Abstract
Background:
Neoadjuvant chemoradiotherapy (NCRT) combined with surgery is the standard treatment option for patients with locally advanced esophageal squamous cell carcinoma (ESCC). However, whether adjuvant chemotherapy (AC) administered postoperatively has a survival benefit remains inconclusive.
Objectives:
To investigate whether AC is necessary after NCRT and esophagectomy and determine which patients might benefit from it.
Design:
A retrospective study.
Methods:
This retrospective study examined patients with ESCC treated with NCRT followed by radical esophagectomy at three hospitals between March 2016 and December 2022. Patients were assigned into the adjuvant and non-adjuvant therapy groups based on whether they received postoperative AC, allowing the comparison of disease-free survival (DFS) and overall survival (OS) between the two groups. In addition, based on whether postoperative pathology indicated pathological complete response (pCR), patients were classified into the pCR and non-pCR populations, with DFS and OS separately analyzed for each subgroup.
Results:
Overall, 218 eligible patients were enrolled. No significant advantage was found in DFS (p = 0.540) and OS (p = 0.058) between the adjuvant and non-adjuvant therapy groups. In the non-pCR population, the adjuvant therapy groups had a significant advantage in DFS (p = 0.046) and OS (p = 0.011) compared to the non-adjuvant therapy group. However, in the pCR population, no significant advantage was found in DFS (p = 0.490) and OS (p = 0.110) analyses between the adjuvant and non-adjuvant therapy groups.
Conclusion:
In the real world, patients with ESCC who underwent NCRT combined with radical esophagectomy and whose postoperative pathology was pCR did not benefit from AC. However, AC significantly improved DFS and OS in patients whose postoperative pathology did not reach pCR.
Keywords
Introduction
Esophageal cancer ranks as the ninth most common cancer globally and the sixth leading cause of cancer-related mortalities. Over 50% of patients with esophageal cancer live in East Asia, where 90% of them have esophageal squamous cell carcinoma (ESCC). 1 Surgery is widely recognized as the primary treatment for locally advanced resectable ESCC; however, the prognosis remains poor, with a 5-year survival rate of 25% following surgery alone.2–4 Multimodal treatment strategies for locally advanced esophageal cancer have significantly evolved over the past decade to improve the likelihood of surgical resection, thereby increasing the probability of pathological complete response (pCR) and improving overall survival (OS). Several clinical trials have demonstrated that patients with esophageal cancer who receive multimodal treatment after surgical resection have a significantly improved prognosis compared to those who only undergo surgery.5–10 Notably, the ChemoRadiotherapy for Oesophageal cancer followed by Surgery Study (CROSS) trial revealed that preoperative neoadjuvant chemoradiotherapy (NCRT) for esophageal cancer doubled the median survival time, increasing it from 24 to 49 months, compared to surgery alone. 11
While neoadjuvant therapy is highly effective, distant progression occurs in 39% of patients.11,12 Therefore, the need for subsequent adjuvant chemotherapy (AC) to improve patient survival has become a critical issue. Research has shown that administering AC to patients receiving initial treatment and after esophagectomy can lead to an improvement in survival rates.13–15 A retrospective study based on the National Cancer Database (NCDB) revealed that patients who received AC had significantly better long-term survival than controls (median OS: 38.5 vs 32.3 months, hazard ratio = 0.78, 95% confidence interval (CI): 0.71–0.87, p < 0.001), and they benefited from AC irrespective of N-stage (yp) and margin status. 16 However, another retrospective study based on the NCDB showed that AC did not improve the long-term prognosis of patients with stage pT2-4aN0M0 esophageal cancer after radical resection. 17 Therefore, no definitive conclusion has been reached regarding whether AC should be added after NCRT combined with surgery or which patients would benefit from AC. This study aimed to investigate whether the administration of AC after NCRT combined with radical esophagectomy is associated with improved disease-free survival (DFS) and OS and explore how postoperative pathology affects the efficacy of AC.
