Abstract
Background:
The prognosis for patients with ultra high-risk gestational trophoblastic neoplasia (GTN) is significantly worse, and there is currently no consensus regarding the optimal treatment strategies for this specific patient population.
Objectives:
This study aims to investigate the clinical characteristics, treatment responses, outcomes, and prognostic risk factors in ultra high-risk GTN patients with an International Federation of Gynecology and Obstetrics (FIGO) score of 13 or higher.
Design:
Retrospective study.
Methods:
Medical records of 36 GTN patients with FIGO score ⩾ 13, treated at the first affiliated hospital of Zhengzhou University, China, from January 2015 to June 2024, were retrospectively reviewed. Chi-square tests, univariate analysis, and Kaplan-Meier survival analysis were employed for data analysis.
Results:
Etoposide, methotrexate, actinomycin D, cyclophosphamide, and vincristine (EMA-CO) was the most commonly used chemotherapy regimen (17/36, 47.2%). Twenty-one patients responded well to the initial chemotherapy regimen and achieved complete remission (CR). One patient was switched to salvage chemotherapy due to resistance to the initial regimen and subsequently achieved CR. Four patients experienced relapse; of these, three attained CR. The median follow-up time was 46 months (range 12–85 months). The CR rate was 69.4% (25/36). A total of 20 patients (55.6%) received adjuvant treatments, including surgery and radiotherapy. Stage IV disease, liver metastases, uncommon distant metastatic sites, and ⩾3 metastatic sites were significant predictors of mortality.
Conclusion:
In our study, the overall CR rate for ultra high-risk GTN patients with FIGO score ⩾ 13 was 69.4%. Five patients (45.5%) experienced early mortality. All patients with brain metastases who received chemotherapy in conjunction with whole-brain or stereotactic radiotherapy achieved CR. Immune checkpoint inhibitors demonstrated potential efficacy in treating chemotherapy-resistant GTN.
Plain language summary
In this study, we retrospectively investigated the clinical characteristics, treatment responses, outcomes, and prognostic risk factors in 36 ultra high-risk gestational trophoblastic neoplasia (GTN) patients with a FIGO score of 13 or higher. The median follow-up time was 46 months (range 12-85 months). (EMA-CO) was the most commonly used chemotherapy regimen. A total of 20 patients (55.6%) received adjuvant treatments, including surgery and radiotherapy. The CR rate was 69.4% (25/36). Five patients (45.5%) experienced early mortality. Stage IV disease, liver metastases, uncommon distant metastasis sites, and ⩾3 metastatic sites were significant predictors of mortality. Our study provided valuable data regarding treatments, outcomes, and prognostic risk factors for rare ultra high-risk GTN.
Keywords
Introduction
Gestational trophoblastic neoplasia (GTN) arises from the aberrant proliferation of trophoblastic tissue and encompasses conditions such as invasive mole, choriocarcinoma, placental site trophoblastic tumor (PSTT), and epithelioid trophoblastic tumor (ETT). In the year 2000, the International Federation of Gynecology and Obstetrics (FIGO) published the GTN guidelines, integrating the fundamental FIGO anatomic staging (stages I−IV) with the modified World Health Organization (WHO) risk factor scoring system. 1 GTN exhibits marked sensitivity to chemotherapy. The FIGO 2000 scoring system is utilized to predict prognosis and guide the treatment of GTN patients. Patients with a FIGO score of less than 7 are classified into the low-risk group and can be treated with single-agent chemotherapy. Conversely, those with a score of 7 or higher are categorized as high-risk and require multiagent chemotherapy. 2
The cure rates for low-risk GTN patients approach 100%. 3 For high-risk GTN patients, the cure rate ranges from 80% to 95%, indicating a high likelihood of successful treatment.4–7 However, a subgroup of patients with a FIGO score exceeding 12 or 13 indicates a poor prognosis. Complete sustained remission was ultimately achieved in only 67.9% to 68.4% of GTN patients with a FIGO score of 12 or higher,7,8 which is significantly lower than that observed in the overall high-risk GTN population. Patients with a FIGO score ⩾ 13 had a 6.4-fold increased risk of mortality compared to those classified as high-risk. 9 Several studies have demonstrated that patients with FIGO scores ⩾ 13 exhibit a substantially elevated mortality rate, ranging from 30% to 52%.6,10 Therefore, patients with a FIGO score⩾ 13 are classified as ultra high-risk GTN.11,12
To date, limited information is available regarding ultra high-risk GTN owing to its rarity. There remains no consensus on the optimal treatment strategies for these specific patients. Herein, we conducted a retrospective study to report our decade-long experience in treating ultra high-risk GTN patients at the First Affiliated Hospital of Zhengzhou University. The aim of this study was to investigate the clinical characteristics, treatment responses, outcomes, and prognostic risk factors in ultra high-risk GTN patients.
