Abstract
Background:
Soft tissue sarcomas (STS) are rare, heterogeneous tumors with poor prognosis, often characterized by high recurrence rates and limited response to conventional chemotherapy in advanced stages. Vascular-targeting agents (VTA) have shown promising efficacy for STS in numerous clinical trials; however, the optimal agent and combination strategies remain undetermined.
Objectives:
This work aims to compare the clinical efficacy and adverse events of different VTA-containing regimens for patients with STS.
Design:
This study is a systematic review and network meta-analysis.
Data sources and methods:
We searched PubMed, Embase, and the Cochrane Library for eligible randomized clinical trials. All trials with VTA as monotherapy or VTA-included regimens in the experimental or control groups were selected, except for some specific histological subtypes. Pooled hazard ratios (HRs) for survival data and odds ratios for objective response rate (ORR), disease control rate (DCR), and treatment-related adverse events with credible intervals were generated using R software. A Bayesian random-effects model was applied to rank different treatments based on the surface under the cumulative ranking curve (SUCRA) values.
Results:
Seventeen articles covering 15 studies were included. Pairwise comparisons demonstrated prolonged progression-free survival (PFS) for tyrosine kinase inhibitor (TKI) versus placebo (HR 0.50, 95% confidence interval (CI) 0.32–0.83) and prolonged overall survival (OS) for monoclonal antibody plus chemotherapy versus placebo (HR 0.42, 95% CI 0.19–0.89). Vascular-disrupting agents (VDA) plus chemotherapy were ranked highest for OS (87.05%) and ORR (89.66%). TKI plus chemotherapy and TKI plus immunotherapy had higher SUCRA values for PFS (68.21%) and DCR (82.29%). TKI-based regimens were associated with higher incidences of hypertension, diarrhea, and elevated transaminase levels, whereas chemotherapy-based strategies resulted in higher incidences of hematological toxicity and constipation.
Conclusion:
VTA-containing regimens showed promising activity and tolerability in patients with advanced STS. Combination regimens with TKI showed better efficacy for PFS and DCR. Novel VDA combined with chemotherapy showed potential to prolong OS and improve ORR.
Trial registration:
PROSPERO website (registration number: CRD42024588134).
Introduction
Soft tissue sarcomas (STS) are rare, heterogeneous tumors originating from mesenchymal precursors. They are characterized by local invasiveness, infiltrative growth, and high metastatic potential. Although the incidence rate is low (1% in adults with cancer), 11 major categories and over 100 pathological types exist including leiomyosarcoma, liposarcoma, angiosarcoma, synovial sarcoma, fibrosarcoma, and undifferentiated pleomorphic sarcoma. This diversity presents significant challenges in determining optimal therapeutic strategies in clinical practice. 1
Traditional therapeutic options for advanced STS have relied on standard chemotherapy, including anthracyclines, ifosfamide, cyclophosphamide, gemcitabine, dacarbazine, eribulin, trabectedin, platinum, and others. 2 However, the efficacy of chemotherapy regimens is limited, with a 2-year survival rate of 20%–30%.3–6 In addition, elderly patients with comorbidities are poorly tolerant to chemotherapy. These limitations have highlighted the urgent need for alternative therapeutic options.
