Abstract
Background:
While cancer-specific mortality has decreased with therapeutic advances, a growing proportion of survivors are at risk of treatment-related adverse effects (AEs).
Objectives:
To assess cardiopulmonary disease-specific mortality risks in patients with non-small-cell lung cancer (NSCLC) based on pathological subtypes.
Design:
This was a retrospective cohort study. Mortality data from the Surveillance, Epidemiology, and End Results Program database and AE data from the FDA Adverse Event Reporting System were analyzed in NSCLC patients. In addition, an independent validation cohort comprising 161 NSCLC patients was enrolled.
Methods:
The piecewise regression was established using the Joinpoint software to characterize temporal trends. Univariate and multivariate analyses for specific mortality were performed using the Cox risk regression model.
Results:
From 2012 to 2016, patients with lung adenocarcinoma (LADC) showed a 2.06% annual reduction in mortality. Conversely, this population exhibited a 3.82% yearly increase in cardiovascular disease (CVD)-specific mortality and a 6.54% rise in pulmonary disease-specific mortality during the same period. Patients with lung squamous cell carcinoma (LSCC) may have benefited more from the initiation of immunotherapy, with a gradual decrease in incidence (2016–2019, −5.19%). The incidence of large-cell lung cancer (LCLC) declined significantly (2008–2012, −14.52%; 2012–2016, −7.93%; 2016–2019, −6.42%), with decrease in overall mortality (2008–2012, −10.63%; 2012–2016, −8.76%; 2016–2019, −10.27%) and cancer-specific mortality (2008–2012, −12.17%; 2012–2016, −10.35%; 2016–2019, −10.78%). LCLC showed no significant rise in CVD-specific mortality (2012–2016: CVD-specific mortality, −4.83%; pulmonary disease-specific mortality, −0.82%. 2016–2019: CVD-specific mortality, −21.82%; pulmonary disease-specific mortality, −12.78%) compared to LSCC and LADC. The reporting odds ratio (ROR = 47.80, 95% CI, 24.44–93.50) of endocarditis of LADC was the top-reported AE. Multivariate analysis showed that immune checkpoint blockade was associated with increased CVD death (HR = 4.602, 95% CI, 1.154–18.359, p = 0.031).
Conclusion:
Our analyses revealed a temporal increase in cardiopulmonary disease-specific mortality among NSCLC patients during the study period. This trend coincided with the introduction of targeted therapies and immunotherapies, particularly in LADC patients. CVD-specific mortality risk requires extensive attention during anticancer therapy, regardless of the geographical population.
Introduction
While therapeutic advances in lung cancer have driven sustained declines in cancer-related mortality, 1 it remains the leading cause of cancer-specific deaths globally.1,2 Meanwhile, the growing population of survivors may develop short-term and long-term treatment-related complications. Consequently, non-cancer-specific mortality in cancer patients cannot be ignored. Analysis of cause-specific mortality among lung cancer patients revealed cardiovascular disease (CVD)-related deaths as clinically significant, ranking as the second leading cause of mortality after cancer-specific deaths.3,4 Previous studies indicate that cancer patients experience high mortality correlated with CVD and pulmonary diseases. Non-cancer deaths may result from cardiac and pulmonary toxicity.5,6 Therefore, CVD and pulmonary disease risks require extensive attention during anticancer therapy.
The emergence of targeted therapies and immunotherapies has improved survival outcomes in lung cancer. However, treatment-related toxicities and resistance mechanisms remain challenges across histological subtypes. A 2009 Iressa Pan-Asia Study (IPASS) confirmed that only patients with epidermal growth factor receptor (EGFR)-activating mutations in non-small-cell lung cancer (NSCLC) receiving gefitinib experienced a survival benefit, particularly those with LADC harboring specific hotspot mutations.7,8 However, cancer-directed therapies can adversely affect normal cellular functions, particularly in cardiomyocytes and alveolar epithelial cells. For instance, Oxitinib (a third-generation EGFR tyrosine kinase inhibitor) inhibits EGFR signaling in cardiomyocytes, which may result in reduced myocardial contractility, cardiac dysfunction, or electrophysiological abnormalitie.9,10 In addition, EGFR inhibition in both alveolar epithelium and vascular endothelium may trigger apoptosis pathways, potentially leading to pulmonary interstitial fibrosis and pulmonary embolism.11,12 According to National Comprehensive Cancer Network (NCCN) guidelines, immunotherapy is recommended as the first-line treatment for advanced lung squamous cell carcinoma (LSCC) and extensive small-cell lung cancer (SCLC),13,14 but the treatment process may accelerate coronary atherosclerosis and myocardial infarction.15 –18 Immunotherapy-related adverse effects (AEs), including immune pneumonia and idiopathic interstitial pneumonia, are also frequent. 19 While current therapeutic modalities have significantly improved survival outcomes in NSCLC patients, heterogeneous treatment responses to targeted agents and immunotherapies persist across histological subtypes. Of particular concern is the increasing incidence of cardiopulmonary disease-specific mortality among NSCLC patients, which may reflect an emerging epidemiological trend. This trend underscores the need for a comprehensive investigation of treatment-associated cardiovascular and pulmonary mortality.
Hence, this study aims to assess cardiopulmonary-specific mortality risks in NSCLC patients stratified by pathological subtypes. We employed incidence-based mortality to evaluate trends in CVD and pulmonary disease-specific mortality among LSCC, lung adenocarcinoma (LADC), and large-cell lung cancer (LCLC) from 2001 to 2019. Continued monitoring of these trends may help identify potential influencing factors and provide epidemiological evidence to inform prevention strategies.
