Abstract
Background:
Current biomarkers for predicting pathological response to neoadjuvant chemoimmunotherapy in esophageal squamous cell carcinoma (ESCC) remain limited.
Objective:
This study investigates the potential of the pan-immune-inflammation value (PIV) as a biomarker for predicting pathological response after neoadjuvant chemoradiotherapy combined with anti-programmed death protein-1 (PD-1) therapy in ESCC.
Design:
A multicenter, real-world, retrospective clinical study conducted across five centers in Southern China (January 2021–March 2024).
Methods:
A multicenter retrospective study included 334 patients with ESCC, divided into pathological complete response (pCR) and non-pathological complete response (non-pCR) groups. Clinical and laboratory data were analyzed using univariate and multivariate logistic regression to evaluate the relationship between post-treatment PIV and pathological response. The threshold effect of PIV was explored using restricted cubic spline analysis.
Results:
Subgroup analysis showed no significant interactions across clinical subgroups. Post-treatment PIV was positively associated with non-pCR risk (odds ratio = 1.002; 95% confidence interval: 1.001–1.003, p < 0.005). A positive association was observed in the high-PIV stratum (⩾280), where elevated PIV levels significantly correlated with increased non-pCR risk. Receiver operating characteristic analysis showed an area under the curve of 0.86 for predicting non-pCR, with a sensitivity of 86.6% and specificity of 72% at an optimal cutoff of 438.04. The high-PIV group exhibited inferior survival outcomes with significantly increased mortality risk.
Conclusion:
Post-treatment PIV shows a nonlinear relationship with pathological response in patients receiving neoadjuvant chemoradiotherapy combined with anti-PD-1 therapy and may serve as a predictive biomarker.
Keywords
Background
Esophageal cancer is the sixth leading cause of cancer-related deaths and the seventh most common cancer worldwide. 1 Despite the development of multimodal treatments, including surgery, radiotherapy, chemotherapy, and chemoradiotherapy, patient outcomes remain poor.2,3 The limited effectiveness of these traditional therapies has led to a growing interest in innovative strategies, such as immune checkpoint inhibitors (ICIs). In particular, inhibitors targeting programmed death protein-1 (PD-1) and programmed death ligand-1 (PD-L1) have shown promising therapeutic effects across several malignancies, including esophageal cancer.4,5 As a result, understanding both systemic and tumor-specific immune status is crucial, underscoring the need for novel biomarkers to improve prognostic stratification and predict treatment outcomes in esophageal cancer.6,7 Blood-based biomarkers derived from routine complete blood counts are readily available, cost-effective, and reflect the host’s immune status. In recent years, several inflammatory markers, such as the systemic immune-inflammation index (SII) and the neutrophil-to-lymphocyte ratio (NLR), have been widely used to predict neoadjuvant treatment outcomes in esophageal squamous cell carcinoma (ESCC), demonstrating strong associations with both pathological response and survival.8,9 Unlike these individual markers, the pan-immune-inflammation value (PIV) is an integrated marker that combines neutrophil, platelet, monocyte, and lymphocyte counts, providing a comprehensive reflection of the systemic immune and inflammatory status. 10 Previous studies have validated PIV’s prognostic value in various cancers, including esophageal and colorectal cancers, where it is an independent predictor of both pathological response and survival.11,12 However, the predictive role of post-treatment PIV in esophageal cancer treated with neoadjuvant chemoradiotherapy combined with anti-PD-1 therapy remains unexplored. This study seeks to fill this gap by evaluating PIV’s potential to predict pathological response in ESCC following neoadjuvant chemoradiotherapy combined with anti-PD-1 therapy, providing a scientific basis for personalized treatment strategies.
