Abstract
Background:
Platinum-based chemotherapy has demonstrated superior efficacy as a first-line treatment for metastatic triple-negative breast cancer (mTNBC), at a cost of higher toxicity. Reliable prognostic tools are needed to refine risk stratification and facilitate individualized treatment discussions.
Objectives:
To develop and validate prognostic nomograms for overall survival (OS) and progression-free survival (PFS) in patients with mTNBC receiving first-line platinum-based chemotherapy.
Design:
Patient-level pooled analysis of prospective trial data, with internal and external validation.
Methods:
Individual patient-level data of 777 mTNBC patients from 5 prospective clinical trials were collected. In total, 445 patients who received first-line platinum-based chemotherapy at our center were included as the PFP-TNBC cohort, which was randomly divided (7:3) into training and validation sets. An independent real-world cohort of 216 mTNBC patients was used for external validation. Multivariable Cox regression was used to construct OS and PFS models based on clinical characteristics, laboratory biomarkers, and regimen choice. Model performance was evaluated using concordance index (C-index), time-dependent area under the curve (AUC), calibration, and decision curve analysis.
Results:
Both nomograms included the number of metastatic sites and the choice of platinum combination agent. The OS model additionally incorporated baseline cancer antigen 125 and lactate dehydrogenase; the PFS model included liver metastasis, baseline pain, and CA153 and carcinoembryonic antigen. For OS, the C-indexes were 0.67, 0.66, and 0.65 in the training, internal validation, and external cohorts, with AUCs of 0.71–0.74, 0.71–0.75, and 0.69–0.71 for 1-, 2-, and 3-year OS. For PFS, the C-indexes were 0.66, 0.62, and 0.61 in the training, internal validation, and external cohorts, with AUCs of 0.69–0.80, 0.66–0.74, and 0.63–0.72 for 6-, 12-, and 24-month PFS, respectively. Both models demonstrated good calibration and risk stratification identified groups with significantly different survival outcomes (log-rank p < 0.001).
Conclusion:
The PFP-TNBC-OS and PFP-TNBC-PFS nomograms provide practical tools to estimate outcomes in mTNBC patients treated with first-line platinum chemotherapy. While C-index values indicated modest discrimination, time-dependent AUCs showed good accuracy at clinically relevant intervals. These models may aid risk stratification, patient counseling, and shared decision-making in chemotherapy-only settings. Future studies are warranted to evaluate their applicability in the evolving era of chemoimmunotherapy.
Plain Language Summary
Keywords
Introduction
Triple-negative breast cancer (TNBC) accounts for approximately 15% of all breast cancers and is characterized by aggressive clinical behavior and poor prognosis. 1 Although immunotherapy and poly(ADP-ribose) polymerase inhibitors have shown promising results in selected subgroups of TNBC, these options are limited to patients with specific biomarkers.2–4 For the majority of patients, chemotherapy remains the mainstay of treatment. Increasing evidence highlights the pivotal role of platinum-based chemotherapy in metastatic TNBC (mTNBC). Our previous phase III CBCSG006 trial demonstrated that first-line cisplatin plus gemcitabine significantly prolonged progression-free survival (PFS) compared with paclitaxel plus gemcitabine in patients with mTNBC (7.73 vs 6.47 months, p = 0.009). 5 A comprehensive Bayesian network meta-analysis involving 125 first-line clinical trials and 37,812 patients with mTNBC further confirmed that platinum-based regimens ranked highest for objective response rate, PFS, and overall survival (OS) based on surface under the cumulative ranking curve values. 6 However, the benefits of platinum must be weighed against its higher toxicity burden, including nausea/vomiting, peripheral neuropathy, and hematotoxicity. Identifying patients who are most likely to benefit from platinum-based treatment has therefore become a critical priority in mTNBC management.
Recent advances have also underscored the substantial heterogeneity of TNBC and the need for refined prognostication.7–13 Molecular immunohistochemistry-based classifications have revealed biologically distinct subgroups with significantly different survival outcomes.11,14 Meanwhile, large-scale registry analyses have identified clinical factors such as age, marital status, extent and sites of metastases, and receipt of systemic therapy as key determinants of survival.7,8,10 Collectively, these findings emphasize the need for prognostic tools that integrate multiple aspects of patient information to enhance risk stratification. This need is especially important in the platinum-based chemotherapy setting, where clinicians currently lack personalized prognostic models to guide individualized treatment decisions.
