Abstract
Background:
In the era of targeted therapy, whether patients with human epidermal growth factor receptor 2 (HER2)-positive breast cancer are exempted from anthracycline usage in the neoadjuvant setting is controversial.
Objectives:
Our objective was to retrospectively analyze the differences in pathological complete remission (pCR) rates between the anthracycline group and the nonanthracycline group.
Design:
The CSBrS-012 study (2010–2020) included female primary breast cancer patients with neoadjuvant chemotherapy (NAC) who underwent standard breast and axillary surgery post-NAC.
Methods:
A logistic proportional hazard model was applied to estimate the association of covariates with pCR. Propensity score matching (PSM) was performed to balance the differences in baseline characteristics, and subgroup analyses were performed using the Cochran–Mantel–Haenszel test.
Results:
A total of 2507 patients were enrolled: the anthracycline group (n = 1581, 63%) and the nonanthracycline group (n = 926, 37%). A pCR was recorded in 17.1% (271/1581) of patients in the anthracycline group and in 29.3% (271/926) in the nonanthracycline group, and the difference in the pCR rate between the two groups was statistically significant [odds ratio (OR) = 2.00, 95% confidence interval (CI) (1.65–2.43); p < 0.001). In the subsequent subgroup analysis, substantial differences in pCR rates between the anthracycline and nonanthracycline groups were detected in the nontargeted [OR = 1.91, 95% CI (1.13–3.23); p = 0.015] and dual-HER2-targeted populations [OR = 0.55, 95% CI (0.33–0.92); p = 0.021) before PSM, whereas differences vanished after PSM. The pCR rates between the anthracycline and nonanthracycline groups did not differ for the single target population, either before or after PSM.
Conclusion:
In the presence of trastuzumab and/or pertuzumab, the pCR rate of patients with HER2-positive breast cancer receiving anthracycline was not superior to that of patients receiving nonanthracycline. Thus, our study further provides clinical evidence for exempting anthracycline treatment in HER2-positive breast cancer in the era of targeted therapy.
Keywords
Introduction
Breast cancer is the most common female malignancy worldwide. 1 Human epidermal growth factor receptor 2 (HER2)-positive breast cancer is a more aggressive and rapidly progressive subtype with poor prognosis. 2 Anti-HER2-targeted therapy can significantly improve the prognosis of these patients, reduce recurrence, and prolong survival time. 2 Trastuzumab when added to chemotherapy has shown improvement in disease-free survival (DFS), overall survival (OS), and pathological complete remission (pCR) rate in patients with advanced and early HER2-positive breast cancer in several adjuvant and neoadjuvant trials.3–14 Currently, trastuzumab plus pertuzumab combined with taxane is the preferred neoadjuvant therapeutic scheme for HER2-positive breast cancer.15,16
However, trastuzumab is associated with a risk of cardiac toxicity, particularly when combined with high-dose chemotherapy containing anthracyclines.10,17 A BCIRG006 trial reported that incidences of congestive heart failure were 2.0% and 0.4% in the anthracycline and nonanthracycline arms based on trastuzumab plus chemotherapy, respectively. 10 Higher cardiac event rates were also found in studies of anthracycline-containing regimens with trastuzumab compared to those without trastuzumab.18,19 Subsequently, the results of the TRAIN-2 and TRYPHAENA trials9,13 showed a similarity in the pCR rate between nonanthracycline and anthracycline regimens with targeted therapy in patients with HER2-positive breast cancer. Therefore, it is currently controversial whether patients with HER2-positive breast cancer can be exempted from anthracyclines during the neoadjuvant period.
Data of this study were from a multicenter and 10-year retrospective study in China based on patients with HER2-positive breast cancer treated with neoadjuvant chemotherapy (NAC). Our objective was to retrospectively analyze the differences in pCR rates between the anthracycline group and the nonanthracycline group, hoping to add additional evidence of exempting anthracycline in NAC in patients with HER2-positive breast cancer.
Methods
Study design and data collection
The Chinese Society of Breast Surgery study (CSBrS-012) (2010–2020) is a nationwide, multicenter, 10-year retrospective clinical epidemiological study across 20 hospitals in China. The CSBrS-012 study embodied woman primary breast cancer patients with NAC who underwent standard breast and axillary surgery post NAC between January 2010 and December 2020. These hospitals were from seven different geographic regions of China, which cover the majority of China and represent different levels of breast cancer burden.
