Abstract
Background:
Neoadjuvant therapy improves survival benefits in patients with locally advanced non-small cell lung cancer but increases tissue density, presenting challenges for surgeons.
Objectives:
To compare the differences in surgical complexity and short-term prognostic outcomes between neoadjuvant targeted therapy (NTT) and neoadjuvant chemoimmunotherapy (NCI).
Design/methods:
This study enrolled 106 patients underwent curative surgery after neoadjuvant therapy between January 2020 and December 2023 at the National Cancer Center of China. Differences in surgical complexity and short-term prognostic outcomes between the two neoadjuvant therapy cohorts were evaluated. The pathological indicators such as pathological response rate and lymph node upstaging/downstaging were then analyzed.
Results:
In total, 33 patients underwent NTT and 73 underwent NCI preoperatively. Patients who received NTT showed a higher minimally invasive surgery rate (84.8% versus 53.4%, p < 0.01), shorter operative time (144 versus 184 min, p < 0.01), lower conversion rate (3.3% versus 17.8%, p = 0.03), less postoperative drainage (day 3: 140 versus 200 mL, p = 0.03), and lower incidence of postoperative complications including arrhythmias (6.1% versus 26%, p = 0.02). The pathological response rate in the NTT and NCI groups was 70% and 75%, respectively, with the latter group showing a higher complete pathological response rate. The two groups had no significant differences in major pathological response and lymph node pathological response rate.
Conclusion:
Patients who received NTT presented fewer surgical challenges for surgeons and had better surgical outcomes than those who received NCI therapy, with comparable pathological response rates between the two cohorts. Accordingly, NTT is the preferred induction regimen for patients harboring mutation status.
Keywords
Introduction
Non-small cell lung cancer (NSCLC) accounts for approximately 85% of all lung cancer cases, with 22% are diagnosed at a locally advanced stage. The feasibility of surgical resection is constrained by a limited patient demographics, contributing to high cancer-related mortality. 1 Theoretical benefits are ascribed to neoadjuvant therapies administered before surgery, including tumor burden reduction, occult micrometastases mitigation, and decreased postoperative recurrence. Despite these benefits, preoperative chemotherapy alone has only increased the 5-year survival rate of patients with stage IB–IIIA NSCLC by approximately 5%. 2 Targeted inhibitors for epidermal growth factor receptor (EGFR) mutation have become the foundation of tyrosine kinase inhibitor (TKI) therapy. 3 Recently, an increasing number of immunotherapies and targeted therapeutic regimens have been developed. Clinical researches have reported that neoadjuvant targeted therapy (NTT) and immunotherapy significantly improve the complete pathological response (CPR) and major pathological response (MPR) rates, as well as disease-free survival (DFS) and overall survival (OS) in patients with lung cancer, compared to single chemotherapy.4–6 Accordingly, neoadjuvant chemotherapy combined with immunotherapy (NCI) or NTT is gradually becoming the first-line approach for managing locally advanced NSCLC.
Although there is growing enthusiasm for induction therapy for resectable NSCLC, concerns among surgeons are intensifying regarding its potential to increase procedural complexity and the incidence of complications in subsequent surgical resections. Some clinical studies have reported that neoadjuvant immunotherapy can exacerbate inflammatory reactions in the surgical field, subsequently leading to fibrosis of the hilum and mediastinum. Such developments may require increased reliance on thoracotomy or conversion rate.7–9 Current research on the surgical prognosis after NTT remains limited. Bian et al. reported surgical outcomes in patients with stage III EGFR-mutant NSCLC treated with afatinib, highlighting that targeted therapy significantly increases the rate of CPR and pathological downstaging, it also disrupts normal cellular signaling pathways, leading to cell death and subsequent extracellular matrix fibrosis. Such adverse effects complicate the identification of tumor margins and relevant anatomical structures during operative procedures, thus increasing the risk of intraoperative bleeding and the complexity of postoperative recovery. 10
The choice between targeted therapy and chemoimmunotherapy is influenced by many factors, such as clinical staging, tumor histology, genetic mutations, and patient comorbidities. A clinical neoadjuvant regimen must balance the benefits of short-term surgical outcomes with long-term patient survival. Severe complications or mortality resulting from induction therapy are not acceptable to surgeons. Despite being the predominant induction strategies, little research has been conducted on the surgical outcomes following treatment with NCI or NTT. Furthermore, differences in surgical challenge parameters, surgical outcomes, and tumor bed and lymph node pathological response rates between these two therapies have not been reported. This study aimed to assess the safety and feasibility of minimally invasive surgery (MIS) following these two neoadjuvant therapies and to explore the differences in short-term outcomes after pulmonary resection.