Materials and methods
Patient selection
We retrospectively analyzed patients with locally advanced ESCC who underwent NCRT combined with radical esophagectomy at the Affiliated Cancer Hospital of Shandong First Medical University, Shandong Proton Center, and the Second Affiliated Hospital of Shandong First Medical University between March 2016 and December 2022. Specifically, all eligible patients met the following criteria: aged 18–75 years, had an Eastern Cooperative Oncology Group score of 0–1, confirmed pathology of squamous esophageal cancer, and had undergone NCRT before surgical treatment. Patients were excluded if they received other neoadjuvant therapies (such as targeted therapies and immunotherapies), if the dose of radiotherapy was not between 40 and 50.4 Gy (in units of 1.8–2.0 Gy), or if their information records were incomplete. Finally, 218 patients with locally advanced ESCC treated with NCRT and esophagectomy were eligible for enrollment. The study protocol was approved by the Ethics Committee of the Affiliated Cancer Hospital of Shandong First Medical University, Shandong Proton Center, and the Second Affiliated Hospital of Shandong First Medical University.
Neoadjuvant treatment regimens
Patients undergoing NCRT typically receive a radiation dose ranging from 40.0 to 50.4 Gy, delivered in fractions of 1.8–2.0 Gy. The radiotherapy methods used were intensity-modulated radiation therapy and three-dimensional conformal radiotherapy. All patients were localized under large-aperture computed tomography (CT). The radiotherapy target area was predominantly located in the involved field. In addition, the primary preoperative chemotherapy regimen involved the administration of platinum-based medications in combination with fluorouracil (PF regimen) or paclitaxel/albumin paclitaxel (TP regimen) through intravenous injection. The PF regimen consisted of platinum-based drugs (nedaplatin at 75 mg/m2, carboplatin at an area under the curve of 5, or cisplatin at 25 mg/m2 on days 1–3) along with fluorouracil at 500 mg/m2 on days 1–5. By contrast, the TP regimen consisted of paclitaxel at 135–175 mg/m2 or albumin–paclitaxel at 260 mg/m2. Patients received 1–3 preoperative chemotherapy (PF or TP regimens) every 3 weeks, and the average usage cycle was two. Neoadjuvant radiotherapy and neoadjuvant chemotherapy were performed simultaneously in all patients in principle.
Surgical treatment
The qualifying patients underwent esophagectomy and mediastinal lymph node dissection under general anesthesia 4–8 weeks after completing their last neoadjuvant therapy. Specifically, the surgeries were performed using thoracotomy or minimally invasive techniques with two or three incisions. Double-field lymph node dissection was standard. In cases of suspected cervical lymph node enlargement, patients underwent standard three-field lymph node dissection.
Adjuvant therapy
All patients included in this study who received adjuvant therapy received three to four cycles of AC after surgical treatment with the same chemotherapy regimen as in the neoadjuvant regimen.
Follow-up
All selected patients received regular outpatient reviews and telephone follow-ups since admission, and physical examination, chest and abdominal enhanced CT, and esophagography were performed regularly during the follow-up period. Ultrasound, endoscopy, positron emission tomography/CT, magnetic resonance imaging, or whole-body bone imaging were performed when necessary. For patients whose last case system record was more than 1 month before the study cutoff, a telephone follow-up was conducted to gather details about their progress and survival. The follow-up period ended on May 1, 2024, and the median follow-up time for all patients was 24 months, with a range of 4–96 months.
Study endpoints
This study’s endpoints were DFS and OS. DFS was defined as the time from radical resection of esophageal cancer to the first recording of recurrence, all-cause mortality, or the last follow-up. By contrast, OS was defined as the time from the start of the first cycle of neoadjuvant therapy to all-cause mortality or the last follow-up. Two trained pathologists evaluated all postoperative pathological findings. pCR was defined as the absence of residual tumor cells in the primary site of the surgical specimen and the resected lymph nodes (ypT0N0M0). DFS and OS were analyzed for patients who received postoperative adjuvant therapy and those who did not across the total population, as well as the pCR and non-pCR subgroups.