Numerous studies have explored the efficacy and safety of anti-angiogenic (AA) tyrosine kinase inhibitors (TKIs) as monotherapy in patients with advanced STS. As the first clinical trial to evaluate the efficacy of TKI in STS, the PALETTE study demonstrated a progression-free survival (PFS) prolongation from 1.6 months with placebo to 4.6 months with pazopanib (hazard ratio (HR) 0.31, 95% confidence interval (CI) 0.24–0.40, p < 0.0001). 7 Based on these results, pazopanib was approved by the Food and Drug Administration (FDA) for patients with STS except gastrointestinal stromal tumors (GIST) and adipocytic sarcomas. Other TKIs also showed favorable trends for improving PFS in advanced STS when compared with placebo.8–11
Other studies have explored combinations of vascular-targeting agents (VTA), including TKI, large-molecule monoclonal antibodies, and novel vascular-disrupting agents (VDA). A single-arm phase II clinical study of anlotinib with epirubicin for first-line treatment of advanced STS demonstrated an objective response rate (ORR) of 13.3%, a disease control rate (DCR) of 80%, and a median PFS of 11.5 months. 12 However, randomized clinical trials (RCTs) comparing chemotherapy with or without VTA have yielded inconsistent results. Some studies reported improved PFS with the addition of the VTA, 13 whereas others achieved very minor differences.14–16 The combination of AA TKI with immunotherapy also showed preliminary efficacy against metastatic STS. 17
Given the multiple treatment algorithms for VTA, there is no existing consensus on the selection of specific agents and optimal combination strategies. Therefore, we conducted this systematic review and network meta-analysis to explore the best therapeutic regimens and shed light on the issues discussed above.
Materials and methods
The study protocol for this meta-analysis was registered on the PROSPERO website (https://www.crd.york.ac.uk/PROSPERO/) with registration number CRD42024588134. During the entire process of this research, we followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension statement (PRISMA-NMA). 18
Selection and exclusion criteria
On July 20, 2024, we searched PubMed, Embase, and the Cochrane Library using the search terms “soft tissue sarcoma,” “advanced,” and “vascular-targeting agents,” including relevant synonyms and MeSH terms. Additionally, we manually screened the relevant abstracts reported at conferences of the American Society of Clinical Oncology, European Society of Medical Oncology, San Antonio Breast Cancer Symposium, and Chinese Society of Clinical Oncology over the past 5 years. The detailed literature search process is presented in Supplemental Table 1.
Two researchers (Y.W. and H.X.) independently conducted the literature screening, focusing on phase Ib–III RCTs on STS in which VTAs were used in either the experimental or control arms. Eligible studies were required to report at least one of the following endpoints: PFS, overall survival (OS), ORR, or treatment-related adverse events (TRAEs). The main exclusion criteria were as follows: (1) non-randomized, single-arm, or retrospective studies; (2) exploratory analyses or subgroup reports from the included articles; (3) studies involving early-stage STS, specific subtypes of STS, or other tumor types; and (4) comparisons conducted between regimens of the same category. All articles were initially screened based on their titles and abstracts, after which a full-text review was conducted. Discrepancies between the two researchers were resolved through discussion.
Data collection and quality assessment
All baseline characteristics of the included RCTs, including study name, publication year, study phase, and treatment strategies in the interventional and control arms, were collected and documented in a standard data extraction form. The primary outcome was PFS, which was the time interval between randomization and first recurrence, progression, or death. Secondary endpoints included OS, ORR, DCR, and TRAEs. OS was defined as the time interval between randomization and death from any cause. ORR was defined as the proportion of patients with a complete response (CR) or partial response (PR) to treatment according to the Response Evaluation Criteria in Solid Tumors. DCR was defined as the proportion of patients with the best response of CR, PR, or stable disease. Time-to-event outcomes (PFS and OS) were recorded as HRs and 95% CIs. Dichotomous outcomes (ORR, DCR, and TRAEs) were extracted as the exact number of events and total number of patients in each treatment arm.
The revised Cochrane risk-of-bias tool (RoB 2) was used to assess the quality of included RCTs. 19 Five domains including the randomization process, deviations from intended interventions, missing outcome data, measurement of the outcome, and selection of the reported result were considered. Each study was independently rated by two authors as “high risk,” “low risk,” or “some concerns.” Discrepancies were resolved through discussion to reach consensus.