Materials and methods
Patient selection
We retrospectively collected data from patients who underwent curative surgery after NTT and NCI at the National Cancer Center of China between January 2020 and December 2023. The protocols deployed for clinical evaluation and T-stage assessment included enhanced computed tomography (CT) of the neck, thorax, and upper abdomen, magnetic resonance imaging of the brain, bone scanning, and positron emission tomography-CT (PET-CT). N staging was performed using endobronchial ultrasonography and transbronchial needle aspiration. All patients underwent pathological biopsy guided by CT or bronchoscopy to confirm histological classification and driver gene mutation status. The inclusion criteria were as follows: (1) patients diagnosed with stage I–III NSCLC; (2) patients who received immunotherapy with chemotherapy or targeted therapy prior to surgical intervention; (3) patients considered suitable for curative surgery after neoadjuvant therapy assessment. The following exclusion criteria were applied: (1) history of other malignancies; (2) history of lung surgery; (3) patients who had received chemotherapy or radiation as monotherapy; (4) preoperative use of immunosuppressive medications; and (5) patients who had undergone palliative tumor resection. Therapeutic response to neoadjuvant treatment was assessed using CT or PET-CT imaging by applying the Response Evaluation Criteria in Solid Tumors scoring criteria. 11 Our retrospective study conforms to the Reporting of Observational Studies in Epidemiology statement 12 (Supplemental Table 1).
Genomic mutation analysis
The polymerase chain reaction (PCR) tube gene mutation detection kits were used to detect 10 targeted gene alterations (including epidermal growth factor receptor (EGFR) 18–20 exon, kirsten rats arcomaviral oncogene homolog (KRAS) 2 exon, B-raf proto-oncogene, serine/threonine kinase (BRAF)15 exon, human epidermal growth factor receptor 2 (HER2) 20 exon, neuroblastoma RAS viraloncogene homolog (NRAS) 3 exon, phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha (PIK3CA) 20/9 exon, anaplastic lymphoma kinase (ALK) fusion gene, ROS proto-oncogene 1, receptor tyrosine kinase (ROS1) fusion gene, rearranged during transfection (RET) fusion gene, and mesenchymal-epithelial transition factor (MET) 14 exon jump mutation). The reaction mixture was prepared in accordance with these instructions.13,14 PCR was performed using Stratagene Mx3000P™ system (Amoy Diagnostics Co, Xiamen, China).
Preoperative adjuvant therapy evaluation
All cohort patients underwent neoadjuvant therapy before surgery, with preoperative assessments conducted by a multidisciplinary team of cancer specialists. Prior to treatment, each patient had tumor or lymph node biopsies – guided by CT, bronchoscopy, or ultrasound fine needle aspiration. Following pathological classification, driver mutation analyses were performed. Patients with positive gene mutations and matched targeted drugs received NTT. While those without detectable mutations received NCI.
Analyses of prognostic and pathological parameters
The same senior thoracic surgeon performed all surgical procedures. Surgical procedures included minimally invasive or open lobectomies, extended lobectomies, pneumonectomies, and sleeve resections, followed by systematic mediastinal lymphadenectomy. Comprehensive information was collected, including preoperative examination results and short-term perioperative indicators such as surgical duration, MIS rate, conversion to thoracotomy, chest tube removal time, postoperative drainage volume, and postoperative complications. Two experienced pathologists independently evaluated the pathological response rates of the samples. In cases of disagreement, a third pathologist was consulted, with a majority consensus determining the final judgment. The evaluation of surgical specimens involved quantification of the percentage of residual viable tumor cells, necrosis, and stroma (fibrosis and inflammatory lesions). When the proportion of residual viable tumor cells approached the criteria for MPR or CPR, additional sampling of the tumor bed was required to reexamine the gross specimen. 15 All resected lymph nodes were sampled and the presence or absence of tumor cells and response to induction treatment were recorded. Microscopic observation of tumor tissue necrosis, accompanied by necrosis, mucin production, granulomatous inflammation, and hyalinization, was described as a response to treatment for metastatic lymph node carcinoma. 16 The pathological tumor, node, metastasis stage classification was based on the ninth edition of the American Joint Committee on Cancer Staging Manual. 17 CPR was characterized by the complete absence of residual viable tumor cells within the tumor bed and lymph nodes after neoadjuvant therapy. MPR was diagnosed when residual viable tumor cells comprised 10% or less of the tumor bed after neoadjuvant intervention. 18
Statistical analysis
Means and standard deviations were used to describe continuous variables that conformed to a normal distribution, and t tests were used to compare differences between the groups. Non-normally distributed data were represented by median and interquartile ranges (IQR), and statistical differences between the targeted therapy and chemotherapy immunotherapy groups were compared using the Mann–Whitney U test. Categorical variables were presented as percentages, and the Fisher’s exact test was used for intergroup comparisons. Statistical significance was established at p < 0.05. All statistical analyses were performed using the IBM SPSS Statistics for Social Sciences (version 25 software; IBM Corporation, Armonk, NY, USA).