Materials and methods
Study design and patients
This retrospective cohort study follows the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines (Supplemental File S1). 20 This study employed a tripartite approach to evaluate cardiopulmonary mortality risks in NSCLC: (1) Surveillance, Epidemiology, and End Results (SEER) registry-based analysis across histological subtypes (LSCC, LADC, and LCLC); (2) FDA Adverse Event Reporting System (FAERS) pharmacovigilance assessment of treatment-associated toxicities; and (3) clinical validation using a stage IV NSCLC cohort examining associations between anticancer therapies and non-cancer outcomes.
Study population data were extracted from the SEER database (17 Registries, a substantial segment of the U.S. populace). Incidence, cancer-specific, and non-cancer mortality were calculated. The AE data for NSCLC patients were obtained from the FAERS database. The reports of the FAERS database are coded using preferred terms (PTs) from the Medical Dictionary for Standardized Medical Dictionary for Regulatory Activities Queries (MedDRA Version 26.0; International Council for Harmonisation, Geneva, Switzerland). 21 Cases were identified as reports of pulmonary AEs and cardiac AEs. Lastly, we reviewed the medical records of patients diagnosed with stage IV NSCLC at the Shandong Cancer Hospital and Institute between 2018 and 2022.
Data collection
This study contains an analysis of the April 2023 SEER registry data submission to the National Cancer Institute (NCI). We utilized the SEER*Stat software (version 8.4.1; National Cancer Institute, Bethesda, MD, USA) to extract data using cause of death (COD) to site recode from International Classification of Diseases for Oncology Third Edition (ICD-O-3). The study population was chosen from the SEER database (17 Registries, Nov 2022 Sub (2000–2020)), which has been updated in April 2023. Age-standardized rates were calculated using SEER*Stat software, expressed per 100,000 and age-adjusted to the 2000 US Std Population (19 age groups – Census P25-1130) standard; confidence intervals (CIs) (Tiwari mod) are 95% for rates. To classify NSCLC subtypes accurately, we utilize the classification system developed by Lewis et al., which employs the ICD-O-3 morphological code. LSCC subtypes are defined as 8051, 8052, 8070–8076, 8078, 8083, 8084, 8090, 8094, 8120, and 8123; the subtypes of LADC are defined as 8015, 8050, 8140, 8141, 8143–8145, 8147, 8190, 8201, 8211, 8250–8255, 8260, 8290, 8310, 8320, 8323, 8333, 8401, 8440, 8470, 8471, 8480, 8481, 8490, 8503, 8507, 8550, 8570–8572, 8574, and 8576. LCLC subtypes are defined as 8012–8014, 8021, 8034, and 8082. Patients were considered to have died of CVD-specific death and pulmonary-specific death following International Classification of Diseases (ICD) 10th revision codes (available at URL: https://seer.cancer.gov/codrecode/icdo3_d2023/index.html). COD to site records are used to analyze the specific mortality. Patients were considered to have died of CVD when SEER COD recodes were hypertension disease (I10–I15); ischemic heart disease (I20–I25); cerebrovascular diseases (I60–I69); diseases of arteries, arterioles, and capillaries (I70–I78); other and unspecified disorders of the circulatory system (I00–I09, I30–I51, and I80–I99), while pulmonary disease-specific death were identified with COD including pneumonia (J09–J18), chronic obstructive pulmonary disease and allied conditions (J40–J47), or pulmonary heart disease and disease of pulmonary circulation (I26–I28). Patients lacking survival information were excluded from the analysis. In addition, data were characterized by standardized mortality ratios, which indicated the relative risk of death compared to the general population.
We retrospectively analyzed patients diagnosed with stage IV NSCLC between 2018 and 2022. The study cohort selection process is detailed in Supplemental Figure 1. The study cohort was selected through strict inclusion criteria: (1) histologically confirmed NSCLC, (2) American Joint Committee on Cancer (AJCC) 8th edition stage IV disease, (3) Eastern Cooperative Oncology Group (ECOG) performance status 0–2, and (4) complete treatment records. Exclusion criteria comprised the following: (1) age < 18 years, (2) history of prior malignancies, (3) receipt of localized therapies, and (4) incomplete follow-up data. In all, 161 patients were included in the final analysis. Comprehensive clinical data were collected, including demographic variables (age, gender, smoking history, comorbidity profile), clinicopathological data (pathological subtypes, tumor stage, location), treatment strategies (immune checkpoint blockade, chemotherapy, targeted therapy), and survival outcomes.
Data analysis
This study presents data by year, detailing the incidence of histological subtypes, as well as cancer-specific mortality, cardiopulmonary disease-specific mortality, and overall mortality (Supplemental Files S2–S6). The annual percentage change (APC) in incidence, overall mortality, cancer-specific mortality, and cardiopulmonary disease-specific mortality was calculated using Joinpoint software (Version 5.3.0; National Cancer Institute, Bethesda, MD, USA). Temporal trends were examined, and significant inflection points were identified through Joinpoint regression analysis.22 –24 This method helps understand the dynamic changes in disease development, providing important insights for formulating prevention and intervention strategies. We utilized this approach to analyze trends in cardiopulmonary disease-specific mortality over time across histological subtypes. Percentages represent the calculated APC for each specified temporal segment. An asterisk (*) denotes statistical significance (p < 0.05) compared to zero. 22 Rate trends were displayed using a dual-axis approach to ensure comparability, consistent with previously validated methodologies. 8
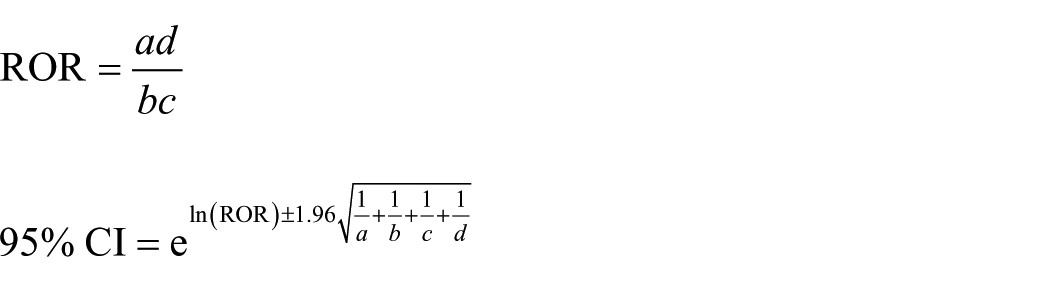
The reporting odds ratio (ROR) 25 is a classic disproportionality analysis method widely used in detecting adverse drug reaction (ADR) signals. The ROR compares the drug exposure among cases of AEs with that of other reported AEs, as a measure of disproportionality analysis to identify potential signals in pharmacovigilance. According to the two matric contingency table (Supplemental Table 1), the ROR value and its corresponding 95% CIs can be calculated using the following formulas:
The ADR signal detection results are classified based on the case count and the lower limit of the 95% CI. Positive signals require ⩾3 cases and a lower limit of 95% CI > 1.