Statistical analysis
This study was based on a Bayesian random-effects model, utilizing the “gemtc” and “rjags” packages in R software (version 4.3.3, R Development Core Team, Vienna, Austria). HRs and odds ratios (ORs) with 95% credible intervals (CrIs) were generated to summarize time-to-event and binary outcomes. Treatment rankings were estimated using the surface under the cumulative ranking curve (SUCRA), which reflects the probability of each intervention being the most effective (100%) or least effective (0%). Network plots were generated to visualize the structure of treatment comparisons. The Markov Chain Monte Carlo methods were employed for the network analysis, with 10,000 burn-in iterations and 50,000 sampling iterations. Convergence was assessed using trace plots, density plots, and Brooks-Gelman-Rubin diagnostics. Comprehensive results were presented using league tables and forest plots. Statistical significance was set at p < 0.05. Heterogeneity among the included studies was quantified using the I2 statistic and Cochran’s Q test, with I2 >50% or a p < 0.10 indicating significant heterogeneity.
Results
Study selection and baseline characteristics
A total of 4378 articles were initially retrieved. After removing 1240 duplicates, we screened the remaining 3138 records. Of these, 2795 articles were excluded based on titles and abstracts, and an additional 326 articles were excluded after full-text review. Finally, 17 articles reporting on 15 studies were included in the final analysis. The study selection process is shown in Figure 1. Study characteristics (including study name, publication time, study phase, and interventions) and predefined outcomes (including PFS, OS, ORR, and DCR) are documented in Table 1. A quality assessment of the included RCTs is depicted in Figure 2.

The flowchart of the detailed literature screening process.
The baseline characteristics and predefined outcomes of included RCTs.

The HR and 95% CI of the study conducted by Hensley et al. were reported for the control group (gemcitabine plus docetaxel) over the interventional group (gemcitabine, docetaxel, and bevacizumab) in the original publication.
The results of study REGOSARC were reported in three articles, 1.1–1.4 were the data of first publication in 2016, representing the subgroups with different pathological subtypes, which were liposarcoma, leiomyosarcoma, synovial sarcoma, and other sarcomas, respectively. 2 and 3 were the updated report in 2018 and 2020, revealing the outcomes of patients with non-adipocytic soft tissue sarcoma, included from 2013 to 2014 and 2015 to 2017, respectively. Only the data of 1.1, 2, and 3 were used for our analysis.
The ANNOUNCE 2 study was divided into two subgroups, which enrolled Olaratumab-naïve and Olaratumab-pretreated patients.
CI, confidence interval; DCR, disease control rate; HR, hazard ratio; m, months; NA, not available; No., number; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; RCT, randomized clinical trials; w, weeks.

Quality assessment for the included 15 RCTs (risk of bias summary).
For further analysis, all interventions were categorized into eight groups: placebo; chemotherapy; chemotherapy plus placebo; AA TKI; chemotherapy plus TKI; chemotherapy plus AA monoclonal antibody; immunotherapy plus TKI; and chemotherapy plus VDA. The specific agents were as follows: (1) Chemotherapy: gemcitabine, docetaxel, cisplatin, paclitaxel, doxorubicin, methotrexate, vinblastine, actinomycin-D, ifosfamide, and dacarbazine. (2) AA TKI: pazopanib, imatinib, regorafenib, anlotinib, cediranib, nintedanib, and cabozantinib. (3) AA monoclonal antibody: bevacizumab and olaratumab. (4) Immunotherapy: nivolumab and ipilimumab. (5) VDA: ombrabulin.
Survival outcomes
A total of 15 PFS comparisons from 13 RCTs and 13 OS comparisons from 12 RCTs were included in the analysis. The REGOSARC study was divided into adipocytic and non-adipocytic STS subgroups, while the ANNOUNCE 2 study was divided into Olaratumab-naïve and Olaratumab-pretreated arms. Seven types of interventions were included in the survival outcome analysis (Figure 3(a) and (b)). The results of pairwise comparisons are shown in Table 2 (lower-left triangle for PFS and upper-right triangle for OS). Considering PFS, only TKI monotherapy showed significantly better efficacy than placebo (HR 0.50, 95% CrI 0.32–0.83). For OS, a significant improvement was observed in chemotherapy plus monoclonal antibody versus placebo (HR 0.42; 95% CrI 0.19–0.89).

Network plots of PFS (a), OS (b), ORR (c), and DCR (d). Every node represents one kind of intervention, and the numbers marked in lines represent the quantity of direct comparisons.