Next, a total of 3927 female patients with HER2-positive breast cancer were selected from 10713 patients of the CSBrS-012 study. Among 3927 patients, we excluded 1420 patients who met the following criteria: (a) patients with no complete clinicopathological features, such as unknown clinical T stage or Tis or T0, unknown clinical N status, estrogen receptor (ER) status, progesterone receptor (PR) status, or Ki-67 expression; (b) patients with an unknown NAC regimen or receiving less than four cycles of treatment or modification of the chemotherapy regimen during the neoadjuvant period; (c) patients with an unknown pCR state of breast or axillary lymph nodes; and (d) patients who only received the AC regimen. Eventually, 2507 patients were included in our study (Figure 1). Patients in the anthracycline group received an NAC regimen with TAC, AC-T, TA, AC-TH, or AC-THP. Patients in the nonanthracycline group received a therapeutic regimen with TC, TX (X for capecitabine), TP (P for platinum), THP, or TCbHP. ‘T’ stands for taxane, ‘A’ for anthracycline, ‘C’ for cyclophosphamide, ‘Cb’ for platinum, ‘H’ for trastuzumab, and ‘P’ for pertuzumab.

Flow diagram of the study design.
In this study, HER2 status was confirmed by immunohistochemistry (IHC) or fluorescence in situ hybridization (FISH) assays. HER2 IHC scores of 0 and 1+ were considered HER2 negative, and a HER2 score of 3+ was considered HER2 positive, whereas a HER2 score of 2+ was further estimated by FISH, and HER2 gene amplification was considered HER2 positive.
Outcomes
The primary endpoint was the percentage of pathological complete remissions (pCRs), which was assessed locally at the time of surgery and defined as the absence of invasive tumor cells in the breast and axilla (pCR; ypT0/Tis, ypN0). Patients underwent breast-conserving or ablative surgery, and treatment of axillary lymph nodes included sentinel lymph node biopsy or axillary lymph node dissection.
Statistical analysis
Clinicopathological features were compared between the anthracycline group and the nonanthracycline group using Pearson’s chi-square test or Fisher’s exact test. The pCR rate difference between the two groups and the 95% confidence interval (95% CI) for the difference were estimated using the VassarStats tool 20 (http://vassarstats.net/index.html). Univariate and multivariate logistic proportional hazard models were applied to estimate the association of covariates with pCR and calculate the odds ratio (OR) and 95% CI. Propensity score matching (PSM) was performed to balance the differences in baseline characteristics between groups according to 1:1 using the following predetermined factors: age at diagnosis, clinical T stage, clinical N status, hormone receptor status, and Ki-67 expression level. The standardized mean difference (SMD) is usually used to quantify the balance between the two groups before and after PSM, and SMD > 0.1 is considered inconsistent between the two groups. Therefore, to eliminate the influence of confounding factors, this study conducted a 1:1 PSM analysis between patients receiving nonanthracycline and those receiving anthracycline in subpopulations. PSM and SMD were analyzed using the ‘MatchIt’ and ‘Tableone’ R packages, respectively. Subgroup analyses were performed for the above stratification factors using the Cochran–Mantel–Haenszel test.
A two-sided p value < 0.05 was considered statistically significant. All statistical analyses were performed using SPSS version 25.0 (IBM SPSS Statistics, Chicago, IL, USA) and R software v4.1.1 (http://www.r-project.org).
Results
Demographics and clinical characteristics of the study population
A screening flow diagram of our study population is presented in Figure 1. A total of 1581 patients receiving anthracycline and 926 patients receiving nonanthracycline who met the inclusion criteria were included in our study. As shown in Table 1, the demographics and clinical characteristics between the two treatment groups were generally different. The proportion of patients receiving anthracycline increased with older age, smaller tumor size, positive lymph nodes, ER negative, PR negative, and Ki-67 > 30%. The population receiving a nonanthracycline regimen was more likely to receive targeted therapy than those receiving anthracycline (H, 58.5% versus 22.8%; HP, 32% versus 4.8%); however, there was no difference in breast-conserving surgery rate between the two groups (11.5% versus 13.4%; p = 0.117). Hence, an interesting phenomenon was discovered in this study: the pCR rate of the nonanthracycline group was higher than that of the anthracycline group [29.3% versus 17.1%; OR = 2.00, 95% CI (1.65–2.43); p < 0.001] (Figure 2), and the difference in the pCR rate between the two groups was also statistically significant [rate difference (RD) = 12.2%, 95% CI (8.6–15.7); p = 0.0002] (Figure 2).
