvant intensity-modulated radiotherapy (Neo-IMRT) plus surgery with surgery plus adjuvant IMRT (Adj-IMRT) in patients with cl-HCC.
Design:
This was a retrospective cohort study.
Methods:
Data from previous prospective studies were analyzed. Propensity score matching (PSM) was used to balance the baseline characteristics between the Neo-IMRT group and the Adj-IMRT group.
Results:
Before PSM, the Neo-IMRT group had a higher proportion of larger tumor size (>5 cm), multiple nodes, and higher α-fetoprotein levels than the Adj-IMRT group (all p < 0.05). There were no significant differences in 5-year OS (69.1% for the Neo-IMRT group vs 72.2% for the Adj-IMRT group), DFS (41.0% for vs 51.6%), or recurrence pattern (all p > 0.05). The Neo-IMRT group had less leukopenia (50.0% vs 68.7%), more bilirubin increased (42.1% vs 22.4%), and wider surgical margins (median, 1.0 vs 0 cm; all p < 0.05). After PSM, 31 patients were matched in each group. The 5-year OS was 75.9% for the Neo-IMRT group and 66.3% for the Adj-IMRT group (p = 0.532), and the 5-year DFS was 45.8% and 46.0% (p = 0.970), respectively. There were no significant differences in recurrence pattern, radiation-related acute adverse events, operative variables, or postoperative complications, except that the Neo-IMRT group had wider surgical margins (p < 0.001).
Conclusion:
Neo-IMRT plus surgery was as effective and safe as surgery plus Adj-IMRT for patients with cl-HCC. Further prospective trials are warranted to confirm the findings.
Keywords
Introduction
Liver cancer is the sixth most common malignancy, causing over 780,000 deaths worldwide in 2020. 1 Centrally located hepatocellular carcinoma (cl-HCC) is a special subgroup of liver cancer. Currently, hepatectomy is the mainstay of treatment for cl-HCC.2,3 However, the prognosis of patients with cl-HCC is unsatisfactory when treated with surgery alone. The cumulative rate of recurrence was as high as 70%–80%, and the 5-year overall survival (OS) was about 40%.3–6 Various adjuvant and neoadjuvant therapies have been tested to reduce recurrence and improve long-term survival, including local and systemic therapies such as transarterial chemoembolization, targeted therapy, and immunotherapy, but none have been recognized as standard treatment.7–9
With the advances in radiation technology and the promotion of comprehensive treatment based on multidisciplinary team (MDT) discussions, the application of radiotherapy (RT) in HCC has become more prevalent over the past decades.10,11 Radiotherapy has also been explored as adjuvant or neoadjuvant therapy for HCC. Previous retrospective and prospective studies suggested that adjuvant RT could increase disease-free survival (DFS) and OS of patients with cl-HCC.12,13 In addition, we recently reported an elevation in OS and DFS by applying neoadjuvant RT for patients with cl-HCC in a prospective phase II trial. 14 Thus, these data have shown that both adjuvant RT and neoadjuvant RT are beneficial in treating cl-HCC. But which modality is superior remains unknown in terms of survival, recurrence, adverse events, or postoperative complications.
Evidence guiding the selection between neoadjuvant RT and adjuvant RT for HCC is scarce. Two retrospective studies analyzed data from the Surveillance, Epidemiology, and End Results (SEER) program and found a better OS in patients who received preoperative RT than those who received postoperative RT.15,16 However, these studies were limited by the retrospective study design, a lack of detailed information regarding tumors, the reasons for applying RT, as well as treatment-related adverse events. In this study, we aim to compare the therapeutic effects and safety profiles of neoadjuvant and adjuvant intensity-modulated radiotherapy (Adj-IMRT) combined with hepatectomy in cl-HCC. Data from two previous prospective phase II trials were reviewed and analyzed to compare prognosis, recurrence pattern, and treatment-related adverse events between the two modalities. To make outcomes comparable, one-to-one propensity score matching (PSM) was performed to balance the basic clinical characteristics.