Pairwise comparisons among seven interventions for PFS and OS (HR, 95% CrI).

Every square has the corresponding horizontal and vertical interventions. On the lower triangle, the figures lower than 1 could reveal the preferential tendency for horizontal regimens, while on the upper triangle, figures lower than 1 predicted preferential tendency for longitudinal regimens. Statistically significant differences were marked in bold.
CrI, credible intervals; HR, hazard ratio; MA, monoclonal antibody; OS, overall survival; PFS, progression-free survival; TKI, tyrosine kinase inhibitor; VDA, vascular-disrupting agents.
To identify the most effective treatment strategies for advanced STS, SUCRA values were calculated to rank interventions based on their impact on PFS and OS (Table 3). Regarding PFS, AA TKI plus chemotherapy ranked highest with a SUCRA value of 68.21%, followed by monoclonal antibody plus chemotherapy (66.54%), VDA plus chemotherapy (61.63%), and TKI monotherapy (61.55%). Chemotherapy and/or placebo ranked lower, with SUCRA values ranging from 10.07% to 44.34%. For OS, the favorable effect of VDA or AA monoclonal antibody plus chemotherapy was particularly evident (81.29%–87.05%). Chemotherapy with or without placebo revealed a tendency toward prolonged OS (71.45% and 41.66%). AA TKI with or without chemotherapy and placebo tended to perform poorly (6.63%–33.49%).
SUCRA values of different interventions for PFS, OS, and ORR.
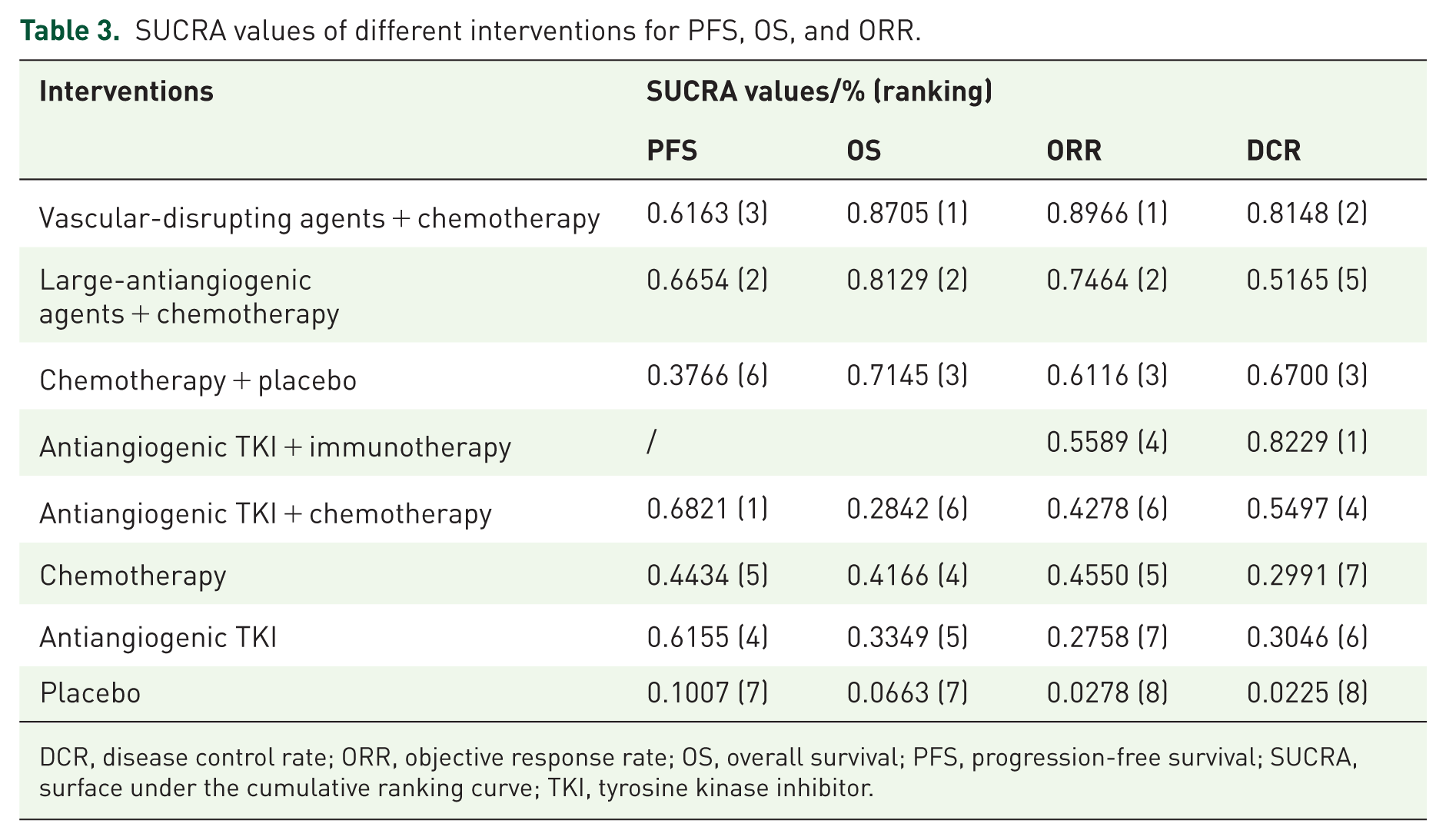
DCR, disease control rate; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; SUCRA, surface under the cumulative ranking curve; TKI, tyrosine kinase inhibitor.
Therapeutic efficacy