Materials and methods
Data sources and study population
This multicenter retrospective study collected data from January 2021 to March 2024 from five centers, including Fujian Medical University Union Hospital, Quanzhou First Hospital Affiliated to Fujian Medical University, Ganzhou Cancer Hospital, Quanzhou Guangqian Hospital, and Anxi County Hospital. All patients underwent comprehensive diagnostic evaluations before treatment, including esophagogastroduodenoscopy, chest and abdominal CT scans, upper gastrointestinal angiography, neck ultrasound, and PET-CT when necessary. Pre- and post-treatment laboratory tests, such as complete blood count, full biochemical panel, and coagulation tests, were also performed. The inclusion criteria were as follows: (1) histologically confirmed ESCC; (2) clinical staging from stage II to IVA; (3) undergoing radical esophagectomy; (4) receiving neoadjuvant chemotherapy combined with immunotherapy before surgery; and (5) complete medical records and follow-up data. The exclusion criteria were as follows: (1) diagnosed with adenocarcinoma or other types of esophageal cancer; (2) neoadjuvant treatments other than chemotherapy and immunotherapy; (3) undergoing emergency surgery due to gastrointestinal bleeding or perforation; (4) concurrent other malignancies; and (5) diagnosed with autoimmune diseases or a history of steroid use for more than 1 month. The study design and patient selection process are illustrated in Figure 1.

The flow chart of the study.
The reporting of this study conforms to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statement. The completed STROBE checklist is provided in Supplemental File 1.
Treatment
The included patients received at least two cycles of treatment, consisting of paclitaxel-based chemotherapy combined with a PD-1 inhibitor. The PD-1 monoclonal antibodies used in this study included nivolumab, pembrolizumab, and others (including sintilimab, tislelizumab, and toripalimab). Surgical resection was conducted after ⩾2 treatment cycles, following response evaluation. All patients underwent McKeown minimally invasive esophagectomy, which involved a two-field lymphadenectomy and stomach-based digestive tract reconstruction. In cases with cervical lymph node involvement, a three-field lymphadenectomy was performed.
Definition
In this study, postoperative pathological response, defined by Tumor Response Grade (TRG), was used to assess the viability of cancer cells in the resected tumor tissue. According to TRG classification, TRG 0 was defined as pathological complete response (pCR), indicating no detectable cancer cells in the resected tissue, while TRG 1 and above were classified as non-pathological complete response (non-pCR). For analytical purposes, patients were categorized into two groups: the pCR group and the non-pCR group. Laboratory data collection timepoints: Pre-treatment values: The most recent laboratory results obtained within 2 weeks before initiating neoadjuvant therapy. Post-treatment values: The first laboratory results collected during the initial follow-up visit after completing neoadjuvant therapy (typically within 1 week prior to surgery).
In addition, the Geriatric Nutritional Risk Index (GNRI) was employed to evaluate patients’ nutritional status, calculated as follows: GNRI = 14.89 × serum albumin (g/L) + 41.7 × (current body weight (kg)/ideal body weight (kg)). Ideal body weight was calculated as 50 + 2.3 × (height (cm) − 152) kg for males, and 45.5 + 2.3 × (height (cm) − 152) kg for females. PIV, an indicator of immune status and inflammation, was calculated as follows: PIV = (neutrophil count × monocyte count × platelet count)/lymphocyte count.
Statistical analysis
Continuous variables were expressed according to their distributional characteristics: normally distributed data as mean ± standard deviation, and skewed data as median and interquartile range (IQR). Categorical variables were reported as percentages. In univariate analysis, normally distributed continuous variables were analyzed using univariate tests, while categorical variables were assessed using χ2 or trend tests.
