Abstract
Background:
Fertility loss due to chemotherapy is a major concern for young patients with breast cancer (BC), influencing treatment decisions and quality of life. Despite established guidelines recommending fertility counseling, access to fertility preservation remains limited in Latin America.
Objectives:
This study evaluated attitudes and preferences regarding fertility-related concerns and chemotherapy decision-making among young Latin American women with early-stage BC.
Design:
A prospective cohort study was conducted at seven institutions in Brazil, Mexico, and Peru.
Methods:
Premenopausal women aged 18–40 years with stage I–III BC requiring (neo)adjuvant chemotherapy completed a fertility questionnaire before treatment, along with validated quality-of-life assessments (EORTC QLQ-C30 and EORTC QLQ-BR23). One year after chemotherapy initiation, the patients were reassessed for ovarian function status and quality of life. Factors associated with chemotherapy acceptance despite potential infertility risks were analyzed using univariate and multivariate Poisson regression models.
Results:
A total of 270 patients were included (mean age, 33.9 years). Prior to diagnosis, 41.5% of the women had children, and 31.1% expressed a desire for future childbearing. Among the participants, 8.5% were unaware of chemotherapy-induced infertility risks, 21.5% would decline chemotherapy if the infertility risk exceeded 25%, and 20.0% would accept treatment despite a 76%–100% infertility risk. In addition, 44.1% of patients required at least a 20% increase in survival probability to accept chemotherapy. In the multivariate analysis, married patients were significantly less likely to refuse chemotherapy (risk ratio: 0.88, 95% confidence interval: 0.82–0.94; p < 0.01). One year post-treatment, 73.1% of the patients experienced chemotherapy-induced amenorrhea.
Conclusion:
Fertility concerns significantly impact chemotherapy decision-making in young Latin American patients with BC. Limited fertility awareness, socioeconomic disparities, and restricted access to fertility preservation contribute to these challenges. Strengthening fertility counseling and improving access to preservation options are essential for supporting informed treatment decisions in this population.
Trial registration
(ClinicalTrials.gov): NCT02862990.
Keywords
Background
Breast cancer (BC) is the leading cause of cancer-related morbidity among young women in Latin America. 1 Younger age is associated with more aggressive disease and poorer outcomes in patients with BC, partly due to delayed diagnosis and the absence of reliable screening methods. 2 These malignancies are typically characterized by adverse pathological features, including high grade and lymphovascular invasion, and predominantly aggressive molecular subtypes, such as luminal B, HER2 positive, and triple negative. 2 In recent years, the indications for neoadjuvant or adjuvant chemotherapy in young patients with BC have been refined, especially in cases of hormone receptor-positive BC, with better risk stratification tools now available. 3 However, in Latin American countries, where most of the population depends on public healthcare systems, access to innovations such as genomic signatures, adjuvant CDK4/6 inhibitors, or olaparib is limited or even nonexistent. 4 Consequently, (neo)adjuvant chemotherapy is still recommended for most premenopausal patients with BC in Latin America. 5 In most Latin American countries, chemotherapy is fully reimbursed by public healthcare systems and private health insurance providers, as part of the standard cancer care coverage. 6 While chemotherapy significantly improves survival in selected early-stage BC, its long-term toxicities—including infertility or premature ovarian failure due to depletion of the ovarian reserve—are major concerns for young patients. Evidence suggests that fertility loss following chemotherapy contributes to increased psychological distress and diminished quality of life in this population.7,8
With the trend of delayed childbearing in Western countries, many young women diagnosed with BC have not yet completed their families.9,10 While advancements in reproductive medicine have provided fertility preservation options before cancer treatment, access to these services remains limited in Latin America. It is essential to recognize that Latin American healthcare systems are characterized by a heterogeneous mix of public and private providers, resulting in substantial coverage disparity. Most of the population relies on public healthcare systems, which provide no coverage for fertility preservation procedures such as oocyte or embryo cryopreservation, ovarian tissue freezing, or gonadotropin-releasing hormone agonists. These systemic barriers may impact patients’ perceptions and willingness to undergo chemotherapy, particularly when weighing potential side effects such as fertility loss. 11
Despite established guidelines for fertility management in BC care, studies have indicated that many patients feel inadequately informed about their infertility risks and available preservation options.10,12 –14 This highlights the need for improved communication and support within the oncological care teams.