Methods
Study design and patients
This was a retrospective cohort study of 36 ultra high-risk GTN patients in the first affiliated hospital of Zhengzhou University (a large teaching hospital in Henan Province, China) from January 2015 to December 2021. All patients were classified according to the FIGO 2000 staging system for GTN, utilizing the modified WHO risk factor scoring system. 13 All 36 patients included in the study had a FIGO score of 13 or higher. All relevant hCG isoforms were quantitatively measured using the electrochemiluminescence immunoassay (ECLIA) method, including intact hCG, free β-hCG, nicked hCG, and the β-core fragment. The basic characteristics (including age, antecedent pregnancy, antecedent interpregnancy interval, pretreatment serum hCG value, stage, FIGO score, and metastatic sites) and the information on treatment (including chemotherapy, radiotherapy, and surgery), resistance, relapse, and survival were extracted from the electronic medical record system. Data from January 2015 to June 2024 were extracted. As this research was conducted retrospectively and involved the analysis of de-identified data, patient consent was not required. The reporting of this study conforms to the STROBE statement 14 (Supplemental Files).
Inclusion and exclusion criteria: Patients who were diagnosed with ultra high-risk GTN were included in the study. Patients diagnosed with low-risk or high-risk GTN and patients with incomplete data or lost to follow-up were excluded.
Treatment
Several different multiagent chemotherapy regimens were adopted as the first-line chemotherapy. A total of 2–3 more cycles of consolidation chemotherapy courses were given after normalization of serum β-hCG. Salvage chemotherapy regimens were used for those who didn’t have a good response to the first-line chemotherapy or relapsed after an initial successful treatment. Adjuvant treatment, including surgery and radiotherapy, was applied to remove the chemotherapy-resistant lesions or deal with emergencies such as hemorrhage. Follow-ups were provided to all of the patients either at outpatient clinics or through telephone interviews.
Evaluation after treatment
Complete remission (CR) was defined as normalization of hCG level for at least four consecutive weeks. Resistance was diagnosed by a plateau or an increase in hCG level after two or three courses of chemotherapy. Relapse was diagnosed when hCG levels increased again after CR. Early death was defined as that which occurred within 4 weeks after treatment initiation. The survival time was calculated from the date of diagnosis to the date of last follow-up or death.
Statistical analysis
Data are shown as a number (percentage). The chi-square test or Fisher’s exact test was used to compare categorical variables as appropriate. Kaplan-Meier survival curves were used to describe the survival of patients after treatment. Univariate analysis was performed to find out prognostic risk factors for survival. We performed statistical analyses with SPSS v.24.0 software (IBM Corp., Armonk, NY, USA), and a p value less than 0.05 was considered statistically significant.
Results
Between January 2015 and December 2021, a total of 38 patients were diagnosed with ultra high-risk GTN. Two patients were excluded because of incomplete medical records. A total of 36 participants were analyzed.
The detailed characteristics of these patients are shown in Table 1. The mean age of the group was 42 years (range 24–70). A total of 22 (61.1%) patients were older than 40. The most common antecedent pregnancy was term delivery in 19 cases (52.8%), followed by mole in 9 (25%), and non-molar abortion in 8 (22.2%). 29 (80.6%) patients had a time interval from antecedent pregnancy to treatment of more than 1 year. According to the FIGO 2000 staging system, most patients (33, 91.7%) were classified as advanced stage (FIGO III and IV). The common metastatic sites were the lungs (80.6%), brain (36.1%), and liver (25%). In addition, there were several unusual metastatic sites, including the kidney, spleen, intestine, bone, pancreas, atrium dextrum, bladder, and adrenal gland. 14 patients (38.9%) presented with isolated site metastases, 11 patients (30.6%) with double-site distant metastases, 5 (13.9%) patients with triple-site distant metastases, and 3 patients (8.3%) with more than 3 sites of distant metastases.