To address this unmet need, we developed and validated two prognostic nomograms based on individual patient-level data from five prospective clinical trials.5,15–17 These models aim to estimate outcomes with first-line platinum-based chemotherapy in patients with mTNBC. The nomograms were internally validated and externally tested in an independent real-world cohort, offering clinically applicable tools to inform risk stratification and facilitate personalized treatment discussions.
Patients and methods
Population selection
Data were retrieved from clinical trials conducted at our institution, including the published CBCSG006, GAP, GP, AP, and GPGC trials.5,15–18 Across these studies, a total of 777 patients with metastatic breast cancer were screened, of whom 596 had mTNBC and received first-line platinum-based chemotherapy. For the present analysis, 445 patients treated at Fudan University Shanghai Cancer Center (FUSCC) were included to ensure data completeness and consistency. For patients enrolled at other centers, only trial-mandated laboratory tests were consistently available, while other indicators required for model development were either missing or measured inconsistently. In addition, variation in assay platforms and reagents across centers could have introduced measurement bias. To minimize these issues, model construction was restricted to the FUSCC subset, designated as the PFP-TNBC cohort (Prospective study of First-line Platinum-based chemotherapy in metastatic Triple Negative Breast Cancer, PFP-TNBC). This cohort was used for model development and internal validation.
Demographics, clinicopathologic, and laboratory results were extracted from electronic medical records. All patients provided written informed consent for the use of their clinical data, and all studies were approved by the institutional review board. The study was conducted in accordance with the Declaration of Helsinki. The reporting of this study conforms to the TRIPOD statement (Supplemental File 1). 19
To enhance generalizability, an independent real-world cohort (n = 216) from our previously published study was used for external validation. 20
Statistical analysis
Categorical variables were compared using either the chi-square test or Fisher’s exact test, and continuous variables were compared using either the Wilcoxon test or independent sample t-tests. Survival curves were generated by the Kaplan–Meier method, and differences in PFS and OS were assessed with the log-rank test.
The PFP-TNBC cohort was randomly divided into training and validation cohorts in a 7:3 ratio. Prognostic factors were first screened in the training set using univariate Cox proportional hazards regression; variables with p < 0.05 were entered into multivariate models. Independent prognostic factors identified in the final multivariable model were used to construct the nomograms. All candidate variables assessed during model construction are listed in Table 1, encompassing clinical characteristics, baseline laboratory biomarkers, and the choice of first-line treatment regimen. Missing data were minimal and were handled using multiple imputation with the “mice” package in R. Model performance was evaluated using the concordance index (C-index), time-dependent receiver operating characteristics (ROC) curve, and calibration curve. Clinical utility was assessed by decision curve analysis (DCA). Based on nomogram-derived total risk scores, an optimal cutoff value was determined to stratify patients into high- and low-risk groups using the R package CatPredi. Kaplan–Meier analysis was then performed to compare outcomes between subgroups. All p values were two-sided, and p < 0.05 was considered statistically significant. Statistical analyses were performed using R software 4.3.1 (R Foundation for Statistical Computing, Vienna, Austria).
Baseline characteristics of all variables collected and assessed for model development.
