Abstract
The introduction of anti-HER2 therapies to the treatment of patients with HER2-positive breast cancer has led to dramatic improvements in survival in both early and advanced settings. Despite this breakthrough, nearly all patients with metastatic HER2-positive breast cancer eventually progress on anti-HER2 therapy due to
Keywords
Introduction
A better understanding of tumor biology and HER2 signaling has led to the development and approval of new HER2-targeted agents that, together with the use of continued anti-HER2 therapy beyond progression, have resulted in unpreceded survival outcomes in patients with advanced HER2-positive breast cancer. 1
The addition of trastuzumab to standard therapy dramatically improved prognosis for patients with HER2-positive breast cancer, and became a landmark in the treatment of these patients.2,3 The second anti-HER2 agent that was incorporated into routine practice of advanced HER2-positive disease was lapatinib, an oral tyrosine kinase inhibitor (TKI) that reversibly inhibits HER1 or epidermal growth factor receptor (EGFR) and HER2 kinases. The approval of lapatinib was based on the improvement in progression-free survival (PFS) found in a phase III trial when combined with capecitabine
Despite the outstanding improvement in survival with the introduction of anti-HER2 therapies alone or as dual HER2-blockade in the standard treatment of advanced disease, most patients ultimately develop progressive disease and die. Furthermore, up to 40–50% of patients with advanced HER2-positive breast cancer will develop brain metastases during their disease course. Better options for the prevention and treatment of brain metastases are clearly needed. 9 A growing understanding of the underlying mechanisms of primary and acquired resistance to anti-HER2 therapies and compensatory pathways as well as tumor heterogeneity and the tumor microenvironment is essential for the development of novel therapeutic strategies. A substantial number of novel anti-HER2 treatments are being investigated extensively in the preclinical and clinical settings to further improve patient outcomes. Here, we review the rationale and latest evidence of those novel treatments and approaches to overcome resistance in advanced HER2-positive breast cancer.
Mechanisms of resistance and response heterogeneity to anti-HER2 therapy
Many potential resistance mechanisms to anti-HER2 therapy have been described that ultimately lead to reactivation of the HER2 pathway or its downstream signaling, through pathway redundancy or stimulation of alternative survival pathways.
10
Some of these mechanisms include incomplete blockade of the HER2 receptor that activates compensatory mechanisms within the HER family (such as HER3), activation of alternative receptor tyrosine kinases (RTKs) or other membrane receptors outside of the HER family [such as insulin-like growth factor 1 receptor (IGF-1R)
11