The detailed clinical characteristics of the 36 ultra high-risk GTN patients.
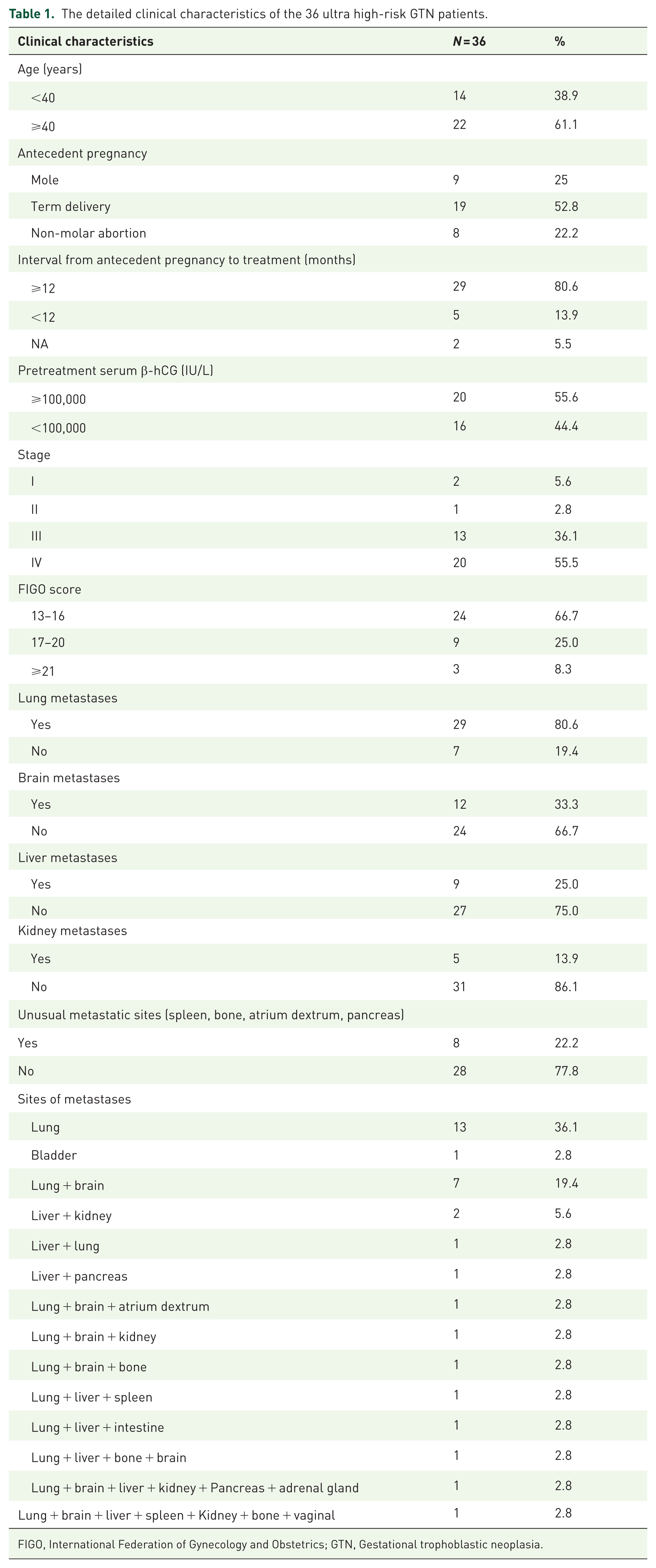
FIGO, International Federation of Gynecology and Obstetrics; GTN, Gestational trophoblastic neoplasia.
Table 2 summarizes the treatment and outcomes of the ultra high-risk GTN patients. Etoposide, methotrexate, actinomycin D, cyclophosphamide, and vincristine (EMA-CO) were the most commonly used chemotherapy regimens (17/36, 47.2%). Floxuridine-dactinomycin (FA) was given to 19.4% (7/36) of patients. Low-dose etoposide and cisplatin (EP) induction chemotherapy was administered to six patients. Nevertheless, three of the six patients had an early death. Among the remaining three patients, two of them received EMA-CO chemotherapy regimens and one received floxuridine-dactinomycin-etoposide-vincristine (FAEV) following induction chemotherapy. In addition, several regimens, including FAEV, etoposide-platinum alternating with EMA (EP/EMA), bleomycin-etoposide-cisplatin (BEP), methotrexate-cyclophosphamide-etoposide (MCE), and floxuridine-methotrexate-vincristine (FMV) were also administered as the initial chemotherapy regimens.
