Abstract
Background:
Platinum-based chemotherapy is the first-line treatment choice for advanced thymic epithelial tumors (TETs), with the expected objective response rate (ORR) ≈of 50% in thymoma and ≈20% in thymic carcinoma.
Objective:
To evaluate the impact of relative dose intensity (RDI) on first-line treatment outcomes in TET patients.
Design:
Retrospective cohort referred between 2016 and 2022 at the University of Naples Federico II, Italy.
Methods:
Advanced TETs treated with first-line platinum chemotherapy; RDI calculated as delivered/planned dose intensity and categorized as low (<85%) or high (⩾85%). Outcomes: ORR, time to next treatment (TTNT), overall survival (OS).
Results:
Thirty-three patients (15 thymoma, 18 carcinoma); 22 low RDI, 11 high RDI. RDI was not associated with ORR. High RDI showed longer TTNT (6.6 vs 5.0 months; p = 0.042) and numerically longer OS (86.4 vs 32.2 months; p = 0.361).
Conclusion:
Maintaining ⩾85% RDI during first-line platinum chemotherapy may offer clinical benefits and warrants further validation in larger cohorts.
Plain language summary
Thymic epithelial tumors are rare cancers that develop in the chest near the thymus gland. For patients with advanced disease, the usual first treatment is chemotherapy with drugs that contain platinum. Sometimes doctors need to lower the dose or delay treatment to manage side effects or when the treatment does not seem effective. It is not well known whether reducing the planned dose affects treatment results. In this study, we reviewed the medical records of 33 patients treated at the University of Naples Federico II in Italy between 2016 and 2022. All patients received platinum-based chemotherapy as their first treatment. We compared patients who received at least 85% of the planned dose (high dose intensity) with those who received a lower dose (low dose intensity). We found that patients who received the higher dose intensity did not have a higher chance of tumor shrinkage. However, they stayed longer on their treatment before needing another therapy, and they also lived longer on average, even though this difference was not statistically significant. These findings suggest that keeping close to the planned chemotherapy dose, when possible, may bring important benefits for patients with thymic tumors. Further studies are needed to confirm these results.
Introduction
Thymic epithelial tumors (TETs) are rare malignancies of the thymic gland, including thymomas (T) and thymic carcinomas (TC). 1 These tumors exhibit distinct biological behaviors, as T typically progress slowly and have a more favorable prognosis, whereas TC are significantly more aggressive. 2 Platinum-based chemotherapy is the standard treatment for unresectable metastatic TETs. Regimens for T include CAP (cisplatin, doxorubicin, and cyclophosphamide) or KIM (cyclophosphamide, doxorubicin, cisplatin, and prednisone), whereas carboplatin-paclitaxel is commonly used for TC. Carboplatin-etoposide is an alternative option for patients unfit for cisplatin and/or anthracyclines. 3 Notably, these tumors are associated with autoimmune disorders, such as Myasthenia Gravis (MG), Pure Red Cell Aplasia, and Good’s syndrome (GS), which can worsen baseline frailty and require a multidisciplinary approach. 4 Overall, the objective response rate (ORR) reported for first-line platinum-based chemotherapy is 50% for T and 20% for TC. 5 A pooled analysis by Okuma et al. 6 demonstrated the superior efficacy of anthracycline-cisplatin-based regimens in T compared to non-anthracycline therapies, while showing comparable effectiveness in TC. This supports the use of anthracyclines in T, whereas their role in TC remains unclear. Maintaining full chemotherapy doses is a widely accepted strategy in oncology for improving patient outcomes. 7 However, in TETs, it lacks evidence due to a few large studies. Maintaining treatment intensity is often reduced by patient-related factors or adverse events, and starting with dose reduction helps assess tolerability in frail patients. 8 Relative dose intensity (RDI), the ratio of delivered to planned chemotherapy, affects outcomes in various cancers. 9 Nielsen et al. found that an RDI of 80%–85% or higher improves survival in solid tumors treated with regimens such as carboplatin, FOLFOX, FOLFIRI, or FOLFIRINOX. Conversely, lower RDI correlates with poorer efficacy and survival, likely from reduced cytotoxic exposure. 10
However, the clinical impact of RDI in TETs remains unclear due to the rarity of the disease and the absence of prospective trials. Thus, evaluating RDI may help optimize treatment and improve outcomes. This study examined whether RDI is associated with clinical outcomes in TET patients receiving first-line platinum-based chemotherapy.