In the clinical cohort, we compared the differences in demographics, clinicopathology, and treatment, and divided patients according to histological subtypes. Continuous variables were expressed as mean ± standard deviation (SD), while categorical variables were presented as counts. In addition, univariate and multivariate analyses for CVD death were performed using the Cox risk regression model. For multivariate analysis, variables with p < 0.10 in univariate analysis were included in the multivariate regression model. Analyses were performed using SPSS Statistics for Windows version 25, R 4.3.3, and Microsoft Excel 2019. All statistical tests were two-sided, and statistical significance was set at p < 0.05.
Results
Discordance trends in the cancer-specific mortality and non-cancer-specific mortality
We analyzed 2-year cancer-specific mortality and non-cancer-specific mortality from 2001 to 2019, as depicted in Figure 1(a). The cancer-specific mortality has shown a consistent decline, particularly post-2012. It decreased by 8.5% from 2012 (65.4%) to 2018 (56.9%) and by 3.2% from 2006 (68.6%) to 2012 (65.4%). By contrast, the non-cancer-specific mortality exhibited a consistent increase, particularly since 2012, rising from 2.6% to 3.1% between 2012 and 2018. The top three COD are presented in Figure 1(b). From 2001 to 2019, cancer-specific mortality accounted for the majority of deaths among lung cancer patients, followed by cardiovascular and pulmonary disease. Cancer-specific mortality has decreased while non-cancer-specific mortality has risen, exhibiting inconsistencies. It is essential to further investigate and prioritize attention on non-cancer-specific mortality. Given the differential treatment responses across histological subtypes, we conducted refined analyses of overall mortality, cancer-specific mortality, and cardiopulmonary disease-specific mortality.
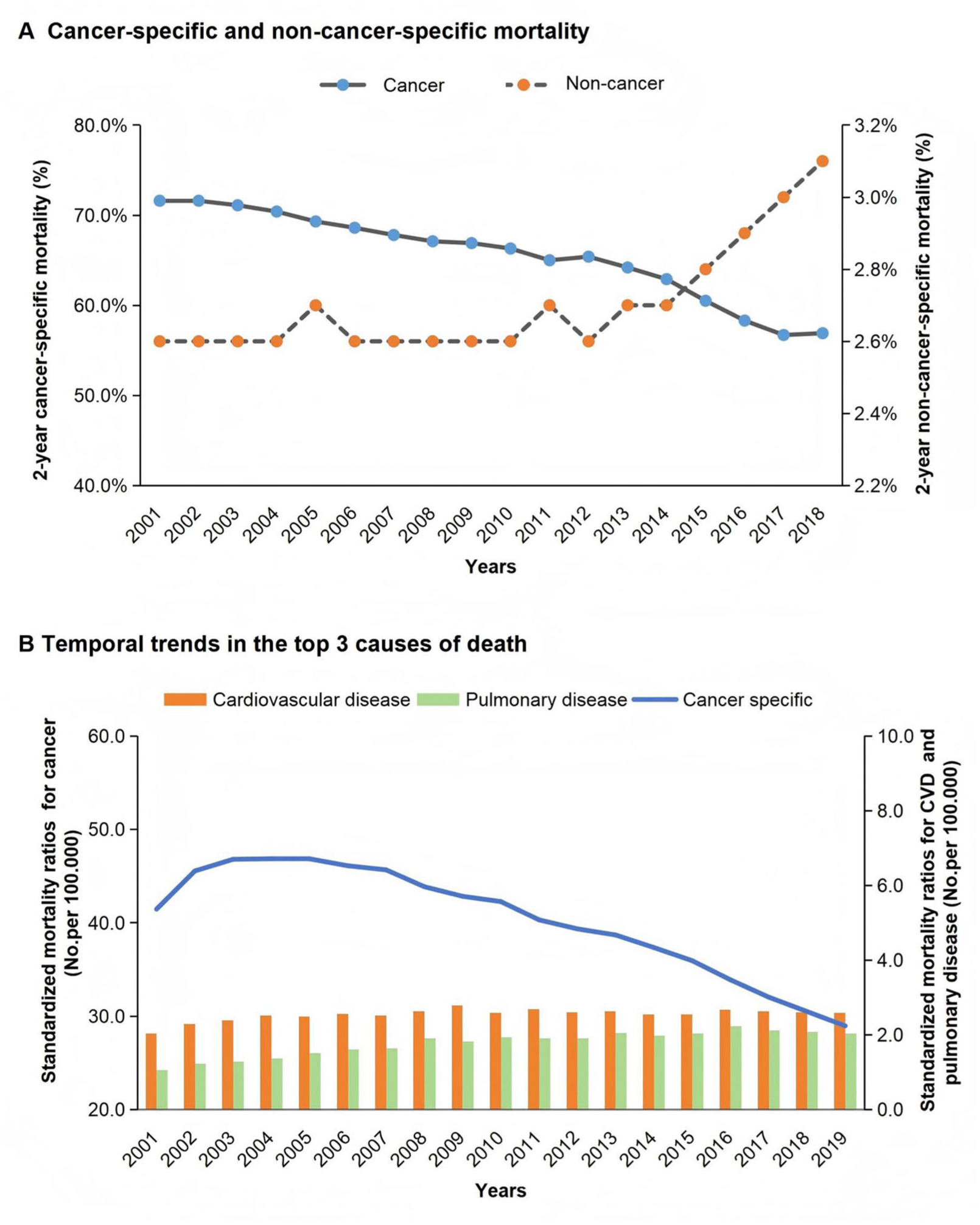
Discordance trends in the cancer-specific mortality and non-cancer-specific mortality. (a) Temporal trends of 2-year cancer-specific mortality (left y-axis, blue circle) and non-cancer-specific mortality (right y-axis, orange circles) from 2001 to 2019. (b) Temporal trends in the top three causes of death among patients with non-small-cell lung cancer. Cancer-specific mortality (left y-axis, blue line), cardiovascular disease mortality (right y-axis, orange line), and pulmonary disease mortality (right y-axis, green line).