and MET
12
], and alterations in downstream signaling pathways, such as hyperactivation of the PI3K/AKT/mTOR pathway13,14 by reduced levels of tumor suppressor genes (like
It has also been demonstrated that the cyclin D1-CDK4 pathway can mediate resistance to HER2-targeting therapies
Substantial preclinical and clinical studies support the bidirectional cross-talk between HER2 and estrogen receptor (ER) signaling when both receptors are expressed in breast cancer cells. 24 Tumors that express both ER and HER2 are less sensitive to endocrine therapy than ER-positive and HER2-negative tumors, and ER can act as an escape pathway to HER2 inhibition.25,26 Concurrent inhibition of ER together with dual anti-HER2 therapy can improve outcomes, as demonstrated in several trials in early and advanced HER2-positive breast cancer.27–29
The HER2Δ16 splice variant is a major oncogenic driver that promotes trastuzumab resistance. Preclinical data suggest trastuzumab-resistant HER2Δ16 cells are sensitive to the SRC kinase inhibitor dasatinib and data from a phase I/II (GEICAM/2010-04) study suggest there may be a signal for activity when dasatinib is combined with trastuzumab and paclitaxel in the first-line treatment for patients with advanced HER2-positive breast cancer.30,31 In addition, SRC activation by itself has been associated with trastuzumab resistance. 32
The mechanisms that contribute to T-DM1 resistance are not fully understood. There are multiple components to consider when identifying mechanisms of resistance for ADCs, such as the ones related to the antibody, the linker or the payload. Preclinical studies have shown that CDK1/cyclin B1 activity is needed for T-DM1 action. Silencing cyclin B1 induces resistance to T-DM1 while increasing the levels of cyclin B1 in resistant cells partially restores sensitivity. 33 Other potential mechanisms of T-DM1 resistance have been proposed including the reduction of the intracellular DM1 payload due to upregulation of multidrug resistance proteins (e.g. MDR1), 34 impaired lysosomal proteolytic activity 35 or lysosomal transporter loss (e.g. SLC46A3). 34
Interestingly, molecular imaging seems promising not only to further our understanding of tumor heterogeneity in advanced HER2-positive breast cancer but also to identify patients who will unlikely benefit from T-DM1. 36 In the prospective ZEPHIR trial, striking levels of inter- and intrapatient heterogeneity in HER2 expression were observed, with one-third of patients having little or no trastuzumab-zirconium uptake (HER2-Positron emission tomography (PET)/computed tomography (CT) scan [PET/CT scan]) across their metastatic sites. Moreover, the combined use of HER2-PET/CT scan and early fluorodeoxyglucose-PET/CT scan discriminated patients treated with T-DM1 with a median time to treatment failure (TTF) of 2.8 months from those with 15 months of TTF. 36 Despite the extensive translational research being conducted, most of the mechanisms of HER2 resistance and potential biomarkers of response or resistance either have not been clinically validated, or the results are contradictory. 