Materials and methods
This retrospective study included consecutive TET patients treated with first-line platinum-based chemotherapy at the University of Naples Federico II between January 2016 and December 2022. Data were extracted from the institutional electronic medical records. Key inclusion criteria were age 18 years or older, a histologically confirmed diagnosis of TET, and evaluation of the presence of the most frequent autoimmune conditions, including MG and GS, defined as previously reported. 11
Chemotherapy regimens included CAP or KIM, carboplatin and paclitaxel, or cisplatin/carboplatin and etoposide, depending on the histological subtype and physician preference. The RDI was calculated as the delivered dose intensity ratio (dose intensity: total dose (mg/m²)/time to complete chemotherapy) to the planned dose intensity based on standard treatment protocols (RDI = (delivered dose intensity/planned dose intensity) × 100%). 12 The planned treatment schedule consisted of six cycles administered every 21 days. Patients were stratified according to the established thresholds in previous RDI studies 9 : high RDI (h-RDI, ⩾85%) and low RDI (l-RDI, <85%).
Patient inclusion in each of the two RDI groups was performed retrospectively, based on the actual dose intensity delivered during treatment. Patients who experienced dose reductions, treatment delays, or early discontinuation, resulting in an RDI below 85%, were categorized as having a l-RDI. These adjustments reflected real-world factors such as hematologic toxicity, treatment intolerance, or physician decisions in clinically frail patients.
ORR was assessed from radiological and clinical evaluations according to RECIST 1.1. OS was defined as the time from chemotherapy initiation to death from any cause, and TTNT as the time from chemotherapy initiation to second-line treatment, documented progression, or death. Baseline characteristics were summarized with descriptive statistics. OS and TTNT were estimated using Kaplan–Meier curves and compared with the log-rank test. Associations between categorical variables, including RDI and ORR, were evaluated using Fisher’s exact test. A p-value < 0.05 was considered statistically significant. All analyses and figures were generated in R (version 4.3.0; R Foundation for Statistical Computing, Vienna, Austria), and a multivariable logistic regression model was used to assess independent associations between RDI and key clinical variables.
Results
A total of 33 patients were enrolled in this study. The study cohort included 22 males (66.7%) and 11 females (33.3%) with a median age of 59 years (range, 33–79). Fifteen patients (45.5%) were diagnosed with T, and 18 (54.5%) were diagnosed with TC. The T subtypes were distributed as follows: B1 (9.1%), B2 (9.1%), B2–B3 (9.1%), B3 (9.1%), AB (3.0%), and unspecified T (6.1%). Among the TC, one was a neuroendocrine carcinoma. Based on the eighth edition of the TNM classification for TETs, 5 (15.2%) patients had stage III, 9 (27.3%) had stage IVA, and 19 (57.5%) had stage IVB disease at the time of diagnosis. Patient characteristics are summarized in Table 1.
Baseline characteristics of study population.

The KIM scheme refers to a chemotherapy regimen consisting of cyclophosphamide, doxorubicin, cisplatin, and prednisone.
The CAP scheme refers to a chemotherapy regimen consisting of cisplatin, doxorubicin, and cyclophosphamide.
RDI, relative dose intensity.
Platinum-based chemotherapy regimens were heterogeneous across the cohort. CAP was administered to 12 patients (36.4%), of whom 2 (6.1%) received h-RDI, and 10 (30.3%) received l-RDI. Carboplatin-paclitaxel was administered to 10 patients (30.3%), equally divided between the h-RDI and l-RDI groups (five patients each). The KIM regimen was used in seven patients (21.2%), with 2 (6.1%) receiving h-RDI and 5 (15.2%) receiving l-RDI. Finally, 4 patients (12.1%) were treated with platinum-etoposide, with equal distribution between the h-RDI and l-RDI groups (two patients each, 6.1%; Table 1). Twenty patients (60.6%) had a performance status (PS) recorded at treatment initiation = 1, and 13 (39.4%) had PS = 0. Autoimmune conditions were identified in 12 patients (36.4%), including GS in 11, MG in 2, and Isaacs’ syndrome in one. Notably, two patients presented with two concomitant autoimmune conditions.