Clinically relevant pre-specified candidate variables—including age, sex, weight, body mass index (BMI), smoking history, alcohol history, comorbidities, tumor location, American Society of Anesthesiologists Physical Status Classification System (ASA), clinical stage, neoadjuvant treatment regimen and cycles, surgical approach, lymphadenectomy extent, and pre-/post-treatment laboratory values (albumin, GNRI, PIV)—were included in univariate analysis. Variables associated with pCR at p < 0.1 in univariate analysis were entered into multivariate logistic regression. Covariates were retained in the final model if significantly associated with the outcome or if they altered the effect estimate (odds ratio (OR)) of key variables (e.g., PIV) by >10%. Adjusted ORs with 95% confidence intervals (CIs) were calculated. Restricted cubic splines (three knots) explored potential nonlinear relationships between post-treatment PIV and pathological response. When nonlinearity was detected, a two-piecewise linear regression model identified the threshold (inflection point). The inflection point’s statistical significance was verified using likelihood ratio tests with Bootstrap resampling (1000 repetitions). Pre-specified subgroup analyses (stratified by age, sex, BMI, alcohol use, smoking history, comorbidities, tumor location, ASA grade, and clinical stage) assessed the consistency of the PIV-pathological response association. All subgroup analyses adjusted for core covariates (age, sex, clinical stage) in logistic models, with interaction effects evaluated by likelihood ratio tests. Receiver operating characteristic (ROC) analysis evaluated post-treatment PIV’s predictive performance for non-pCR. The area under the curve (AUC) was calculated, with the Youden index determining the optimal cutoff’s sensitivity and specificity. Using the ROC-derived optimal PIV cutoff, patients were stratified into high/low PIV groups. Kaplan–Meier curves depicted overall survival (OS), with between-group differences assessed via the log-rank test.
All analyses used R software (v4.2.3) and Z stats v0.90 (www.medsta.cn/software), with two-tailed p < 0.05 considered significant.
Results
Clinical characteristics of ESCC patients
A total of 334 patients were included in this study, categorized into the pCR and non-pCR groups based on postoperative pathological TRG classification, as shown in Table 1. The median age of the patients was 61 years (IQR 55.2–66.8 years), with 81.1% being male. The median pre-treatment weight was 60.0 kg (IQR 52.0–65.0 kg), which showed no significant change after treatment, with a median post-treatment weight of 60.0 kg (IQR 52.5–65.0 kg). The median post-treatment BMI was 21.5 (IQR 19.8–23.3). The majority of patients had tumors located in the middle thoracic (53.9%) and lower thoracic (38.0%) esophagus, and most were classified as ASA grade II (82%). The clinical stages were predominantly stage II (26.65%) and stage III (53.38%). Among the included patients, 104 (31.13%) achieved pCR. No statistically significant differences were observed in baseline characteristics between the pCR and non-pCR groups, including smoking history (p = 0.505), alcohol consumption (p = 0.442), and comorbidities such as hypertension (p = 0.325), diabetes (p = 0.536), coronary heart disease (p = 0.240), chronic obstructive pulmonary disease (COPD; p = 1.000), and cerebrovascular disease (p = 0.314). Furthermore, treatment-related variables, including neoadjuvant regimens (p = 0.395), number of neoadjuvant cycles (p = 0.809), surgical approaches (p = 0.298), and lymph node dissection (p = 0.579), also showed no significant intergroup differences.
Clinical characteristics in 334 esophageal squamous cell carcinoma patients.

ASA, American Society of Anesthesiologists; BMI, body mass index; CHD, coronary heart disease; COPD, chronic obstructive pulmonary disease; CVD, cerebrovascular disease; pCR, pathological complete response; TP, taxane + platinum.
Inflammatory and nutritional changes pre- and post-treatment
The experimental results indicated that the pre-treatment albumin level was 40.7 g/L, decreasing to 39.3 g/L post-treatment, representing a reduction of 0.6 g/L. The albumin levels in the pCR group were significantly higher than those in the non-pCR group prior to treatment (p = 0.002) and were maintained at significantly higher levels (p < 0.001). The pCR group exhibited a rising trend in albumin levels after treatment, whereas the non-pCR group showed a declining trend, with a significant difference in the magnitude of change between the two groups (p = 0.002).
Regarding nutritional indices, the pre-treatment GNRI was 101, decreasing to 99.6 after treatment, indicating a decline in nutritional status by 0.48. There was no significant difference in nutritional status between the pCR and non-pCR groups before treatment (p = 0.088). However, post-treatment, the nutritional status of the pCR group showed a slight increase, while the non-pCR group experienced a notable decline, with a significant statistical difference in the magnitude of change between the two groups (p < 0.001).