Statistical analysis
Continuous variables were analyzed using the independent samples t-test or rank-sum test. Subtypes of variables were compared using the Chi-square or Fisher’s exact test. The DFS and OS of patients were assessed using the Kaplan–Meier method and compared using the log-rank test. Univariate and multivariate analyses were performed to identify independent prognostic factors for DFS and OS using the Cox proportional risk model. All tests were two-sided, and statistical significance was considered at p < 0.05. Statistical analysis was performed using R software (version 4.2.3; R Foundation, Vienna, Austria).
Reporting guideline
The reporting of this study conforms to Strengthening the reporting of observational studies in epidemiology (STROBE; Supplemental Table 1). 18
Results
Patients’ baseline characteristics
A total of 218 patients with resectable ESCC who underwent NCRT combined with radical esophagectomy at three hospitals between March 2016 and December 2022 were enrolled. Among these, 95 (43.6%) achieved pCR, while 123 (56.4%) were categorized as non-pCR. Of the 218 eligible patients, 63 (28.9%) and 152 (69.7%) had TNM clinical stages II and III, respectively. In addition, 107 (49.1%) patients received postoperative adjuvant therapy, while 111 (50.9%) did not (Table 1).
Baseline characteristics of patients.

Comorbidities include hypertension, diabetes mellitus, chronic obstructive pulmonary disease, and coronary atherosclerotic heart disease.
LVSI, lymphovascular space invasion; MIE, minimally invasive esophagectomy; No., number; PF, platinum-based drugs and fluorouracil; SD, standard deviation; TNM, tumor-node-metastasis; TP, platinum-based drugs and paclitaxel/albumin paclitaxel.
Progression and survival outcomes
Univariate and multivariate analyses were performed to identify the factors associated with OS and DFS in all patients. Our result demonstrated that pCR was related to longer OS and DFS in ESCC patients in univariate and multivariate analyses (Supplemental Table 2). In the total population (n = 218), the median DFS of the adjuvant and non-adjuvant therapy groups was 63 and 58 months, respectively. As shown in Figure 1(a), compared with the non-adjuvant therapy group, no significant advantage in 3-, 4-, and 5-year DFS analyses was found in the adjuvant therapy group (3-year DFS, 68.8% vs 69.4%; 4-year DFS, 54.4% vs 55.7%; and 5-year DFS, 38.4% vs 48.4%; p = 0.540). Median OS was not reached in the adjuvant or non-adjuvant therapy group. No significant advantage in the 3-, 4-, and 5-year OS analyses was found in the adjuvant therapy group compared with the non-adjuvant therapy group (3-year OS, 75.5% vs 83.0%; 4-year OS, 72.6% vs 73.6%; and 5-year OS, 55.6% vs 71.9%; p = 0.058; Figure 1(b)).

Kaplan–Meier survival analysis of DFS (a) and OS (b) for patients in the adjuvant and non-adjuvant therapy groups in the total population.
Patients were categorized into the pCR and non-pCR populations based on whether the postoperative pathology achieved pCR. In the pCR population, neither the adjuvant therapy group nor the non-adjuvant therapy group achieved the median DFS. No significant advantage in the 3-, 4-, and 5-year DFS analyses was found in the adjuvant therapy group compared with the non-adjuvant therapy group (3-year DFS, 90.8% vs 94.0%; 4-year DFS, 67.0% vs 81.9%; and 5-year DFS, 67.0% vs 81.9%; p = 0.490; Figure 2(a)). In addition, neither the adjuvant therapy group nor the non-adjuvant therapy groups achieved a median OS. Compared with the non-adjuvant therapy group, no significant advantage in the 3-, 4-, and 5-year OS analyses was found in the adjuvant therapy group (3-year OS, 81.2% vs 100.0%; 4-year OS, 78.4% vs 80.3%; and 5-year OS, 71.8% vs 80.3%; p = 0.110; Figure 2(b)).