Baseline characteristics of patients with HER2-positive breast cancer in the anthracycline group and nonanthracycline group.
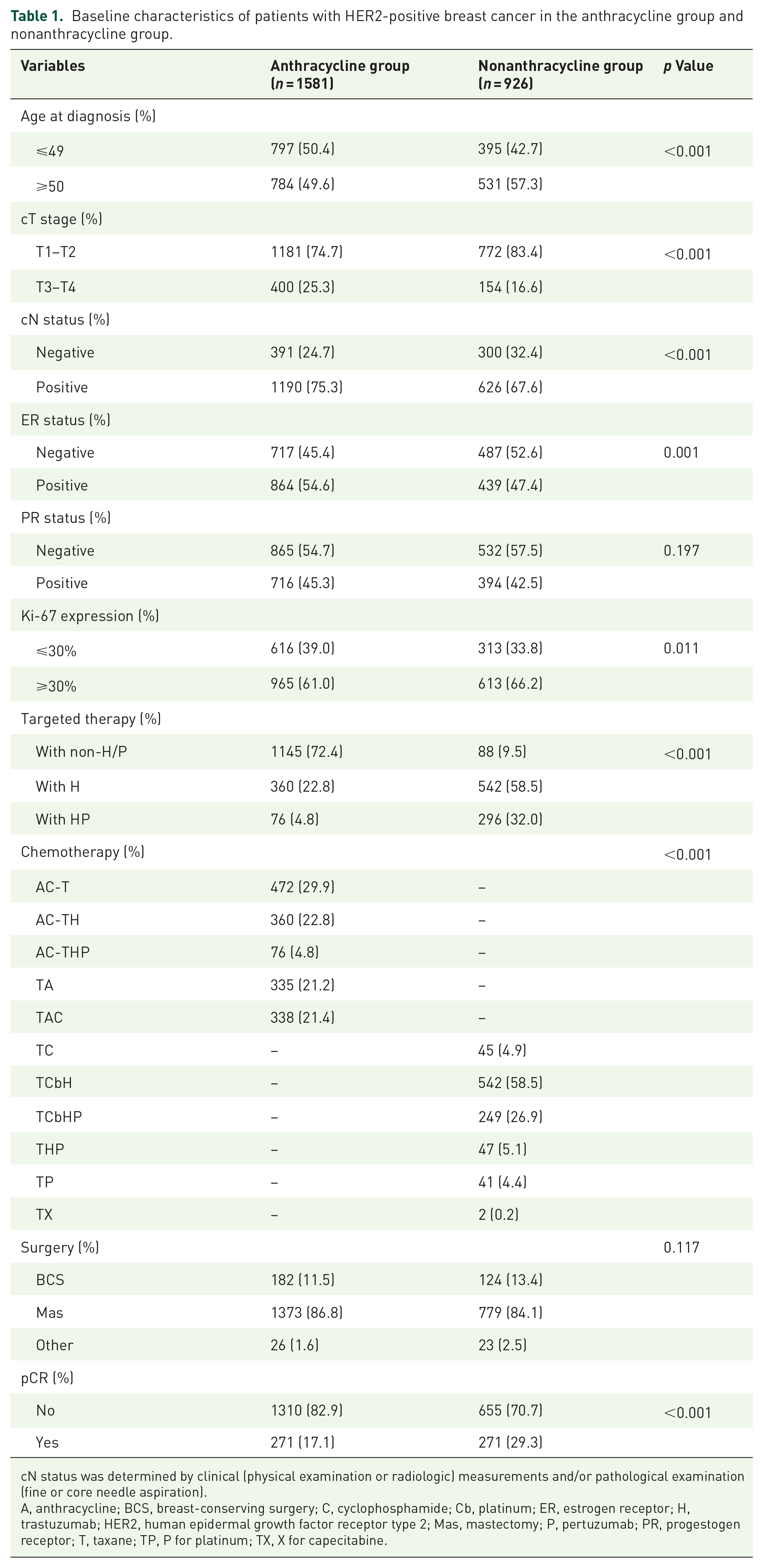
cN status was determined by clinical (physical examination or radiologic) measurements and/or pathological examination (fine or core needle aspiration).
A, anthracycline; BCS, breast-conserving surgery; C, cyclophosphamide; Cb, platinum; ER, estrogen receptor; H, trastuzumab; HER2, human epidermal growth factor receptor type 2; Mas, mastectomy; P, pertuzumab; PR, progestogen receptor; T, taxane; TP, P for platinum; TX, X for capecitabine.