Results
Clinical and pathological characteristics of the study cohort
This cohort study enrolled 106 patients with lung cancer who underwent surgical intervention following neoadjuvant therapy after meeting the specified inclusion and exclusion criteria (Figure 1). Clinical and pathological characteristics of the study cohort of patients undergoing surgery after NTT versus NCI were showed in Table 1. Of these, 73 (68.87%) underwent NCI and 33 (31.13%) were treated with NTT. The prevalent neoadjuvant chemoimmunotherapy (NCI) protocols included albumin-bound paclitaxel 400 mg day1 + carboplatin 350 mg day1 + atezolizumab 200 mg day1/q21d, pembrolizumab 200 mg day1 + albumin-bound paclitaxel 400 mg day1 + cisplatin 40 mg day1/250 mg day3/q21d, and albumin-bound paclitaxel 200 mg day1/300 mg day8 + carboplatin 450 mg day1 + nivolumab day1 360 mg/q21d, with a median treatment duration of two cycles. In the targeted therapy cohort, EGFR mutations were detected more frequently (n = 31, 93.94%) in patients treated with osimertinib, erlotinib, gefitinib, and apatinib. Furthermore, one patient with an ALK fusion mutation received lorlatinib, and another patient with a MET exon 14 skipping mutation was treated with savolitinib. The median treatment duration for the targeted therapy protocols was 4 months.
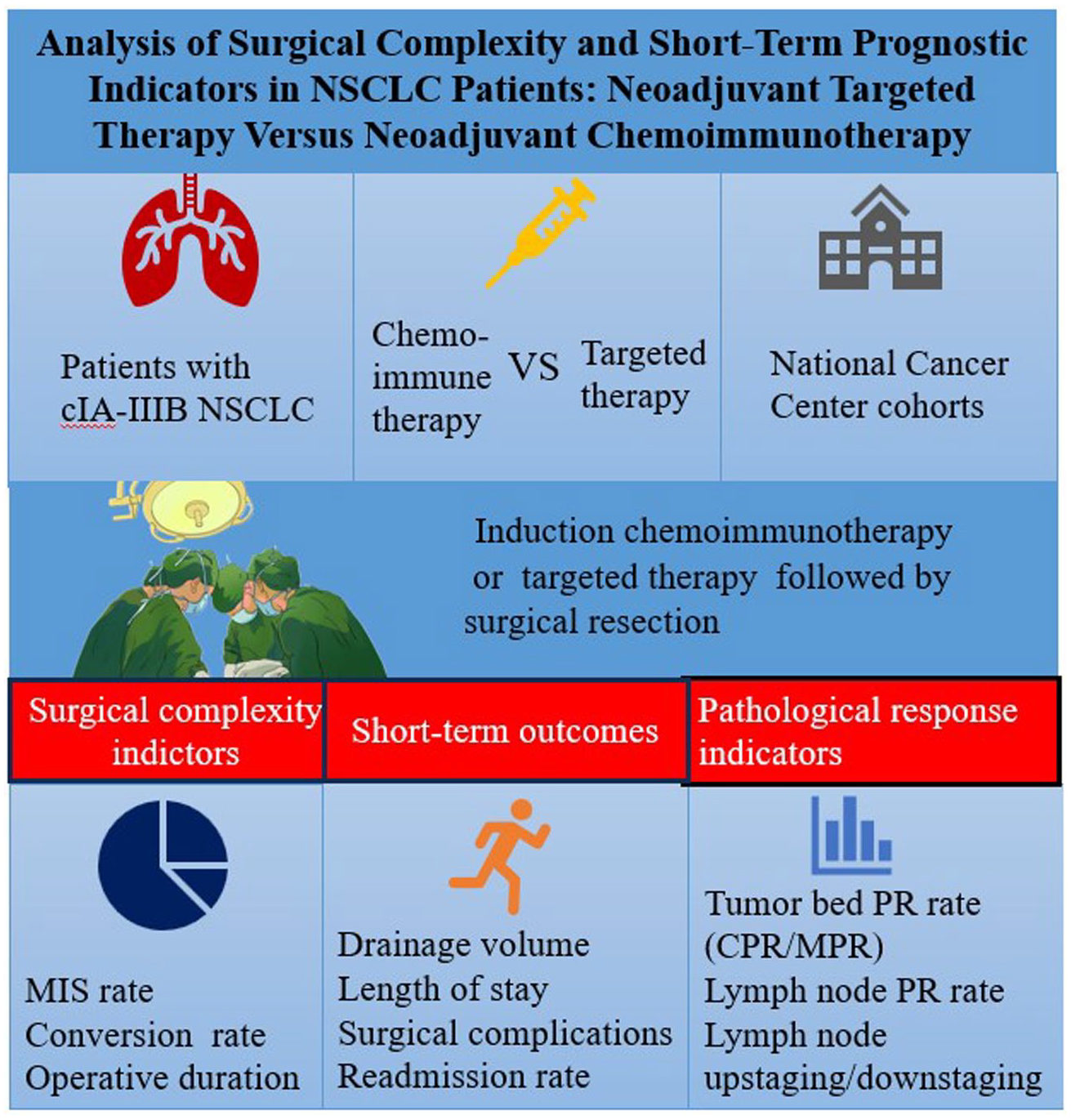
Flow diagram detailing schema of study subject selection. This study assessed the safety and feasibility of minimally invasive surgery following NTT and NCI. Differences in surgical complexity, patient prognosis, and pathological response were analyzed to guide optimal preoperative induction regimen.
