Abstract
The treatment landscape for patients with advanced gastric or gastroesophageal junction adenocarcinoma is evolving, driven by a deeper understanding of molecular profiling and the introduction of novel anticancer drugs. Systemic chemotherapy has improved survival compared to supportive care; however, overall survival worldwide remained limited to approximately 1 year. Recently, the introduction of upfront immune checkpoint inhibitors (ICIs) and/or targeted agents in biomarker-enriched populations has led to clinically meaningful survival improvements. The expansion of treatment options for metastatic disease, combined with the identification of biomarker-selected populations, underscores the importance of tailoring first-line, maintenance, and sequential therapies across multiple lines. In this context, the delivery of an effective first-line treatment is crucial, as less than half of patients retain fitness for additional anti-cancer therapies due to the morbidity associated with disease progression to the initial regimen even within modern clinical trials regardless of the use of targeted therapy or immunotherapy combined with chemotherapy. On the other hand, the expanding therapeutic armamentarium including ICIs, antibody-drug conjugates, and targeted therapies for molecularly selected subgroups, will enable the development of sequential treatment strategies across multiple lines of therapy. This review summarizes the current knowledge about maintenance strategies and subsequent lines of treatment in either biomarker selected and unselected populations.
Keywords
Introduction
Advanced gastric (GC) or gastroesophageal junction (GEJ) cancer represents a hard-to-treat disease, and survival of patients is still poor. The introduction of upfront immune checkpoint inhibitors (ICIs) and claudin 18.2 (CLDN18.2)-targeted treatments widened the first-line therapeutic options in patients with HER2-negative gastroesophageal adenocarcinoma and significantly improved survival in biomarker-enriched subgroups.1–5 Moreover, the clinical development of several novel drugs including antibody-drug conjugates (ADCs), matched drugs in biomarker-selected populations, or chimeric antigen receptor T cells (CAR-T cells) is widening the therapeutic scenario beyond the first line. In this context, delivery of an effective first-line treatment strategy is crucial, as less than half of patients retain fitness for additional anti-cancer therapies due to the morbidity associated with disease progression to the initial regimen even in more recent trials. Additionally, an effective first-line maintenance strategy may significantly prolong the benefit of an induction treatment, delay performance status deterioration allowing patients to receive sequential treatments.
Methods
We developed a narrative review summarizing the current knowledge about maintenance strategies and subsequent lines of treatment in either biomarker selected and unselected populations. To provide a comprehensive and clinically oriented overview, a literature search on PubMed was performed. Abstract from American Society of Clinical Oncology Annual Meeting, American Association for Cancer Research, and European Society for Medical Oncology Congress was included as well. The narrative review was conducted according to the Scale for the Assessment of Narrative Review Articles. 6
First-line maintenance strategies
Maintenance therapy is the continuation of treatment following the completion of induction therapy, administered until disease progression or unacceptable toxicity. It typically involves a de-escalation strategy, in which the less toxic agent of a combination regimen is continued to prolong the efficacy of first-line chemotherapy while minimizing cumulative toxicity over time. Switch maintenance strategy involves the use of different agents administered immediately after a pre-specified induction therapy in patients who do not experience disease progression. The rationale behind this strategy is that early administration of a non-cross-resistant regimen may delay progression and ultimately improve survival outcomes. A “stop and go” strategy refers to the administration of a limited chemotherapy course followed by a planned temporary suspension to relieve from toxicity and the reintroduction of the same regimen when disease progression occurs. Agents used in the maintenance regimen should be safe, well-tolerated, cost-effective, and have an easy route of administration. Maintenance regimens have already demonstrated to improve outcomes in some cancer types, including colorectal, lung, and breast cancer, with significant heterogeneity depending on cancer type.7–10 However, the efficacy of maintenance therapies in advanced GC or GEJ cancers remained unclear until the most recent evidence (Table 1). 11
Randomized clinical trials evaluating different maintenance strategies in patients with advanced gastric cancer.

PFS defined as the interval from the first-line treatment to disease progression or death.
Refers to TRAEs.
5-FU, 5-fluorouracil; AEs, adverse events; CAPOX, capecitabine and oxaliplatin; CDDP, cisplatin; CI, confidence interval; ECF, epirubicin, cisplatin and continuous 5-fluorouracil; EOF, epirubicin, oxaliplatin and continuous 5-fluorouracil; FLO, fluorouracil and oxaliplatin; FLOT, fluorouracil, oxaliplatin and docetaxel; FOLFOX, fluorouracil, folinic acid and oxaliplatin; HR, hazard ratio; irPFS, immune related PFS; OS, overall survival; PFS, progression-free survival; SOX, S-1 and oxaliplatin; TRAEs, treatment related AEs; UFT, uracil and tegafur.