In this context, our study aimed to evaluate the attitudes and preferences of young Latin American women with early-stage BC requiring (neo)adjuvant chemotherapy regarding fertility-related concerns and treatment decision-making.
Methods
A prospective cohort study was conducted at seven institutions in Brazil, Mexico, and Peru from March 2015 to September 2018. The study was approved by the Ethics Committees in all participating countries (Supplemental Material—Institutional Review Boards) in accordance with national regulations and was registered on ClinicalTrials.gov (NCT02862990).
We included premenopausal women aged 18–40 years at diagnosis with stage I–III BC and an indication for (neo)adjuvant chemotherapy. Eligible patients receiving care from their attending physicians at the participating study sites were invited to participate in the study. Written informed consent was obtained from all participants prior to study enrollment.
Before chemotherapy administration, patients completed a previously pilot-tested EORTC Fertility Questionnaire, which included eight questions related to pregnancy history and awareness of infertility risk. 15 In addition, they completed two validated quality-of-life questionnaires: the EORTC QLQ-C30 and EORTC QLQ-BR23. Sociodemographic characteristics, BC diagnosis, clinicopathological features, and treatment details were collected.
After 1 year of follow-up from the first chemotherapy administration, patients were assessed for disease recurrence and ovarian function status (amenorrhea or resumption of menses) and were asked to complete the EORTC QLQ-C30 and EORTC QLQ-BR23 questionnaires again.
The primary endpoint was to determine the proportion of patients who would decline (neo)adjuvant chemotherapy due to concerns regarding fertility loss. The secondary endpoints included assessing the accepted risk of chemotherapy-induced infertility and examining the relationships between patients’ attitudes and their clinical characteristics, family situation, and treatment details.
The sample size was calculated to determine the chemotherapy acceptance rate in the BC population. Based on an expected adjuvant treatment acceptance rate of 85%, with a significance level of 5% and a power of 80%, a sample size of 450 patients was required. To determine the treatment refusal rates, considering an expected rejection rate of 15% with a significance level of 5%, a minimum of 196 patients was needed. The larger sample size of 450 patients adequately covered both objectives. The association between explanatory variables and the outcome variable (acceptance of chemotherapy) was evaluated using univariate and multivariate Poisson regression models to estimate unadjusted and adjusted risk ratios (RRs) with 95% confidence intervals (CI). The multivariate model initially included all variables with p < 0.05 in the univariate analysis, and a backward selection method was used to remove non-significant variables to obtain the final model. All p-values were two-sided. Statistical analyses were performed using SAS version 9.4 (SAS Institute, Cary, NC, USA).
The reporting of this study conforms to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines (Supplemental Material—STROBE Checklist). 16
Results
Patients’ characteristics
A total of 270 patients were included in this study. Table 1 summarizes their characteristics. The mean age at diagnosis was 33.9 years (range: 22–40). Regarding education, 83.2% of the participants had completed at least a secondary degree. At baseline, 37.2% were married, and 41.5% had children before their cancer diagnoses.
Patients’ characteristics at baseline.