In terms of inflammatory indices, the pre-treatment PIV was 303, decreasing to 284 post-treatments, reflecting a reduction of 20. There was no significant difference in inflammatory status between the pCR and non-pCR groups prior to treatment (p = 0.089). Post-treatment, the PIV was 198 in the pCR group and 345 in the non-pCR group, revealing a significant statistical difference between the two groups (p < 0.001). The inflammatory status in the pCR group significantly decreased, while the non-pCR group showed a slight decrease, with a statistically significant difference in the magnitude of change between the two groups (p < 0.001, see Table 2).
The comparisons of laboratory investigations, inflammatory, and nutritional markers between pre-CIT and post-CIT.

ALB, albumin; CIT, chemoimmunotherapy; GNRI, Geriatric Nutritional Risk Index; PIV, Pan-Immune-Inflammation Value.
Univariate and multivariate analyses
The findings from univariate and multivariate logistic regression analyses are summarized in Table 3. In the univariate analysis, no statistically significant differences were observed among the clinical baseline data. However, significant differences were detected in albumin, GNRI, and PIV before and after treatment. Therefore, these variables were included in the multivariate logistic regression model for further evaluation. The analysis revealed that post-treatment PIV is an independent risk factor for postoperative pathological response. Specifically, after adjusting for multiple variables, each 10-unit increase in PIV was associated with a 2% increase in the risk of non-pCR (OR = 1.002; 95% CI: 1.001–1.003; p < 0.005; see Table 3).
Univariate and multivariate logistic regression analyses of pathological response.

ALB, albumin; ASA, American Society of Anesthesiologists; BMI, body mass index; CHD, coronary heart disease; CI, confidence interval; COPD, chronic obstructive pulmonary disease; GNRI, Geriatric Nutritional Risk Index; OR, odds ratio; PIV, Pan-Immune-Inflammation Value.
Subgroup analyses
The results of the subgroup analyses are illustrated in Figure 2. Stratification based on age, sex, BMI, alcohol consumption, smoking history, hypertension, diabetes, coronary heart disease, COPD, cerebrovascular disease, tumor location, ASA classification, and clinical staging revealed no significant interactions among the subgroups (all p > 0.05).

Subgroup analysis between PIV and the pathological response.
Nonlinear relationship between PIV and pCR
After accounting for covariates, we established a nonlinear dose–response relationship between the PIV and postoperative pathological response (likelihood ratio test, p = 0.01; see Figure 3). Using a two-segment linear regression model, the threshold for PIV was determined to be 280. As indicated in Table 4, notably, a positive association was observed in the high-PIV stratum (⩾280), where elevated PIV levels significantly correlated with increased non-pCR risk (OR = 1.01, 95% CI: 1.006–1.015; p < 0.001). Conversely, no statistically significant association was detected in the low-PIV stratum (<280; OR = 1.00, 95% CI: 0.996–1.005; p = 0.870).

Nonlinear relationship between PIV and pathological complete response. Adjustment factors included age, sex, smoking, alcohol, diabetes, weight, BMI, hypertension, CHD, COPD, CVD, location of tumor, ASA, clinical stage, and GRNI. The red curve represents the principal curve, depicting the fitted nonlinear relationship between pathological response and PIV. The principal curve illustrates the nonlinear trend of adverse pathological response with increasing PIV. The red shaded area indicates the 95% CI of the principal curve. The black dashed line (Y = 1) serves as the risk reference baseline (OR = 1). The position of the principal curve and its CI relative to this line determines the statistical significance of risk changes.
Threshold effect analysis of the relationship between PIV and the incidence of non-pCR.

CI, confidence interval; OR, odds ratio; pCR, pathological complete response; PIV, Pan-Immune-Inflammation Value.
ROC curve analysis and survival analysis
We conducted ROC curve analysis for PIV values above the threshold to predict the risk of postoperative non-pCR. Relevant data are presented in Figure 4 and Table 5. The AUC was 0.86 (95% CI: 0.79–0.92), with an optimal cutoff value of 438.04. The sensitivity was 86.6% (95% CI: 74%–97%), and the specificity was 72% (95% CI: 64%–79%). Using the ROC-derived optimal cutoff (438.04), patients were stratified into high-PIV (n = 95) and low-PIV (n = 239) cohorts. Kaplan–Meier analysis demonstrated significant separation of survival curves (log-rank p = 0.039; Figure 5). The high-PIV group exhibited inferior survival outcomes with significantly increased mortality risk (hazard ratio (HR) = 1.416; 95% CI: 1.016–1.975).