37 To date, no biomarker beyond HER2 exists for patient selection for anti-HER2 therapy in HER2-positive breast cancer. Of note, the interpretation of mechanisms of resistance based solely in preclinical models can be challenging due to tumor heterogeneity, the complex nature of drug resistance and compensatory pathways, and the use of different tumor cell lines. Moreover, multiple mechanisms of resistance may coexist in the same cell.
Novel strategies to overcome resistance to HER2-targeted therapy
Replacement of current anti-HER2 therapies for improved anti-HER2 drugs
ADCs
ADCs are a therapeutic class that provide wider therapeutic window by more efficient and specific drug delivery. ADCs exploit target selectivity of monoclonal antibodies (MAbs) to deliver cytotoxic drugs to antigen-expressing cells to improve tumor selectivity and reduce damage to normal cells. 38 The success observed with the first-in-class T-DM1 has led to a rapid and extensive development of new ADCs. Table 1 lists several anti-HER2 ADCs in clinical development.7,39–41
HER2-directed ADCs in clinical development.
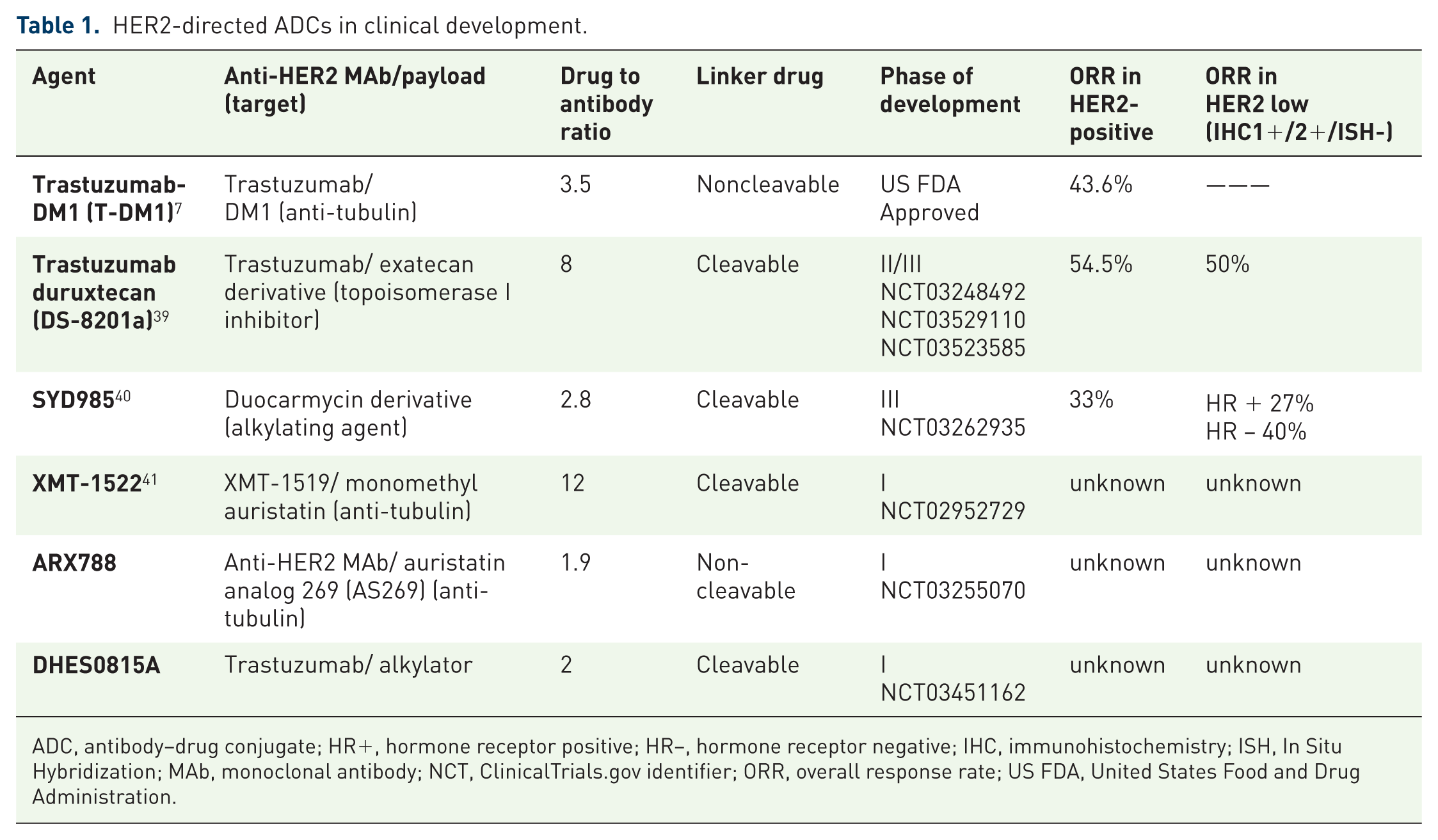
ADC, antibody–drug conjugate; HR+, hormone receptor positive; HR−, hormone receptor negative; IHC, immunohistochemistry; ISH, In Situ Hybridization; MAb, monoclonal antibody; NCT, ClinicalTrials.gov identifier; ORR, overall response rate; US FDA, United States Food and Drug Administration.
ADCT-502 (ADC Therapeutics) is also a novel pyrrolobenzodiazepine (PBD)-based ADC that targets HER2-expressing solid tumors, including breast cancers. 46 However, based on data from the phase I study (ClinicalTrials.gov identifier: NCT03125200) that showed that ADCT-502 did not meet the necessary efficacy and safety profile required for patient benefit, clinical testing of this drug was recently halted.
In preclinical models, another bivalent biparatopic HER2-targeting ADC that targets two nonoverlapping epitopes on HER2 and is conjugated with microtubule inhibitor demonstrated superior activity than T-DM1 in breast cancer models and was able to overcome T-DM1 resistance. This biparatopic ADC also demonstrated bystander killing activity. 47
In contrast with T-DM1, most of these new ADCs have a cleavable drug linker (see Table 1) that mediates the bystander killing effect. This is the passive diffusion of the free cytotoxin from target-positive cancer cells into the tumor microenvironment, killing neighboring cancer cells that are insensitive to the ADC because of the lack or limited target expression. This desired feature of those novel HER2-targeting ADCs, given that heterogeneity is frequent in HER2-positive breast cancer, may be however, a double-edged sword with an increased toxicity.