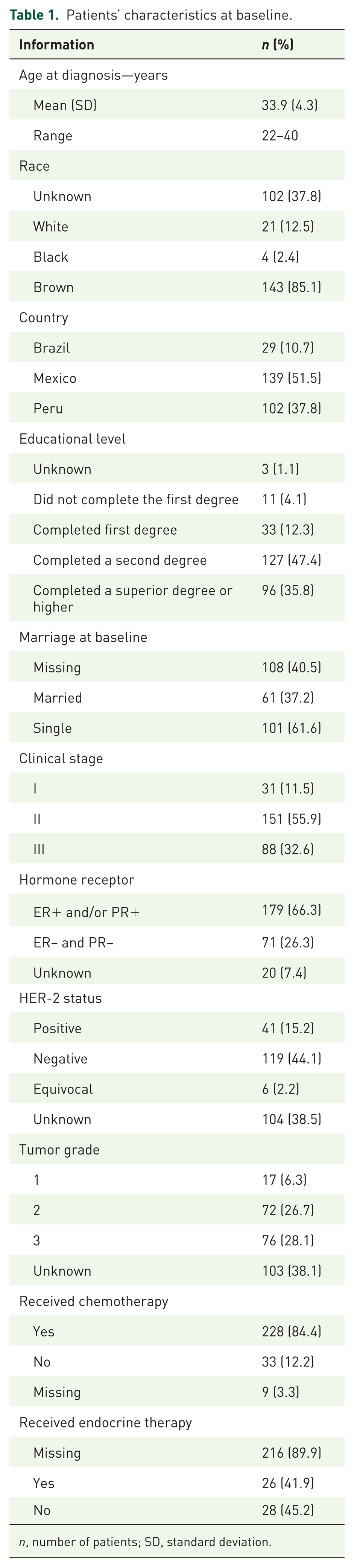
n, number of patients; SD, standard deviation.
Regarding the clinical stage, 11.5% had stage I, 55.9% had stage II, and 32.6% had stage III disease. Regarding tumor characteristics, 66.3% of patients had hormone receptor-positive (ER+ and/or PR+) tumors, 15.2% had HER2-positive tumors, and 28.1% had high-grade (G3) tumors.
A proportion (8.5%) of patients were unaware that chemotherapy could reduce fertility, and 15.2% stated that they would not accept chemotherapy if they knew it could reduce the chance of having children. Detailed responses to the fertility questionnaire are presented in Table 2.
Answers to the fertility questionnaire.

Patients could agree with more than one sentence.
Desire to have (more) children
Before BC diagnosis, 41.5% of patients had children. When asked about their future reproductive plans, 31.1% of patients expressed a desire to have children. Among those who did not want children, 20% made this decision before diagnosis.
Interestingly, the desire to have children was more common among women who had already had children before diagnosis (16.3%). Among those who were not mothers, 12% wished to have children.
Acceptance of chemotherapy
Influence of infertility risk
When evaluating fertility risks associated with chemotherapy, responses varied considerably: 20.0% of patients would accept chemotherapy even if there was a 76%–100% risk of infertility, whereas 21.5% would decline chemotherapy if the infertility risk exceeded 25%.
Expected cure benefit required for chemotherapy acceptance
Regarding the minimum survival benefit required for chemotherapy acceptance, responses varied: 7.4% would accept treatment for a 1%–5% increase in survival, and 3.0% would accept treatment for a 6%–20% increase. Most (44.1%) would only accept chemotherapy if it increased their survival probability by >20%.
Factors influencing chemotherapy acceptance
Table 3 presents the factors associated with the refusal of chemotherapy. In the univariate analysis, married patients were significantly less likely to refuse chemotherapy (RR: 0.88, 95% CI: 0.82–0.94; p < 0.01). This association remained significant in the multivariate analysis (Table 3). By contrast, prior motherhood, future childbearing desires, clinical stage, educational level, and geographic differences did not significantly affect chemotherapy acceptance (Table 3).
Factors associated with chemotherapy refusal: univariate and multivariate analysis.