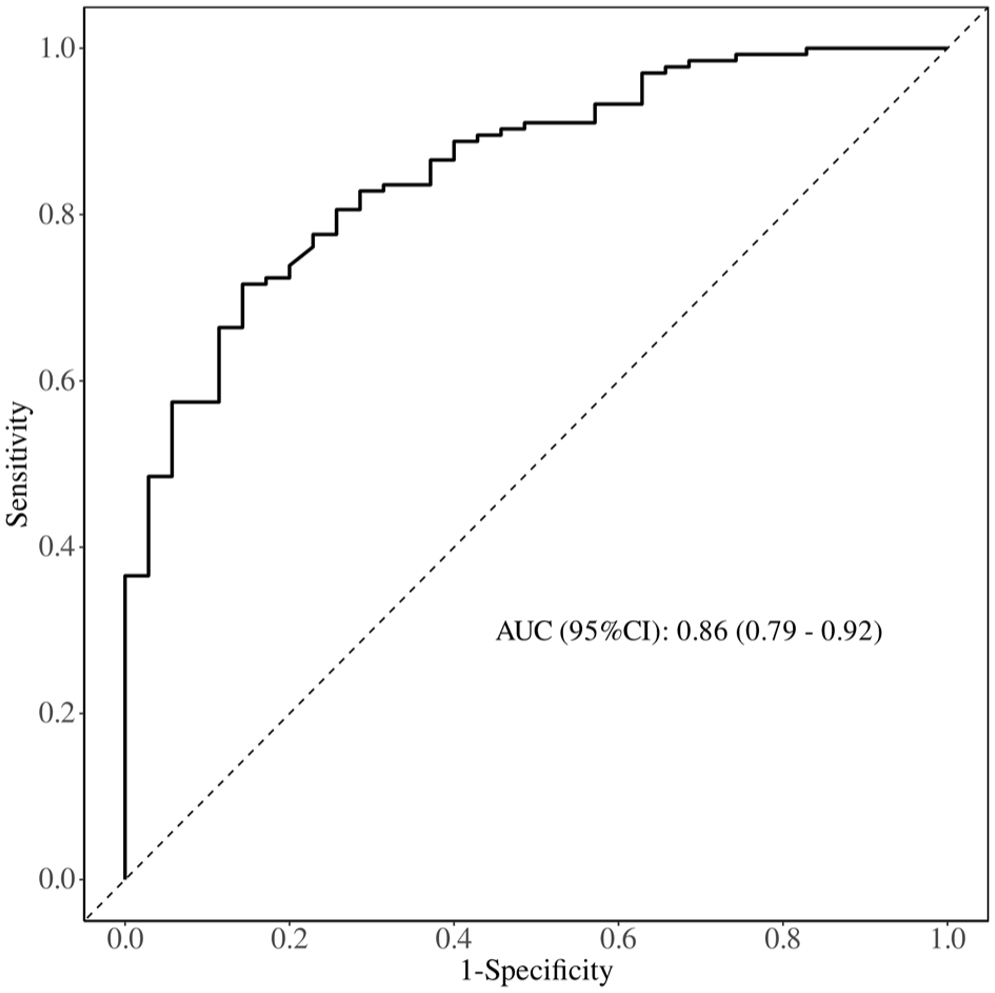
Receiver operating characteristic analysis of PIV in predicting the risk of postoperative non-pCR.
Information on the receiver operating characteristic curve in Figure 4.

AUC, area under the curve; CI, confidence interval; PIV, Pan-Immune-Inflammation Value.