Novel anti-HER2 antibodies
Margetuximab (MGAH22, MacroGenics) is an Fc-optimized chimeric monoclonal antibody that binds to the same epitope as trastuzumab. Margetuximab has enhanced Fcγ receptor-binding properties with an increased affinity for CD16A polymorphisms and a decreased affinity for FcγRIIB (CD16B), an inhibitory receptor, which allows it to bind more tightly to effector cells and increase antibody-dependent cell-mediated cytotoxicity (ADCC); it also preserves the antiproliferative properties of trastuzumab. 48 A first-in-human phase I study demonstrated promising single-agent activity of margetuximab in heavily pretreated patients with HER2-positive solid tumors. Among 24 patients with metastatic breast cancer, the ORR was 17% and 3 out of the 4 responders remained on treatment for 39–54 months. 49 The most common AEs were grade 1–2 constitutional symptoms and no cardiotoxicity was observed. Margetuximab is currently being evaluated in the randomized phase III SOPHIA trial (ClinicalTrials.gov identifier: NCT02492711) that compares margetuximab plus chemotherapy with trastuzumab plus chemotherapy as a third-line therapy in patients with HER2-positive breast cancer after prior treatment with trastuzumab, pertuzumab, and T-DM1. The US FDA has granted Fast Track designation for the investigation of margetuximab for the treatment of patients with metastatic or locally advanced HER-positive breast cancer previously treated with anti-HER2-targeted therapy.
Bispecific antibodies
Bispecific antibodies (BsAbs) combine the functionality of two MAbs that target two different targets or epitopes, either in the same or in different receptors. BsAbs can interfere with two or more RTK signaling pathways, by inactivating either the RTKs or their ligand. Several are currently being studied in patients with advanced HER2-positive disease.
T-cell bispecific antibodies (TCBs) are engineered molecules that include, within a single entity, binding sites to the invariant CD3 chain of the T-cell receptor (TCR) and to tumor-associated or tumor-specific antigens. Binding to the tumor antigen results in crosslinking of the TCR and subsequent lymphocyte activation and tumor cell killing. However, on-target off-tumor effects caused by redirected lymphocytes can result in severe toxicities. Several are currently in clinical development:
Expression of the tumor-specific antigen p95HER2, a truncated form of HER2, occurs in about 40% of HER2-positive tumors. Rius Ruiz and colleagues have developed a TCB against p95HER2 (p95HER2-TCB) that has a potent anti-tumor effect on breast tumors expressing p95HER2, both
Novel TKIs
TKIs are orally bioavailable small molecules developed to further block the HER receptor family, acting on the intracellular domain. Those HER-directed TKIs have a lower molecular weight compared with MAbs, allowing them a more efficacious penetration through the blood–brain barrier and therefore, theoretically may be more effective for the treatment of HER2 brain metastases. Lapatinib was the first TKI approved in HER2-positive advanced breast cancer (and to date, remains the only one) based on the results described above. Dual blockade with lapatinib plus trastuzumab without chemotherapy also demonstrated benefit in OS in heavily pretreated patients with advanced disease, when compared with lapatinib alone. 55 Initial results with these molecules, however, have not been the ones initially expected, even in the treatment of brain metastases.56,57As a first-line therapy, lapatinib was found to be inferior to trastuzumab when combined with paclitaxel. 58 In the same way, afatinib was found to be not as effective as trastuzumab and less tolerated when each was combined with vinorelbine in a phase III trial (LUX-Breast1 study). 59 No difference was detected between lapatinib-capecitabine and trastuzumab-capecitabine for the incidence of brain metastases in the phase III CEREBEL (EGF111438) study. 56 In the phase II LUX-Breast3 trial, patients with HER2-positive breast cancer and progressive brain metastasis previously treatment with trastuzumab, lapatinib or both, were randomized to afatinib alone, afatinib plus vinorelbine or the investigator’s choice of treatment. Similarly, afatinib-containing regimens not only did not show better activity than investigator-selected treatments but also seemed to be less tolerated. No further development of afatinib for HER2-positive breast cancer is currently planned. There are, however, several novel TKIs in clinical development (Table 2).60–66
HER2-directed TKIs in clinical development.