Incidence, overall mortality, cancer-specific mortality, and cardiopulmonary disease-specific mortality of LADC
Figure 2 presents the temporal trends in LADC incidence, overall mortality, cancer-specific mortality, and cardiopulmonary disease-specific mortality between 2001 and 2019. A gradual increase in LADC incidence was observed prior to 2012 (Figure 2(a)). Subsequently, an annual decrease of 0.49% in LADC incidence was recorded between 2012 and 2016. During this period, cancer-specific mortality declined notably by 2.06% per year, followed by further decreases (−4.97%) from 2016 to 2019. Possibly due to genetic testing and targeted therapy, the overall mortality decreased significantly from 2012 to 2016 (−1.14%). As presented in Figure 2(b), there was a significant annual increase in CVD-specific mortality, particularly a 3.82% annual increase from 2012 to 2016. Similarly, pulmonary disease-specific mortality increased more sharply compared to CVD. Over the same period, pulmonary disease-specific mortality increased by 6.54% annually. From 2016 to 2019, LADC incidence (−5.02%), overall mortality (−3.43%), and cancer-specific mortality (−4.97%) decreased significantly, but there was no consistent change in CVD-specific mortality (0.6%). Since the introduction of new models of recommended genetic testing and targeted therapy in 2012, advances in treatment and control of incidence have contributed to a significant reduction in cancer-specific mortality, while cardiopulmonary disease-specific mortality has increased significantly.

Temporal trends in incidence, overall mortality, cancer-specific mortality, and cardiopulmonary disease-specific mortality of LADC from 2001 to 2019. (a) Temporal trends in modeled incidence (blue line), modeled overall mortality (red line), and modeled cancer-specific mortality (green line), observed incidence of LADC (blue square), observed overall mortality (red circle), and observed cancer-specific mortality (green circle) of LADC are presented. (b) Temporal trends in modeled cancer-specific mortality (green line), modeled CVD-specific mortality (orange line), modeled pulmonary disease-specific mortality (purple line), observed cancer-specific mortality (green circle), observed CVD-specific mortality (orange diamond), and pulmonary disease-specific mortality (purple triangle) of LADC are also depicted. The left y-axis represents cancer-specific mortality, while the right y-axis displays cardiopulmonary disease-specific mortality. The attached percentage represents the annual percentage change over the given time range. Annual percentage changes with a significant difference from zero (p < 0.05) are denoted by asterisks.
Incidence, overall mortality, cancer-specific mortality, and cardiopulmonary disease-specific mortality of LSCC
Figure 3 depicts temporal trends in LSCC incidence, overall mortality, cancer-specific mortality, and cardiopulmonary disease-specific mortality from 2001 to 2019. In Figure 3(a), the incidence, overall mortality, and cancer-specific mortality showed a gradual decline. Specifically, the incidence of LSCC exhibited an annual decline of 3.26% during 2012–2016, followed by a slightly attenuated decrease of 2.91% per year from 2016 to 2019. However, overall mortality demonstrated a significant downward trend, with annual declines accelerating from −2.89% (2012–2016) to −3.65% (2016–2019). Concurrently, cancer-specific mortality showed a pronounced reduction of 5.19% per year during 2016–2019. In Figure 3(b), the cancer-specific mortality of LSCC patients increased slowly before 2012 and then decreased from 2012 to 2019, declining from 4.07% annually to 5.19% annually. Nevertheless, cardiopulmonary disease-specific mortality increased annually, though less markedly than in LADC. The CVD-specific mortality showed the most significant increase from 2008 to 2012, with an annual rise of 5.23%. Pulmonary disease-specific mortality increased by 2.74% annually from 2008 to 2012, peaking at 5.31% annually during 2012–2016. Notably, the cancer-specific mortality of LSCC has generally decreased, potentially correlated with the implementation of immunotherapy approaches. However, cardiopulmonary disease-specific mortality showed no significant increase, but rather exhibited an overall downward trend from 2016 to 2019.

Temporal trends in incidence, overall mortality, cancer-specific mortality, and cardiopulmonary disease-specific mortality of LSCC from 2001 to 2019. (a) Temporal trends in modeled incidence (blue line), modeled overall mortality (red line), and modeled cancer-specific mortality (green line), observed incidence (blue square), observed overall mortality (red circle), and observed cancer-specific mortality (green circle) of LSCC are presented. (b) Temporal trends in modeled cancer-specific mortality (green line), modeled CVD-specific mortality (orange line), modeled pulmonary disease-specific mortality (purple line), observed cancer-specific mortality (green circle), observed CVD-specific mortality (orange diamond), and pulmonary disease-specific mortality (purple triangle) of LSCC are also depicted. The left y-axis represents cancer-specific mortality, while the right y-axis displays cardiopulmonary disease-specific mortality. The attached percentage represents the annual percentage change over the given time range. Annual percentage changes with a significant difference from zero (p < 0.05) are denoted by asterisks.