Eight interventions were included in the network meta-analysis, involving 16 direct comparisons for ORR and 14 for DCR (Figure 3(c) and (d)). Compared with placebo, monoclonal antibody plus chemotherapy and VDA plus chemotherapy significantly improved ORR (OR 0.07, 95% CrI 0.01–0.67; OR 0.02, 95% CrI 0.00–0.63, Table 4). TKI combined with chemotherapy or immunotherapy also significantly improved DCR compared with placebo (OR 0.17, 95% CrI 0.04–0.80; OR 0.07, 95% CrI 0.01–0.44)). TKI monotherapy was also superior to placebo in terms of DCR (RR 3.36, 95% CrI 1.50–6.66).
Pairwise comparisons among eight interventions for ORR and DCR (OR, 95% CrI).

Every square has the corresponding horizontal and vertical interventions. On the left lower triangle, the figures lower than 1 could reveal the preferential tendency for horizontal regimens, while on the right upper triangle, figures lower than 1 predicted preferential tendency for longitudinal regimens. Statistically significant differences were marked in bold.
Chemo, chemotherapy; CrI, credible intervals; DCR, disease control rate; Immuno, immunotherapy; MA, monoclonal antibody; TKI, tyrosine kinase inhibitor; VDA, vascular-disrupting agents.
The SUCRA ranking of interventions for ORR was VDA plus chemotherapy (89.66%), monoclonal antibody plus chemotherapy (74.64%), chemotherapy plus placebo (61.16%), TKI plus immunotherapy (55.89%), chemotherapy (45.50%), TKI plus chemotherapy (42.78%), TKI monotherapy (27.58%), and placebo (2.78%; Table 3). For DCR, TKI plus immunotherapy, VDA plus chemotherapy, and chemotherapy plus placebo were the top three regimens (67.00%–82.29%). TKI and monoclonal antibodies plus chemotherapy followed, with SUCRA values of 54.97% and 51.65%, respectively. Chemotherapy alone, TKI, and placebo ranked lowest (2.25%–30.46%).