Among the 33 patients, 11 (33.3%) had an h-RDI and 22 (66.7%) had an l-RDI. No significant correlation was observed between the RDI and ORR (Fisher’s exact test p = 0.66).
However, patients receiving h-RDI showed a statistically longer median TTNT than those receiving l-RDI (6.67 vs 5.00 months; p = 0.0425; HR = 0.48, 95% CI 0.23–1.00; Figure 1). Among the study subjects, the number of administered chemotherapy cycles ranged from 2 to 6. Notably, 9 out of 22 patients (40.9%) in the l-RDI group received ⩽4 cycles, with early discontinuation primarily due to hematologic toxicities (three patients, 9%), lack of response (four patients, 12%), or treatment intolerance (one patient, 3%). In contrast, most patients in the h-RDI group completed the planned treatment course (Table 2). The multivariable logistic regression analysis did not show any associations between RDI and disease stage, PS, age, or relevant comorbidities (Supplemental Table 1).

Time to next treatment by RDI.
Patient characteristics according to RDI group.

PS, performance status; RDI, relative dose intensity.
Similarly, patients treated with h-RDI exhibited a numerically longer OS, with a median OS of 86.4 months compared to 32.2 months in those receiving l-RDI treatment. However, this difference did not reach statistical significance (HR = 0.60, 95% CI: 0.22–1.65, p = 0.361; Figure 2).
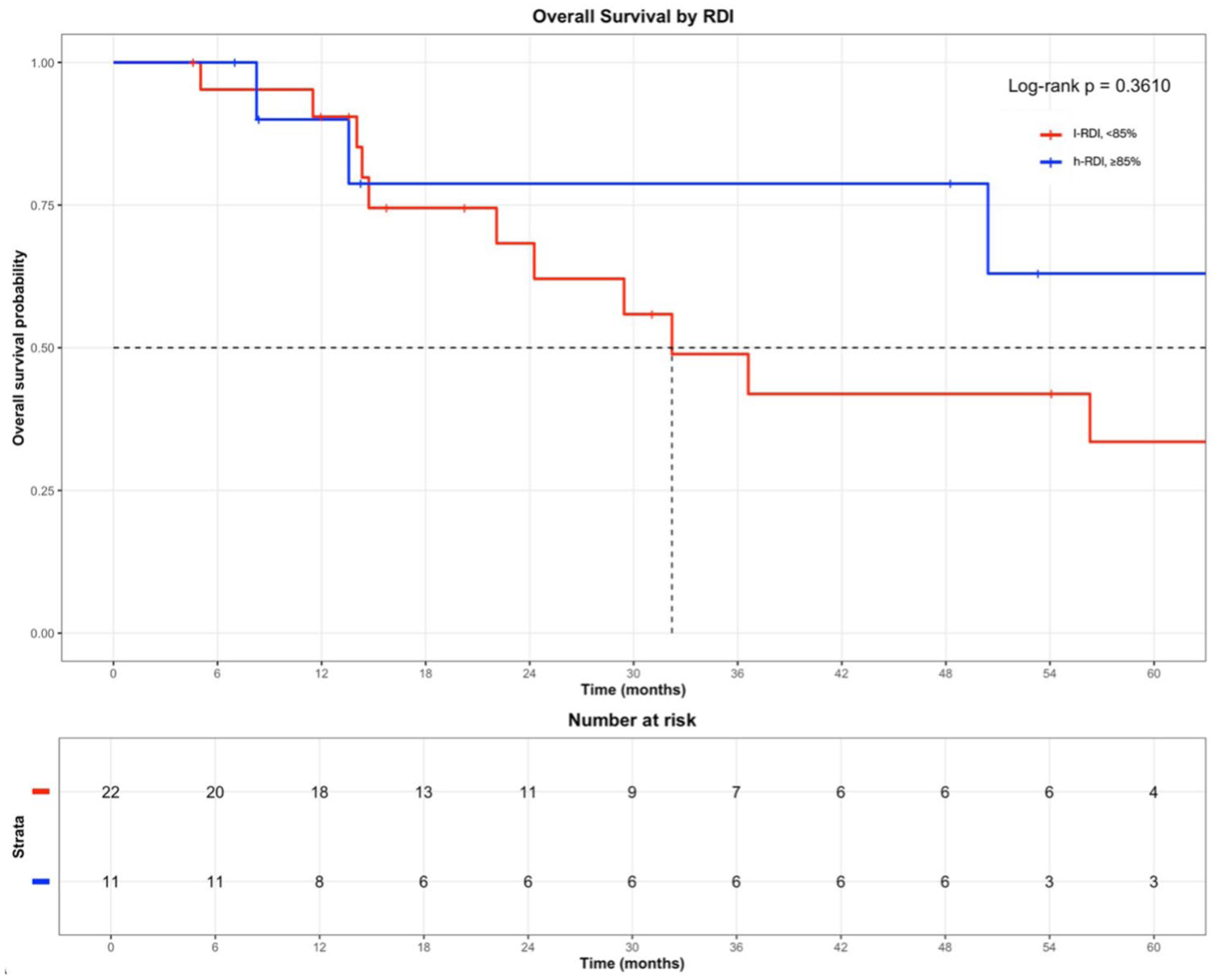
Overall survival by RDI.
Discussion
Dose reduction and delay are important strategies implemented by oncologists to limit adverse events, preserve treatment maintenance, and limit the negative impact of anticancer treatments on the quality of life. This retrospective analysis suggests the potential clinical impact of maintaining h-RDI platinum-based chemotherapy as a first-line treatment for TETs. We found a statistically significant association between h-RDI and prolonged TTNT, suggesting that maintaining a higher chemotherapy dose intensity may delay disease progression and the need for subsequent therapies. This underscores the potential therapeutic advantage of dose adherence in this challenging malignancy, where available treatment options are scarce. To further support