The treatments and outcomes of the 36 ultra high-risk GTN patients.

EMA-CO, Etoposide, methotrexate, actinomycin D, cyclophosphamide, and vincristine; VAC, vincristine-dactinomycin-cyclophosphamide.
Twenty-one patients responded well to the initial chemotherapy regimen and achieved CR. One patient was switched to receive salvage chemotherapy EMA-CO due to resistance to the initial chemotherapy regimen (FA) and achieved CR. Five patients ultimately died due to progressive disease despite initial chemotherapy and subsequent salvage chemotherapy. Among these five patients, three had adjuvant interventions in addition to chemotherapy. One patient underwent a hysterectomy. Another one who received multi-line salvage chemotherapy regimens underwent hysterectomy and pulmonary lobectomy. The other one with pancreas and liver metastasis received pancreas particle implantation, radiotherapy, and hepatic artery embolization. Four patients suffered a relapse following an initially successful treatment. Among these four patients, three attained CR through salvage chemotherapy subsequent to relapse. Unfortunately, the other recurrent patient ultimately died despite undergoing salvage chemotherapy and hysterectomy. The salvage chemotherapy regimens encompassed EMA-CO, FAEV, EP/EMA, vincristine-dactinomycin-cyclophosphamide (VAC), and docetaxel-cisplatin.
Adjuvant treatment, such as surgery and radiotherapy, is utilized based on the specific disease status of each patient to prevent lethal bleeding, reduce tumor burden, or eliminate isolated drug-resistant lesions. A total of 20 patients (55.6%) received adjuvant treatment, with eight patients undergoing more than one intervention. Emergency craniotomy was carried out in three patients due to the life-threatening high intracranial pressure resulting from hemorrhage and edema. Other adjuvant surgical treatments encompassed hysterectomy, resection of vaginal or bladder lesions, pulmonary lobectomy, and radiofrequency ablation of pulmonary lesions. Furthermore, seven patients underwent either whole-brain radiotherapy or stereotactic radiotherapy for brain metastases. Two patients who had developed resistance to multiple lines of chemotherapy were subsequently treated with immune checkpoint inhibitor (ICI) therapy. One patient received the anti-PD-1 agent sintilimab and achieved a complete response, remaining in remission. The other patient, despite receiving the anti-PD-1 agent camrelizumab, continued to progress. However, this patient subsequently achieved a complete response following salvage chemotherapy with FAEV in combination with stereotactic radiotherapy.
In our study, the median follow-up time was 46 months (range 12–85 months). The complete response (CR) rate was 69.4% (25/36). Among the 11 patients who died, nearly half (5/11, 45.5%) died early, occurring within one month of initiating treatment. Five patients died after initial and salvage treatments, and one patient died after relapse. The detailed clinical characteristics, treatment, and causes of death of the five patients who experienced early deaths are presented in Table 3. Kaplan-Meier survival curves were used to describe the survival of patients after treatment (Figure 1). Almost all of the mortalities occurred within the first year after treatment (9/11, 81.8%). The median time interval from treatment initiation to death was 3 months (range from 1 to 40 months).
The detailed clinical characteristics, treatment, and causes of death of the five patients who experienced early deaths.