Kaplan–Meier survival analysis of DFS (a) and OS (b) for patients in the adjuvant and non-adjuvant therapy groups in the pCR population.
In the non-pCR population, the median DFS was 43 and 28 months in the adjuvant and non-adjuvant therapy groups, respectively. Compared with the non-adjuvant therapy group, a significant advantage in the 3-, 4-, and 5-year DFS analyses was found in the adjuvant therapy group (3-year DFS, 48.1% vs 57.2%; 4-year DFS, 39.8% vs 47.2%; and 5-year DFS, 21.2% vs 39.8%; p = 0.046; Figure 3(a)). The median OS was not achieved in the adjuvant therapy group, and it was 55 months in the non-adjuvant therapy group. Compared with the non-adjuvant therapy group, a significant advantage in the 3-, 4-, and 5-year OS analyses was found in the adjuvant therapy group (3-year OS, 58.7% vs 79.2%; 4-year OS, 50.0% vs 59.3%; and 5-year OS, 38.8% vs 53.8%; p = 0.011; Figure 3(b)).

Kaplan–Meier survival analysis of DFS (a) and OS (b) for patients in the adjuvant and non-adjuvant therapy groups in the non-pCR population.
In the non-pCR population, we separately analyzed the prognosis of patients with residual disease in the primary focus or lymph nodes. For patients with residual lesions in the primary foci, the adjuvant therapy group had a significant advantage in the 3-, 4-, and 5-year DFS analyses compared with the non-adjuvant therapy group (3-year DFS, 61.4% vs 47.1%; 4-year DFS, 47.2% vs 38.3%; and 5-year DFS, 47.2% vs 19.3%; p = 0.039; Figure 4(a)). Compared with the non-adjuvant therapy group, a significant advantage in the 3-, 4-, and 5-year OS analyses was found in the adjuvant therapy group (3-year OS, 52.7% vs 79.4%; 4-year OS, 46.2% vs 61.3%; and 5-year OS, 34.2% vs 55.9%; p = 0.006; Figure 4(b)).

Kaplan–Meier survival analysis of DFS (a) and OS (b) for patients in the adjuvant and non-adjuvant therapy groups in a population with residual disease in the primary lesion.
For patients with residual lymph node lesions postoperatively, we calculated the ratio of positive lymph nodes to the total number of lymph nodes resected (PEN) and established a cutoff value of 0.05 for PEN based on DFS (Supplemental Figure 1). In patients with PEN >0.05, compared with the non-adjuvant therapy group, the adjuvant therapy group had a significant advantage in the DFS analysis (3-year DFS, 52.4% vs 13.1%; p = 0.013; Figure 5(a)). The adjuvant therapy group also had a significant advantage in the OS analysis compared with the non-adjuvant therapy group (3-year OS, 74.4% vs 45.7%; p = 0.041; Figure 5(b)). In patients with PEN ⩽0.05, the adjuvant therapy group had no significant advantage in the DFS and OS analyses compared with the non-adjuvant therapy group (Supplemental Figure 2).