Clinical and pathological characteristics of the study cohort of patients undergoing surgery after NTT versus NCI.
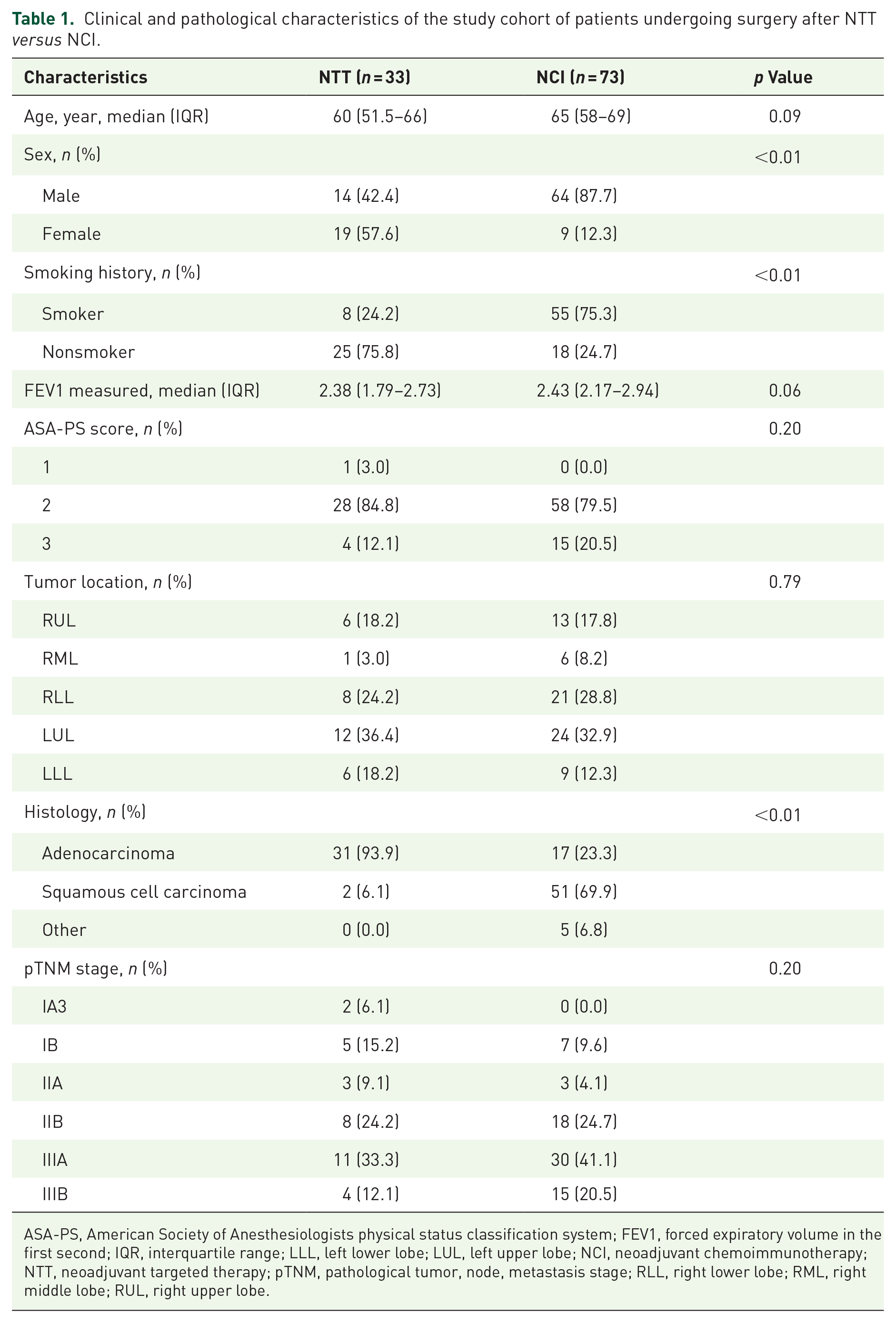
ASA-PS, American Society of Anesthesiologists physical status classification system; FEV1, forced expiratory volume in the first second; IQR, interquartile range; LLL, left lower lobe; LUL, left upper lobe; NCI, neoadjuvant chemoimmunotherapy; NTT, neoadjuvant targeted therapy; pTNM, pathological tumor, node, metastasis stage; RLL, right lower lobe; RML, right middle lobe; RUL, right upper lobe.