Materials and methods
Patients
Data from patients who received neoadjuvant IMRT (Neo-IMRT) followed by hepatectomy (n = 38, Neo-IMRT group) and those who received hepatectomy followed by Adj-IMRT (n = 76, Adj-IMRT group) were prospectively collected as reported in the two previous studies,12,14 and retrospectively analyzed in this research. All patients were diagnosed with cl-HCC, which was defined as “a carcinoma adjoined hepatic portals, less than 1 cm from major vascular structures (including the main portal branches, the main trunks of the hepatic veins, as well as the inferior vena cava) which are usually located in Couinaud segments Ⅰ, Ⅳ, Ⅴ, Ⅷ, or at the junction of the central segments.”2,4 Other detailed information about inclusion and exclusion criteria was described in each corresponding study.12,14 The reporting of this study conforms to Strengthening the reporting of observational studies in epidemiology (STROBE; Supplemental Table 1). 17
Treatment
All patients in both groups were treated by the same surgical team and radiotherapy team. MDT discussion was held before carrying out IMRT and hepatectomy. The procedures of IMRT and hepatectomy have been described previously.12,14 Briefly, for patients in the Neo-IMRT group, the prescription dose to 95% planned target volume (PTV) was 50–60 Gy in 25–30 fractions over 5–6 weeks, and surgery was performed 4–12 weeks post-IMRT. For patients in the Adj-IMRT group, hepatectomy was the primary treatment, followed by adjuvant radiotherapy 4–12 weeks after resection. The prescription dose to 95% PTV was also 50–60 Gy in 25–30 fractions over 5–6 weeks, mainly depending on the dose constraints of organs at risk. Additional details regarding surgical procedures and radiotherapy target definition, treatment planning, and dose delivery were described in the previous studies.12,14
Follow-up, toxicity, and evaluation
After hepatectomy (Neo-IMRT group) or the completion of IMRT (Adj-IMRT group), patients were scheduled for follow-up visits every 3 months during the first 2 years, every 6 months during the next 3 years, and annually thereafter. Follow-up examinations included serum α-fetoprotein (AFP), liver biochemistry, blood routine, coagulation test, chest radiography, and CT and/or MRI of the abdomen.
Radiation-related acute toxicities were evaluated weekly during RT and monthly after RT for 3 months and were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events (version 4.03). Operative complications were graded according to the Clavien–Dindo classification system. 18 Tumor responses were evaluated according to the modified Response Evaluation Criteria in Solid Tumors guidelines. 19 A narrow margin was defined as a <1 cm macroscopic distance from the edge of the tumor to the line of transection. 12
PSM and statistical analysis
PSM was performed using the MatchIt package in R to balance the differences in the basic clinical characteristics between groups. Factors used for matching included gender, age, AFP level, lesion number, diameter of lesion, and distance to major vessel. Patients were matched at a 1:1 ratio without replacement using 1:1 nearest-neighbor matching. The caliper was set to 0.20.
The primary endpoint was a 5-year OS. The secondary endpoints were 5-year DFS, recurrence pattern, and treatment-related adverse events. OS was calculated as the period from the date of treatment initiation to death from any cause or the last follow-up. DFS was defined as the time interval between the date of surgery and that of the first detection of recurrence, death from any cause, or the last follow-up. Intrahepatic recurrence-free survival (IHRFS) and distant metastasis-free survival (DMFS) were defined as the time interval between the date of surgery and that of the first detection of intrahepatic recurrence or distant metastasis, as appropriate, death from any cause, or the last follow-up.
Continuous variables are expressed as median (range) or mean ± standard deviation, as appropriate. Categorical variables are expressed as n (proportion). The Kaplan–Meier estimator was performed to calculate the median survival time and survival rates (DFS and OS). Survival curves were compared using the Log-rank test. p < 0.05 was considered significant. All statistical analyses were performed using SPSS Statistics (v24.0; IBM, Armonk, NY, USA) and R (v4.2.1; R Foundation, Vienna, Austria).
