Abstract
Background:
Although systemic inflammation- and nutrition-related indices have demonstrated predictive value for treatment response and prognosis in various malignancies, their role in locally advanced gastrointestinal stromal tumors (LA-GIST) undergoing neoadjuvant imatinib therapy (NIT) remains unclear.
Objectives:
To evaluate the predictive value of the Systemic Inflammation-Nutrition Index (SINI) for pathological response and prognosis in patients with LA-GIST receiving NIT.
Designs:
A retrospective, two-center study was conducted to evaluate the prognostic value of high pathological response, and to develop a nomogram incorporating SINI for predicting high pathological response in patients with LA-GIST undergoing NIT.
Methods:
A retrospective analysis was performed on 200 patients with LA-GIST receiving NIT. Least Absolute Shrinkage and Selection Operator regression was used to construct the SINI from key hematological markers and to evaluate its association with relapse-free survival (RFS). A nomogram for predicting high response was established based on independent predictors identified by multivariate analysis, and the performance and prognostic values were validated in an external cohort.
Results:
High response was independently associated with improved 5-year RFS. The 5-year RFS showed a significant stepwise gradient across the low-, medium-, and high-SINI groups (46.0% vs 75.3% vs 94.8%, all p < 0.05). SINI independently predicted high response. A nomogram incorporating preoperative imatinib duration, tumor size, and SINI demonstrated good discrimination in both the training and validation cohorts (area under the curve = 0.798 and 0.849, respectively), with well-fitted calibration curves and favorable net clinical benefit per decision curve analysis. Patients predicted to have a low response showed significantly shorter RFS than those with a high response (p < 0.01).
Conclusion:
The SINI is an effective predictor of pathological response and prognosis in LA-GIST patients receiving NIT. The validated nomogram offers a practical tool for individualized treatment decision-making.
Keywords
Introduction
Gastrointestinal stromal tumors (GISTs) are the most common mesenchymal neoplasms of the digestive tract. 1 Surgical resection remains the only curative option for patients with resectable GISTs.2,3 However, in cases involving large tumors, anatomically complex locations, or close proximity to surrounding structures, surgery alone is often insufficient and associated with poor prognosis. In such scenarios, neoadjuvant imatinib therapy (NIT) followed by surgery is commonly recommended.4,5 NIT can reduce tumor volume, thereby increasing the likelihood of R0 resection, minimizing surgical extent, preserving organ function, and improving quality of life. Moreover, NIT suppresses tumor activity and reduces the risk of metastasis and iatrogenic dissemination.6 –8 Despite its benefits, the pathological response to NIT is highly variable, ranging from minimal to high therapeutic effects.9,10 Studies have shown that patients with locally advanced GIST (LA-GIST) who achieve high pathological response to NIT have significantly better prognoses than those who do not. 11 Therefore, the degree of pathological response following NIT is crucial for predicting prognosis and guiding individualized treatment strategies. Accurately identifying patients likely to achieve a high response to NIT holds significant clinical value.
Inflammation plays a pivotal role in tumor initiation and progression. As a key component of the tumor microenvironment, it affects pathological response and prognosis after neoadjuvant therapy by modulating the host’s systemic inflammatory status and nutritional metabolism.12,13 Peripheral blood tests provide a means to evaluate tumor-associated inflammation. Basic hematological parameters—including white blood cells (WBC), lymphocytes (LYM), neutrophils (NEU), and platelets (PLT)—and composite indices such as the platelet-to-lymphocyte ratio (PLR), neutrophil-to-lymphocyte ratio (NLR), and prognostic nutritional index (PNI) have been linked to prognosis and treatment response in various malignancies.14 –17 Ding et al. 11 reported that the systemic immune-inflammation index and PNI are significantly associated with pathological response and prognosis in patients with LA-GIST undergoing NIT. 11
This study employed pre-NIT systemic blood markers to select 5 key biomarkers from 22 candidates using Least Absolute Shrinkage and Selection Operator (LASSO) regression, which were used to develop the Systemic Inflammation-Nutrition Index (SINI). The predictive value of SINI for pathological response and prognosis in patients with LA-GIST receiving NIT was subsequently assessed. Furthermore, a nomogram incorporating SINI was constructed and externally validated to predict high pathological efficacy.
Materials and methods
Patients
This retrospective study included 200 patients with LA-GIST receiving NIT at Fujian Medical University Union Hospital (FMUUH) from May 2010 to January 2025 and Fujian Cancer Hospital (FCH) from November 2012 to January 2025. LA-GIST refers to a group of patients with tumors confined to the primary site without distant metastasis (M0), in whom curative surgery is rendered difficult by high-risk anatomical factors.18 –20 Inclusion criteria were (1) histopathological diagnosis of GIST; (2) preoperative assessment identifying one or more high-risk anatomical features, such as large tumor size, the need for multivisceral resection, and tumor location in anatomically complex sites associated with potential functional impairment (e.g., lower rectum, gastroesophageal junction, and duodenum). Exclusion criteria included (1) recurrent or metastatic disease; (2) presence of other malignancies; (3) incomplete clinical data. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: guidelines for reporting observational studies. 21
Treatment strategy
The NIT regimen was developed following a multidisciplinary team assessment. Treatment duration was guided by international authoritative guidelines, including National Comprehensive Cancer Network (NCCN), European Society for Medical Oncology (ESMO), and Asian consensus recommendations,18 –20 with regular imaging evaluations to optimize the treatment course. Surgical intervention was contingent upon meeting two criteria: (1) radiological evidence of disease stabilization, defined as no tumor volume change on two consecutive imaging assessments; and (2) feasibility of curative resection or preservation of organ function as determined. Regardless of pathological response, all patients were advised to initiate adjuvant imatinib therapy within 2–4 weeks postoperatively.