Safety
The nine most commonly reported TRAEs in advanced STS were hypertension, diarrhea, increased transaminase levels, anemia, neutropenia, constipation, fatigue, nausea, and decreased appetite. Given the low incidence of ⩾3 grade safety events, only all-grade TRAEs were compared across subgroups. Due to the limited number of trials, no significant disparity between the different interventions was observed. Compared with chemotherapy, the TKI-included regimens tended to exhibit a higher risk of hypertension, diarrhea, and elevated transaminase levels (Supplemental Figures 1–3). In contrast, chemotherapy was more likely to cause hematological toxicity, including neutropenia (OR 3.9; 95% CrI 0.41–38.0) and anemia (OR 3.0; 95% CrI 0.59–16.0), than TKI (Supplemental Figures 4 and 5). Chemotherapy-based regimens tended to increase the risk of constipation (Supplemental Figure 6). No notable differences in fatigue, nausea, or decreased appetite were found among all regimens (Supplemental Figures 7–9).
Discussion
Advanced STS is a highly aggressive malignancy with poor prognosis and limited treatment options. Management of STS remains challenging due to its heterogeneity and resistance to conventional therapies. Tumor angiogenesis plays a central role in the growth, local invasion, and metastasis of advanced tumors. 29 However, no preferred VTA or combination strategy is recommended by clinical guidelines. And the efficacy of these agents, alone or in combination with chemotherapy or immunotherapy, remains unclear. Although multiple single-arm studies and RCTs have explored these strategies, no consensus has been established.
A major clinical challenge in STS management is selecting effective treatment options after progression on first-line anthracycline-based chemotherapy. The PAPAGEMO study investigated whether TKI combined with chemotherapy agents improved efficacy compared to TKI monotherapy. 27 This study enrolled patients with STS who had received at least one prior line of therapy and randomized them to either the pazopanib plus gemcitabine or pazopanib groups. The combination group demonstrated significantly improved PFS and DCR, although no significant differences in OS or ORR were found. Notably, this benefit was most pronounced in the liposarcoma subgroup, of which PFS increased from 1.5 to 8.6 months, OS from 5.4 to 25.4 months, and the risk of death was reduced by 61%. Another single-arm, phase II clinical trial evaluated the activity of gemcitabine plus pazopanib followed by pazopanib maintenance as a second-line treatment for advanced leiomyosarcoma, 30 reporting a median PFS of 6.5 months OS of 22.4 months. The main TRAEs during combination therapy were myelosuppression and hepatic toxicity, both of which were reversible and significantly decreased during the TKI maintenance phase, supporting the feasibility of this treatment regimen. In our study, TKI plus chemotherapy ranked highest for PFS but relatively lower for OS and ORR. Given the relatively low response rate and limited long-term survival benefit indicated by SUCRA values, caution should be exercised when considering this strategy in clinical practice.
Blay et al. 13 conducted a randomized phase III study evaluating the efficacy of a VDA in patients with advanced STS. This study demonstrated that ombrabulin combined with cisplatin statistically prolonged PFS compared with cisplatin alone (HR 0.76; 95% CI 0.59–0.98) in patients who had progressed after at least two prior lines of therapy, including anthracyclines and ifosfamide. Additionally, a trend toward improved OS was observed (HR 0.85; 95% CI 0.67–1.09), contributing to high SUCRA rankings of OS and ORR. Moreover, the combination of TKIs and immunotherapy demonstrated remarkable advantages for DCR. Multiple studies confirmed that AA agents could enhance the effects of immunotherapy by modulating the tumor immune microenvironment. 31 However, only one related study has evaluated the combination of VDAs and immunotherapy, and the absolute gains for PFS and OS were limited. Furthermore, efficacy comparisons with other regimens were mainly based on indirect evidence. Therefore, the results should be interpreted with caution.