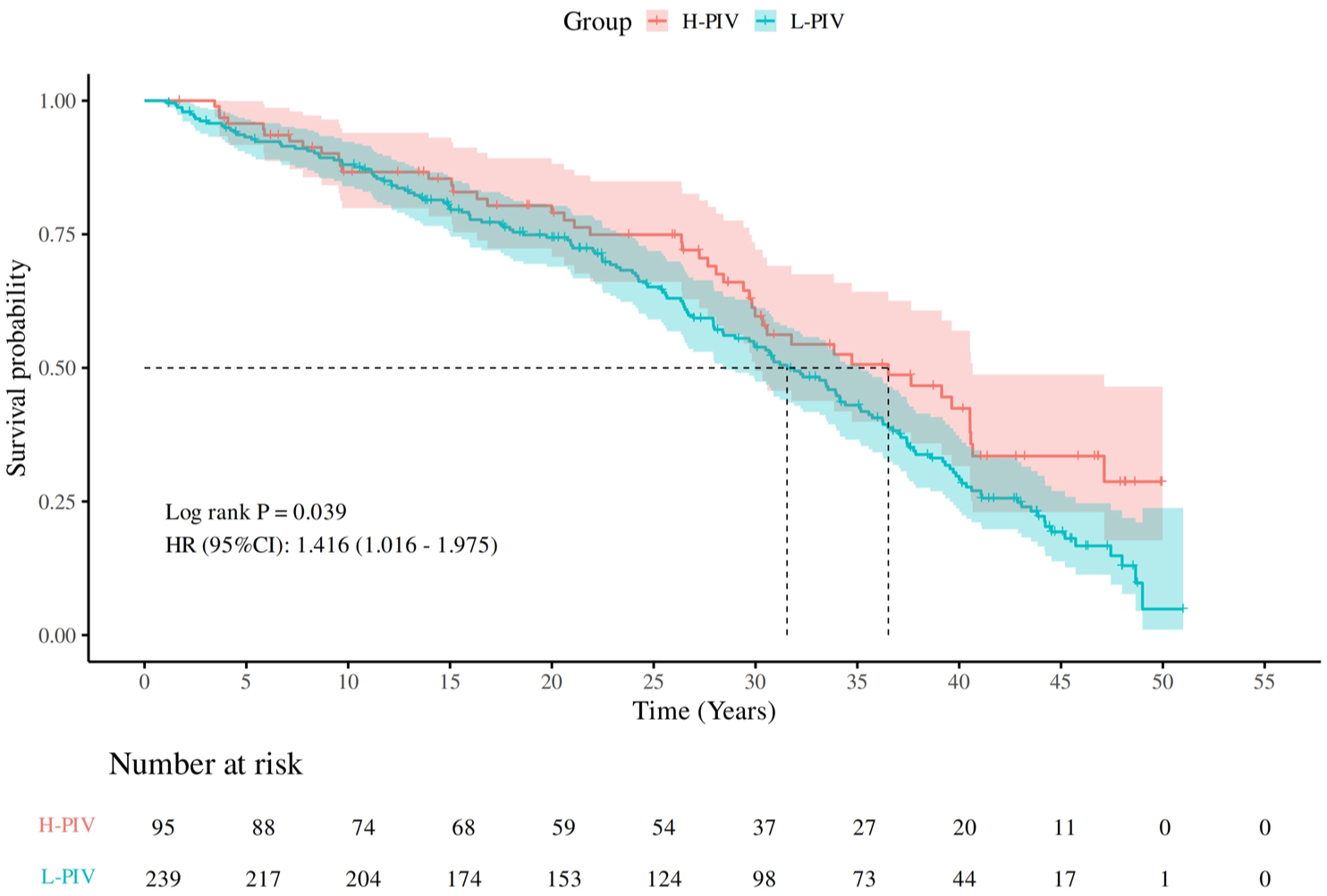
Kaplan–Meier analysis of PIV groups classified using the ROC-optimized cutoff value.
Discussion
In this multicenter retrospective study conducted in southern China, we observed an independent correlation between postoperative pathological response in ESCC and PIV. Specifically, post-treatment PIV was positively associated with the risk of non-pCR. Notably, this association remained consistent and significant even after accounting for potential covariates and confounding factors. To the best of our knowledge, this study is the first to demonstrate a significant correlation between post-treatment PIV and postoperative pathological response in patients with esophageal cancer.