Results
Patient characteristics
Before PSM, patients in the Neo-IMRT group (n = 38) had larger baseline tumor sizes (>5 cm), more multiple tumors (n > 1), and higher AFP levels (⩾400 ng/ml) than those in the Adj-IMRT group (n = 76; Table 1). After PSM, there were 31 patients in each group, and all baseline clinical and pathological features were well-balanced (Table 1, Supplemental Figure S1). The median tumor size in the Neo-IMRT group significantly decreased from 5.3 cm at baseline to 3.6 cm before surgery (p = 0.033).
Patient characteristics.

Values are n (%) or median (range).
Adj-IMRT, adjuvant intensity-modulated radiotherapy; AFP, α-fetoprotein; AJCC, American Joint Committee on Cancer; BCLC, Barcelona Clinic Liver Cancer; Neo-IMRT, neoadjuvant intensity-modulated radiotherapy; PSM, propensity score matching; S, surgery.
Survival outcomes
The median follow-up duration was 45.8 months for the Neo-IMRT group and 70.0 months for the Adj-IMRT group. Before PSM, the Neo-IMRT group had a close 5-year OS with the Adj-IMRT group (69.1% vs 72.2%, p = 0.339), and a numerically lower 5-year DFS (41.0% vs 51.6%, p = 0.076), but there was no statistically significant difference (Figure 1(a) and (b)).

Kaplan–Meier analysis of survival. (a) OS before PSM. (b) DFS before PSM. (c) OS after PSM. (d) DFS after PSM.
After PSM, both OS and DFS were similar between the two groups (Figure 1(c) and (d), Supplemental Figure S2), as well as in all subgroups (Supplemental Figure S3). The median OS was not reached in either group. The 1-, 3-, and 5-year OS was 100%, 83.5%, and 75.9% in the Neo-IMRT group, and 96.8%, 80.6%, and 66.3% in the Adj-IMRT group (p = 0.532), respectively. The median DFS was 45.0 months for the Neo-IMRT group, and 46.6 months for the Adj-IMRT group, and the 1-, 3-, and 5-year DFS were 80.6%, 61.3%, and 45.8% in the Neo-IMRT group, and 83.9%, 62.8%, and 46.0% in the Adj-IMRT group, respectively (p = 0.970).
Pattern of recurrence
Before PSM, a total of 20 (52.6%) patients in the Neo-IMRT group and 39 (51.3%) patients in the Adj-IMRT group developed recurrence (p = 0.895). There was no significant difference in the proportion of early recurrence (<2 years), intrahepatic, or extrahepatic recurrence (all p > 0.05; Table 2).