Table 1 compares the baseline characteristics of patients who underwent surgery after NTT and NCI. Fourteen males (42.4%) in the NTT group and 64 males (87.7%) in the NCI group were included. The NCI group showed a significantly higher proportion of male patients (p << 0.01). Significant differences were detected in smoking history between the two groups (p << 0.01). The predominant disease location was the upper lobe of the left lung, with an incidence of 36.4% (n = 12) in the targeted therapy group and 32.9% (n = 24) in the chemoimmunotherapy group. Most patients who underwent neoadjuvant therapy were diagnosed with stage IIIA disease, comprising 33.3% (n = 11) in the targeted therapy group and 41.1% (n = 30) in the chemoimmunotherapy group. Adenocarcinoma was the main pathological type among patients receiving NTT (n = 31, 93.9%), whereas squamous cell carcinoma was predominant among patients undergoing NCI (n = 51, 69.9%, p << 0.01).
Short-term prognostic characteristics of the surgical study cohort
All patients underwent curative resection and systemic lymph node dissection after neoadjuvant therapy. Table 2 compares the indicators of surgical difficulty and short-term prognostic outcomes in patients who underwent lobectomy after NTT and NCI. Single-port video-assisted thoracoscopic surgery was performed in most patients (84.8% in the NTT group and 53.4% in the NCI group), with significantly higher MIS in patients who received neoadjuvant-targeted regimens (p < 0.01). Lobectomy was the most common surgical procedure (81.8% in the NTT group and 67.1% in the NTT and NCI groups), followed by extended lobectomy (12.1%) and combined lobectomy (67.1%). Interestingly, patients treated with NCI had a significantly higher conversion rate (17.8% versus 3.3%, p = 0.03) than those treated with NTT. In terms of surgical duration, patients in the NTT group had shorter operative times compared to the NCI group [median, 155.18 min (IQR, 139.06–171.30) versus 195.33 min (IQR, 183.95–206.71); p < 0.01].
Short-term prognostic characteristics of the surgical study cohort.

ICU, intensive care unit; IQR, interquartile range; NCI, neoadjuvant chemoimmunotherapy; NTT, neoadjuvant targeted therapy; VATS, video-assisted thoracic surgery.
In terms of postoperative outcomes, patients treated with NTT showed significantly reduced drainage volumes on the third day after surgery compared to those who received NCI [median, 150.15 mL (IQR, 186.29–266.72) versus 226.51 mL (IQR, 186.29–266.72); p = 0.03], with no differences observed during the first 2 days of postoperative life. Furthermore, the duration of chest tube drainage was significantly shorter in the NTT cohort than the NCI cohort [median, 5.91 days (IQR, 4.72–7.10) versus 7.00 days (IQR, 5.69–8.31); p = 0.04]. There was no significant difference in the length of hospitalization between the two groups.
Regarding postoperative complications, patients who underwent preoperative chemo immunotherapy showed an increased incidence of postoperative arrhythmias, such as atrial fibrillation and supraventricular tachycardia (n = 19, 26.0% versus n = 2, 6.1%, p = 0.02). For other prevalent complications, such as postoperative fever, persistent air leaks, chylothorax, and intensive care unit transfers, along with hospital readmission rates, no significant disparities were identified between the two cohorts.