Furthermore, we noted a nonlinear and non-equidistant pattern of effect size variation across different PIV groups. Particularly, when the PIV threshold exceeded 280, the correlation between PIV and postoperative pathological response exhibited significant changes. Therefore, when PIV values exceed 280, further increases in PIV significantly heighten the risk of non-pCR. These findings indicate that PIV holds promise as a potential biomarker for predicting the degree of postoperative pathological response. Fu et al. 13 demonstrated in unresectable advanced ESCC that a pretreatment PIV ⩾415.885 was significantly associated with inferior objective response rate (34.8% vs 62.5%, p = 0.003), disease control rate (65.2% vs 87.5%, p = 0.008), and shorter median OS (8.9 vs 21.2 months, p < 0.001). Although their cohort (unresectable advanced disease) differed from ours (neoadjuvant-treated operable cases), both studies support PIV’s predictive utility. Notably, Fu et al. identified the SII, not PIV, as an independent prognostic factor, whereas our study—using pathologic response assessment—established PIV (cutoff: 438.04) as an independent predictor of neoadjuvant chemoimmunotherapy outcomes, providing complementary evidence across disease stages.
Neoadjuvant chemotherapy induces tumor cell apoptosis and antigen release, activates antigen-presenting cells and immune cells, and inhibits immune evasion mechanisms, thereby enhancing the efficacy of immunotherapy.14,15 In this combined treatment approach, immune-inflammatory responses play a crucial role in evaluating treatment outcomes, predicting prognosis, and managing complications. 16 Widely used immune-inflammatory markers, such as the NLR, platelet-to-lymphocyte ratio (PLR), and SII, have proven effective in predicting the outcomes of neoadjuvant chemotherapy combined with immunotherapy.10,17 Studies have shown that elevated NLR and PLR are generally associated with poorer pathological response and prognosis, whereas lower levels often correspond to higher pCR rates.18,19 In addition, further research has demonstrated that the combination of neoadjuvant chemotherapy and immunotherapy not only boosts immune cell activity within the tumor microenvironment but may also improve long-term survival by modulating the expression of inflammatory markers. 20 However, the association between post-treatment PIV and postoperative pathological response has not been explored in prior studies. This investigation is the first to evaluate the clinical utility of PIV in the context of neoadjuvant chemotherapy combined with immunotherapy for esophageal cancer.
Notably, compared to other singular markers, PIV serves as an integrated biomarker, reflecting systemic immune and inflammatory status by combining several indices, including neutrophil, platelet, monocyte, and lymphocyte counts. 10 This characteristic makes PIV a potentially reliable predictor of postoperative pathological response. In this study, we observed that the nutritional status of the pCR group improved after treatment, while the non-pCR group showed a declining trend. Although multivariate regression analysis did not identify PIV as an independent risk factor, this finding warrants further investigation. We hypothesize that tumor regression and alleviation of esophageal obstruction in the pCR group may have facilitated increased food intake, thereby improving nutritional status; however, the underlying mechanisms require further exploration.
A 2024 study by Feng et al. 21 identified the GNRI as an effective dynamic monitoring indicator of nutritional status during neoadjuvant chemotherapy, demonstrating its predictive value for pathological response in patients with esophageal cancer. GNRI changes not only reflect improvements in nutritional status but are also closely associated with enhanced immune responses. Another study noted that a low GNRI correlated with poor immunotherapy response, while a high GNRI was predictive of better treatment response and OS. 22
Several studies have established PIV as an independent risk factor for poor prognosis across various cancers, representing systemic immune-inflammatory levels. Elevated PIV typically indicates worse outcomes, including reduced OS and disease-free survival, as observed in head and neck squamous cell carcinoma, breast cancer, and other malignancies.23,24 The prognostic significance of PIV receives broader validation from a 2024 meta-analysis by Kuang et al. 25 Their results demonstrated that elevated PIV was significantly associated with worse OS (HR = 1.895, 95% CI: 1.548–2.318) and progression-free survival (HR = 1.582, 95% CI: 1.324–1.890). These findings align closely with our data, where ROC analysis revealed PIV’s strong predictive value for non-pCR (AUC = 0.86; cutoff = 438.04), with high PIV linked to increased mortality risk (HR = 1.416). Kuang et al. further proposed the PILE score (integrating PIV, ECOG status, and LDH), suggesting that multidimensional models may enhance predictive accuracy in future research.