De-escalation maintenance
The first randomized trial to evaluate fluoropyrimidine maintenance in patients with advanced GC who had not progressed after first-line fluoropyrimidine-based chemotherapy (epirubicin, cisplatin and continuous 5-fluorouracil/epirubicin, oxaliplatin and continuous 5-fluorouracil or fluorouracil, folinic acid and oxaliplatin (FOLFOX)/capecitabine and oxaliplatin (CAPOX)) was a phase II randomized study comparing maintenance with oral uracil/tegafur (UFT) to observation. Recruitment was prematurely interrupted due to negative results at the interim analysis: progression-free survival (PFS) was slightly higher in the observation arm (3.2 vs 3.6 months, p = 0.752), while overall survival (OS) was equivalent in both arms (14.2 vs 14.2 months, p = 0.983). 12 The continuation of capecitabine was compared to observation in two different randomized trials. Capecitabine was evaluated as a maintenance versus observation after up to six cycles of paclitaxel and capecitabine. Among the 60 randomized patients, PFS and OS favored the maintenance arm (median PFS: 11.0 vs 7.0 months, p < 0.05; median OS: 17.0 vs 11.0 months, p < 0.05). There were no significant differences in adverse events (AEs) between the two groups. 13 Capecitabine was compared to observation in 63 patients with partial response or disease stabilization after six cycles of CAPOX. A significant difference in PFS in favor of capecitabine arm (6.3 vs 4.1 months, p = 0.010) was observed. Moreover, median OS was numerically higher in the experimental arm, although the difference was not significant (18.2 vs 16.5 months, p = 0.624). 14
A phase II trial explored the continuation of an intensive first-line regimen versus a “stop-and-go” arm. Here, 121 patients who received 6 cycles of S1 and oxaliplatin (SOX) induction were randomized to the continuation of SOX or a chemotherapy-free interval. PFS was significantly longer in the continuation arm (5.0 vs 2.7 months; hazard ratio (HR) = 0.57; 95% confidence interval (CI): 0.38–0.83; p = 0.003) while OS was similar in the two arms (22.6 vs 22.7 months; HR = 0.78; 95% CI: 0.50–1.23; p = 0.284). Expectedly, a higher incidence of grade 3 or higher AEs was observed in the chemotherapy continuation arm, especially regarding fatigue (28.8% vs 8.1%, p = 0.003) and sensory neuropathy (25.4% vs 9.7%, p = 0.022). Focusing on quality-of-life assessment during the maintenance phase, a significant overall difference was reported favoring the “stop-and-go” arm in terms of health status, physical functioning, role functioning, and fatigue. 15
The phase II German AIO MATEO trial randomized 165 patients with advanced HER2-negative GC without disease progression after a 12-week induction doublet or triplet chemotherapy to demonstrate the non-inferiority for OS of S1 maintenance versus the continuation the same induction chemotherapy. S1 maintenance led to non-inferior outcomes compared to the continuation of platinum-based chemotherapy. Both PFS and OS from randomization did not significantly differ among the two arms (median PFS: 4.3 vs 6.1 months; HR = 1.10; 80% CI: 0.86–1.39; p = 0.62; median OS: 13.4 vs 11.4 months; HR = 0.97; 80% CI: 0.76–1.23; p = 0.86), as well as frequency of grade 3 or higher AEs (40.6% vs 42.9%), while serious AEs were more frequent in chemotherapy continuation arm (14.2% vs 22.4%). Discontinuation of cytotoxic agent was lower in S-1 maintenances arm (10.4% vs 44.9%). Moreover, an improvement in the global health status 9 weeks after randomization was observed in 41.9% of the patients in the maintenance group, compared to the 15.8% in the controls. 16
Switch maintenance strategies
Early clinical trials investigated several non-cross-resistant regimens in the context of switch maintenance strategies. Marimastat, a matrix metalloproteinase inhibitor, was administrated to patients with metastatic GC after 5-FU-based chemotherapy versus placebo. In patients receiving the switch maintenance, a modest significant benefit for marimastat in terms of OS was reported (253 vs 175 days; HR = 1.68; 95% CI: 1.16–2.44; p = 0.006). 17
Several trials also investigated the role of ICIs as a switch maintenance. Ipilimumab was the first ICI investigated in patients with advanced GC as a maintenance treatment after induction chemotherapy with platinum and fluoropyrimidine-based combinations. In a phase II trial, 114 patients were randomized to receive ipilimumab or therapeutic holiday/continuation of chemotherapy as per investigator choice. The rate of progression in the first 6 weeks after randomization was higher in the ipilimumab arm. The primary endpoint was the immune-related PFS, which was lower in ipilimumab group (2.92 vs 4.90 months; HR = 1.44, 80% CI: 1.09–1.91; p = 0.097). Moreover, OS did not differ in the two cohorts. 18
In the PLATFORM phase II trial 205 patients who had disease control after 18 weeks of platinum and fluoropyrimidine-based chemotherapy were randomized to receive switch maintenance with durvalumab or surveillance. PFS was similar between the two groups (3.0 vs 3.2 months; HR = 0.84; 95% CI: 0.62–1.14; one-sided p = 0.13) as well as OS (11.3 vs 11.4 months; HR = 0.98, 95% CI: 0.70–1.36; one-sided p = 0.45). 19
The JAVELIN Gastric 100 was a phase III randomized trial that investigated switch maintenance therapy with avelumab after a 12-week course of FOLFOX/CAPOX versus chemotherapy continuation. Four hundred ninety-nine patients were enrolled in the trial; the primary endpoint of OS was not met (median OS 10.4 vs 10.9 months; HR = 0.91; 95% CI: 0.74–1.11; p = 0.1779) irrespectively of the pre-specified PD-L1 cut off (i.e., ⩾1% by 73-10 assay). In patients with PD-L combined positive score (CPS) ⩾1 median OS was 16.2 months with avelumab and 17.7 months with chemotherapy (HR = 1.13; 95% CI: 0.57–2.23; p = 0.6352). As expected, a consistent benefit from avelumab was described in the Microsatellite Instability (MSI)-High subgroup. Furthermore, no difference in PFS or objective response rate (ORR) was detected between the two groups. 20 Consistently, sequential chemotherapy followed by early ICI does not seem to be supported by other trials such as the recently reported MOONLIGHT platform study, which investigated short-course FOLFOX followed by low-dose ipilimumab plus nivolumab compared to FOLFOX until progression. 21 It should be pointed out that these trials were conducted before the introduction of high PD-L1 expression as a selection biomarker for chemo-ICI upfront combinations.
Two poly ADP ribose polymerase inhibitors (PARPi), pamiparib and rucaparib, were also investigated as maintenance treatment after first-line chemotherapy in patients with molecularly unselected advanced GC. In the PARALLEL-303 phase II trial, 136 patients who received an induction phase with chemotherapy up to 28 weeks were randomized to either pamiparib or placebo. Median PFS was 3.7 months in the experimental arm compared to 2.1 months in control arm (HR = 0.8; 95% CI: 0.5–1.2; p = 0.1428). OS was 10.2 months with pamipanib versus 12.0 months with placebo. A significant increase in toxicity was reported in the experimental arm with an incidence of grade 3 or higher AEs in 26.8% of patients receiving pamiparib compared to 9.2% in controls. 22 In the phase II randomized PLATFORM trial, maintenance rucaparib was compared to surveillance after 18 weeks of platinum-based chemotherapy. Among the 125 patients randomized, a numerically higher median PFS was reported with rucaparib (4.2 vs 2.8 months; HR = 0.70; 97.5% CI: 0.46–1.08; p = 0.063), which did not translate into improved OS. 23 The lack of efficacy of PARPi in the maintenance or second-line setting 24 may be related to the rapid resistance to platinum agents and lack of selection potentially based on homologous recombination deficiency (HRD). A genomic HRD signature (HRDSig) has been described in approximately 10% of gastric cancers. 25 Importantly, HRDSig-positive tumors displayed enhanced sensitivity to PARPi and oxaliplatin even in the context of wild-type genes involved in the homologous recombination pathway such as BRCA, ATM, CDK12, or PALB2. 26 Therefore, prospective data are needed to assess the efficacy of PARPi in patients enriched for HR repair-related alterations.