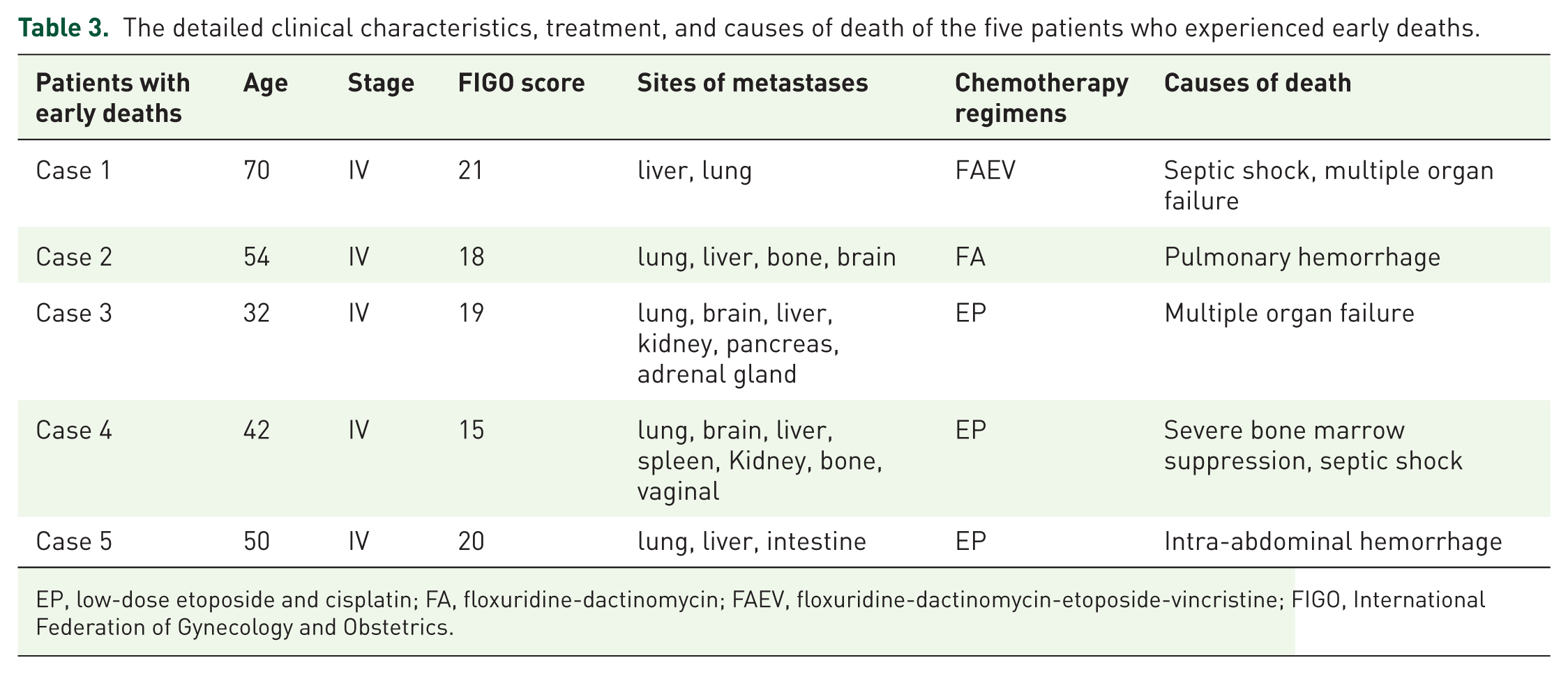
EP, low-dose etoposide and cisplatin; FA, floxuridine-dactinomycin; FAEV, floxuridine-dactinomycin-etoposide-vincristine; FIGO, International Federation of Gynecology and Obstetrics.

Kaplan-Meier survival curves were used to describe the survival of patients after treatment.
The univariate analysis of prognostic risk factors is presented in Table 4. As illustrated in Table 4, stage IV disease, liver metastases, uncommon metastatic sites (including kidney, pancreas, spleen, bone, and heart), and a number of metastases ⩾ 3 were significant predictors of mortality.
The univariate analysis of prognostic risk factors in the 36 ultra high-risk GTN patients.