Beyond inflammatory markers, other biomarkers such as PD-L1 expression (combined positive score, CPS) and tumor mutational burden (TMB) have been explored as predictive markers for immunotherapy in ESCC. While PD-L1 (CPS) demonstrated predictive value in trials like KEYNOTE-590, its interpretation lacks standardization and may be confounded by tumor heterogeneity. 26 TMB, though associated with immunotherapy response in some cancers, lacks a validated cutoff in ESCC and involves high testing costs. 27 Mismatch repair deficiency is rare in ESCC (<2%), 28 limiting its clinical utility. By contrast, blood-based inflammatory indices like PIV offer distinct advantages: they are easily accessible and measurable (via routine blood tests), allow dynamic monitoring, and are unaffected by tumor heterogeneity. Our study confirms PIV’s predictive efficacy (AUC = 0.86) in neoadjuvant chemoimmunotherapy, providing a more practical and complementary tool for clinical use.
Limitation
This study has several limitations. First, the cross-sectional design requires caution when interpreting the observed associations as causal. Second, inherent to observational studies, there may be residual confounding factors that could not be completely controlled. Third, the study population was restricted to esophageal cancer patients who had undergone surgery, with surgical indications based primarily on imaging-assessed tumor regression. Therefore, the applicability of these findings to the broader esophageal cancer population remains uncertain and should be interpreted with caution.
Conclusion
A nonlinear relationship was observed between the PIV and postoperative pathological response in ESCC patients treated with neoadjuvant chemotherapy combined with anti-PD-1 therapy. Post-treatment PIV emerged as a potential biomarker for predicting postoperative pathological response, offering a more robust basis for preoperative assessment of treatment efficacy.
Supplemental Material
sj-doc-1-tam-10.1177_17588359251378883 – Supplemental material for Pan-immune-inflammation value as a novel predictor of pathological response to neoadjuvant chemotherapy combined with anti-PD-1 therapy in esophageal squamous cell carcinoma: a multicenter real-world retrospective clinical study
Supplemental material, sj-doc-1-tam-10.1177_17588359251378883 for Pan-immune-inflammation value as a novel predictor of pathological response to neoadjuvant chemotherapy combined with anti-PD-1 therapy in esophageal squamous cell carcinoma: a multicenter real-world retrospective clinical study by Jiang-shan Huang, Qi-hong Zhong, Gang Wang, Zi-lu Tang, Bing-Lin Shen, Wei-nan Liu, Fei-long Guo, Jing-yu Wu, Zhen-yang Zhang and Jiang-bo Lin in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251378883 – Supplemental material for Pan-immune-inflammation value as a novel predictor of pathological response to neoadjuvant chemotherapy combined with anti-PD-1 therapy in esophageal squamous cell carcinoma: a multicenter real-world retrospective clinical study
Supplemental material, sj-docx-2-tam-10.1177_17588359251378883 for Pan-immune-inflammation value as a novel predictor of pathological response to neoadjuvant chemotherapy combined with anti-PD-1 therapy in esophageal squamous cell carcinoma: a multicenter real-world retrospective clinical study by Jiang-shan Huang, Qi-hong Zhong, Gang Wang, Zi-lu Tang, Bing-Lin Shen, Wei-nan Liu, Fei-long Guo, Jing-yu Wu, Zhen-yang Zhang and Jiang-bo Lin in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We highly acknowledge all the staff from our unit who participated in this study. We are particularly grateful to Dr Dan-wei Zhang for her expert review of our statistical methodology. We would also like to express our heartfelt appreciation to the family members of the first author, especially his wife, for her unwavering support and understanding throughout the completion of this study.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