CBR, clinical benefit rate; CNS, central nervous system; DCR, disease control rate; EGFR, epidermal growth factor receptor; m, months; NA, not applicable; NCT, ClinicalTrials.gov identifier; ORR, overall response rate; PFS, progression-free survival; TKI, tyrosine kinase inhibitor; US FDA, United States Food and Drug Administration; w, weeks.
Combinations of anti-HER2 agents with other drugs
Immunotherapy
Preclinical and clinical data suggest that HER2-positive breast cancer is immunogenic.
71
In contrast with luminal tumors, HER2-positive tumors have a higher mutational burden, and harbor higher numbers of TILs and programmed cell death protein 1 ligand (PD-L1) positivity.
72
In addition, mechanisms of action of anti-HER2 MAbs include not only ADCC but also the generation of adaptive immunity.
73
Together, these data support the rationale of combining anti-HER2 therapies with immune checkpoint blockade (anti-PD-1 or anti-PD-L1 agents). Results from the JAVELIN phase I study,
74
however, were disappointing and no responses were seen with single-agent avelumab in the subgroup of patients with advanced pretreated HER2-positive breast cancer. The first study to evaluate the addition of pembrolizumab to trastuzumab in patients with trastuzumab-resistant HER2-positive breast cancer was the PANACEA (IBCSG 45-13/BIG 4-13/KEYNOTE-014) study. This phase Ib/II study demonstrated that the combination was associated with an ORR of 15.2% and a median of PFS and OS of 2.7 months and 16 months, respectively, in PD-L1 positive patients.
75
However, these data also highlight the limitations of this combination, as no responses were seen in the PD-L1 negative cohort and most of the PD-L1 positive patients who initially responded eventually developed resistant disease. Several randomized studies are ongoing to further evaluate the role of immune checkpoints inhibitors in HER2-positive metastatic breast cancer. Results from the phase II KATE2 trial (ClinicalTrials.gov identifier: NCT02924883) assessing the efficacy and safety of T-DM1 in combination with atezolizumab or placebo in pretreated patients with HER2-positive advanced breast cancer were recently presented.
76
In this trial, the addition of atezolizumab to T-DM1 did not demonstrate a significant PFS benefit in the ITT population (8.2
Immunotherapy with HER2-targeting vaccines are also being currently investigated in clinical trials. The HER2 vaccine NeuVaxTM (Nelipepimut-S or E75 peptide combined with granulocyte macrophage-colony stimulating factor) 77 is being evaluated in two phase II clinical trials in combination with trastuzumab in breast cancer patients with HER2–3+ (ClinicalTrials.gov identifier: NCT02297698) and in HER2–1+/2+ (ClinicalTrials.gov identifier: NCT02297698), respectively. ETBX-021 is another HER2-targeting vaccine comprising an Ad5 vector and a modified HER2 gene insert that is being evaluated in a phase I clinical trial with locally advanced or metastatic HER2-low-expressing (IHC 1+/2+) breast cancer.
CDK4/6 inhibitors
There is a strong rationale to evaluate CDK4/6 inhibitors in HER2-positive breast cancer. Activity of CDK4/6 is regulated by several mechanisms that include mitogenic signaling pathways (such as HER2) by increasing
PI3K/Akt/mTOR inhibitors
As mentioned previously, dysregulations in the PI3K/AKT/mTOR pathway seem to play an important role in trastuzumab resistance. PI3K inhibition results in an enhanced HER2 signaling in HER2-overexpressing breast cancer, especially in an increased expression of HER2 and HER3.