this observation, we evaluated treatment discontinuations for reasons unrelated to the disease progression. In the l-RDI group, 8 out of 22 patients (36.4%) discontinued chemotherapy due to hematologic toxicity (n = 3), poor treatment tolerance (n = 1), or lack of response (n = 4). In contrast, early discontinuation was less frequent in the h-RDI group, where all patients received at least five cycles or completed the planned treatment course without major toxicity. These findings suggest that a low RDI was often the result of tolerability issues rather than tumor progression, reinforcing the hypothesis that suboptimal dose intensity may compromise treatment efficacy in this setting.
Although the multivariable analysis did not show statistically significant associations between RDI and key clinical variables, the small sample size precluded drawing definitive conclusions.
Despite the difference in median OS observed in our cohort between h-RDI and l-RDI groups was numerically longer (86.4 vs 32.2 months), this did not reach statistical significance. This suggests that dose intensity may play a role in extending survival in TET patients. However, the relatively small sample size and short follow-up period likely limited the power of this study to detect statistically significant differences in OS. Notably, most patients in our cohort received reduced platinum-based chemotherapy doses or experienced treatment delays, primarily due to poor PS, comorbidities, or the physician’s choice. This highlights the critical importance of administering the appropriate chemotherapy dose on schedule in first-line treatment whenever feasible, as it could substantially influence clinical outcomes.
Interestingly, no significant correlation was observed between RDI and ORR, suggesting that response evaluation based on conventional radiological criteria might not fully capture the benefits of dose intensity in TETs, at least in a real-world setting. Alternative tumor monitoring approaches, such as circulating biomarkers (e.g., circulating tumor DNA), could complement standard radiological evaluations and provide a more comprehensive strategy for assessing treatment efficacy.