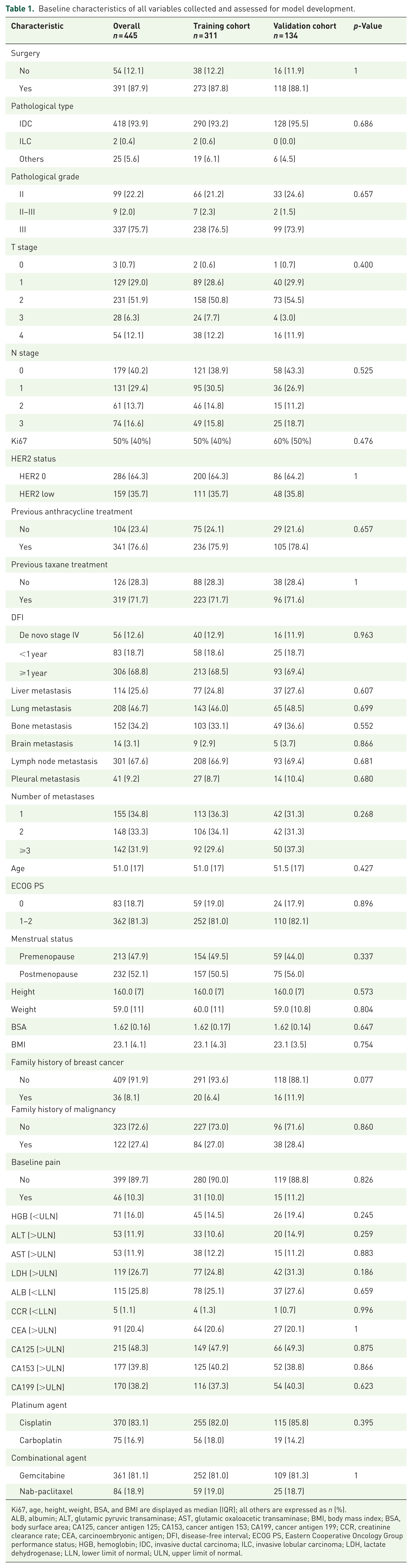
Ki67, age, height, weight, BSA, and BMI are displayed as median (IQR); all others are expressed as n (%).
ALB, albumin; ALT, glutamic pyruvic transaminase; AST, glutamic oxaloacetic transaminase; BMI, body mass index; BSA, body surface area; CA125, cancer antigen 125; CA153, cancer antigen 153; CA199, cancer antigen 199; CCR, creatinine clearance rate; CEA, carcinoembryonic antigen; DFI, disease-free interval; ECOG PS, Eastern Cooperative Oncology Group performance status; HGB, hemoglobin; IDC, invasive ductal carcinoma; ILC, invasive lobular carcinoma; LDH, lactate dehydrogenase; LLN, lower limit of normal; ULN, upper limit of normal.
Results
Baseline characteristics of patients
The baseline clinical data for the total PFP-TNBC cohort (n = 445), the training cohort (n = 311), and the validation cohort (n = 134) are summarized in Table 1.
In the PFP-TNBC cohort, 87.9% of patients had recurrence or metastasis after surgery. More than 70% had previously received anthracycline and taxane-based chemotherapy regimens. The most frequent metastatic sites were lymph nodes (67.6%), lungs (46.7%), bones (34.2%), liver (25.6%), pleura (9.2%), and brain (3.1%). Approximately one-third of the patients had 1, 2, or ⩾3 metastatic sites.
Baseline hematological parameters were normal in most patients, as determined by the eligibility criteria of the clinical trials. Lactate dehydrogenase (LDH) and tumor marker levels reflected disease burden, with 26.7% of patients showing elevated LDH at baseline. The incidences of abnormal tumor markers were 48.3% for CA125, 39.8% for CA153, 38.2% for CA199, and 20.4% for CEA. Regarding treatment regimens, 83.1% received a cisplatin-based regimen, and 16.9% carboplatin-based regimen. In terms of combinational agents, 81.1% received gemcitabine and 18.9% received albumin-paclitaxel.
Construction and validation of the OS prognostic model
The median follow-up time was 68.2 months (95% CI, 62.2–78.6). During this period, 358 deaths (80.5%) occurred, and the median OS was 22.0 months (95% CI, 20.2–23.7).
The univariable Cox regression results, including hazard ratios (HRs) and 95% CIs for all candidate predictors, are provided in Table S1. Multivariate Cox regression identified the number of metastatic sites, elevated baseline CA125, and elevated baseline LDH as independent prognostic factors for worse OS, while the use of albumin–paclitaxel as the platinum partner was an independent prognostic factor for better OS (Figure S1). These four factors were incorporated into the PFP-TNBC-OS nomogram (Figure 1).

The nomogram of the PFP-TNBC-OS model.