Anti-vascular monoclonal antibodies or tyrosine kinase inhibitors (TKIs) were used within switch maintenance contexts. The phase II a-MANTRA study assessed the efficacy of regorafenib, an oral multi-targeted TKI, as maintenance therapy in patients with advanced GC after a first line of platinum and fluoropyrimidine-based therapy compared to placebo. The study was prematurely closed due to the introduction of immunotherapy in the first-line setting, and the trial did not meet the primary endpoint of PFS (5.19 vs 3.91 months; HR = 0.736; 80% CI: 0.51–1.04; p = 0.1318). 27 Ramucirumab plus capecitabine was assessed as a switch maintenance versus observation in patients completing 18-week induction with platinum-based chemotherapy within the PLATFORM trial. Herein, median PFS was increased in the experimental arm (5.5 vs 2.5 months; HR = 0.33; 95% CI: 0.17–0.63; p < 0.001) with a manageable safety profile. 28
The ARMANI phase III trial demonstrated the superiority of this non-cross-resistant regimen used in the switch maintenance setting. The trial investigated paclitaxel plus ramucirumab, as a switch maintenance versus continuation of chemotherapy in 280 patients with disease control after an induction phase of 3-month oxaliplatin and fluoropyrimidine in patients with advanced HER2-negative GC. The primary endpoint of PFS was met since median PFS were 6.6 versus 3.5 months in the experimental and control arm, respectively (HR = 0.61; 95% CI: 0.48–0.79; p = 0.0002). The PFS gain translated into improved OS and PFS-2, the latter being defined as the time from random assignment to evidence of tumor progression on the next-line treatment regimen or death, suggesting that the initial benefit of this upfront strategy was retained throughout the subsequent therapeutic lines. Importantly, a similar proportion of patients in the two arms received any additional second-line therapy (58% vs 56%), and only 45% of patients in the control arm were fit enough to receive paclitaxel plus ramucirumab as subsequent regimen. The paclitaxel plus ramucirumab regimen was associated with an increase in grade 3 or higher AEs, mostly neutropenia and ramucirumab-specific toxicities. However, a similar rate of grade 3 or higher peripheral neuropathy was reported. 29
Conclusions and future perspectives on maintenance strategies
Overall, these data show that continuation maintenance with fluoropyrimidines is generally not effective because most patients rapidly develop chemotherapy resistance with modest life expectancy (Table 1).
Some of the presented studies reported OS exciding the historical benchmark typically observed in unselected patients with metastatic gastric cancer. This discrepancy can be largely attributed to trial design as many of these studies randomized patients only after achieving disease control following first-line induction chemotherapy and thus intrinsically selecting a population with better prognosis compared to the broader metastatic population. Moreover, most trials were conducted in molecularly unselected cohorts, and biomarker data were either unavailable or inconsistently reported, further complicating comparisons between studies.
The ARMANI trial demonstrated a clear survival benefit of a switch maintenance or early second-line strategy and may represent a therapeutic option in patients with advanced HER2-negative GC who are non-eligible for upfront immunotherapy or ICIs (Figure 1). This benefit may be even higher in those patients with lack of RECIST response to oxaliplatin and fluoropyrimidine doublets, as documented in the trial subgroup analyses conducted in patients with measurable disease. 29 Moreover, molecular selection is crucial for switch maintenance with targeted agents or immunotherapy, as inadequate patient selection may account for the failure of many trials. 26 Nowadays, a substantial proportion of patients is eligible for chemotherapy in combination with PD-1, CLDN18.2, or FGFR2b inhibitors depending on ongoing study results and regulatory approvals, and the next generation of clinical trials may assess the optimal maintenance strategies in these biomarker-selected subgroups.

Algorithm of first-line therapy options divided per induction treatment and maintenance treatment. Although chemotherapy de-escalation was not assessed in most of randomized clinical trials, interruption of platinum agent administration can be considered in everyday clinical practice.
Second- and later-line treatments
The morbidity associated with disease progression during first-line chemotherapy significantly impacts the patient’s ability to receive subsequent anticancer therapies. In modern trials, the proportion of patients eligible for second-line therapy is below 50% irrespective of what they received in the first-line setting (Table 2). Longer duration of upfront chemotherapy, no weight loss, younger age, HER2 positivity, and synchronous disease were significantly associated with higher chance of receiving second-line therapy in a large real-world dataset. 30 In fact, patients who experience early failure of first-line chemotherapy (i.e., <6 months) have a significantly poorer median OS with second-line therapy compared to those who had a more effective first line treatment (>9 months; 4.0 vs 7.1 months). 31 Importantly, the survival benefit of second line therapy is supported by several randomized clinical trials with either single agents or combination regimens compared to best supportive care and retrospective datasets. 30
Proportion of patients receiving subsequent therapies in modern first-line clinical trials.

n defined as number of patients who received subsequent therapies/number of patients enrolled in each arm.
Data for pembrolizumab only arm (n = 254) are not reported.
Data for Nivolumab + ipilimumab arm (n = 409) are not reported.
CAPOX, capecitabine and oxaliplatin; FOLFOX, folinic acid, fluorouracil, and oxaliplatin; TFOX, docetaxel, folinic acid, fluorouracil, and oxaliplatin.
Current guidelines recommend the combination of ramucirumab with or without paclitaxel as second-line therapies irrespective of molecular selection moving from the results of the RAINBOW and REGARD trials.32–35 The RAMIRIS phase II trial randomized patients to receive second-line FOLFIRI in combination with ramucirumab or paclitaxel plus ramucirumab. The trial failed to meet the primary endpoint of 6-month OS rate for FOLFIRI and ramucirumab. However, this combination is favorably compared to standard paclitaxel plus ramucirumab, and the magnitude of benefit was higher in patients who received docetaxel in the first line or perioperative setting. 36 Additional data are reported in Table 3.
Pivotal randomized trials evaluating subsequent lines of therapy.