Characteristics of the postoperative pathological response in tumor beds and lymph nodes among cohort patients
The pathological outcomes of patients who underwent lobectomy after NTT and NCI are shown in Figure 2 and Table 3. Twenty patients (27.4%) achieved CPR in the NCI group, whereas only one (3%) achieved CPR following NTT. The difference in CPR rates between the two groups was statistically significant (p = 0.004). However, the MPR rate did not show statistically significant differences between our two cohorts (p = 0.46). The average pathological response rates within the tumor bed were 64.48% and 69.86% in the NTT and NCI groups, respectively. The NTT and NCI groups had lymph node pathological response rates of 4.18% (IQR, 1.62–6.74%) and 4.42% (IQR, 2.50–6.34%), respectively, with no significant differences. Furthermore, the analysis did not reveal significant differences in the number of N1 and N2 lymph nodes dissected, or the rates of lymph node upstaging or downstaging between the two study cohorts.

Differences in therapeutic response between NTT and NCI. (a, b) The radiological shrinkage of primary tumor and metastatic lymph node (indicated by red arrow) in two representative patients after receiving NTT and NCI, respectively. (c, d) Figures show no difference in the pathological response rates of tumor bed and metastatic lymph node between the NTT and NCI groups, respectively. (e) Differences in MPR and CPR between the two groups, with the NCI group showing a significantly higher CPR rate than NTT group (p = 0.004).
Characteristics of the postoperative pathological response in tumor beds and lymph nodes among cohort patients.

CPR, complete pathologic response; IQR, interquartile range; LN, lymph node; MPR, major pathologic response; NCI, neoadjuvant chemoimmunotherapy; NTT, neoadjuvant targeted therapy; PR, pathological remission.
Subgroup analysis of lobectomy outcomes after NTT versus NCI
A subgroup analysis was conducted to analyze further the surgical results of lobectomy, the most common curative procedure for lung cancer after neoadjuvant therapy (Table 4). A total of 27 patients underwent lobectomy with systematic lymph node dissection after receiving NTT compared to 49 patients in the NCI group. Among them, patients in the NCI group were more likely to undergo open lobectomy [n = 22 (44.9%) versus n = 3 (11.1%); p < 0.01]. There were no differences in lymph node dissection, lymph node upstaging/downstaging, or conversion rates between the two groups. Furthermore, the duration of surgery was shorter in the NTT group than in the NCI group [median 144 min (IQR, 125–160) versus 180 min (IQR, 161–210); p < 0.01]. No statistically significant differences were observed between the groups regarding postoperative drainage volume, time to chest tube removal, length of hospital stay, or incidence of postoperative complications.
Subgroup analysis of lobectomy outcomes after neoadjuvant targeted therapy versus neoadjuvant chemoimmunotherapy.

CPR, complete pathologic response; ICU, intensive care unit; IQR, interquartile range; LN, lymph node; MPR, major pathologic response; NCI, neoadjuvant chemoimmunotherapy; NTT, neoadjuvant targeted therapy; PR, pathological remission; VATS, video-assisted thoracic surgery.
In terms of tumor pathological response rates, patients who received NCI were more likely to achieve CPR than those who receiving NTT (32.7% versus 3.7%, p < 0.01). The two groups had no statistically significant differences in the MPR rates, overall tumor bed response, or lymph node response.
Subgroup analysis of stage III lung cancer patients: short-term outcomes following NTT versus NCI
Targeted as the principal demographic for neoadjuvant intervention, patients diagnosed with stage III NSCLC showed distinct surgical and pathological outcomes after two different neoadjuvant therapies (Table 5). Our cohort included 30 patients (41.1%) with stage IIIA NSCLC and 15 (20.5%) with stage IIIB NSCLC. Surgical outcome analysis revealed that patients who underwent NTT had shorter operative times [median, 143 min (IQR, 135–157) versus 185 min (IQR, 168–248); p < 0.01] and length of stay [median, 7 days (IQR, 6–11) versus 10 days (IQR, 7–12); p = 0.048] than those who underwent NCI. Furthermore, the targeted therapy group showed significantly lower drainage volumes on the first (p = 0.03) and third (p = 0.02) postoperative days than the chemoimmunotherapy group. There were no statistically significant differences between the two groups in terms of surgical approach, conversion rates, chest tube removal times, or postoperative complication rates.
Subgroup analysis of stage III lung cancer patients: short-term outcomes following NTT versus NCI.

CPR, complete pathologic response; ICU, intensive care unit; IQR, interquartile range; LN, lymph node; MPR, major pathologic response; NCI, neoadjuvant chemoimmunotherapy; NTT, neoadjuvant targeted therapy; PR, pathological remission; VATS, video-assisted thoracic surgery.
Regarding pathological outcomes, patients treated with NCI showed a higher tumor bed pathological response rate [median, 80% (IQR, 55–100)] than those treated with NTT [median, 70% (IQR, 60–70); p = 0.04]. Furthermore, the CPR rate was higher in the NCI group than in the NTT group (n = 15, 33.3% versus 0; p = 0.01). No significant differences were observed in the MPR rate, lymph node pathological response rate, or lymph node upstaging/downstaging.