The C-indexes of the PFP-TNBC-OS model were 0.67 (95% CI, 0.63–0.71) in the training cohort, 0.66 (95% CI, 0.61–0.71) in the validation cohort, and 0.65 (95% CI, 0.61–0.69) in the external real-world validation cohort. The time-dependent area under the curves (AUCs) for 1-, 2-, and 3-year OS were 0.74, 0.73, and 0.71 in the training cohort; 0.75, 0.71, and 0.75 in the validation cohort; and 0.69, 0.70, and 0.71 in the real-world validation cohort (Figure 2(a)). The calibration plot demonstrated good agreement between the predicted and observed survival rates at 1, 2, and 3 years (Figure 2(b)). DCA showed that the model provided net clinical benefits across all three cohorts (Figure 2(c)).

PFP-TNBC-OS model’s time-dependent ROC curves, calibration plots, and DCA curves. (a) Time-dependent ROC curves of the PFP-TNBC-OS model for the training cohort, validation cohort, and real-world validation cohort. (b) Calibration plots of the PFP-TNBC-OS model for the training cohort, validation cohort, and real-world validation cohort. (c) DCA curves of the PFP-TNBC-OS model for predicting 2-year OS in the training cohort, validation cohort, and real-world validation cohort.
Using the R package CatPredi, an optimal cutoff of 170.6 was identified. Patients were stratified into low-risk (⩽170.6) and high-risk (>170.6) groups. Kaplan–Meier curves showed that high-risk patients had significantly shorter OS compared with low-risk patients in the training cohort, validation cohort, and real-world validation cohort (all p < 0.001, Figure 3(a)).

Kaplan–Meier curves of (a) OS and (b) PFS for risk stratification in the training, validation, and real-world validation cohorts.
Construction and validation of the PFS prognostic model
At the time of analysis, 378 patients (84.9%) had experienced PFS events. The median PFS was 7.8 months (95% CI, 7.5–8.4).
The univariable Cox regression results are provided in Table S2. Multivariate analysis identified liver metastasis, number of metastatic sites, elevated baseline CEA, elevated baseline CA153, baseline pain, and the use of albumin–paclitaxel as the platinum partner as independent prognostic factors for PFS (Figure S2). These factors were used to develop the PFP-TNBC-PFS nomogram (Figure 4).