Pattern of recurrence.
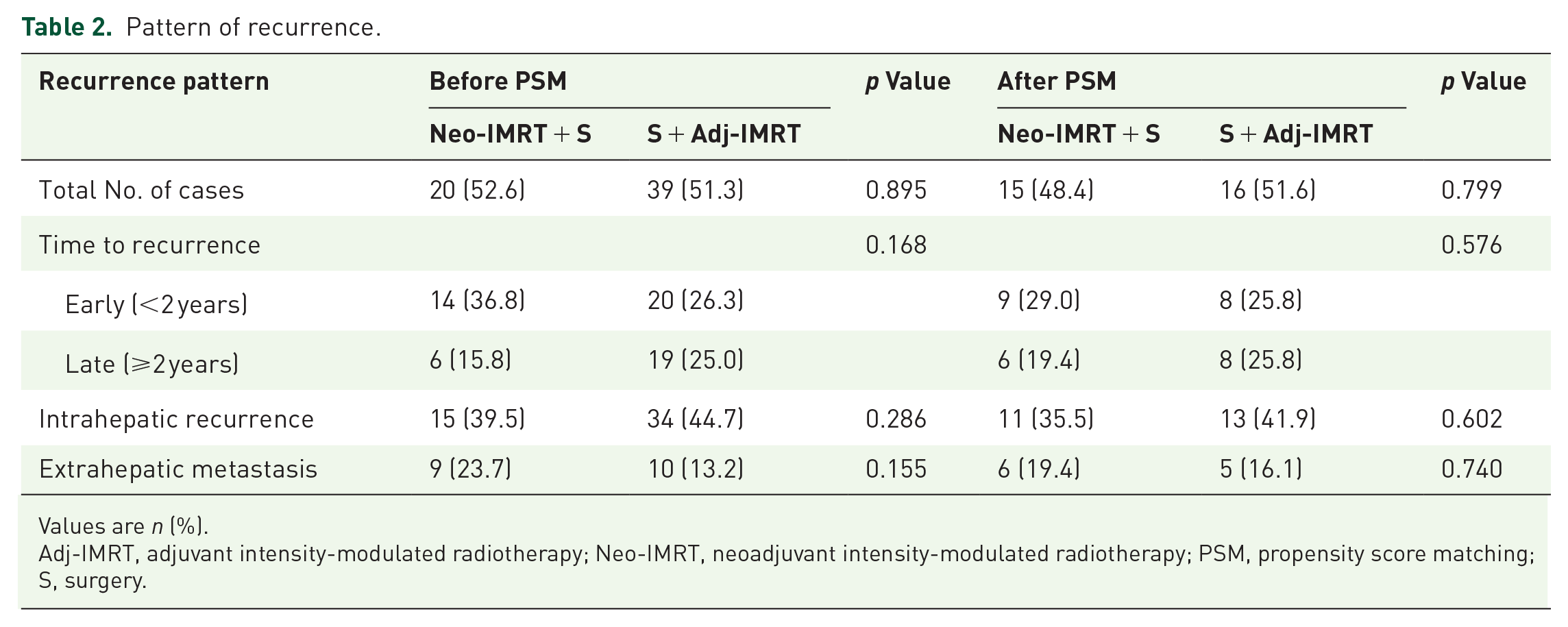
Values are n (%).
Adj-IMRT, adjuvant intensity-modulated radiotherapy; Neo-IMRT, neoadjuvant intensity-modulated radiotherapy; PSM, propensity score matching; S, surgery.
After PSM, 15 (48.4%) patients in the Neo-IMRT group and 16 (51.6%) in the Adj-IMRT group experienced recurrence. No, except one patient in the Neo-IMRT group showed marginal recurrence, which was defined as recurrence within 2 cm from the resection plane. There was no significant difference in the proportion of early recurrence (29.0% vs 25.8%, p = 0.576), intrahepatic (35.5% vs 41.9%, p = 0.602), or extrahepatic recurrence (19.4% vs 16.1%, p = 0.740; Table 2). There was neither a significant difference in IHRFS (p = 0.941) nor in DMFS (p = 0.562; Supplemental Figure S4).
Radiation-related acute adverse events
Among all patients, 33 (86.8%) in the Neo-IMRT group and 69 (90.8%) in the Adj-IMRT group experienced radiation-related acute adverse events, most of which were grade I–II (78.9% in the Neo-IMRT group and 80.3% in the Adj-IMRT group; Table 3). Grade 3 radiation-related acute adverse events occurred in 3 (7.9%) patients in the Neo-IMRT group and 8 (10.5%) patients in the Adj-IMRT group (p = 0.517). No grade 4 or 5 acute toxicities or radiation-induced liver disease occurred in either group. In the Neo-IMRT group, no patient experienced surgery delay or was unable to undergo surgery due to radiation-related adverse events. Before PSM, the Neo-IMRT group had a lower rate of leukopenia (50.0% vs 68.7%, p = 0.039), and a higher rate of bilirubin increase than the Adj-IMRT group (42.1% vs 22.4%, p = 0.047), while there were no significant differences in the proportion of other adverse events. After PSM, we found no significant differences in the proportions of all radiation-related acute adverse events (Table 3).
Acute radiation-related adverse events.

Values are n (%). For n < 5, statistical analysis was not performed.