pCR rate of the total population according to two treatment groups.
Predictive factors for pCR
Furthermore, univariate and multivariate logistical analyses were successively utilized to determine the predictive factors of pCR. As shown in Table 2, the results revealed that patients with a smaller tumor size, nodal status negativity, ER negativity, and PR negativity were more likely to achieve pCR, whereas age at diagnosis and Ki-67 expression level were not independently associated with the pCR rate. In addition, in terms of treatment administration, patients receiving targeted therapy were more likely to achieve pCR than those receiving nontargeted therapy. There was also no independent association between anthracycline treatment and the pCR rate.
Logistic regression analysis of independent predictors for pCR possibility.

cN status was determined by clinical (physical examination or radiologic) measurements and/or pathological examination (fine or core needle aspiration).
ER, estrogen receptor; H, neoadjuvant chemotherapy regimen with trastuzumab; HER2, human epidermal growth factor receptor type 2; HP, neoadjuvant chemotherapy regimen with trastuzumab plus pertuzumab; non-H/P, neoadjuvant chemotherapy regimen without trastuzumab and pertuzumab; PR, progestogen receptor.
Comparison of clinicopathological characteristics and pCR rates in the non-H/P, H, and HP subpopulations of patients before PSM and after PSM
A detailed comparison of clinicopathological characteristics with/without anthracycline administration was further conducted in the non-H/P, H, and HP subpopulations of patients. As presented in Table 3, in the non-H/P population, there was no difference in the distribution of clinicopathological features between patients receiving anthracycline and those receiving nonanthracycline; in the H population, patients with T1-T2 (84.5% versus 74.2%, p < 0.001) and Ki-67 > 30% (68.8% versus 58.9%, p = 0.003) had higher proportions in the nonanthracycline group than in the anthracycline group; in the HP population, compared to the nonanthracycline group, patients with youger (63.2% versus 44.6%, p = 0.006), positive nodal status (80.3% versus 64.2%, p = 0.011), and PR positivity (52.6% versus 35.1%, p = 0.008) had a higher proportion in those receiving anthracycline.
The clinicopathological features of the anthracycline and nonanthracycline groups were compared in three subpopulations before PSM.

SMD > 0.1 was considered inconsistent between the two groups.