Our findings align with the REMORA trial, a multicenter phase II study evaluating Lenvatinib in patients with metastatic or recurrent TC. In this exploratory analysis, patients with an RDI ⩾ 75% in the first 8 weeks had a longer median OS than those with RDI < 75% (38.5 vs 17.3 months; HR 0.46; p = 0.0406), suggesting that higher early treatment exposure may result in meaningful clinical benefit. The 75% RDI threshold was selected after testing several levels between 50% and 100%, choosing the one with the clearest survival difference. No significant difference in ORR was seen between RDI ⩾ 75% and <75% (44.4% vs 33.3%, p = 0.5314), but a higher ORR was observed in patients maintaining full dose in the first 8 weeks (75.0% vs 29.4%, p = 0.0379). 13 Although obtained with a targeted agent, these findings further support the hypothesis that maintaining adequate treatment intensity may be a key prognostic factor in TETs, especially in the early treatment phase.
One limitation of our study is the histologic heterogeneity of the cohort, which included both T and TC. These entities differ in biology, chemosensitivity, and prognosis, and our findings were largely driven by TC, which accounted for 54.5% of cases and is generally less responsive to chemotherapy and associated with a higher unmet clinical need. Moreover, we retained the single thymic neuroendocrine carcinoma, as comparative series report outcomes comparable to TC; in the joint ESTS/ITMIG analysis, 5-year overall and recurrence-free survival did not differ significantly between these subtypes, and histology was not an independent prognostic factor, supporting a unified analysis. 14 The small sample size and limited follow-up reduced the analysis’s statistical power and the strength of the conclusions and generalizability. Larger studies are needed to validate these findings and strengthen evidence on RDI’s prognostic impact in TETs.
Conclusion
To our knowledge, this is the first study to assess the impact of RDI on outcomes in TETs treated with first-line platinum-based chemotherapy. Our findings suggest that maintaining adequate RDI, when feasible, may improve outcomes and should be supported by expert multidisciplinary management to avoid unnecessary dose modifications or early discontinuation. Future prospective studies are needed to confirm the RDI as a meaningful predictor of outcomes in TETs.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251411122 – Supplemental material for Impact of relative dose intensity of first-line platinum-based chemotherapy in patients with thymic epithelial tumors: a retrospective study
Supplemental material, sj-docx-1-tam-10.1177_17588359251411122 for Impact of relative dose intensity of first-line platinum-based chemotherapy in patients with thymic epithelial tumors: a retrospective study by Erica Pietroluongo, Pietro De Placido, Roberto Buonaiuto, Aldo Caltavituro, Giuseppe Neola, Margaret Ottaviano, Marianna Tortora, Grazia Arpino, Giovannella Palmieri, Carmine De Angelis, Marina Chiara Garassino, Roberto Bianco, Alberto Servetto and Mario Giuliano in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251411122 – Supplemental material for Impact of relative dose intensity of first-line platinum-based chemotherapy in patients with thymic epithelial tumors: a retrospective study
Supplemental material, sj-docx-2-tam-10.1177_17588359251411122 for Impact of relative dose intensity of first-line platinum-based chemotherapy in patients with thymic epithelial tumors: a retrospective study by Erica Pietroluongo, Pietro De Placido, Roberto Buonaiuto, Aldo Caltavituro, Giuseppe Neola, Margaret Ottaviano, Marianna Tortora, Grazia Arpino, Giovannella Palmieri, Carmine De Angelis, Marina Chiara Garassino, Roberto Bianco, Alberto Servetto and Mario Giuliano in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors acknowledge the European Reference Network (ERN-EURACAN) as a powerful resource for transnational collaboration on rare cancers. Erica Pietroluongo and Pietro De Placido performed research related to this study within the PhD Program in Advanced Biomedical and Surgical Therapies at the Department of Clinical Medicine and Surgery, University “Federico II,” Naples, Italy. Erica Pietroluongo is supported by a scholarship from Associazione TUTOR, an Italian patient organization dedicated to rare thoracic tumors, for the 2024–2025 academic year.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