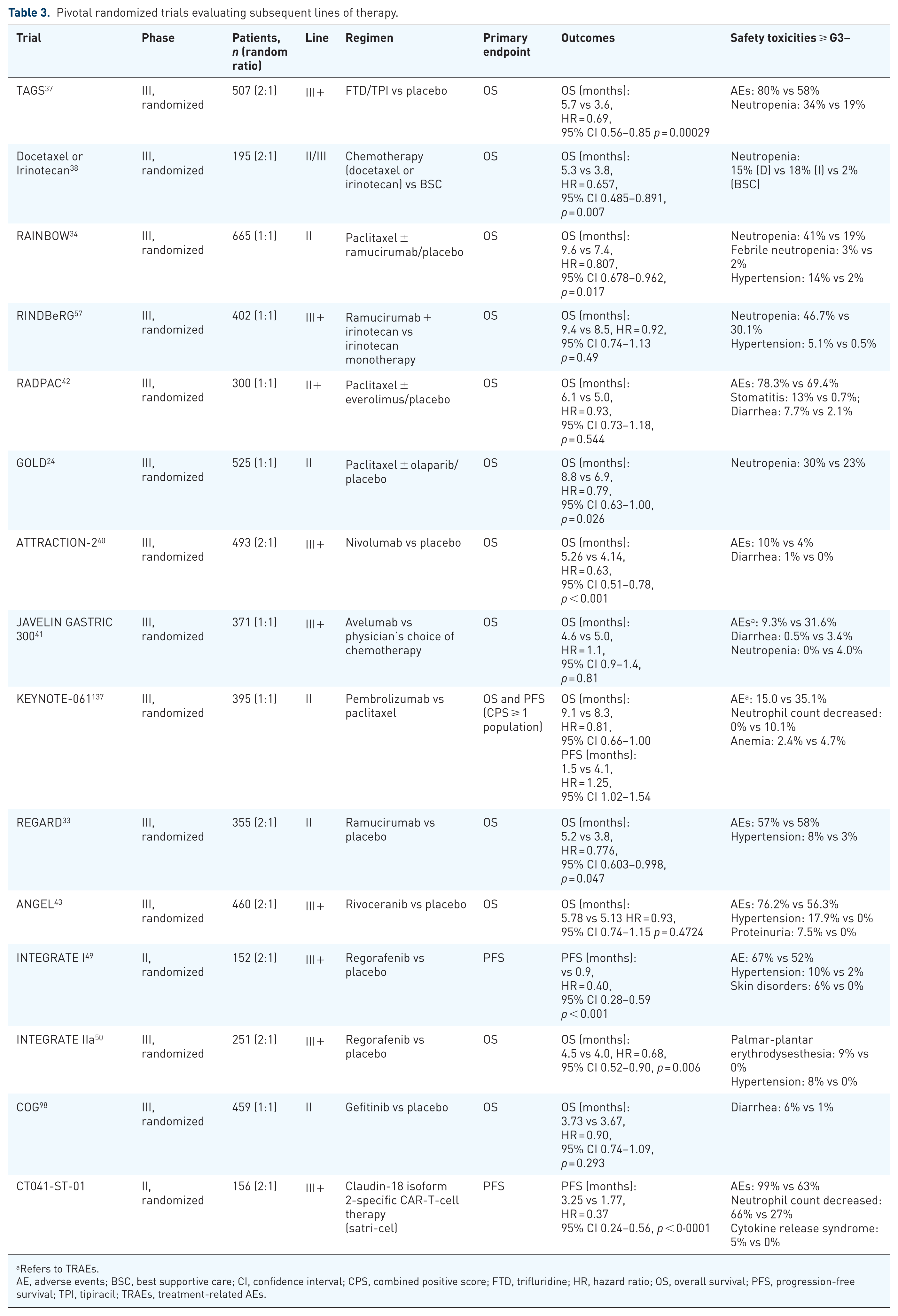
Refers to TRAEs.
AE, adverse events; BSC, best supportive care; CI, confidence interval; CPS, combined positive score; FTD, trifluridine; HR, hazard ratio; OS, overall survival; PFS, progression-free survival; TPI, tipiracil; TRAEs, treatment-related AEs.
Importantly, the magnitude of benefit of systemic treatment in patients with heavily pretreated GEJ cancer is quite modest in later lines of therapy. However, even in third or later lines, the advantage of administering single-agent or combination chemotherapy is clear when compared to BSC alone (Table 3). Currently, the standard of care in later line mainly includes single-agent chemotherapy. Trifluridine/tipiracil (FTD/TPI), an oral fluoropyrimidine that combines thymidine-based nucleoside analog combined to a thymidine phosphorylase inhibitor, is administrated from the randomized TAGS trial in patients who had received at least two prior regimens. 37 Additional options include irinotecan or docetaxel. 38 Before immunotherapy entered the clinical practice as first-line treatment in combination with chemotherapy for PD-L1 positive patients, ICIs were investigated in heavily pretreated patients, demonstrating promising benefit in this population.39–41 Moreover, several trials tested other compound in advanced lines, including a combination of paclitaxel and everolimus, a kinase inhibitor of the mammalian target of rapamycin serine/threonine kinase signal transduction pathway, or mono-agent rivoceranib, a vascular endothelial growth factor receptor-2 (VEGFR-2) inhibitor. However, none of these agents demonstrated an advantage in terms of survival and thus are not considered a standard of care.42,43
VEGF/VEGFR axis targeting
VEGF plays a pivotal role in the progression of GC by promoting tumor angiogenesis, a process essential for supplying oxygen and nutrients to the rapidly expanding tumor mass. Effective VEGF/VEGFR blockade may represent an option in patients who are non-eligible for specific biomarker-driven treatments. In fact, while the integration of antiangiogenetic drugs ramucirumab and bevacizumab with first-line platinum and fluoropyrimidine chemotherapy did not lead to superior outcomes over chemotherapy in several trials, the use of these agents may be important in the subsequent lines of treatment.44–48
In second or later treatment lines, different antiangiogenic agents beyond ramucirumab have been evaluated, either as single-agent or in combination with chemotherapy or immune checkpoint inhibition. The INTEGRATE I phase II trial demonstrated a PFS gain with regorafenib compared to placebo (median PFS 2.6 vs 0.9 months; HR = 0.40; 95% CI: 0.28–0.59; p < 0.001). 49 In a pooled analysis of the phase III randomized INTEGRATE IIa and INTEGRATE regorafenib showed a statistically significant but numerically modest increase in median OS (HR = 0.70; 95% CI: 0.56–0.87; p = 0.001). 50 The ANGEL phase III trial did not meet its primary endpoint, failing to demonstrate a survival advantage for rivoceranib, a VEGFR-2 inhibitor versus placebo. 43
Several trials evaluated potential strategies for implementing the use of antiangiogenic drugs beyond the first line. These studies included early introduction of ramucirumab as switch maintenance as mentioned before in the ARMANI or PLATFORM trials. Additional strategies evaluated the combination of antiangiogenics with ICIs or a sequential VEGF/VEGFR axis blockade approach.