Incidence, overall mortality, cancer-specific mortality, and cardiopulmonary disease-specific mortality of LCLC
We analyzed temporal trends in LCLC incidence, overall mortality, cancer-specific mortality, and cardiopulmonary disease-specific mortality. As demonstrated in Figure 4(a), LCLC incidence showed consistent annual declines (2008–2012, −14.52%; 2012–2016, −7.93%; 2016–2019, −6.42%), along with a downward trend in overall mortality (2008–2012, −10.63%; 2012–2016, −8.76%; 2016–2019, −10.27%) and cancer-specific mortality (2008–2012, −12.17%; 2012–2016, −10.35%; 2016–2019, −10.78%). The 2016–2019 period marked significant improvements, with LCLC overall mortality declining 6.42% annually and cancer-specific mortality dropping 10.78% per year. As observed in Figure 4(b), CVD-specific mortality and pulmonary disease-specific mortality also exhibited a downward trend. This trend was most pronounced from 2016 to 2019, with CVD-specific mortality decreasing by 21.82% annually and pulmonary disease-specific mortality declining by 12.75% annually. From 2008 to 2019, there was a decline in the LCLC incidence, cancer-specific mortality, CVD-specific mortality, and pulmonary disease-specific mortality. Compared to LSCC and LADC, LCLC did not exhibit a significant increase in cardiopulmonary disease-specific deaths, possibly due to limited utilization of treatment improvement.

Temporal trends in incidence, overall mortality, cancer-specific mortality, and cardiopulmonary disease-specific mortality of LCLC from 2001 to 2019. (a) Temporal trends in modeled incidence (blue line), modeled overall mortality (red line), and modeled cancer-specific mortality (green line), observed incidence (blue square), observed overall mortality (red circle), and observed cancer-specific mortality (green circle) of LCLC are presented. (b) Temporal trends in modeled cancer-specific mortality (green line), modeled CVD-specific mortality (orange line), modeled pulmonary disease-specific mortality (purple line), observed cancer-specific mortality (green circle), observed CVD-specific mortality (orange diamond), and pulmonary disease-specific mortality (purple triangle) of LCLC are also depicted. The left y-axis represents cancer-specific mortality, while the right y-axis displays cardiopulmonary disease-specific mortality. The attached percentage represents the annual percentage change over the given time range. Annual percentage changes with a significant difference from zero (p < 0.05) are denoted by asterisks.
Clinical patient data validation
While these population-based analyses reveal distinct temporal patterns in NSCLC subtype-specific mortality, particularly concerning cardiopulmonary outcomes, the underlying treatment-associated toxicities require systematic evaluation. Based on the counts of AE reports in the FAES database, the reporting distribution of immune checkpoint blockade (ICB) and targeted therapy was summarized. The cardiac AEs and pulmonary AEs reported by patients has been increasing annually (blue line) in the general lung cancer population (Figure 5(a) and (b)), among NSCLC patients (Figure 5(c) and (d)), and specifically in the LADC subgroup (Figure 5(e) and (f)). The proportion of cardiac AEs linked to targeted therapies has risen among all cardiac adverse events recorded from 2012 to 2014 (orange bars), with an increase in NSCLC from 41.57% to 58.93%. The proportion of cardiac AEs associated with ICB agents among all cardiac AEs showed an increasing trend from 2015 to 2019 (green bars) in lung cancer (Figure 5(a)), with the proportion in NSCLC rising from 13.46% to 47.07% (Figure 5(c)) and in LADC rising from 8.24% to 32.65% (Figure 5(e)). These findings underscore the association between therapeutic regimens and AEs. A similar pattern was observed in pulmonary AEs. While overall AEs reports remained relatively stable during 2010–2014, targeted therapy-associated AEs gradually increased, with a particularly evident increase in NSCLC reporting (from 38.58% to 55.98%). An increase in pulmonary AEs was observed beginning in 2015. The subset attributable to ICB demonstrated consistent annual growth, escalating from 25.56% to 65.41% in NSCLC (Figure 5(d)) and from 3.61% to 44.26% in LADC (Figure 5(f)). In addition, the top 30 most frequently reported AEs at the high-level term classification are presented in Figure 5(g). Among other conditions (non-tumor-related), endocarditis demonstrated the highest reporting frequency (ROR = 47.80, 95% CI, 24.44–93.50), followed by site-specific necrosis and vascular insufficiency (ROR = 12.98, 95% CI, 7.92–21.26), aortic infections and inflammations (ROR = 12.73, 95% CI, 6.03–26.84), lower respiratory tract inflammatory and immunologic conditions (ROR = 11.93, 95% CI, 10.85–13.12), noninfectious myocarditis (ROR = 10.29, 95% CI, 8.54–12.40), parenchymal lung disorders (ROR = 8.88, 95% CI, 8.34–9.46).