ER, estrogen receptor; H, neoadjuvant chemotherapy regimen with trastuzumab; HP, neoadjuvant chemotherapy regimen with trastuzumab plus pertuzumab; non-H/P, neoadjuvant chemotherapy regimen without trastuzumab and pertuzumab; PR, progestogen receptor; PSM, propensity score matching; SMD, standardized mean difference.
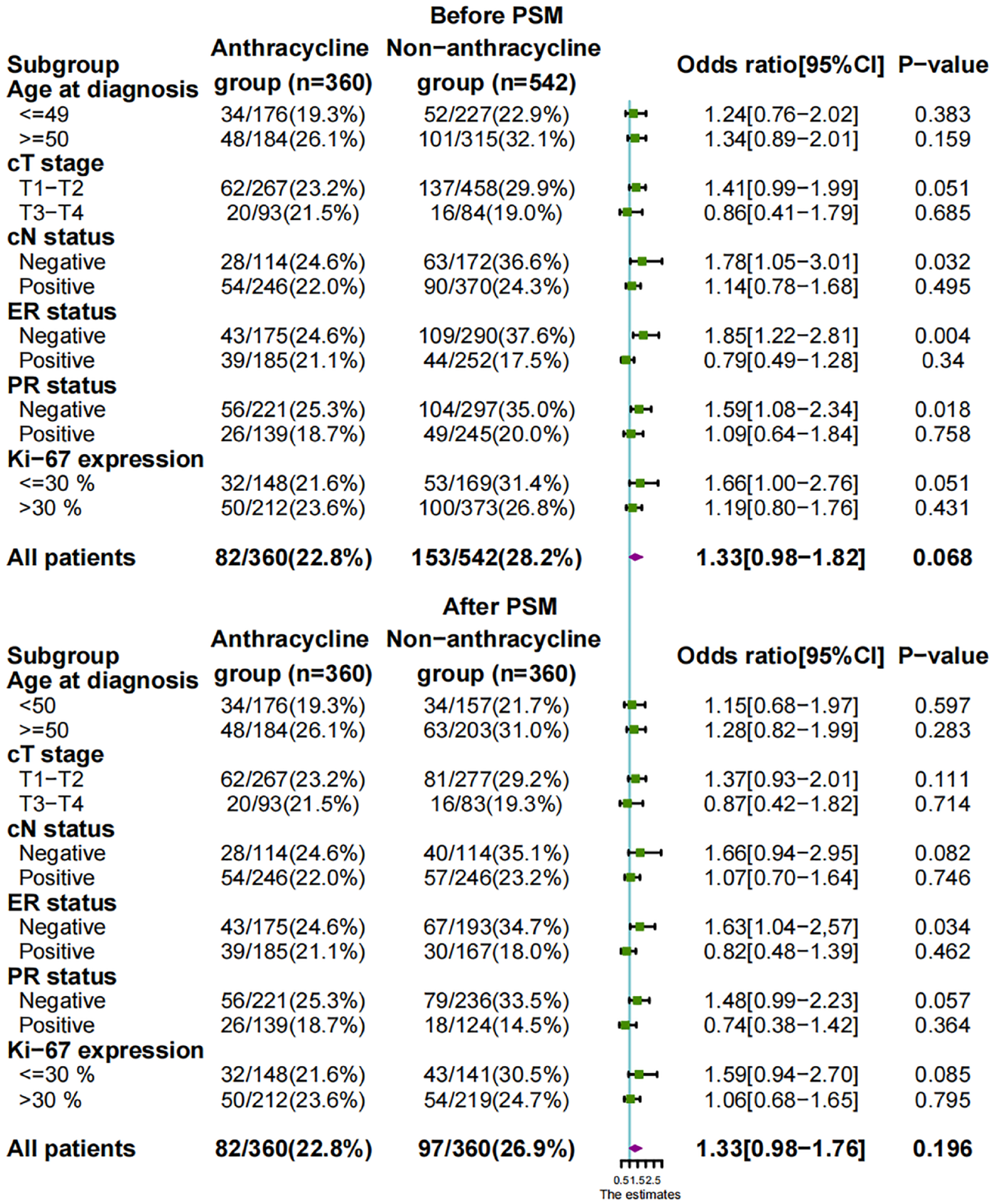
However, it was interesting that the pCR rate of patients receiving nonanthracycline therapy (22.7%) was higher than that of those receiving anthracycline (13.4%) in the non-H/P population [OR = 1.91, 95% CI (1.13–3.23); p = 0.015], corresponding to an absolute benefit of 9.3% [95% CI (1.1–19.9); p = 0.0148] in the nonanthracycline group (Figure 3(a)). The pCR rate of patients receiving anthracycline therapy (47.4%) was higher than that of patients receiving nonanthracycline therapy (33.1%) in the HP population [OR = 0.55, 95% CI (0.33–0.92); p = 0.021], corresponding to an absolute benefit of 14.3% [95% CI (1.5–27.1); p = 0.0209] in the anthracycline group (Figure 3(e)). Nevertheless, in the H population, there was a similar pCR rate between the nonanthracycline and anthracycline groups of patients [28.2% versus 22.8%, OR = 1.33, 95% CI (0.98–1.82), p = 0.068; RD = 5.4%, 95% CI (−0.6 to 11.2), p = 0.0677] (Figure 3(c)).

pCR rates of the three subpopulations before PSM and after PSM. (a, b) The non-H/P population received neoadjuvant chemotherapy regimens without trastuzumab and pertuzumab before PSM (a) and after PSM (b). (c, d) The H population received a neoadjuvant chemotherapy regimen with trastuzumab before PSM (c) and after PSM (d). (e, f) The HP population received a neoadjuvant chemotherapy regimen with trastuzumab plus pertuzumab before PSM (e) and after PSM (f).
After confounding factors were eliminated by PSM (Table 4), we found that pCR rate differences between the anthracycline group and the nonanthracycline group in the non-HP [OR = 1.43, 95% CI (0.68–3.02), p = 0.345; RD = 5.7%, 95% CI (−7.0 to 18.2), p = 0.3452] (Figure 3(b)) and HP [OR = 0.69, 95% CI (0.36–1.31), p = 0.251; RD = 9.2%, 95% CI (−7.3 to 25.1), p = 0.251] (Figure 3(f)) subpopulations disappeared. In addition, there was no difference in the pCR rate between the nonanthracycline group and anthracycline group in the H population after PSM [OR = 1.33, 95% CI (0.98–1.76), p = 0.196; RD = 4.1%, 95% CI (−2.4 to 10.6), p = 0.196], which was similar to the result before PSM [OR = 1.33, 95% CI (0.98–1.82), p = 0.068; RD = 5.4%, 95% CI (−0.6 to 11.2), p = 0.0677] (Figure 3(d)).