Several trials assessed the combination of antiangiogenic drugs and ICIs since a potential synergy of dual VEGFR and PD-(L)-1 blockade relies on the immunomodulatory effect of VEGF/VEGFR-targeting agents within the tumor microenvironment. 51 Regorafenib was combined with nivolumab in the phase Ib REGONIVO trial. Twenty-five patients with metastatic GC who had previously received at least two lines of therapy were evaluated in the trial. Although the primary endpoint of the study was to determine the maximum tolerated dose of regorafenib when combined to nivolumab, promising results in terms of ORR (44%), PFS (5.6 months), and OS (12.3 months) were reported as well. 52 Moving from these results the phase III INTEGRATE IIb is ongoing to evaluate the combination compared to investigators’ choice of chemotherapy. 53
In the AIO-STO-0218 trial, treatment paclitaxel in combination with ramucirumab and avelumab in the second-line setting after oxaliplatin and fluoropyrimidine therapy resulted in 6-month OS rate of 71.2%. 54 Similarly, in the phase I/II UMIN-CTR trial with second-line paclitaxel, ramucirumab and nivolumab demonstrated a median OS of 13.1 months. 55 As expected, in both trials, numerically higher survival was observed in patients with higher PD-L1 expression. Similarly, higher ORR was reported for regorafenib and nivolumab in patients with PD-L1 CPS ⩾1 compared to those with PD-L1-negative tumors (60% vs 25%).
Sequential VEGF/VEGR axis inhibition
Preclinical models and these exploratory analyses support the hypothesis that sequential VEGF/VEGFR inhibition may overcome adaptive resistance mechanisms, such as VEGF-independent angiogenesis and hypoxia-driven pathways. Combining agents with distinct pharmacokinetic profiles or mechanisms of action may sustain anti-angiogenic pressure and improve survival. Despite a promising background, the clinical relevance of this approach has been largely disappointing so far. The phase II RE-ExPEL study evaluated ramucirumab beyond progression in combination with FTD/TPI yielding promising results, with a median OS of 9.07 months.37,56 However, the large Japanese RINDBeRG trial randomized 402 patients pre-treated with ramucirumab to receive irinotecan with or without ramucirumab. The OS data of this study did not show a survival benefit with the addition of ramucirumab even if a modest increase in PFS was reported. 57 The results of this trial challenge the use of ramucirumab beyond progression in patients with gastric cancer, at least in the Western population and different antiangiogenic drugs may be used in this context. In fact, the subgroup analyses of ANGEL and INTEGRATE trials showed that the efficacy of treatment with either rivoceranib or regorafenib was retained irrespective of prior ramucirumab.
Biomarkers for VEGF/VEGFR axis blockade
To date, no validated predictive biomarkers of response have been identified. Several studies, conducted in patients receiving second-line treatment with ramucirumab and paclitaxel, have suggested a correlation between high baseline VEGF-A levels and reduced OS. Conversely, elevated VEGFR-2 levels 1 week after treatment initiation were associated with prolonged PFS and OS, positioning VEGFR-2 as a potential predictive biomarker, though unvalidated.58,59 However, exploratory analyses from the RAINBOW and INTEGRATE trials failed to identify predictive biomarkers for ramucirumab or regorafenib, and thus the identification of predictive biomarkers for antiangiogenic drugs remains elusive. 53
In conclusion, a sequential VEGF/VEGFR blockade does not represent a standard strategy in today’s management of advanced GC. However, even after progression on prior anti-angiogenic agents such as ramucirumab, continued inhibition of angiogenic pathways by innovative drugs can still confer a clinically meaningful survival benefit.
Specific strategies in HER2-positive disease
Overexpression of HER2 is observed in 7%–20% gastric cancers worldwide. 60 The combination of doublet chemotherapy, trastuzumab and pembrolizumab, represents a guideline-recommended option in patients with HER2-positive and PD-L1 CPS ⩾1 based on the results of the KEYNOTE-811 trial. 61
An anti-HER2-based strategy beyond the first line with trastuzumab deruxtecan (T-DXd) is currently recommended. 32 In fact, longitudinal HER2 blockade is a relevant strategy for HER2-addicted tumors. However, its clinical application has been challenging since several anti-HER2 agents have failed to show a survival benefit compared to standard second-line treatments after disease progression following first-line trastuzumab therapy. In fact, the use of trastuzumab across different lines of treatment appears to be challenging due to the temporal heterogeneity of HER2 under the therapeutic pressure of anti-HER2 agents. Retrospective data showed that the chance of HER2 down-scoring as acquired resistance driver to trastuzumab (i.e., loss of HER2 amplification or 2/3+ on IHC) occurs in up to 30% of patients and is particularly relevant in initially HER2 2+ tumors. 62 Similarly, a drop in HER2 somatic copy-number alterations after trastuzumab was detected by circulating tumor DNA (ctDNA) analysis. 63 Therefore, HER2 temporal heterogeneity under the therapeutic blockade is likely to impact on the efficacy continuous HER2 blockade. These findings have been recapitulated in several second-line trials with disappointing results. The phase II T-ACT study investigated paclitaxel with or without trastuzumab in 91 trastuzumab-refractory patients. The study found no significant differences in either PFS or OS (median PFS 3.68 vs 3.19 months; HR = 0.91; 80% CI: 0.67–1.22; p = 0.33; median OS 10.2 vs 9.95 months; HR = 1.2; 95% CI: 0.75–2.0; p = 0.20). However, in the small subset of evaluable patients, only 31% retained HER2 positivity (either IHC 3+ or 2+/FISH positive) after progression to first-line trastuzumab and before randomization. 64 Trastuzumab beyond progression in combination with paclitaxel plus ramucirumab was evaluated in the phase Ib/II Korean HER-RAM trial. ORR was 54% and median PFS 7.1 months. 65 Here, loss of HER2 expression after first-line trastuzumab was reported in 34.8% of evaluable patients. No significant association between HER2 dynamics and clinical outcomes was observed; however, these data may have been influenced by the limited number of patients and the confounding effect of concurrent paclitaxel plus ramucirumab administration. The ASPEN-06 trial evaluated the combination of paclitaxel, ramucirumab, and trastuzumab beyond progression with or without evorpacept, an anti-CD47 myeloid checkpoint inhibitor in the second or third line of treatment. The combination regimen yielded higher ORR (40.3% vs 26.6%; p = 0.095) and duration of response (15.7 vs 7.6 months). The magnitude of benefit for evorpacept was higher in the subset of patients with HER2 positivity retained after first-line trastuzumab (54.8% vs 23.1%). 66 Collectively, these data highlight the importance of longitudinal HER2 assessment to guide any sequential HER2 blockade. Moreover, the introduction of novel agents beyond trastuzumab allows an effective HER2 blockade across different treatment lines.