Discussion
This retrospective study assessed the safety and feasibility of MIS following two common neoadjuvant treatments (NCI versus NTT) and explore the differences in surgical complexity and patient prognosis after pulmonary resection. Our findings revealed that patients in the NTT group had a higher MIS rate, shorter operative duration, and lower conversion rate compared to those in the NCI group. These outcomes reflect a decreased occurrence of ‘frozen mediastinum’ after NTT, suggesting that surgical complexity is reduced for surgeons compared with NCI. In terms of short-term prognosis, post-targeted therapy surgical patients showed lower postoperative drainage volumes, shorter chest tube durations, fewer postoperative complications, and shorter hospital stays than those who received induction chemoimmunotherapy. Our research indicated that the patients had better surgical outcomes after targeted induction therapy than after chemoimmunotherapy, with no significant differences observed in the MPR and lymph node pathological response rates between the two groups.
In recent years, research on mechanisms and clinical trials focusing on immunotherapy and targeted therapy for locally advanced NSCLC has expanded exponentially. Neoadjuvant immune checkpoint inhibitors have shown promising benefits in inducing robust tumor-specific T-cell responses. Integrating traditional chemotherapy with immunotherapy significantly improves pathological response rates and prolongs patient survival. Provencio et al. assembled a cohort of patients with stage IIIA resectable NSCLC, indicating that 90% of patients achieved clinical downstaging after neoadjuvant chemotherapy combined with nivolumab. Among them, 35 of 41 patients (85.4%) did not report recurrence after surgery. 19 Even patients diagnosed with stage IIIB resectable NSCLC could benefit significantly from PFS (median PFS: 27.5 versus 16.7 months), which allowed opting for surgical intervention after neoadjuvant immunotherapy compared to radiation and chemotherapy. 20 This approach indicates that surgery may be a reasonable strategy for advanced-stage lung cancer when complete resection is feasible. Moreover, the ongoing evolution of next-generation sequencing technology has allowed the precise application of targeted therapies after identifying specific oncogenic drivers in NSCLC, significantly altering the treatment paradigm for this malignancy.21,22 Particularly among Asian patients, the EGFR 19 del and exon 21 L858R mutation rate was 30%, which is more than in Caucasians.23,24 In such cases, EGFR-TKI therapy has demonstrated superior survival benefits and drug tolerability compared with chemotherapy. 25 Xiong et al. 26 and Andrews et al. 27 revealed that patients with persistent N2 nodal metastasis who underwent curative surgery after EGFR-TKI induction therapy showed no significant difference in OS compared with patients who achieved mediastinal lymph node downstaging.
However, as a crucial part of neoadjuvant treatment efficacy assessment, inflammatory responses triggered by induction therapy have also raised concerns regarding the feasibility of subsequent surgical interventions. Bott et al. 7 first reported that neoadjuvant immunotherapy could cause necessitating open approaches and intraoperative conversions, with a notable 54% of patients undergoing video-assisted thoracoscopic lobectomies subsequently requiring conversion. However, the cohort size of this study was limited and quantitative parameters for evaluating surgical complexity were lacking. Further investigations by Chaft et al. 28 revealed that neoadjuvant immunotherapy induces thoracic inflammatory reactions, resulting in tissue fibrosis and extensive adhesions, which in turn complicate the anatomical resection process.
The main challenge in neoadjuvant therapy is choosing a suitable treatment that ensures the safety and efficacy for patients at various stages and physical statuses. Detailed reporting of the adverse events, surgical complications, and mortality associated with these therapies is crucial. A comprehensive understanding and effective management of these risks in clinical practice is crucial to maximize benefits and minimize risks without compromising antitumor efficacy. Currently, research on short-term outcomes after induction therapy has primarily focused on neoadjuvant immunotherapy and chemotherapy. There is still little research on surgical complexity-related outcomes between the NTT and NCI protocols. Our study first investigated the differences between these two neoadjuvant treatments in terms of surgical difficulty metrics, short-term prognostic indicators, and pathological response rates with the aim of guiding the selection of clinical strategies for which little information is available in the literature.