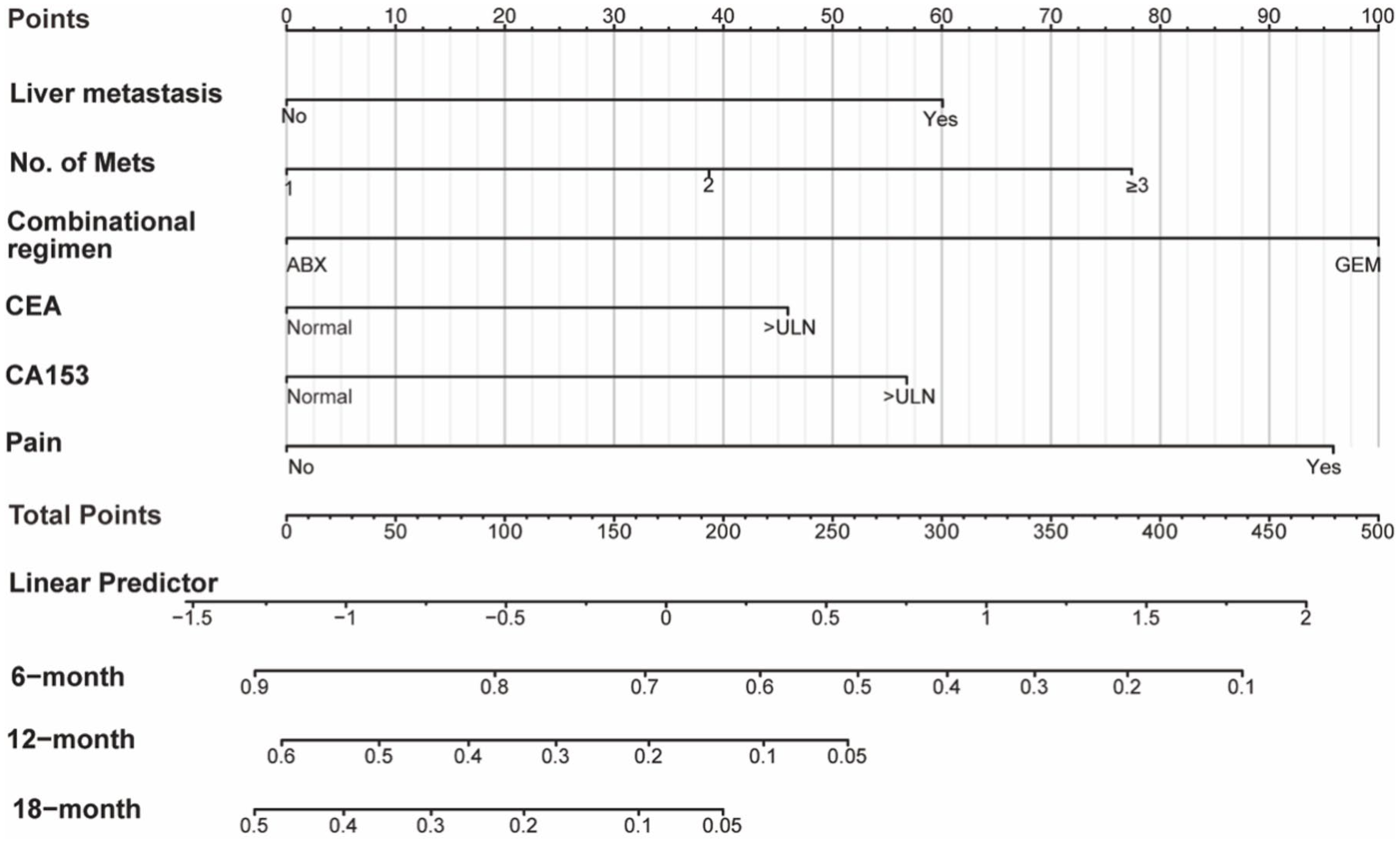
The nomogram of the PFP-TNBC-PFS model.
The C-indexes of the PFP-TNBC-PFS model were 0.66 (95% CI, 0.63–0.69) in the training set, 0.62 (95% CI, 0.57–0.67) in the validation set, and 0.61 (95% CI, 0.56–0.66) in the external real-world cohort. The time-dependent AUCs for 6-, 12-, and 24-month PFS were 0.69, 0.75, and 0.80 in the training cohort; 0.70, 0.66, and 0.74 in the validation cohort; and 0.63, 0.72, and 0.71 in the real-world validation cohort, respectively (Figure 5(a)). PFP-TNBC-PFS model was well-calibrated (Figure 5(b)) and exhibits favorable net benefits (Figure 5(c)).

PFP-TNBC-PFS model’s time-dependent ROC curves, calibration plots, and DCA curves. (a) Time-dependent ROC curves of the PFP-TNBC-PFS model for the training cohort, validation cohort, and real-world validation cohort. (b) Calibration plots of the PFP-TNBC-PFS model for the training cohort, validation cohort, and real-world validation cohort. (c) DCA curves of the PFP-TNBC-PFS model for predicting 12-month PFS in the training cohort, validation cohort, and the real-world validation cohort.
Using the CatPredi package, an optimal cutoff of 158.6 was determined. Patients were stratified into high-risk (>158.6) and low-risk (⩽158.6) groups. Significant differences in PFS were observed between risk groups across the training, validation, and external cohorts (all p < 0.001; Figure 3(b)).
Discussion
Platinum remains a crucial treatment option for TNBC. To our knowledge, this is the first study to develop and validate nomograms to estimate the prognosis of first-line platinum-based chemotherapy in mTNBC. The models were further tested in an external real-world cohort, where they retained a C-index of 0.65 and demonstrated good calibration.
The baseline characteristics of patients in the PFP-TNBC cohort reflected the clinical profile of mTNBC in China. Compared with Western patients in the SEER database, Chinese patients were younger at diagnosis (51 vs 59 years), more frequently presented with invasive ductal carcinoma (93.9% vs 79.0%), and had a greater metastatic burden (⩾2 metastases: 65.2% vs 29.5%). (Chen, Liu and Yi, 2022).