Specifically, ADCs represent a promising strategy in the continuum of care of HER2-positive tumors. Trastuzumab emtansine (T-DM1), trastuzumab conjugated with the microtubule inhibitor emtansine as payload, failed to improve OS and PFS in the GATSBY trial in previously treated HER2-positive advanced GC (median OS 7.9 vs 8.6 months; HR = 1.15; 95% CI: 0.87–1.51; p = 0.86) compared to paclitaxel or docetaxel. In this trial, up to 80% of patients received prior trastuzumab. Even if no clinical subgroup showed an overall treatment benefit with T-DM1, the worst outcomes were observed in patients with initially HER2 2+/ISH positive tumors receiving T-DM1. 67
T-DXd, an ADC that combines trastuzumab with a novel topoisomerase I inhibitor, overcomes the limitations of T-DM1 due to its pharmacological properties. Specifically, T-DXd has a stable and homogeneous drug-to-antibody ratio, a novel potent payload and by-stander demonstrated effect. 68 Due to its specific properties, T-DXd may demonstrate activity also in HER2 heterogeneous tumors. Moreover, preclinical data showed that T-DXd may bypass genomic driver of primary resistance to either anti-HER2 monoclonal antibodies and TKIs, such as downstream MAPK alterations (e.g., KRAS amplification). 69 The clinical development of T-DXd recapitulated these preclinical findings. The results from the DESTINY-Gastric01 trial and DESTINY-Gastric02 led to global approval for T-DXd after failure of prior trastuzumab treatment. In the phase II DESTINY-Gastric01 study, conducted in an Asian population, 187 patients with advanced GC who had been previously treated with at least two lines of therapy including trastuzumab were randomized 2:1 to receive T-DXd or the physician’s choice chemotherapy. The primary endpoint was ORR, while the key secondary endpoint was OS. Notably, the experimental treatment demonstrated a significantly higher ORR compared to chemotherapy (42.9% vs 12.5%, p < 0.01). This benefit was also reflected in median OS (12.5 vs 8.4 months, p = 0.0097). The notable AEs associated with T-DXd included myelosuppression and interstitial lung disease, which were managed through dose reduction or treatment interruption. 70 Translational analysis of this trial showed that an enrichment of tumor responses was seen in patients with IHC 3+ versus 2+ tumors (58.2% vs 28.6%). Importantly, HER2 status was mostly assessed in trastuzumab-naïve samples and may have changed under the selective pressure of previous HER2 blockade. ctDNA, on the other hand, can provide real-time information on HER2 status. In fact, higher HER2 mRNA gene expression, presence of plasma HER2 amplification, and high plasma HER2 copy number were associated with increased tumor response after T-DXd in this patient population. Tumor responses were observed regardless of the presence of MET or EGFR co-amplification (though to a lesser extent), or RAS co-mutations, which are drivers of primary resistance to trastuzumab. 71 These results were further supported by the single-arm phase II DESTINY-Gastric02 study conducted in a Western population. This study enrolled patients treated with trastuzumab in the first line and assessed for HER2 status after progression. The observed ORR was 33%, with a median OS of 12.1 months. In this trial, ORR was higher in IHC 3+ versus 2+ tumors (47.1% vs 10.0%). 72 The DESTINY-Gastric04 trial compared T-DXd to standard paclitaxel plus ramucirumab in patients with retained HER2 positivity after progression to a trastuzumab-containing first line regimen. T-DXd significantly improved OS over paclitaxel plus ramucirumab (median OS 14.7 vs 11.4 months; HR = 0.70; 95% CI: 0.55–0.90; p = 0.004). The treatment effect was consistent across all the key clinical and molecular subgroups including patients with HER2 2+/ISH+ tumors. 73 These latter patients represented a limited proportion of the trial population.
These data support the use of T-DXd as a preferred second-line regimen in patients with retained HER2 positivity after trastuzumab exposure to allow an effective longitudinal HER2 blockade across treatment lines. Additional trials, such as DESTINY-Gastric03, will properly assess the role of T-DXd in the first line and allow the design of innovative sequencing trials. 74 In fact, even if T-DXd or novel ADCs represent the individual agent with the higher activity as monotherapy in patients with HER2-positive advanced gastric cancer and may bypass several genomic drivers of resistance to trastuzumab, a lower degree of HER2 expression (e.g., IHC 2+) may impair its activity. Importantly, patients with HER2 2+ have been underrepresented in clinical trials, and more data are required to draw definitive conclusions in this subgroup.
Additional HER2 ADC are under development. There were some promising results with Disitamab vedotin (RC48) in combination with toripalimab, a PD-1 inhibitor in a recent phase I trial. The trial enrolled patients with advanced GC with HER2 IHC ⩾1 or ISH positive. Overall, ORR was 43% with a median PFS of 6.2 months. Activity was observed regardless of HER2 IHC score or prior anti-HER2s. 75 Additional promising results were observed in the phase I trial of ARX788, anti-HER2 ADC with AS269 as cytotoxic payload, which showed an ORR of 37.9% in patients who received trastuzumab. The confirmed ORR was 41.2% (7/17) in the HER2-positive patients who were centrally confirmed. 76
Additional data will address optimal sequencing of anti-HER2 monoclonal antibodies versus ADCs and degree of HER2 expression to enrich for activity of selected agents.