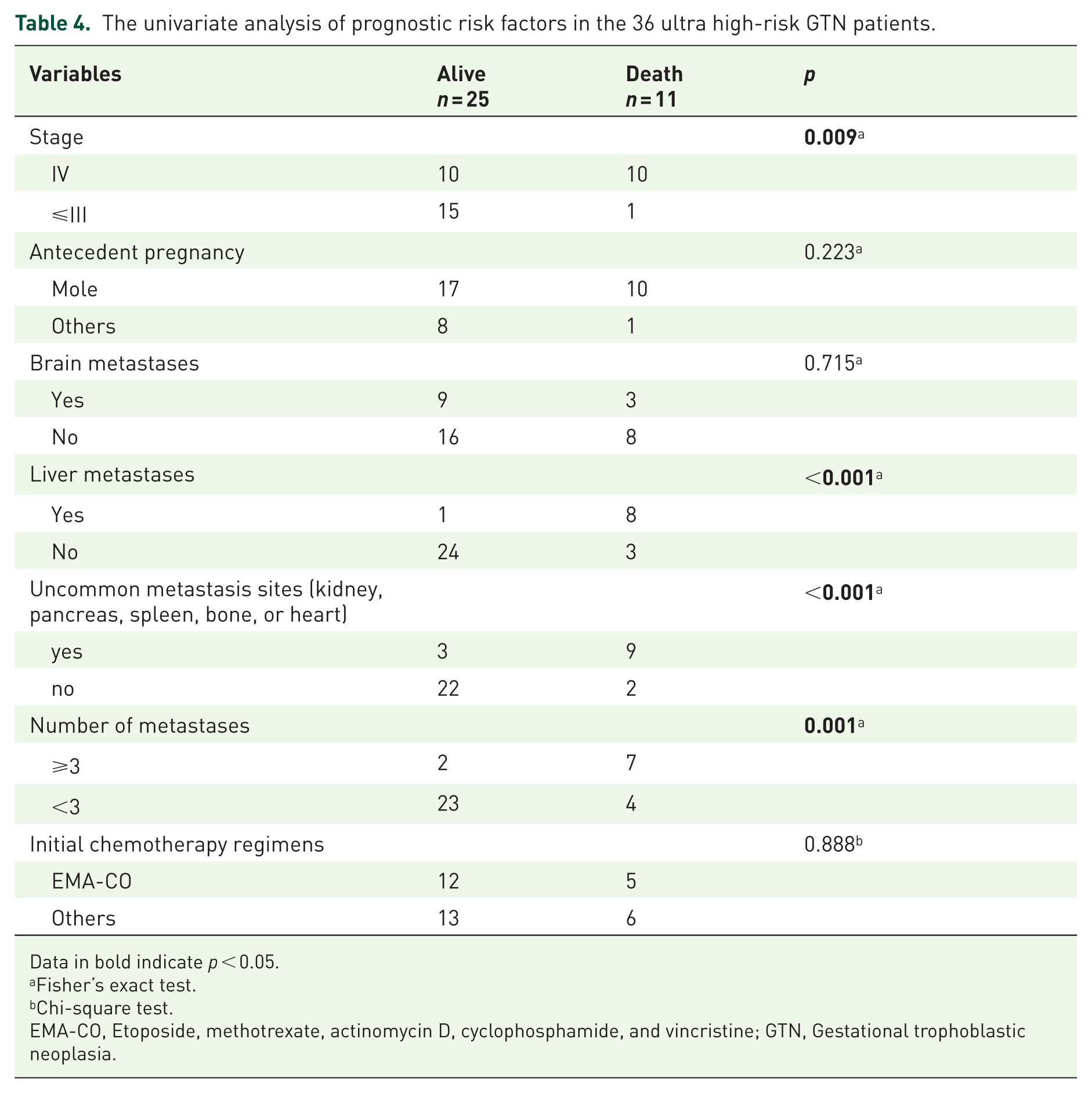
Data in bold indicate p < 0.05.
Fisher’s exact test.
Chi-square test.
EMA-CO, Etoposide, methotrexate, actinomycin D, cyclophosphamide, and vincristine; GTN, Gestational trophoblastic neoplasia.
Discussion
Main findings
Our study conducts a retrospective analysis of the treatment and outcomes for ultra high-risk GTN patients with a FIGO score of 13 or higher. The complete response rate for these 36 ultra high-risk GTN patients was 69.4%. Five patients (45.5%) experienced early death. The CR rate for ultra high-risk GTN patients with brain metastases was 75%. All patients who received chemotherapy in conjunction with whole-brain or stereotactic radiotherapy achieved CR without cognitive impairment. Liver metastases, uncommon distant metastatic sites, and multiple-site metastasis were associated with poorer outcomes.
Strengths and limitations
Given the rarity of ultra high-risk GTN, the primary limitations of this study include its retrospective design and the relatively small sample size. Despite these limitations, this study reports our decade-long valuable therapeutic experience with ultra high-risk GTN at the largest teaching hospital in Central China.