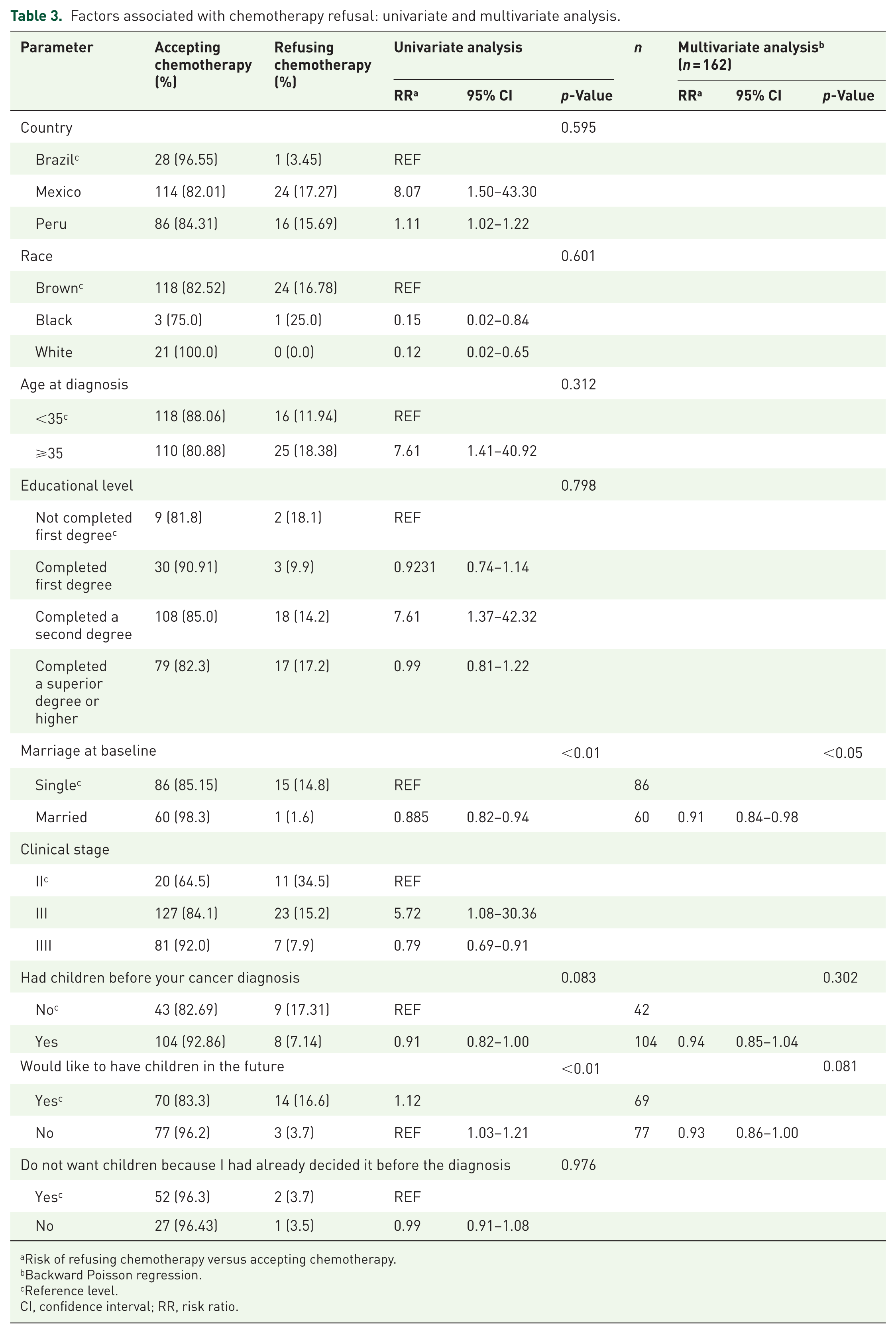
Risk of refusing chemotherapy versus accepting chemotherapy.
Backward Poisson regression.
Reference level.
CI, confidence interval; RR, risk ratio.
One-year follow-up and chemotherapy-induced amenorrhea
At the 1-year follow-up, 73.1% of the patients experienced chemotherapy-induced amenorrhea, and 23.9% resumed menstruation.
Due to missing data in the follow-up assessments for ovarian function and quality of life, the exact number of patients who completed both baseline and follow-up questionnaires could not be determined.