Numerous studies explored the therapeutic effect of large-molecule monoclonal antibodies for STS treatment. A phase II non-RCT investigated the activity of bevacizumab in combination with doxorubicin. 32 The trial reported an ORR of only 12% among 17 patients, which was a lack of improvement over doxorubicin. Therefore, the study was terminated prematurely. Other studies comparing the efficacy of conventional chemotherapy with or without bevacizumab found no conspicuous discrepancy in both PFS and OS.14,20,33 Only the BERNIE study detected an 18% increase in ORR (from 36% to 54%) with the addition of bevacizumab. 20 These findings indicated that bevacizumab provides limited survival benefits when compared with chemotherapy for metastatic STS. Subsequently, a novel platelet-derived growth factor receptor α (PDGFRα) inhibitor, olaratumab, which was developed by Eli Lilly and Company, started to be used in clinical trials. The preliminary phase I/II clinical trials found a clear trend for improving PFS with the addition of olaratumab to chemotherapy, with a 31%–33% reduction in the risk of disease progression.15,16 Tap et al. 16 demonstrated that olaratumab plus doxorubicin significantly improved OS in patients with advanced STS (26.5 vs 14.7 months; HR 0.46; 95% CI 0.30–0.71). Therefore, the U.S. FDA approved olaratumab combined with doxorubicin as first-line therapy for patients with advanced STS. 34 However, the subsequent phase III ANNOUNCE trial failed to find prolonged PFS or OS in STS patients treated with doxorubicin plus olaratumab. 25 Based on these results, Olaratumab was removed from the global market, which is one of the largest medical reversals in sarcoma therapy history. Although combining monoclonal antibodies with chemotherapy ranked top 2 for PFS, OS, and ORR in SUCRA values, the relevant studies have been overturned by large-scale phase III RCTs for the inefficacy of investigational drugs. Therefore, this class of agents is not a preferable clinical option.
The incidence of hypertension, diarrhea, and elevated transaminase levels was relatively higher in the TKI-included regimens. This finding is consistent with a previous meta-analysis of AA agents for advanced STS, 35 which only compared the efficacy and toxicity between regimens with and without AA agents, without performing a network across different VTA categories or combination strategies. In particular, only all-grade TRAEs were analyzed in our study. The incidence of grade ⩾3 TRAEs was too low to allow indirect comparisons, indicating generally mild and well-tolerated adverse effects of all included regimens.
Our study had some limitations. First, all the statistical analyses were based on previously published RCTs, which differed in inclusion criteria and reported outcomes, leading to wide CrIs in pairwise comparisons and forest plots. Second, due to the limited number of eligible studies, subgroup analyses based on treatment lines and pathological types could not be conducted. Moreover, several interventions were represented by single studies, limiting the robustness of indirect comparisons. To reduce heterogeneity as much as possible, we excluded certain histological subtypes, including GISTs, desmoid tumors, and angiosarcomas. GISTs are characterized by specific KIT/PDGFR mutations and follow a distinct treatment algorithm. 36 Desmoid tumors have a significantly better prognosis, rarely metastasis, and do not impact OS. 37 Angiosarcomas also show distinct biological behaviors and respond differently to AA agents due to unique mechanisms of action. 38 Finally, SUCRA values could not reflect the absolute discrepancy among the regimens, which was reported as the ranking sequence and should be interpreted with caution. However, to our knowledge, this is the first network analysis to comprehensively evaluate the efficacy and tolerability of all the VTA-based treatment strategies. Despite these limitations, the generally low I2 values observed indicate acceptable heterogeneity and support the credibility of our findings.
Conclusion
Altogether, our study combines current RCTs and provides updated treatment insights for patients with advanced STS. All VTA-containing regimes demonstrated improved efficacy over placebo for either PFS or OS in patients with advanced STS. AA TKI and VDAs plus chemotherapy were associated with improved efficacy and survival. TKI combined with immunotherapy showed promising efficacy in improving ORR. TKI-containing regimens were more likely to cause hypertension and diarrhea, whereas chemotherapy-containing strategies resulted in higher incidences of anemia, fatigue, and constipation. Overall, these findings indicated that further research and clinical trials involving VTA and chemotherapy for patients with STS are warranted.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251378934 – Supplemental material for Efficacy and safety of vascular-targeting agents in advanced soft tissue sarcoma: a systematic review and network meta-analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359251378934 for Efficacy and safety of vascular-targeting agents in advanced soft tissue sarcoma: a systematic review and network meta-analysis by Yan Wang, Hangcheng Xu, Qiang Sa, Yiran Zhou, Hong Cheng, Renchi Gao, Binghe Xu and Jiayu Wang in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