Interpretation
Due to their high chemosensitivity, the cure rate ranged from 80% to 95% for the high-risk GTN patients.4–7 However, the prognosis of ultra high-risk GTN patients is notably poorer. Several studies have suggested that a FIGO score of 12 or higher is an independent risk factor for poor prognosis.15–18 Kong et al. 8 reported that the five-year overall survival (OS) rate for the cohort of ultra high-risk GTN patients with a FIGO score ⩾ 12 was 67.9%. Maestá et al. 7 also found that complete sustained remission was ultimately achieved in 68.4% of ultra high-risk GTN patients (score ⩾ 12) by 18 months following the completion of therapy. In addition, Bolze et al. 10 demonstrated that patients with a FIGO score ⩾ 13 had a significantly higher 5-year mortality rate compared to those with a FIGO score < 13 (38.4% vs 4.9%). 10 In our study, the CR rate for ultra high-risk GTN patients with a FIGO score ⩾ 13 reached 69.4%, which is consistent with previous findings. In our study, 5 (45.5%) patients experienced early death, occurring within 1 month of initiating treatment, which is consistent with the findings of Hassan et al. 9 The causes of mortality include pulmonary or cerebral hemorrhage, severe bone marrow suppression, electrolyte imbalances, and multiple organ dysfunction. Compared with other patients, the five patients who experienced early death exhibited a higher tumor burden, as evidenced by multiple sites of distant metastases. Three out of the five patients had more than three sites of distant metastases. One patient even had metastases at seven distinct sites, including the brain, lungs, vagina, liver, spleen, kidney, and bones.
EMA-CO was first described by the Charing Cross group in 1986. 19 Currently, EMA-CO has become the most common and preferred first-line chemotherapy regimen for high-risk GTN.4,20 In addition, it is now formally recommended for use in ultra high-risk GTN patients by the National Comprehensive Cancer Network. 11 Adequate first-line treatments are emphasized to reduce the occurrence of drug resistance in ultra high-risk GTN patients. However, there is no consensus on the optimal chemotherapy regimen for ultra high-risk GTN due to its rarity and the lack of prospective randomized data. In a multi-study analysis, EMA-CO, EP/EMA, and FAEV were found to be the three most commonly used first-line regimens for GTN patients with a FIGO score ⩾ 12. 21 The complete response (CR) rate was 55.2%–60% for EMA/CO.21,22 In our country, FAEV is also considered the first-line chemotherapy for ultra high-risk GTN patients at Peking Union Medical College Hospital and has yielded a 63.1% CR rate. 8 In addition, the CR rate for EP/EMA was 60%–66.7%.21,23 In our study, several first-line chemotherapy regimens were administered to 36 ultra-high-risk GTN patients, with EMA-CO being the most commonly used regimen (17/36, 47.2%). Historically, in northern China, floxuridine-dactinomycin (FA) was the preferred first-line chemotherapy regimen for high-risk GTN patients 24 ; therefore, FA was employed as the second most frequently used regimen in this cohort. Notably, three patients were referred to the oncology department due to a variety of primary clinical manifestations, including hemoptysis, chest pain, headache, loss of consciousness, nausea, and vomiting. Less commonly used regimens, such as FMV, MCE, and BEP, were selected based on the treating physicians’ clinical judgment and individual patient conditions. Selection bias in the choice of chemotherapy regimens for these patients results in poor comparability among the regimens. Our data demonstrated that the CR rate for the EMA/CO used as a first-line chemotherapy regimen was 52.9% (9 out of 17 cases). There was no statistically significant difference in the CR rate between the EMA/CO and other regimens when used as a first-line setting (52.9% vs 57.9%). Other first-line chemotherapy regimens, such as FA, FAEV, EP/EMA, and BEP, were not inferior to the EMA/CO regimen. However, due to the small sample size, these findings are inconclusive and require further investigation.
Early death is prevalent among ultra high-risk GTN patients, often associated with tumor hemorrhage, metabolic acidosis, septicemia, and/or multiple organ failure during full-dose multiagent chemotherapy.4,10,20 Alifrangis et al. 4 reported that low-dose induction EP chemotherapy significantly reduced the early death rate of high-risk GTN patients from 7.8% to 0.7%. Therefore, to prevent early death, 1–3 cycles of low-dose EP induction therapy were introduced as a preliminary treatment before initiating EMA/CO. 12 In this study, we had limited experience with this protocol, and only six patients received induction low-dose EP chemotherapy. Among these six patients, four died, including three cases experiencing early death. All three cases of early death exhibited multiple-site metastasis and were characterized by a critical clinical condition, with two patients presenting six and seven distant metastatic sites, respectively. Despite administration of low-dose EP chemotherapy, the patients’ clinical conditions rapidly deteriorated, ultimately resulting in death due to multiple organ failure, septic shock, and intra-abdominal hemorrhage, respectively.