Discussion
This study reveals significant gaps in fertility awareness and substantial barriers to fertility preservation among young BC patients in Latin America. Our findings demonstrate that fertility concerns meaningfully impact chemotherapy decision-making, with notable variation in patients’ tolerance for infertility risks and required survival benefits.
A concerning 8.5% of patients were unaware that chemotherapy could reduce fertility, despite 31.1% expressing a desire to have future children. This knowledge gap persists despite international guidelines recommending that all reproductive-aged cancer patients receive information about fertility risks and preservation options.12,17,18 A recent survey of oncologists from Mexico, Bolivia, and Peru revealed that fertility preservation strategies are almost nonexistent in this region. 11
In several countries, such as Brazil, private health insurance plans are not legally mandated to cover in vitro fertilization and related fertility treatments. Consequently, fertility preservation services are not covered by health insurance plans, even among private providers. The substantial out-of-pocket costs of these interventions render them financially inaccessible to most Latin American patients. Socioeconomic disparities further compound these challenges, with studies highlighting stark financial disparities—only 3% of Mexican patients can afford fertility preservation compared to 50%–60% of women in wealthier countries.12,19,20 In addition, the limited availability of specialized reproductive medicine centers, particularly outside major metropolitan areas, creates significant geographic barriers to accessing fertility preservation services. This lack of coverage and accessibility contrasts sharply with that in developed countries, where many health insurance systems include fertility preservation as an integral component of comprehensive cancer care. 21
Our results demonstrated considerable variation in patients’ willingness to accept chemotherapy based on infertility risk. Notably, 21.5% would decline chemotherapy if the infertility risk exceeded 25%, while 20.0% would accept treatment despite a 76%–100% infertility risk. This finding aligns with previous research showing that fertility concerns significantly influence treatment decisions, although the proportion willing to decline treatment in our study appears substantial.8,20,22 Senkus et al. 15 found that 48% of patients would accept a risk of infertility of 76%–100%.
Regarding survival benefits, 44.1% of patients required at least a 20% increase in survival probability to accept chemotherapy, which is higher than that reported in other regions. Cultural differences likely play a role, as similar preferences vary across different populations. In a web-based survey in the United States, Partridge et al. 19 found that 32% of BC patients under 40 years required at least a 20% survival benefit from chemotherapy to accept treatment. Differences in understanding the benefits of chemotherapy, relapse risk perception, and treatment side effects may also contribute to these disparities.
In our analysis, marital status emerged as the only significant predictor of chemotherapy acceptance, with married patients being less likely to refuse treatment (RR: 0.88, 95% CI: 0.82–0.94; p < 0.01). This association remained significant in the multivariate analysis. Interestingly, prior motherhood, future childbearing desires, clinical stage, educational level, and geographic differences did not significantly affect chemotherapy acceptance, indicating that the decision-making process is complex and may be influenced by factors beyond those traditionally considered important. These may include psychosocial elements such as fear of infertility, cultural attitudes toward motherhood, socioeconomic barriers to fertility preservation, and the availability of social support systems. The finding that marital status was the strongest predictor underscores the importance of comprehensive counseling for all young BC patients, ensuring that fertility preservation options are discussed regardless of marital status or provider bias. 20 This suggests that social support systems and partnership dynamics may play crucial roles in treatment decision-making processes.