Adj-IMRT, adjuvant intensity-modulated radiotherapy; ALT, alanine aminotransferase; AST, aspartate aminotransferase; Neo-IMRT, neoadjuvant intensity-modulated radiotherapy; PSM, propensity score matching; S, surgery.
Operative variables and complications
The Neo-IMRT group had a wider median surgical margin before PSM, as well as after PSM (1.0 vs 0 cm, p < 0.001), apart from which all operative variables were comparable between the two groups (Table 4). There was no significant difference in hospital stay, type of resection, and volume of blood loss between the two groups either before or after PSM (all p > 0.05).
Operative variables and complications.

Values are n (%).For n < 5, statistical analysis was not performed.
According to the Clavien–Dindo classification.
Adj-IMRT, adjuvant intensity-modulated radiotherapy; ICG-R15, ICG retention test after 15 min; Neo-IMRT, neoadjuvant intensity-modulated radiotherapy; PSM, propensity score matching; RBC, red blood cell; S, surgery.
Postoperative complications occurred in 13 (34.2%) patients in the Neo-IMRT group and 22 (28.2%) patients in the Adj-IMRT group before PSM (p = 0.566; Table 4). Complications were graded according to the Clavien–Dindo classification, and mainly included grade I–II complications, while grade IIIa complications occurred only in one patient in the Neo-IMRT group and two patients in the Adj-IMRT group. No grade IIIb or higher operative complications were observed in either group. There was no significant difference in postoperative complications between the two groups either before or after PSM (all p > 0.05).
Discussion
This study aimed to compare the efficacy and safety of Neo-IMRT plus surgery versus surgery plus Adj-IMRT in patients with cl-HCC. The results showed that both treatment modalities were similarly effective in terms of OS and DFS, with no significant differences observed between the two groups after PSM. However, the Neo-IMRT group achieved wider surgical margins, which may have contributed to the numerical trend toward higher long-term survival rates.
Previous prospective studies have shown that neoadjuvant or adjuvant RT could improve the prognosis of patients with HCC and portal vein tumor thrombosis or cl-HCC.12,14,20,21 However, direct comparisons between neoadjuvant and adjuvant RT are scarce. Two retrospective studies based on the SEER database suggested that preoperative RT was associated with better OS than postoperative RT.15,16 These findings are inconsistent with our results, likely due to the limitations of the SEER-based studies, such as the lack of detailed information on tumor location and treatment-related adverse events. In contrast, our study used prospectively collected data and balanced baseline characteristics using PSM, providing more reliable evidence.
An important finding of this study was that the Neo-IMRT group achieved significantly wider surgical margins compared to the Adj-IMRT group (median, 1.0 vs 0 cm, p < 0.001). This advantage may be attributed to the reduction in tumor size following neoadjuvant RT. In the Neo-IMRT group, the median tumor size decreased from 5.3 cm at baseline to 3.6 cm before surgery (p = 0.033). Wider surgical margins are generally associated with better long-term survival outcomes in HCC. Therefore, the wider margins achieved in the Neo-IMRT group may have contributed to the numerical trend toward higher survival rates, although this did not reach statistical significance.22–24 Recently, D’Alessio et al. 25 reported that pathological response following neoadjuvant immune therapy was significantly associated with recurrence-free survival (RFS) in patients with HCC. We also analyzed the relationship between pathological response and recurrence time in our study but found no significant correlation. This may be due to the small sample size of our study. Additionally, it may be because different neoadjuvant treatment regimens were used. Radiotherapy is a local treatment modality, and its mechanism of action differs from systemic therapy.
We selected factors such as gender, age, AFP level, lesion number, distance to a major vessel, and diameter of lesion for the matching in PSM, as these are all widely recognized prognostic factors.26–28 After matching, there were no significant differences in all baseline characteristics between the two groups, indicating that it is feasible to select these factors for matching. Some other factors, such as the hepatitis background, were considered not to be factors contributing to the differences in our data. This is because the characteristics of these factors were quite similar between the two groups even before matching.