The IFCT-1601 IONESCO clinical trial was terminated prematurely because of the high postoperative mortality rate induced by durvalumab. Among the 46 patients who underwent surgical procedures, pneumonectomy was performed in 9, lobectomy in 31, combined lobectomy in 3, and exploratory thoracotomy in three patients. Subsequent follow-up revealed a 90-day mortality rate of 9%, with four patients dying after curative surgery. Causes of death included acute respiratory failure, tracheoesophageal fistulas. 29 This study indicates that arbitrarily following a high pathological response rate while neglecting surgery-related complications does not translate into benefits for patients. Mathey-Andrews et al. conducted a neoadjuvant cohort study that included 4229 patients to compare the outcome differences between neoadjuvant immunotherapy and neoadjuvant chemotherapy. They found that neoadjuvant immunotherapy did not increase open lobectomy rates (60.8% versus 51.6%, p = 0.11) or result in worse perioperative outcomes. Additionally, a study revealed that patients who received neoadjuvant chemotherapy showed higher lymph node downstaging rates than those who received neoadjuvant immunotherapy (48% versus 29%, p = 0.04). 9
Our study found that compared to NCI, patients who received NTT experienced higher MIS rates (84.8% versus 53.4%, p < 0.01) and lower conversion rates, indicating that NTT could reduce surgical complexity and improve short-term patient outcomes. The subgroup analysis revealed that patients with stage III NSCLC experienced more pronounced benefits from surgery after NTT. Furthermore, no statistically significant differences were observed between the NTT and NCI groups regarding lymph node downstaging or upstaging. The average pathological response rate was 64.48% in the NTT group and 69.86% in the NCI group, with no significant differences in the pathological response of the lymph nodes or MPR rates, corroborating the results reported by Shu et al. 30 Furthermore, Deng et al. 20 reported that neoadjuvant immunotherapy resulted in a decreased yield of N1 and N2 lymph nodes, which potentially correlated with increased surgical complexity after immunotherapy. Our study reported no difference in the number of N1/N2 lymph nodes dissected between the NTT and NCI groups in patients with stage I–III lung cancer. The complication rates reported in our cohort remained significant, with 27.3% and 35.7% occurrence rates in the targeted induction and chemoimmunotherapy groups, respectively. Notably, patients receiving NCI showed increased susceptibility to postoperative arrhythmias, including atrial fibrillation and supraventricular tachycardia (n = 19, 26.0% versus n = 2, 6.1%, p = 0.02). Jiang et al. 31 evaluated surgical feasibility in 31 patients following neoadjuvant immunotherapy (with or without chemotherapy) and reported a high complication rate of 58%, among which prolonged air leakage (45%) was identified as the most common. In terms of long-term prognosis, Qi et al. 32 performed a DFS analysis among 26 patients receiving NCI, compared to 9 patients treated with NTT, and found no significant differences [9.6 months (range, 4.0–47.9 months) versus 13.2 months (range, 7.5–32.2 months), p = 0.500] in these surgical cohorts. However, the limited sample size of this study requires further validation in future studies. Regarding pathological responses, we found that the NCI group had a higher CPR rate than the NTT group, consistent with the findings of Gu et al. 33
Neoadjuvant immunotherapy and targeted therapy are poised to revolutionize the treatment paradigm for NSCLC. Investigating differences in indicators of surgical difficulty, prognosis, and pathological response among various induction therapies is crucial for optimizing patient outcomes. Future studies should endeavor to clarify the mechanisms underlying the differences in surgical outcomes and pathological response rates between NTT and NCI. Furthermore, studies that focusing postoperative quality of life and survival are crucial for choosing the most effective neoadjuvant regimen for patients with locally advanced NSCLC.
This study had several limitations. First, its retrospective design inherently carries the possibility of unquantifiable confounders. Furthermore, the National Cancer Center conducted this study, which could have caused a selection bias within the patient cohort. Second, the analysis did not use a propensity score matching approach for baseline variables such as age, sex, and preoperative lung function due to limited data. Therefore, we selected data from a single surgical team to reduce variability in surgical outcomes.
Conclusion
Our findings indicate that patients receiving NTT are more likely to undergo MIS, benefit from shorter surgical durations, and experience lower incidences of conversion due to less challenging surgeries. Our study provides evidence that patients receiving NTT have a better short-term prognosis than those receiving NCI. Although NCI shows better control of systemic disease, the balance between surgical feasibility and therapeutic efficacy must guide treatment choices.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241265214 – Supplemental material for Analysis of surgical complexity and short-term prognostic indicators in NSCLC patients: neoadjuvant targeted therapy versus neoadjuvant chemoimmunotherapy
Supplemental material, sj-docx-1-tam-10.1177_17588359241265214 for Analysis of surgical complexity and short-term prognostic indicators in NSCLC patients: neoadjuvant targeted therapy versus neoadjuvant chemoimmunotherapy by Kun Wang, Hang Yi, Zhuoheng Lv, Donghui Jin, Li Fu and Yousheng Mao in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We appreciate the contributions of Guochao Zhang (Department of Thoracic Surgery, National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College) to the methodology section during the revision of the manuscript.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