Ultra high-risk GTN patients with brain metastases frequently exhibit a diverse range of clinical manifestations. Some patients present exclusively with central nervous system symptoms, which may potentially lead to delayed diagnosis or misdiagnosis. In our study, five of the twelve patients with brain metastases exhibited neurological symptoms exclusively, including unilateral limb weakness, headache, loss of consciousness, and blurred vision. Previous studies have demonstrated that brain metastases serve as a poor prognostic factor.8,25 The survival rate for GTN patients with brain metastases ranged from 35% to 70.3%.15,26 In our study, the survival rate for ultra high-risk GTN patients with brain metastases was 75%, which is higher than previously reported rates. In addition, based on our limited sample size, we were unable to conclusively demonstrate that brain metastases serve as a risk factor for survival. To date, there is no established optimal protocol for the treatment of GTN patients with brain metastases. Traditional treatment methods involved systemic chemotherapy in combination with whole-brain radiation therapy. Given the cognitive impairment associated with whole-brain radiation therapy, intrathecal methotrexate administration has been promoted as an alternative to irradiation for the treatment of brain metastases. 27 However, intrathecal methotrexate was not administered in our clinical practice. Among the twelve patients with brain metastases, seven received chemotherapy in conjunction with whole-brain radiotherapy or stereotactic radiotherapy, achieving CR without cognitive impairment. In addition, emergency craniotomy plays a critical role in salvaging patients’ lives.
An overview of the current data on the use of ICI for GTN by Baas et al. revealed that the majority of high-risk GTN patients with chemotherapy resistance (77 out of 118) achieved remission following ICI treatment. 28 Immune checkpoint inhibitors offer a valuable new therapeutic option for high-risk GTN patients who are resistant to chemotherapy. Our experience with ICIs is limited; only two patients received ICI therapy after developing resistance to multiple lines of chemotherapy. One patient achieved a complete response, while the other initially progressed on ICI but subsequently achieved a complete response following salvage chemotherapy. These findings suggest the potential efficacy of ICIs in treating chemotherapy-resistant GTN.
Liver metastases have previously been identified as a significant risk factor associated with poor prognosis in patients with GTN, according to existing literature.16,29 Consistently, univariate analysis in our study demonstrated that the CR rate among ultra-high-risk GTN patients with liver metastases was 11.1%, which was significantly lower compared to those without liver metastases. In addition, our study identified uncommon metastatic sites, including the kidney, pancreas, spleen, bone, and heart, as well as multiple-site metastasis, as factors associated with poor outcomes. The significant disease burden and extensive disease dissemination in these patients pose a considerable challenge for the management of GTN. Similarly, Xiao et al. 16 also found that multiple distant metastatic sites and concurrent kidney metastasis were predictors of poor outcomes in GTN patients. Unfortunately, due to the limited sample size, we were unable to conduct multivariate analysis to identify independent risk factors for poor outcomes in ultra-high-risk GTN patients. Further studies are needed to address this limitation.
Conclusion
In conclusion, the complete response rate for ultra high-risk GTN patients with a FIGO score ⩾ 13 was 69.4% in our study. Notably, five patients (45.5%) experienced early death. There was no statistically significant difference in the CR rate between the EMA/CO regimen and other regimens (such as FA, FAEV, EP/EMA, and BEP) when used as first-line treatments (52.9% vs 57.9%). The CR rate for ultra high-risk GTN patients with brain metastases was 75%. All patients who received chemotherapy in conjunction with whole-brain or stereotactic radiotherapy achieved CR without cognitive impairment. Emergency craniotomy played a critical role in saving patients’ lives. Liver metastases, uncommon distant metastatic sites, and multiple-site metastasis were associated with poorer outcomes.
Supplemental Material
sj-pdf-1-tam-10.1177_17588359251379400 – Supplemental material for Management and prognosis of ultra high-risk gestational trophoblastic neoplasia patients: a long-term retrospective study
Supplemental material, sj-pdf-1-tam-10.1177_17588359251379400 for Management and prognosis of ultra high-risk gestational trophoblastic neoplasia patients: a long-term retrospective study by Meng Mao, Hanlin Fu, Qian Wang, Jing Bai, Ye Zhang and Ruixia Guo in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
This was a retrospective cohort study. The authors thank all women who provided the survey data to help us better understand the treatment and prognosis of ultra-high-risk GTN.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