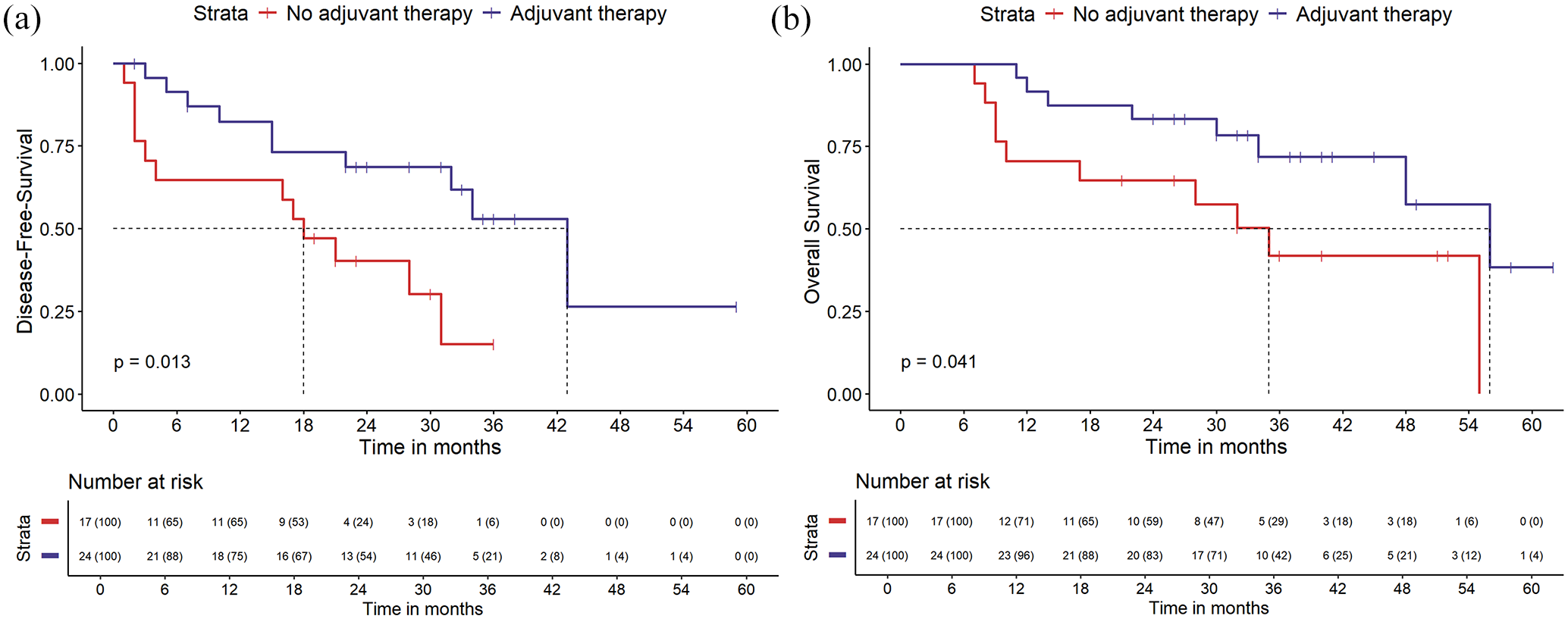
Kaplan–Meier survival analysis of DFS (a) and OS (b) for patients in the adjuvant and non-adjuvant therapy groups in a population with PEN >0.05.
This study also analyzed the prognostic impact of different chemotherapy regimens in patients receiving postoperative AC. The PF regimen showed no significant advantage in the 3-, 4-, and 5-year DFS analysis compared with the TP regimen (3-year DFS, 70.1% vs 71.4%; 4-year DFS, 58.3% vs 48.2%; and 5-year DFS, 58.3% vs 48.2%; p = 0.520; Supplemental Figure 3(A)). Compared with the TP regimen, the PF regimen also showed no significant advantage in the 3-, 4-, and 5-year OS analyses (3-year OS, 80.7% vs 75.4%; 4-year OS, 73.2% vs 68.3%; and 5-year OS, 70.2% vs 68.3%; p = 0.640; Supplemental Figure 3(B)).