The clinicopathological features of the anthracycline and nonanthracycline groups were compared in three subpopulations after PSM.
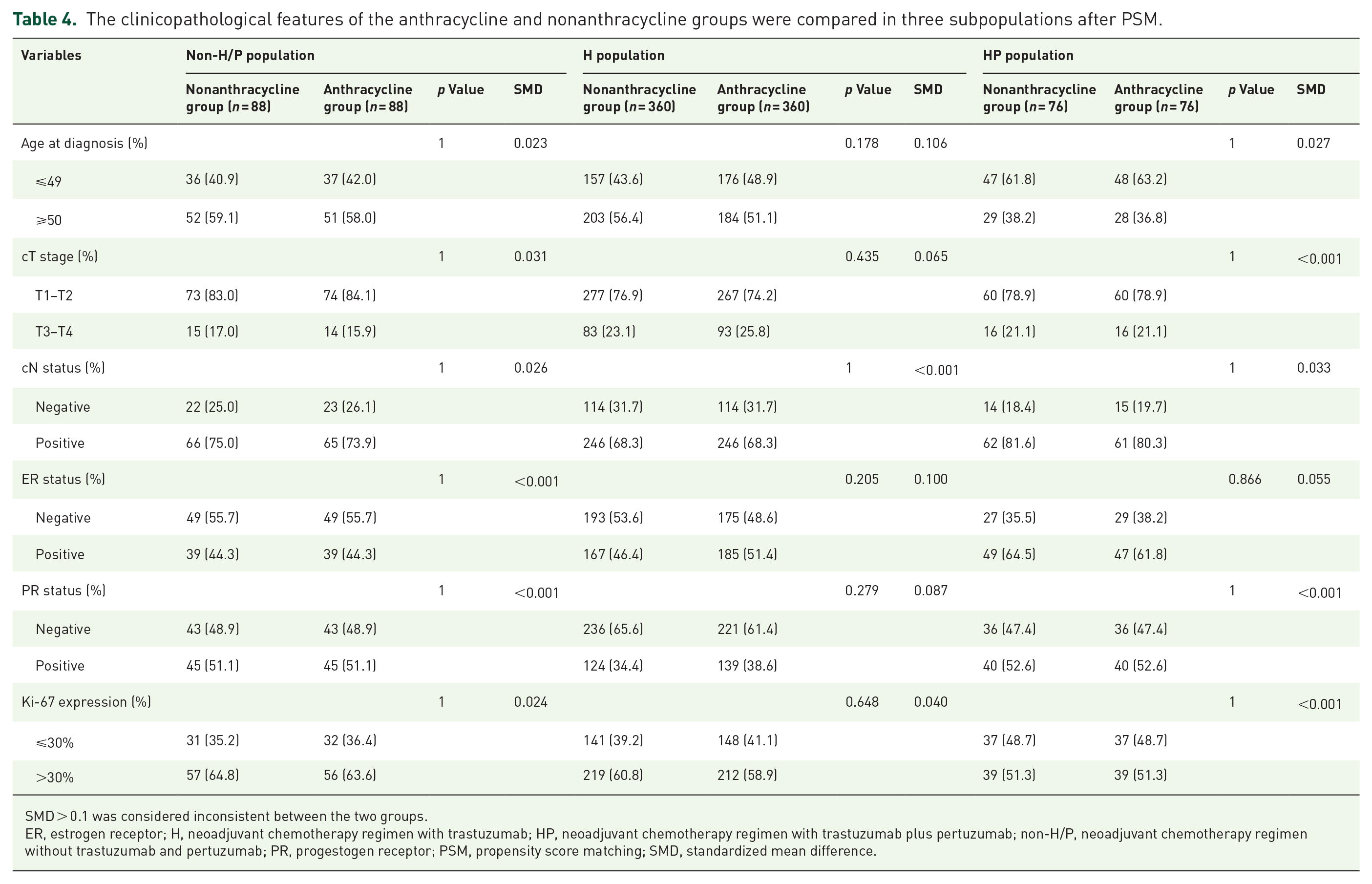
SMD > 0.1 was considered inconsistent between the two groups.