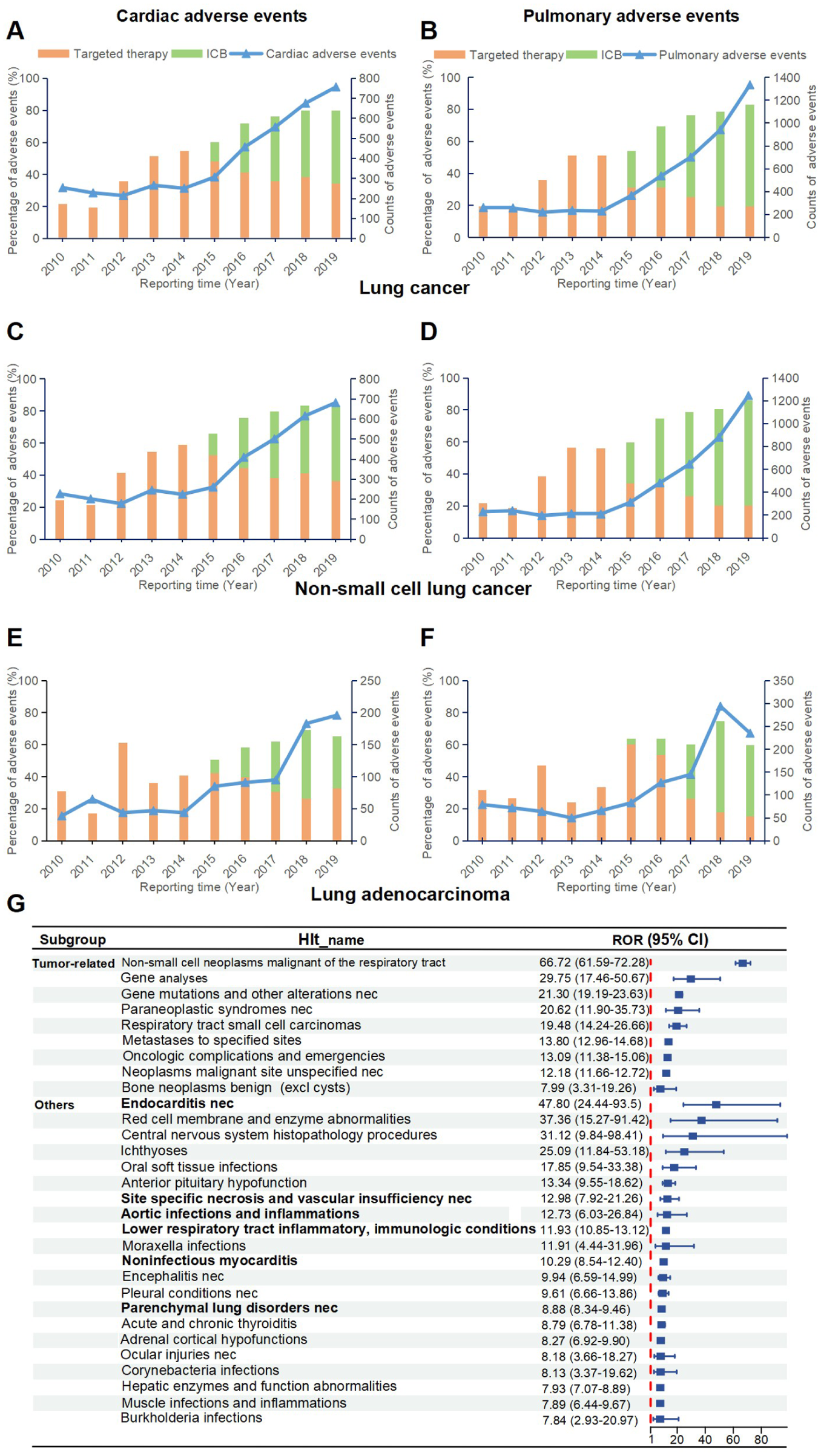
Adverse event reports in the FAERS database of ICB and targeted therapy. (a, c, e) Reporting distribution of cardiac adverse events associated with ICB and targeted therapy in patients with lung cancer, NSCLC, and lung adenocarcinoma. (b, d, f) Reporting distribution of pulmonary adverse events associated with ICB and targeted therapy in patients with lung cancer, NSCLC, and lung adenocarcinoma. (g) The top 30 frequently reported adverse events among patients with lung adenocarcinoma.
Our clinical validation cohort extends the initial population-level findings by providing treatment and non-cancer death in NSCLC patients under real-world therapeutic regimens. The baseline demographic and clinical characteristics of the study cohort (n = 161 stage IV NSCLC patients) are detailed in Supplemental Table 2. The LADC group exhibited a higher proportion of CVD-related deaths (20.34%) compared to LSCC (15.38%) (Figure 6(a)). Consequently, we investigated risk factors for CVD-specific death in the LADC group. Multivariate analysis adjusting for variables (age, gender, smoking history, pathological subtypes, tumor stage, location, treatment strategies) revealed that ICB and age were independent risk factors for CVD-specific death (ICB, HR = 4.602, 95% CI, 1.154–18.359, p = 0.031; age, HR = 5.188, 95% CI, 1.189–22.644, p = 0.029) (Figure 6(b)). Herein, these findings may suggest associations between treatment, histological subtypes, and CVD-specific mortality.

Clinical patient data validation. (a) The proportion of patients suffering CVD death based on histological subtypes and (b) hazard ratio of CVD-specific death in patients with lung adenocarcinoma.
Discussion
To our knowledge, this study provides the first comprehensive evaluation of treatment-associated improvements in cardiopulmonary disease-specific mortality stratified by NSCLC histological subtypes over a two-decade period. Our analysis specifically examined temporal trends in overall mortality, cancer-specific mortality, and cardiopulmonary disease-specific mortality across different NSCLC subtypes. In addition, it sought to clarify the influence of treatment on cardiopulmonary disease-specific mortality. The findings demonstrate a consistent decline in both NSCLC incidence and mortality from 2001 to 2019. By contrast, mortality from CVD and pulmonary diseases exhibited a progressive increase. Most notably, we identified a significant rise in cardiopulmonary disease-specific mortality among LADC patients during 2012–2016, coinciding with the clinical adoption of FDA-approved targeted therapies. These findings suggest that optimal NSCLC treatment strategies should consider both cancer-specific mortality and treatment-associated cardiopulmonary complications.