International efforts are increasingly focused on integrating fertility discussions into cancer care to ensure that young patients receive adequate information and timely specialist referrals. 23 Dedicated programs for young BC patients have been linked to increased fertility preservation referrals.24 –26 In addition, providing written materials and web-based decision aids has improved patient access to information on cancer treatment toxicities and fertility preservation strategies.27,28 However, such resources remain scarce in Latin America, emphasizing the need for culturally adapted educational materials in local languages to address this gap.1,11
This study had several important limitations. First, we assessed fertility attitudes at a single time point, which may not reflect how patients’ perspectives change throughout their cancer journey. Second, we experienced a high proportion of missing data in the 1-year follow-up assessments, particularly regarding ovarian function and quality of life. As a result, we were unable to accurately determine the rate of loss to follow-up, which limited our ability to fully assess long-term outcomes and may have introduced bias. In addition, the study did not include systematic follow-up for post-treatment pregnancies, which prevents us from reporting the proportion of patients who conceived after chemotherapy and menstrual recovery. Third, selection bias may have occurred because patients willing to participate in fertility-focused research may have had greater reproductive concerns than those who declined. Fourth, our actual sample size of 270 patients was smaller than the calculated requirement of 450 patients, which may have reduced the precision of our estimates and the statistical power to detect associations. Finally, our study was conducted at specialized cancer centers in only three countries, which likely overrepresents patients with better healthcare access, higher socioeconomic status, and greater exposure to fertility counseling compared to the majority of Latin American women who receive cancer care in community hospitals or public healthcare systems with limited resources and fertility expertise.
In conclusion, this study highlights the significant influence of fertility concerns on chemotherapy decision-making among young BC patients in Latin America. While 21.5% of patients would decline chemotherapy if the infertility risk exceeded 25%, and 44.1% required at least a 20% survival benefit to accept treatment, marital status emerged as the only significant predictor of treatment acceptance. In addition, 8.5% of patients were unaware that chemotherapy could affect fertility, despite 31.1% expressing a desire for future children, revealing critical knowledge gaps. Systemic barriers, including lack of insurance coverage, high out-of-pocket costs, and limited access to specialized centers, render fertility preservation financially inaccessible for most Latin American patients, despite international guidelines recommending fertility discussions for all reproductive-aged cancer patients. To address these gaps, healthcare providers must prioritize comprehensive fertility counseling, regardless of marital status, promote equitable access to preservation strategies, and develop culturally tailored educational resources. Strengthening these efforts will empower young BC patients to make informed, personalized treatment decisions that align with their oncologic and reproductive goals.
Supplemental Material
sj-pdf-1-tam-10.1177_17588359251378946 – Supplemental material for Factors associated with accepting chemotherapy despite the risk of fertility loss in Latin American breast cancer patients—LACOG 0414 study
Supplemental material, sj-pdf-1-tam-10.1177_17588359251378946 for Factors associated with accepting chemotherapy despite the risk of fertility loss in Latin American breast cancer patients—LACOG 0414 study by Gustavo Werutsky, Cynthia Villarreal-Garza, Henry L. Gomez, Saúl Campos-Gómez, Rosa Ortiz Reyes, Pedro E. R. Liedke, Tomás Reinert, Vanessa Dybal, Jeovany Martinez-Mesa, Paulo Ricardo Nunes Filho, Rafaela Gomes de Jesus, Facundo Zaffaroni, Vitória Silva Garcia, Mariana Fauth Seibel, Pablo Barrios, Matheus Soares Rocha and Carlos H. Barrios in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-2-tam-10.1177_17588359251378946 – Supplemental material for Factors associated with accepting chemotherapy despite the risk of fertility loss in Latin American breast cancer patients—LACOG 0414 study
Supplemental material, sj-pdf-2-tam-10.1177_17588359251378946 for Factors associated with accepting chemotherapy despite the risk of fertility loss in Latin American breast cancer patients—LACOG 0414 study by Gustavo Werutsky, Cynthia Villarreal-Garza, Henry L. Gomez, Saúl Campos-Gómez, Rosa Ortiz Reyes, Pedro E. R. Liedke, Tomás Reinert, Vanessa Dybal, Jeovany Martinez-Mesa, Paulo Ricardo Nunes Filho, Rafaela Gomes de Jesus, Facundo Zaffaroni, Vitória Silva Garcia, Mariana Fauth Seibel, Pablo Barrios, Matheus Soares Rocha and Carlos H. Barrios in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We would like to thank the patients, research team, and investigators for their dedication to this study. We acknowledge the SAS Institute Inc. for providing license-free access to SAS® statistical products.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