Discussion
The treatment of esophageal cancer has always been a clinical challenge. Recently, NCRT has gradually gained wide acceptance as a therapeutic strategy; however, whether chemotherapy administered as adjuvant therapy after R0 resection can provide a survival benefit remains controversial. This study drew key conclusions from the follow-up of patients with ESCC who underwent surgery after NCRT. No noticeable advantage of AC regarding DFS and OS was found for patients who achieved a pCR postoperatively. However, a significant benefit in DFS and OS was observed for patients with primary lesions and lymph nodes with residual disease postoperatively (non-pCR). The study by Burt et al. 19 showed partial similarity to our results, as AC did not lead to a significant decrease in the mortality risk for patients with either no residual disease (ypT0N0) or residual non-lymph node disease (ypT + N0) following NCRT. However, Mokdad et al. 20 conducted a study in 2017 comparing the efficacy of AC with postoperative observations in patients with gastroesophageal adenocarcinoma undergoing NCRT, revealing that AC was significantly associated with improved OS compared to postoperative observations (median survival: 40 months; 95% CI: 36–46 vs 34 months; 95% CI: 32–35 months; p < 0.001). A meta-analysis by Lee et al., 14 which included 10 studies involving 6462 patients, among whom 4646 (71.9%) had adenocarcinoma, 226 (3.5%) had squamous carcinoma, and 1590 (24.6%) had mixed types, showed that the adjuvant group (including AC) had a significantly lower mortality rate at 1 and 5 years than the group receiving neoadjuvant therapy and esophagectomy alone (p < 0.001). These results are inconsistent with our findings. Although some retrospective analyses combining data from patients with adenocarcinoma and squamous cell carcinoma (SCC) have shown some survival benefits, variability between the two histological types and non-standardization of preoperative treatment, and adjuvant treatment regimens have hampered the definitive evaluation of this treatment. In the CROSS trial, 23% of patients with adenocarcinoma achieved a pCR after NCRT, compared to 49% of those with SCC.5,19 We observed that many patients included in the above studies had adenocarcinoma, which is less sensitive to radiotherapy than SCC. This subset of patients may benefit less from NCRT; therefore, AC might provide them with a greater survival benefit. Our study addresses the lack of previous studies on patients with ESCC.
In the CROSS study, the 5-year OS rate for the NCRT combined with the surgery group was 47% in the overall population. 5 Among the pCR group, the 5-year OS rates for both the AC and non-AC subgroups exceeded 47%, with no statistically significant difference between them (80.3% vs 71.8%, p = 0.11). In the non-pCR group, the 5-year OS rate was significantly lower in patients who did not receive AC compared to those who did (34.2% vs 55.9%, p = 0.011). In addition, non-pCR patients who received AC had a higher 5-year survival rate than the overall population in the CROSS study (55.9% vs 47%). Thus, postoperative pathological response holds substantial clinical value in guiding AC administration.
The differing responses of patients with distinct pathological outcomes (pCR or non-pCR) to AC merit attention. Wang et al. 21 demonstrated that immune and metabolic biomarkers within the tumor microenvironment (TME) are closely linked to patients’ pathological responses to NCRT. A high metabolic state is notably associated with poor responses, including non-pCR. They hypothesize that increased glycolysis in a high metabolic state may acidify the TME, activate hypoxia response pathways, and promote immune evasion, thereby contributing to high metastatic potential and therapeutic resistance. Thus, we propose that the pathological response to NCRT may partially indicate the potential for recurrence in patients. For non-pCR patients, AC effectively targets minimal residual disease (MRD), offering survival advantages. Conversely, in pCR patients, the risk of MRD-driven recurrence is minimal, and AC typically offers limited benefit. Nevertheless, the underlying mechanisms warrant further biological investigation.