ER, estrogen receptor; H, neoadjuvant chemotherapy regimen with trastuzumab; HP, neoadjuvant chemotherapy regimen with trastuzumab plus pertuzumab; non-H/P, neoadjuvant chemotherapy regimen without trastuzumab and pertuzumab; PR, progestogen receptor; PSM, propensity score matching; SMD, standardized mean difference.
Subgroup analysis of the pCR rate in the non-H/P, H, and HP subpopulations before PSM and after PSM
We further analyzed the pCR rate of patients with different clinicopathological characteristics in the non-H/P, H, and HP subpopulations. The results from subgroup analysis revealed that in the non-H/P population, patients with age at diagnosis <50 years old (27.8% versus 13.8%, p = 0.021), cT stage T1-T2 (26% versus 14.5%, p = 0.009), clinical nodal positivity (22.7% versus 11.7%, p = 0.009), ER negativity (30.6% versus 16.8%, p = 0.016), PR negativity (32.6% versus 15.1%, p = 0.003), or Ki-67 > 30% (26.3% versus 16.1%, p = 0.048) in the nonanthracycline group had a higher pCR rate than those in the anthracycline group before PSM (Figure 4). Hence, a higher pCR rate was found in the nonanthracycline group than in the anthracycline group in the non-H/P population before PSM (22.7% versus 13.4%, p = 0.015) (Figure 4). In the H population, patients with clinical nodal negativity (36.6% versus 24.6%, p = 0.032), ER negativity (37.6% versus 24.6%, p = 0.004), or PR negativity (35% versus 25.5%, p = 0.018) in the nonanthracycline group had a favorable pCR rate compared with those in the anthracycline group, whereas the difference in the pCR rate was not significant between the two groups before PSM (28.2% versus 22.8%, p = 0.068) (Figure 5). In the HP population, patients with clinical T1-T2 stage disease (50% versus 34.9%, p = 0.009), clinical nodal positivity (47.5% versus 31.1%, p = 0.019), ER negativity (69% versus 40.5%, p = 0.005), or PR negativity (69.4% versus 40.6%, p = 0.001) in the anthracycline group had a higher pCR rate than those in the nonanthracycline group before PSM (Figure 6). Therefore, a lower pCR rate was found in the nonanthracycline group than in the anthracycline group in the HP population before PSM (47.4% versus 33.1%, p = 0.021) (Figure 6). However, after PSM, there was no difference in the pCR rates of patients with different characteristics between the nonanthracycline group and anthracycline group in the non-HP, H, and HP subpopulations (Figures 4–6).

Subgroup analysis of the pCR rate in the non-H/P population before PSM (upper) and after PSM (lower).
Subgroup analysis of the pCR rate in the H population before PSM (upper) and after PSM (lower).

Subgroup analysis of the pCR rate in the HP population before PSM (upper) and after PSM (lower).
Collectively, one interesting finding was that the pCR rates of the non-H/P and HP populations showed opposite trends in response to receiving anthracycline before PSM, which was caused by the inconsistent distribution of clinicopathological factors in the two treatment groups, since the difference disappeared after PSM.
Discussion
In this study, we found no significant difference in the proportion of patients achieving pCR between anthracycline-containing and anthracycline-free chemotherapy regimens in the neoadjuvant setting after controlling for confounding factors. In addition, we found that patients with a smaller tumor size, negative nodal metastasis and negative hormone receptor status were more likely to obtain pCR, which was consistent with most previous reports.21–23
With the advent of precision targeted therapies, it is controversial to exempt patients with HER2-positive breast cancer from anthracycline. Evidence in support of exempting anthracycline comes from the following studies. First, multiple randomized controlled studies and a meta-analysis from the Early Breast Cancer Pilot Collaboration group 6 have confirmed that NAC regimens with trastuzumab effectively improve the pCR rate from 32% to 65.2%,7,14 and adjuvant chemotherapy regimens with trastuzumab prolong DFS and OS in patients with HER2-positive breast cancer.4,8,10,12,24 Next, the JBCRG-16, APT, and BCIRG006 trials elucidated similar DFS and OS at 3, 5, or 10 years among patients with HER2-positive breast cancer after treatment with anthracycline-free versus anthracycline-containing trastuzumab regimens.10,25,26 Second, the results of BERENICE, TRAIN-2, and TRYPHAENA (with trastuzumab plus pertuzumab regimens) suggested that the pCR rate, 3-year event-free survival (EFS), or OS between the anthracycline-free group and the anthracycline-containing group were generally similar.9,11,13,27,28 Our findings are consistent with those of previous studies that have shown that for HER2-positive breast cancer, the pCR rate of the anthracycline-free regimen was similar to that of anthracycline-containing regimens in the H or HP population after PSM.