Impact of therapeutic advances in LADC on population-level cardiopulmonary disease-specific mortality
Our analyses revealed a gradual rise in the LADC incidence along with incidence-based overall mortality and cancer-specific mortality between 2001 and 2012. At the population level, pre-2012 trends may be associated with multiple factors, including the limited efficacy of conventional chemoradiotherapy, heterogeneous responses to targeted therapies among eligible populations, and increased detection rates from early screening initiatives.26 –28 While landmark studies such as IPASS demonstrated that EGFR mutation-selected patients derive significant progression-free survival benefit from gefitinib with acceptable toxicity, 29 real-world outcomes were influenced by several important factors. Therapeutic benefits were modulated by clinical variables such as smoking status, as demonstrated in previous research. 28 Moreover, only approximately 15%–20% of LADC patients harbored actionable EGFR mutations. During the initial phase of targeted therapy adoption, suboptimal molecular testing rates and treatment accessibility may have attenuated the population-level therapeutic impact. Meanwhile, treatment-related toxicities may have partially diminished survival benefits.
Our study revealed a greater annual decrease in overall mortality and cancer-specific mortality from 2012 to 2016 compared to the incidence. However, the cancer-specific mortality decline from 2016 to 2019 correlated more strongly with incidence reduction, a pattern that may reflect the clinical challenges of acquired resistance to targeted agents and limited immunotherapy utilization in LADC. This observation may align with established resistance mechanisms, as Xiang et al. demonstrated that T790M mutations develop in 50%–60% of patients progressing on first-generation EGFR-TKIs, substantially curtailing treatment durability. 24 Furthermore, emerging evidence indicates that KRAS-G12D mutations promote both immunosuppression and ICB resistance. 30 During this period, incidence and mortality demonstrated parallel declines, although the reduction in overall mortality was less pronounced compared to incidence. These findings reflect advancements in treatment efficacy and modifiable disease risk factors in LADC patients while suggesting a relation between overall mortality and non-cancer-specific mortality patterns. Our analysis revealed significantly elevated CVD and pulmonary deaths (2001–2019), peaking during 2012–2016, coinciding with expanded utilization of targeted therapy. The temporal association may reflect potential cardiovascular risks associated with molecularly targeted therapies, consistent with existing mechanistic evidence regarding VEGF signaling-mediated vascular endothelial dysfunction and direct cardiomyocyte effects. 31 KRAS-targeted agents like sotorasib mediate cardiotoxicity via neuraminidase 1-dependent AKT pathway suppression and mitochondrial dysfunction. 32 These observations gain clinical relevance considering reported toxicity increases when combining anlotinib with EGFR-TKIs in treatment-naïve NSCLC patients. 33 These findings underscore the importance of cardiopulmonary disease-specific mortality.
Impact of therapeutic advances in LSCC on population-level cardiopulmonary disease-specific mortality
From 2001 to 2019, our study documented significant reductions in LSCC incidence, overall mortality, and cancer-specific mortality. Notably, between 2016 and 2019, immunotherapy demonstrated a more pronounced mortality reduction relative to incidence compared to the 2012–2016 period. Immunotherapy emerged in 2015, and the KEYNOTE-407 Phase III trial revealed that the pembrolizumab plus chemotherapy yielded superior outcomes in advanced LSCC, with a median PFS of 6.4 months and a median OS of 15.9 months, both significantly exceeding chemotherapy alone. 34 These findings strongly support the notion that immunotherapy confers survival benefits and significantly reduces mortality in patients with LSCC. Notably, CVD-specific mortality showed no significant reduction following the introduction of targeted and immune therapies after 2012. However, LSCC demonstrated a declining trend in cardiovascular mortality from 2012 to 2016 relative to LADC, potentially reflecting the differential therapeutic efficacy of targeted agents between these histological subtypes. Moreover, LADC demonstrated more pronounced adverse reactions, leading to significantly higher CVD-specific mortality during this period. These findings are supported by Wu et al.’s 35 comparative analysis of driver gene mutation frequencies in LSCC and LADC. Although LSCC patients demonstrate improved clinical responses to immunotherapy, no significant reduction in cardiopulmonary disease-specific mortality was observed. Given the recent introduction of immunotherapy, further studies are required to systematically evaluate its associated cardiovascular and pulmonary toxicity profiles.
Impact of therapeutic advances in LCLC on population-level cardiopulmonary disease-specific mortality
In this study, the incidence, overall mortality, and cancer-specific mortality of LCLC declined annually from 2001 to 2019. Notably, a significant reduction in CVD-specific and pulmonary disease-specific mortality was observed during this period, with the most pronounced decreases occurring between 2016 and 2019. For early-stage LCLC patients, surgery remains the primary treatment modality, with adjuvant chemotherapy considered postoperatively to reduce recurrence risk. The optimal management for advanced cases remains undefined, though relevant guidelines recommend radical concurrent radiotherapy. 13 The combination of studies by Leblanc et al. 36 and Meng et al. 37 suggests that the efficacy of targeted therapy and immunotherapy in treating LCLC requires further validation through additional Phase III clinical trials. Limited utilization of immunotherapy and targeted therapy may explain the absence of significant increases in cardiovascular and pulmonary mortality.