Several prognostic factors identified in our study have been consistently reported in previous research. The number of metastatic sites has repeatedly been shown as an independent factor for worse survival in metastatic breast cancer.21,22 LDH, a key enzyme in anaerobic glycolysis, is elevated in hypoxic tumor microenvironments and was previously associated with worse outcomes in both metastatic breast cancer (HR = 1.85, 95% CI: 1.36–2.53) and early TNBC (HR = 1.42, 95% CI: 1.08–1.88).23,24 CEA, CA125, and CA153 are the most commonly used diagnostic markers for breast cancer. Similarly, baseline elevation of CEA, CA125, and CA153 has been linked to inferior outcomes in mTNBC. 25 In addition, the GAP trial demonstrated the superiority of the AP regimen compared with the GP regimen as first-line treatment for mTNBC. 15 Our analysis confirmed the favorable impact of the AP regimen after multivariate adjustment, although residual confounding factors from unmeasured variables cannot be entirely excluded. This finding should therefore be regarded as supportive of existing evidence rather than definitive proof of regimen superiority.
Research on prognostic models for TNBC remains limited.26–28 A recent SEER-based model including 33,654 TNBC patients across early and metastatic stages found that age, race, pathological grade, and TNM stage were key determinants of prognosis. 7 However, the predominance of early-stage patients (>95% of patients) and the limited number of Asian patients restricts its prognostic value for mTNBC patients. Another SEER analysis of 1,962 stage IV TNBC patients identified T stage, N stage, bone metastasis, liver metastasis, brain metastasis, unmarried status, and age as prognostic factors. 8 However, the study lacked treatment details. A retrospective analysis has also suggested that baseline ECOG PS, the number of metastases, and inflammatory indices, such as the Pan-immune-Inflammation Value, may predict outcomes in mTNBC patients receiving platinum-containing treatment. Still, the small sample size and lack of a validation cohort preclude firm conclusions. 9 Collectively, these studies underscore the need for robust, validated models in the metastatic setting.
Our study has several strengths. The PFP-TNBC cohort was derived from prospective clinical trials with high-quality survival data and minimal missing information, providing a reliable basis for model development. The models were subsequently validated in an independent real-world cohort, confirming their reproducibility and generalizability. Notably, the PFP-TNBC-OS nomogram demonstrated consistent prognostic performance, with time-dependent AUCs exceeding 0.70 for 1-, 2-, and 3-year OS. Similarly, the PFP-TNBC-PFS model achieved AUCs above 0.70 at several clinically relevant time points. The PFP-TNBC nomograms serve as practical prognostic tools to estimate OS and PFS probabilities for individual patients receiving platinum-based chemotherapy. High-risk patients identified by the models had poor outcomes despite receiving platinum treatment, highlighting the need for closer monitoring, enrollment in clinical trials, or alternative therapeutic strategies. By contrast, low-risk patients appeared to derive durable benefits and remain appropriate candidates for platinum-based regimens. Furthermore, individualized probability estimates (e.g., 1-, 2-, and 3-year OS; 6-, 12-, and 24-month PFS) may enhance physician–patient communication and support shared decision-making. Currently, these nomograms should be viewed as tools for risk assessment and patient education rather than as devices to determine treatment options. It is worth noting that the models’ discriminatory performance was modest, with C-index values ranging from 0.61 to 0.67, consistent with many published prognostic models in oncology.29,30 However, the time-dependent AUCs at clinically relevant intervals generally exceeded 0.70, indicating good predictive performance at specific time points. Therefore, the models offer clinically meaningful risk stratification while highlighting the heterogeneity of outcomes in mTNBC.
Several limitations should be acknowledged. First, the models were developed from clinical trial participants, who generally had favorable baseline characteristics, including adequate organ function and good ECOG performance status. Consequently, the influence of clinical variables such as unrecovered toxicities and comorbidities could not be fully assessed in this dataset. To address this limitation, we validated the model in a real-world cohort with more heterogeneous characteristics. Second, although immunotherapy has become the standard first-line treatment for patients with PD-L1-positive mTNBC in several countries, the first immunotherapy agent was only approved in China in June 2024. 31 As a result, none of the patients in our analysis received immunotherapy. Thus, the applicability of our model may decline over time in the contemporary era of immunotherapy. An independent prospective study is currently underway to evaluate the efficacy of first-line immunotherapy combined with nab-paclitaxel and cisplatin (NCT04537286). Future analyses are planned to assess the performance of our nomograms in the context of platinum-based chemoimmunotherapy combinations. Third, our models were based on data from a single center, underscoring the need for multicenter validation. In addition, adverse events were documented using different CTCAE versions (v4.0 vs v3.0), which limited pooled toxicity analyses and prevented a comprehensive risk–benefit assessment. Finally, the genomic data were incomplete, especially regarding germline BRCA1/2 status. Only 149 patients had BRCA testing, with 14 identified as mutation carriers. Including this variable would have significantly reduced the effective sample size and precluded external validation. We are currently building a larger cohort with BRCA testing to explore this in future analyses.