Future perspectives
EGFR inhibition
Targeting the EGFR pathway represents a promising strategy in patients with GC e GEJ cancer harboring EGFR amplification. The first trials involving anti-EGFR drugs in a biomarker unselected patient population were mostly disappointing. Several phase II trial evaluated the activity of anti-EGFR-based combinations in patients with metastatic gastroesophageal tumors, including cetuximab, panitumumab, nimotuzumab, erlotinib, gefitinib, matuzumab, and icotinib.77–94 Moving from this background, randomized phase III studies were conducted in the first-line setting of metastatic disease. The EXPAND trial assessed the efficacy of the addition of cetuximab to first-line chemotherapy, 95 while the REAL3 trial evaluated the combination of chemotherapy and panitumumab in patients with previously untreated GC/GEJ cancer. 96 In the EXPAND trial, the median PFS was 4.4 months in patients treated with chemotherapy and cetuximab and 5.6 months in patients treated with chemotherapy (HR = 1.09; 95% CI: 0.92–1.29; p = 0.32), with no apparent benefit from the addition of cetuximab. In the EXPAND trial cohort, the EGFR expression was generally low; however, a survival benefit was shown in patients with high EGFR expression by IHC. In the REAL3 trial, the median OS in patients treated with chemotherapy was 11.3 versus 8.8 months in patients treated with chemotherapy and panitumumab (HR = 1.37; 95% CI: 1.07–1.76; p = 0.013). 97 The phase III COG trial assessed the efficacy of gefitinib in the second line of treatment of unselected patients with esophageal or GEJ cancers. 98 The addition of gefitinib demonstrated only a modest benefit in PFS in the trial population, which was mostly restricted to patients with EGFR-amplified tumors (HR for PFS/OS 0.29/0.21). 99 OS did not differ between the gefitinib and placebo groups, with a median OS of 3.73 and 3.67 months, respectively (HR = 0.90; 95% CI: 0.6–1.18; p = 0.29). Collectively, these trials suggest that EGFR amplification represents the strongest biomarker for EGFR addiction and therapeutic vulnerability to EGFR blockade in advanced GC. In this context, Maron reported on the activity of anti-EGFRs in patients with EGFR amplification. Maron and colleagues identified EGFR amplification in 5% of a selected population of patients with gastroesophageal cancers and treated 7 of them with anti-EGFR-based combinations, either first-line FOLFOX combined with ABT-806 (anti-EGFR monoclonal antibody), second-line FOLFIRI plus cetuximab, or third or later line cetuximab monotherapy.100,101 In this biomarker-selected population, ORR was 58% with a median PFS of 10 months. As anticipated, primary resistance to anti-EGFRs was related to co-occurrence of several genomic alterations that bypass the therapeutic blockade of EGFR, including co-amplification of KRAS or HER2/MET. 102 In a subsequent study, a cohort of 60 patients with advanced gastroesophageal cancer carrying EGFR amplification was treated with on- or off-protocol EGFR inhibitors, of which 31 (52%) with concurrent chemotherapy. The study showed an ORR of 43% with a median PFS of 4.6 months. 103 In conclusion, EGFR amplification should be regarded as one of the most promising targets in the context of metastatic gastric cancer. Other than the aforementioned co-amplification of KRAS, HER2/MET, or other oncogenes involved in the EGFR pathway, other plausible resistance mechanisms include the characteristic spatial and temporal EGFR heterogeneous expression. Novel drugs could tackle some of these issues and increase the efficacy of EGFR inhibition in this subset of patients such as amivantamab, a dual targeting bispecific antibody which simultaneously inhibits EGFR and MET, or ADCs which are also active in heterogeneous tumor tissues to their bystander effects. Moreover, a refined definition of predictive biomarkers will be necessary to select the patients, which are more likely to benefit from this strategy. 104
Novel ADCs
The encouraging results of the first trials on ADCs have led to increased efforts by clinical researchers to bring these new drugs into the first and subsequent lines of therapy. Several ADCs are in clinical development in patients with advanced gastric cancer.
Trophoblast surface antigen 2 (TROP-2) is overexpressed in about 66% of gastric cancers. 105 Sacituzumab govitecan is a conjugate of an antibody active against TROP-2 coupled with SN-38, the active metabolite of irinotecan, which is mainly indicated in the second-line treatment of metastatic triple-negative breast cancer. A phase I/II basket clinical trial evaluated sacituzumab govitecan in various epithelial cancers, including esophageal and GC, demonstrating manageable toxicity. 106 Overall, 495 patients were included in the trial, including 19 patients (3.8%) with esophageal cancer and 5 (1%) patients with gastric adenocarcinoma. 107 A recent phase Ib/II trial specifically tailored for patients with advanced gastroesophageal cancer is currently ongoing and will provide new evidence regarding this specific field of treatment. 108 MK-2870 is another TROP-2 ADC linked to a belotecan-derivative topoisomerase 1 inhibitor, which demonstrated durable responses in monotherapy heavily pretreated GC/GEJ cancers in a phase II trial. 109 Among 41 response-evaluable patients treated with MK-2870, the ORR was 22% and Disease Control Rate (DCR) was 80.5% at 9 weeks of follow-up, with a median Duration of Response (DoR) of 7.5 months and a manageable safety profile. A phase III trial is currently comparing MK-2870 versus treatment of physician’s choice after ⩾2 prior lines of systemic therapy. 110 Closely recalling the history of T-DXd, the assessment of the degree of TROP-2 expression will be crucial to determining the activity of TROP-2 ADCs.
CLDN18.2 overexpression is observed in up to 25% of patients with gastroesophageal cancers making it an attractive target for both monoclonal antibodies (e.g., zolbetuximab) and ADCs. 111 The phase I SHR-A1904 trial evaluated DS-9606a, a novel anti-CLDN18.2 ADC linked with a topoisomerase I inhibitor payload, in patients with metastatic solid tumors demonstrating a favorable safety profile, with no dose-limiting toxicity, dose-dependent increase in AEs, or treatment discontinuations. Preliminary analyses confirmed activity in germ-cell tumors and gastric cancer. 112 Another phase I/II trial assessed the safety and efficacy of RC118, an anti-CLDN18.2 ADC carrying microtubule-disrupting agent monomethyl auristatin E (MMAE). AEs were observed in all 18 enrolled patients, with 38.9% experiencing grade 3 or higher AEs, mostly nausea, vomiting, and decreased appetite. No treatment-related mortality was reported. Among the 17 patients evaluable for efficacy per RECIST v1.1, the ORR was 47.1% and the DCR was 76.5%. The median PFS was 3.9 months with a median follow-up duration of 4.4 months. 113 The phase I KYM901 trial assessed the activity and safety of CMG901, a different ADC composed by an anti-CLDN18.2 monoclonal antibody linked to MMAE in patients with advanced GC/GEJ cancer and other solid tumors. 114 Twenty-seven patients were enrolled in the dose-escalation phase and 107 patients with CLDN18.2 IHC ⩾2+ expression in ⩾5% of tumor cells in the dose-expansion phase. The safety profile was acceptable: G3 or higher AEs were most commonly related to myelosuppression and vomiting. Overall, ORR was 29% which rose slightly to 33% in patients with CLDN18.2 high tumors defined as at least IHC 2+ in at least 20% of tumor cells, which is much lower than the cutoff used for zolbetuximab efficacy. IBI343 is an anti-CLDN18.2 ADC with a payload consisting of the topoisomerase inhibitor exatecan. IBI343 induced tumor responses in patients with CLDN18.2 high tumors (defined by IHC at least 2+ in at least 40% of tumor cells; ORR 37.5%). Higher responses were observed in patients with higher amounts of CLDN18.2. 115
These data emphasize the importance of ADCs with the bystander effect, as it is a key determinant of their efficacy. This effect allows for the distribution of antineoplastic activity even in the presence of heterogeneous expression of a given tumor target. Recalling the history of HER2 longitudinal blockade, future trials will evaluate the optimal therapeutic sequencing of monoclonal antibody-based therapies and ADCs in patients with CLDN18.2-positive tumors, while also uncovering the distinct mechanisms of both primary and acquired resistance that may influence sequencing decisions.