Collectively, our population-based analysis of NSCLC patients revealed distinct patterns in incidence and mortality outcomes, including overall, cancer-specific, and non-cancer-specific mortality across histological subtypes. These observations suggest the need for increased clinical attention to non-cancer mortality, particularly treatment-related cardiopulmonary AEs, though further studies are needed to establish causal relationships between specific therapies and these outcomes. In addition, we incorporated research on cardiopulmonary diseases, representing both the innovative and focal aspects of this study. Second, mortality calculations in this study were incidence-based, reducing potential misclassification of cancer-specific deaths and improving result validity. Third, the 20-year study period enabled comprehensive documentation of incidence and mortality trends following the introduction of various therapeutic modalities, enhancing the epidemiological significance of this research. While this study provides a comprehensive assessment through multiple analytical approaches, several limitations should be acknowledged. First, the observational nature of population-level data may include unmeasured confounding factors, particularly regarding non-cardiac pulmonary mortality. Second, while the clinical validation cohort was carefully characterized, its limited sample size compromised subgroup analyses, and the exclusive inclusion of stage IV patients may restrict generalizability across all disease stages. Third, the temporal alignment between treatment advances and mortality trends, though biologically plausible, requires further mechanistic investigation. Certain limitations could be somewhat offset by our comprehensive analytical strategy and the validated cohort.
Conclusion
From 2001 to 2019, patients across NSCLC histological subtypes demonstrated declining incidence and mortality rates, whereas CVD-specific mortality showed a significant rise. This temporal association aligns with the clinical introduction of targeted therapies and ICB. CVD-specific mortality risk requires extensive attention during anticancer therapy regardless of the geographical population.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251379962 – Supplemental material for Temporal trends in treatment-related cardiopulmonary disease-specific mortality in NSCLC based on pathological subtypes: a retrospective population-based cohort study
Supplemental material, sj-docx-1-tam-10.1177_17588359251379962 for Temporal trends in treatment-related cardiopulmonary disease-specific mortality in NSCLC based on pathological subtypes: a retrospective population-based cohort study by You Mo, Xinyi Liang, Duncan Wei, Dawei Chen and Jinming Yu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359251379962 – Supplemental material for Temporal trends in treatment-related cardiopulmonary disease-specific mortality in NSCLC based on pathological subtypes: a retrospective population-based cohort study
Supplemental material, sj-docx-3-tam-10.1177_17588359251379962 for Temporal trends in treatment-related cardiopulmonary disease-specific mortality in NSCLC based on pathological subtypes: a retrospective population-based cohort study by You Mo, Xinyi Liang, Duncan Wei, Dawei Chen and Jinming Yu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-4-tam-10.1177_17588359251379962 – Supplemental material for Temporal trends in treatment-related cardiopulmonary disease-specific mortality in NSCLC based on pathological subtypes: a retrospective population-based cohort study
Supplemental material, sj-docx-4-tam-10.1177_17588359251379962 for Temporal trends in treatment-related cardiopulmonary disease-specific mortality in NSCLC based on pathological subtypes: a retrospective population-based cohort study by You Mo, Xinyi Liang, Duncan Wei, Dawei Chen and Jinming Yu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-5-tam-10.1177_17588359251379962 – Supplemental material for Temporal trends in treatment-related cardiopulmonary disease-specific mortality in NSCLC based on pathological subtypes: a retrospective population-based cohort study
Supplemental material, sj-docx-5-tam-10.1177_17588359251379962 for Temporal trends in treatment-related cardiopulmonary disease-specific mortality in NSCLC based on pathological subtypes: a retrospective population-based cohort study by You Mo, Xinyi Liang, Duncan Wei, Dawei Chen and Jinming Yu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-6-tam-10.1177_17588359251379962 – Supplemental material for Temporal trends in treatment-related cardiopulmonary disease-specific mortality in NSCLC based on pathological subtypes: a retrospective population-based cohort study
Supplemental material, sj-docx-6-tam-10.1177_17588359251379962 for Temporal trends in treatment-related cardiopulmonary disease-specific mortality in NSCLC based on pathological subtypes: a retrospective population-based cohort study by You Mo, Xinyi Liang, Duncan Wei, Dawei Chen and Jinming Yu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-7-tam-10.1177_17588359251379962 – Supplemental material for Temporal trends in treatment-related cardiopulmonary disease-specific mortality in NSCLC based on pathological subtypes: a retrospective population-based cohort study
Supplemental material, sj-docx-7-tam-10.1177_17588359251379962 for Temporal trends in treatment-related cardiopulmonary disease-specific mortality in NSCLC based on pathological subtypes: a retrospective population-based cohort study by You Mo, Xinyi Liang, Duncan Wei, Dawei Chen and Jinming Yu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-8-tam-10.1177_17588359251379962 – Supplemental material for Temporal trends in treatment-related cardiopulmonary disease-specific mortality in NSCLC based on pathological subtypes: a retrospective population-based cohort study
Supplemental material, sj-docx-8-tam-10.1177_17588359251379962 for Temporal trends in treatment-related cardiopulmonary disease-specific mortality in NSCLC based on pathological subtypes: a retrospective population-based cohort study by You Mo, Xinyi Liang, Duncan Wei, Dawei Chen and Jinming Yu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-9-tam-10.1177_17588359251379962 – Supplemental material for Temporal trends in treatment-related cardiopulmonary disease-specific mortality in NSCLC based on pathological subtypes: a retrospective population-based cohort study
Supplemental material, sj-docx-9-tam-10.1177_17588359251379962 for Temporal trends in treatment-related cardiopulmonary disease-specific mortality in NSCLC based on pathological subtypes: a retrospective population-based cohort study by You Mo, Xinyi Liang, Duncan Wei, Dawei Chen and Jinming Yu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-2-tam-10.1177_17588359251379962 – Supplemental material for Temporal trends in treatment-related cardiopulmonary disease-specific mortality in NSCLC based on pathological subtypes: a retrospective population-based cohort study
Supplemental material, sj-pdf-2-tam-10.1177_17588359251379962 for Temporal trends in treatment-related cardiopulmonary disease-specific mortality in NSCLC based on pathological subtypes: a retrospective population-based cohort study by You Mo, Xinyi Liang, Duncan Wei, Dawei Chen and Jinming Yu in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.