88
Targeting both pathways could prevent the development of resistance. However, results of two phase III trials evaluating the role of everolimus, an mTOR inhibitor, in combination with either trastuzumab plus paclitaxel as first-line treatment (BOLERO-1)
89
or in combination with trastuzumab plus vinorelbine in trastuzumab-resistant (BOLERO-3)
90
advanced HER2-positive breast cancer were quite disappointing with a significant increase in toxicity. Although in the PFS subgroup analysis of both studies, the benefit of adding everolimus to the standard therapy seemed greater in patients who had HR-negative disease.89,90 Moreover, the combined biomarker analyses of the BOLERO-1 and BOLERO-3 trials demonstrate an improved PFS in patients harboring
Conclusion
Although the use of anti-HER2-targeted therapy has dramatically changed the outlook for patients with advanced HER2-positive breast cancer, almost all patients ultimately experience disease progression. Most of them advance to the point where no approved HER2-targeting treatment controls their disease. This might change in the near future as many promising anti-HER2 therapies are being developed in this setting. The newer HER2 ADCs such as DS-8201a and SYD985 may replace T-DM1 in the second-line treatment space or may be utilized in the third-line and beyond setting after progression on T-DM1. Moreover, these newer ADCs, as opposed to T-DM1, are active not only in patients with HER2-positive breast cancer but also in patients with HER2 low-expressing tumors (IHC1+ or 2+/FISH) in whom to date, there are no current anti-HER2 therapies specifically indicated. This desired feature of its bystander effect may be particularly useful in heterogeneous cancer cell populations among HER2-positive disease. On the other hand, the new TKIs, such as neratinib or tucatinib, are being explored with capecitabine and may demonstrate activity in the third-line setting, though may be associated with increased toxicity relative to trastuzumab. And finally, margetuximab plus chemotherapy is being compared with trastuzumab plus chemotherapy also in the third-line setting in a registrational study.
These strategies of combining optimized HER2-targeted therapies could potentially improve outcomes for HER2-positive breast cancer patients but may also allow de-escalation of treatment in selected patients, potentially sparing some from unnecessary treatments and their related toxicities. Hence, potential biomarkers of response or resistance such as intrinsic subtypes, might be helpful to better select patients for these strategies. Moreover, specific strategies for HR-positive, HER2-positive breast cancer are needed, such as the current studies exploring the use of CDK 4/6 inhibitors in the first and later line setting with anti-HER2 therapy and specific trials allowing patients with progressive brain metastases (who are generally excluded from clinical trials) should be enhanced. The underlying mechanisms of resistance to anti-HER2 therapies and compensatory pathways are indeed complex and a wide range of mechanisms of resistance may coexist in the same cell. Therefore, combining clinical strategies and strengthening international collaborations in the translational setting might be needed to validate predictive biomarkers beyond HER2, which will help us to better select patients and improve their outcomes.
Footnotes
Acknowledgements
We thank Kaitlyn T. Bifolck, BA, for her editorial support. Fundación AECC (Asociación Española Contra el Cáncer) and the Spanish Society of Medical Oncology (SEOM) grants (to S. Pernas)
Funding
This manuscript did not receive specific funding.
Conflict of interest statement
S. Pernas has received honoraria for talks and travel grants from Roche, outside of the submitted work and has served on advisory boards for Polyphor. S. Tolaney receives institutional research funding from Eli Lilly, Pfizer, Novartis, Exelixis, Eisai, Merck, Bristol Meyers Squibb, AstraZeneca, Nektar, Nanostring, Cyclacel, and Immunomedics. S. Tolaney has served on advisory boards or as a consultant for Eli Lilly, Pfizer, Novartis, Eisai, Merck, AstraZeneca, Nektar, Nanostring, Immunomedics, and Puma.