HER3 is overexpressed in ~50% of gastric cancers. 116 U3-1402 is an anti-HER3 ADC, which already demonstrated promising activity in other types of tumors, and various other ADCs such as AMT-562, MCLA-128, MM-121, CDX/KTN3379, and GSK-2849330 are currently assessing the activity of this novel treatment in gastric and non-gastric cancer.
Mucin1 (MUC1) is overexpressed in several epithelial cells, including the stomach mucosa. Many studies demonstrated significant in vitro activity of anti-MUC1 ADCs against gastric cancer cells. A few in vivo clinical trials assessed the activity and safety of anti-MUC1 ADCs, specifically targeted against the cancer antigen (CanAg) isoform of MUC1. 117 A phase II trial enrolled six patients with CanAg-positive gastric or GEJ cancer and treated them with huC242-DM4, an anti-MUC1 ADC, demonstrating a partial response in one patient and drug-related ocular toxicities in three patients, and therefore the program was later discontinued. 118
B7-H3 is an emerging biomarker in gastric adenocarcinoma, and its expression is significantly correlated with HER2 over-expression.119,120 Various trials are currently evaluating anti-B7-H3 ADCs in other settings with promising results, and presumably, in the near future, patients with gastric cancer may also be involved in trials.
Nectin-4 is frequently overexpressed in gastric cancer. Various ongoing trials are assessing the activity and safety of anti-Nectin-4 ADCs in advanced solid tumors. 121 A phase I ongoing study is assessing the safety and activity of ABBV-400, an ADC consisting of the c-Met-targeting telisotuzumab conjugated to a novel topoisomerase I inhibitor payload, in advanced gastric/GEJ cancer that progressed after ⩽2 prior chemotherapy lines. The preliminary efficacy results demonstrated a tolerable safety profile and antitumor activity. 122
CAR-T cell therapy
CARs are synthetic receptors specifically engineered to provide T cells the ability to recognize specific target antigens expressed on tumor cells. CAR-T cell therapy is characterized by high on-target specificity and, on the other hand, by significant adverse effects, which often lead to the discontinuation of treatment.123,124 The use of CAR-T cells still lacks evidence in the setting of solid tumors. To date, only two studies involving CAR-T cell therapy in solid tumors have described a range of activity comparable to that observed in hematologic tumors trials.125–127 One of these two studies evaluated CLDN18.2-specific CAR-T cells in patients with GC. After the publication of promising results in preclinical studies, CLDN18.2-redirected CAR-T cells were tested in a phase I, single-arm trial by Qi et al.127,128 in 98 patients with pre-treated advanced CLDN18.2-positive GI cancers, of which 73 (74.5%) with gastric cancer. The final results of the study showed that among 98 patients treated with satricabtagene autoleucel (satri-cel) the ORR was 38.8% and the DCR was 91.8%, with a median PFS and OS of 4.4 and 8.8 months, respectively. Among the 98 patients treated with CLDN18.2-redirected CAR-T cells, 1 had a complete response, specifically in the monotherapy cohort. No dose-limiting toxicities or treatment-related deaths were reported, thus combining an acceptable safety profile with a significant degree of activity. Satri-cel was also tested in the phase Ib ELIMYN18.2 study in heavily pretreated patients with CLDN18.2-positive metastatic GC/GEJ or pancreatic cancer. 129 Nineteen patients received satri-cel at three dose levels, of which seven with gastric/GEJ cancer. All 19 patients experienced at least 1 AE, while no dose-limiting toxicities occurred, and 17 patients experienced cytokine release syndrome. Among the gastric cancer subgroup, the ORR was 42.9% (3/7), the median DoR was 6.9 months, and the clinical benefit rate was 57.1%. These promising data were supported by a single case of a target lesion complete response among the study population. 130 Satri-cel was evaluated in a randomized phase II trial in patients with CLDN18.2-positive tumors (defined as ⩾2+ in at least ⩾40% of tumor cells) refractory to at least two treatment lines, compared to investigator’s choice therapy. The study met its primary endpoint of PFS (median PFS 3.2 vs 1.7 months, HR = 0.37, 95% CI: 0.24–0.56, p < 0.0001). ORR was 35% with long-lasting tumor responses highlighting the ability of Satri-cel to unleashing durable antitumor immunity. 131
Other than CLDN18.2, other targets for CAR-T cell therapy in gastric cancer include, and are not limited to, HER2, TROP2, MUC1, CEA, EpCAM, mesothelin, prostate stem cell antigen, NKG2D, folate receptor 1, and ICAM-1, as these antigens have either higher expression or a different structure in gastric cancer cells. Several phase I/II and preclinical trials are currently ongoing and will eventually pave the way for the deepening of the therapeutic arsenal available to oncologists. 132
Conclusion
At least three decades of clinical trials demonstrated that the efficacy of first-line regimen is crucial since the chance of receiving later-line therapies is low due to performance status worsening or death after disease progression. Moreover, there is now clear evidence that first-line (switch-) maintenance strategies improve patients’ outcomes. These findings should be considered within the framework of modern disease management, which is increasingly defined by the emergence of multiple therapeutic targets beyond HER2 amplification/overexpression, particularly MMR deficiency, high PD-L1 expression, CLDN18.2 positivity, and FGFR2b overexpression. The upfront use of targeted therapies matched to these biomarkers has led to significant survival benefits for these patients. Therefore, key challenges for innovative clinical trials include determining the optimal therapeutic strategies in the context of overlapping biomarkers and integrating novel treatments—such as ADCs, bispecific antibodies, and CAR-T therapies—to address the spatial and temporal heterogeneity of established biomarkers.