Both Neo- and Adj-IMRT were found to be safe and feasible, with no significant differences in radiation-related acute adverse events or postoperative complications between the two groups. The Neo-IMRT group had a lower rate of leukopenia (50.0% vs 68.7%, p = 0.039) before PSM, indicating better tolerance of radiation in treatment-naïve patients. However, the Neo-IMRT group also had a higher rate of bilirubin increase (42.1% vs 22.4%, p = 0.047) before PSM, which may be related to a larger irradiated volume. After PSM, these differences were no longer significant, suggesting that both treatment modalities have similar safety profiles. No cases of radiation-induced hepatic decompensation were observed in our study. To better evaluate radiation-induced liver damage, it may be necessary to use tools such as imaging, in addition to markers in blood, to conduct a more comprehensive analysis. 29
Postoperative complications were mainly grade I–II in both groups, with no significant differences in the proportion of complications or in hospital stay, type of resection, or volume of blood loss. These results indicate that adding Neo-IMRT before surgery does not significantly increase the difficulty of hepatectomy or the incidence of postoperative complications.
Our study provides valuable insights into the choice between neoadjuvant and adjuvant RT for cl-HCC. While both treatment modalities were equally effective in terms of survival outcomes, Neo-IMRT had the advantage of achieving wider surgical margins, which may be associated with better long-term survival.22–24 However, the potential for increased adverse events, such as increased bilirubin, in the neoadjuvant group should also be considered. Future prospective randomized controlled trials with larger sample sizes are needed to confirm these findings and to better understand the respective benefits and risks of each treatment modality.
This study is limited by its retrospective design. As a nonrandomized comparative study based on two separate prospective studies, the baseline characteristics were not always comparable between the two groups, and selection bias was possible despite taking advantage of PSM. In addition, the sample size after PSM was small, and all cases in this study were enrolled from a single center, which could have limited its generalizability.
Conclusion
In conclusion, although Neo-IMRT did not significantly improve survival as compared with Adj-IMRT in cl-HCC, it resulted in wider surgical margins, without increasing the difficulty in hepatectomy or the incidence of postoperative complications. Further prospective randomized studies with a larger sample size are warranted to confirm the findings.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251342686 – Supplemental material for Neoadjuvant versus adjuvant radiotherapy for centrally located hepatocellular carcinoma: a propensity score matching study based on two prospective phase II trials
Supplemental material, sj-docx-1-tam-10.1177_17588359251342686 for Neoadjuvant versus adjuvant radiotherapy for centrally located hepatocellular carcinoma: a propensity score matching study based on two prospective phase II trials by Fan Wu, Bo Chen, Dezuo Dong, Liming Wang, Hongzhi Wang, Weiqi Rong, Jing Jin, Yueping Liu, Hui Fang, Yuan Tang, Yexiong Li, Weihu Wang and Jianxiong Wu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-2-tam-10.1177_17588359251342686 – Supplemental material for Neoadjuvant versus adjuvant radiotherapy for centrally located hepatocellular carcinoma: a propensity score matching study based on two prospective phase II trials
Supplemental material, sj-pdf-2-tam-10.1177_17588359251342686 for Neoadjuvant versus adjuvant radiotherapy for centrally located hepatocellular carcinoma: a propensity score matching study based on two prospective phase II trials by Fan Wu, Bo Chen, Dezuo Dong, Liming Wang, Hongzhi Wang, Weiqi Rong, Jing Jin, Yueping Liu, Hui Fang, Yuan Tang, Yexiong Li, Weihu Wang and Jianxiong Wu in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
This study was funded by Beijing Hospitals Authority Youth Programme (QML20231102), Science Foundation of Peking University Cancer Hospital (BJCH2024GG02), National Natural Science Foundation of China (81902371), Capital’s Funds for Health Improvement and Research (2024-2-2156), Beijing Hospitals Authority’s Ascent Plan (DFL20220902), Young Elite Scientists Sponsorship Program by BAST (BYESS2023002), and Beijing Hope Run Special Fund of Cancer Foundation of China (LC2020L05).
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