Conclusion
By leveraging individual patient data from multiple prospective clinical trials, we developed and internally validated prognostic models for OS and PFS in patients with mTNBC receiving first-line platinum-based chemotherapy. Importantly, the prognostic performance of both models was further confirmed in an external real-world cohort, underscoring their potential clinical utility. These nomograms may facilitate individualized risk stratification and support shared decision-making in routine practice. At the same time, their discriminatory ability was modest, and certain prognostic variables such as BRCA status and immunotherapy exposure were not incorporated due to data limitations. Thus, while these models provide a practical framework for risk stratification in the chemo-only setting, further refinement and validation will be essential to enhance accuracy and ensure applicability in the evolving era of chemo-immunotherapy.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251395906 – Supplemental material for Development and validation of nomograms of outcomes for first-line platinum-based chemotherapy for metastatic triple-negative breast cancer: patient-level analysis of five prospective clinical trials in China
Supplemental material, sj-docx-1-tam-10.1177_17588359251395906 for Development and validation of nomograms of outcomes for first-line platinum-based chemotherapy for metastatic triple-negative breast cancer: patient-level analysis of five prospective clinical trials in China by Chengcheng Gong, Ting Li, Yifan Chen, Jian Zhang, Yannan Zhao, Leiping Wang, Jun Cao, Zhonghua Tao, Mingchuan Zhao, Xichun Hu and Biyun Wang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-2-tam-10.1177_17588359251395906 – Supplemental material for Development and validation of nomograms of outcomes for first-line platinum-based chemotherapy for metastatic triple-negative breast cancer: patient-level analysis of five prospective clinical trials in China
Supplemental material, sj-pdf-2-tam-10.1177_17588359251395906 for Development and validation of nomograms of outcomes for first-line platinum-based chemotherapy for metastatic triple-negative breast cancer: patient-level analysis of five prospective clinical trials in China by Chengcheng Gong, Ting Li, Yifan Chen, Jian Zhang, Yannan Zhao, Leiping Wang, Jun Cao, Zhonghua Tao, Mingchuan Zhao, Xichun Hu and Biyun Wang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-tiff-3-tam-10.1177_17588359251395906 – Supplemental material for Development and validation of nomograms of outcomes for first-line platinum-based chemotherapy for metastatic triple-negative breast cancer: patient-level analysis of five prospective clinical trials in China
Supplemental material, sj-tiff-3-tam-10.1177_17588359251395906 for Development and validation of nomograms of outcomes for first-line platinum-based chemotherapy for metastatic triple-negative breast cancer: patient-level analysis of five prospective clinical trials in China by Chengcheng Gong, Ting Li, Yifan Chen, Jian Zhang, Yannan Zhao, Leiping Wang, Jun Cao, Zhonghua Tao, Mingchuan Zhao, Xichun Hu and Biyun Wang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-tiff-4-tam-10.1177_17588359251395906 – Supplemental material for Development and validation of nomograms of outcomes for first-line platinum-based chemotherapy for metastatic triple-negative breast cancer: patient-level analysis of five prospective clinical trials in China
Supplemental material, sj-tiff-4-tam-10.1177_17588359251395906 for Development and validation of nomograms of outcomes for first-line platinum-based chemotherapy for metastatic triple-negative breast cancer: patient-level analysis of five prospective clinical trials in China by Chengcheng Gong, Ting Li, Yifan Chen, Jian Zhang, Yannan Zhao, Leiping Wang, Jun Cao, Zhonghua Tao, Mingchuan Zhao, Xichun Hu and Biyun Wang in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