Nevertheless, the rate of neutropenic fever and cardiac events in patients was significantly higher in the anthracycline group than in the nonanthracycline group in the TRAIN-2 and BCIRG006 trials.13,26,28 Collectively, given a similar efficacy between the nonanthracycline regimen and anthracycline regimen, plus fewer acute toxic effects and lower risks of cardiotoxicity and neutropenic fever occurring in the nonanthracycline regimen, nonanthracycline treatment could be considered for implementation in the presence of single or dual HER2 blockade in patients with HER2-positive breast cancer.
In previous studies with small samples, wherein trastuzumab was not allowed in neoadjuvant treatment, 5-year OS and DFS were not significantly different between anthracycline-containing TAC and TC regimens in HER2-positive breast cancer patients.29,30 Similarly, an interesting observation in our study of HER2-positive patients without targeted therapy was that the pCR rate of patients with a nonanthracycline regimen was higher than that of the anthracycline group before PSM, and baseline characteristics were consistent between the two groups. However, in the subgroup analysis, patients with age at diagnosis >50 years old, clinical T stage with T1-T2, clinical nodal positive, hormone receptor status negative, or Ki-67 > 30% in the nonanthracycline group had a higher pCR rate than those in the anthracycline group before PSM, but 1:1 after matching, the pCR differences between the two groups disappeared. Therefore, we suspect that this interesting phenomenon is due to the clinicopathological characteristics of patients in the nonanthracycline group that determine their favorable pCR rate. Collectively, there is currently a lack of large randomized clinical evidence to support our conclusion; thus, the treatment decision of exempting anthracycline in the presence of taxane and the absence of targeted therapy should be made with caution in patients with HER2-positive breast cancer.
One point that needs to be stressed: In this study, 335 patients received the TA regimen, and 2 patients received the TX regimen, among which the breast pCR, axillary pCR, and total pCR rates of patients receiving the TA regimen were 14.0% (47/335), 42.7% (143/335), and 9.0% (30/335), respectively. Although TA and TX regiments are not the standard preferred NAC regiments, they are also effective treatment options for hormone receptor-positive and triple-negative breast cancer patients according to the latest Chinese Society of Clinical Oncology and National Comprehensive Cancer Network guidelines. In addition, at the National Cancer Center in Korea, a phase III randomized study showed that TX increased the pCR rate in primary tumors compared to AC (21% versus 10%, p = 0.024). 31 Another study of NAC for stage II and III breast cancer showed that the pCR rates with TA (91 patients) and TX (103 patients) were 15% and 21%, respectively. 32 The above studies showed that these nonstandard schemes are effective.
There are several limitations in our study. First, as a multicenter retrospective study, one of the inherent limitations is the difficulty in gathering comprehensive data on all participants, as 36% of patients had missing data on key factors and/or treatment, such as unknown pCR (n = 153), unknown chemotherapy scheme (n = 619), and changed chemotherapy scheme (n = 392). However, the inclusion of some patients without complete demographic and clinical information may lead to selection bias. Therefore, to ensure the validity of the research results, patients with incomplete information were excluded from this study. Second, our data did not provide follow-up information, leading to the inability to further evaluate the long-term benefits of anthracycline usage. Previous studies or meta-analyses on HER2-targeted therapy have shown that pCR after neoadjuvant therapy is associated with long-term clinical benefits (such as DFS, PFS, EFS and OS).15,33,34 Therefore, we chose pCR as our primary endpoint and used it as a surrogate indicator of long-term prognosis. Third, our study did not document some of the side effects that patients experienced after chemotherapy (such as acute toxic effects, cardiotoxicity, and neutropenic fever), and the chemotherapy dose of the patients in this study was not recorded. We expect that this information can be included in our upcoming prospective studies.
Conclusion
In summary, a similar efficacy between the nonanthracycline regimen and anthracycline regimen in the neoadjuvant setting was found in the presence of single or dual HER2 blockade in patients with HER2-positive breast cancer. Thus, our study further provides clinical evidence for omitting anthracycline treatment in HER2-positive breast cancer in the presence of trastuzumab and/or pertuzumab.