Patients with lymph node disease are at higher risk of experiencing micrometastatic systemic disease. The results of a phase II clinical trial of perioperative chemotherapy and surgery for distal esophageal or esophagogastric junction cancer patients with pN0 had significantly longer DFS than patients with pN+. 22 Raja et al. had a similar view to our study, indicating that patients with esophageal cancer with ypT3-4N0 or ypN2-3 derive a survival benefit from AC. A small group of patients who benefited the most tended to have a higher frequency of lymph node disease and staging, as well as a higher frequency of the ypT4 type and positive margins. 23 Our study showed that patients with non-pCR, either with residual disease in the primary site or lymph nodes, had a survival benefit after undergoing AC, emphasizing the importance of AC in managing the high risk of recurrence associated with incomplete tumor eradication. Non-pCR indicates an incomplete response to initial treatment and the potential for more aggressive tumor biology, which may require further systemic therapy to manage the underlying micrometastatic disease. Previous studies have found a link between elevated PEN and reduced survival rates in esophageal, gastric, and colon cancers, indicating a stronger correlation with survival outcomes compared to traditional lymph node categories.24–26 Our analysis showed that PEN, with a cutoff value of 0.05, could distinguish patients who benefited from receiving postoperative AC. For patients with PEN >0.05, receiving AC was significantly beneficial in terms of DFS and OS. The recommended number of lymph node dissections for locally advanced esophageal cancer after neoadjuvant therapy is currently unclear. In a retrospective study, among 3859 patients with esophageal cancer treated with neoadjuvant therapy, the threshold for long-term prognostic benefit was 25 and 30 lymph nodes for patients with stages ypN0 and ypN+, respectively. 27 Another study by Samson et al. 24 also revealed that a PEN of 0–0.10 was associated with maximum survival benefit in all patients who underwent esophagectomy. Furthermore, for patients with a PEN of 0, the more lymph nodes sampled, the more significant the increase in OS (85.3 months for sampling ⩾20 lymph nodes vs 52.0 months for sampling 1–9 lymph nodes; p < 0.001). 24 The mean number of lymph node dissections in this study was 17, with only a few patients meeting current treatment recommendations. Therefore, increasing the number of lymph node dissections in clinical practice has significant implications for treatment decisions and patient prognosis.
This study had some limitations. First, it was performed at three institutions with a few participants, which may affect the applicability of the findings to a broader population. Second, the retrospective nature of the study could introduce bias and limit the ability to control for confounding variables.
Conclusion
In summary, our study suggests that the need for AC in patients undergoing surgery after NCRT for squamous esophageal cancer should be determined based on whether pCR is achieved postoperatively. In addition, the postoperative pathology of the primary site and lymph nodes is crucial. This study provides valuable information on the need for AC after multimodal therapy for esophageal cancer. Although patients with pCR did not benefit from AC, those who did not achieve pCR in the primary lesion or lymph nodes showed a significant survival advantage with AC treatment. Therefore, this highlights the need for the development of an individualized treatment plan.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251316179 – Supplemental material for Adjuvant treatment after neoadjuvant chemoradiotherapy and surgery in patients with esophageal squamous cell carcinoma: a real-world study
Supplemental material, sj-docx-1-tam-10.1177_17588359251316179 for Adjuvant treatment after neoadjuvant chemoradiotherapy and surgery in patients with esophageal squamous cell carcinoma: a real-world study by Yuxin Geng, Junfeng Zhao, Ying Li and Yintao Li in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251316179 – Supplemental material for Adjuvant treatment after neoadjuvant chemoradiotherapy and surgery in patients with esophageal squamous cell carcinoma: a real-world study
Supplemental material, sj-docx-2-tam-10.1177_17588359251316179 for Adjuvant treatment after neoadjuvant chemoradiotherapy and surgery in patients with esophageal squamous cell carcinoma: a real-world study by Yuxin Geng, Junfeng Zhao, Ying Li and Yintao Li in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359251316179 – Supplemental material for Adjuvant treatment after neoadjuvant chemoradiotherapy and surgery in patients with esophageal squamous cell carcinoma: a real-world study
Supplemental material, sj-docx-3-tam-10.1177_17588359251316179 for Adjuvant treatment after neoadjuvant chemoradiotherapy and surgery in patients with esophageal squamous cell carcinoma: a real-world study by Yuxin Geng, Junfeng Zhao, Ying Li and Yintao Li in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