Data collection and definitions
Blood samples were collected within 2 weeks prior to the initiation of NIT, including measurements of WBC, NEU, PLT, fibrinogen (FIB), LYM, hemoglobin (HB), and albumin (ALB). For missing data, multiple imputation method was used for supplementation. Based on their established associations with cancer progression, these markers were classified into four upregulated indicators (WBC, NEU, PLT, and FIB) and three downregulated indicators (LYM, HB, and ALB). Fifteen composite inflammation-nutrition indices were subsequently calculated by deriving ratios between the upregulated and downregulated markers (Figure 1(a) and (b)).
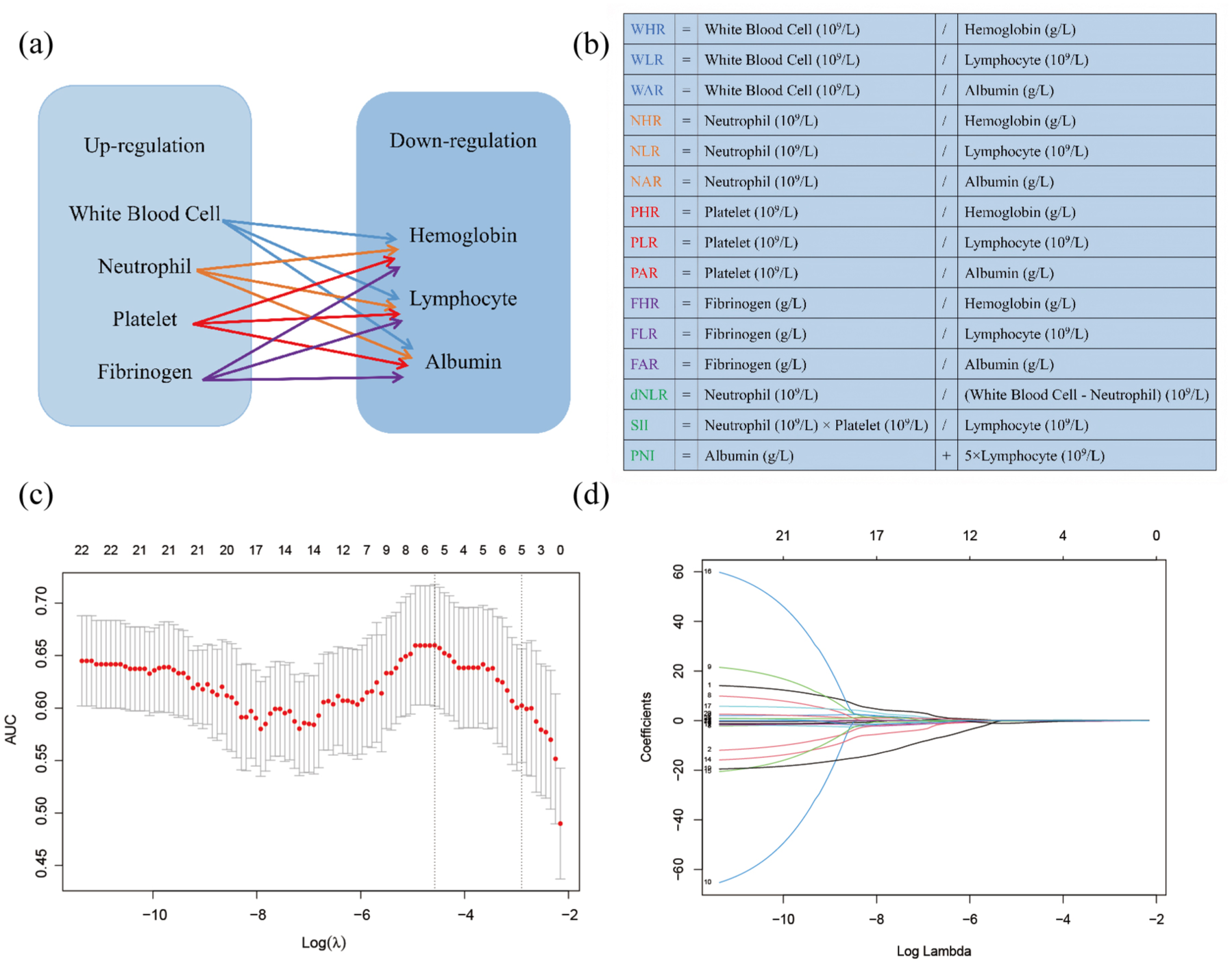
(a, b) Calculation methods for the 15 composite inflammatory markers. (c) LASSO coefficient profiles of the 22 indicators. (d) Ten-fold cross-validation for tuning parameter selection.
Clinical data were extracted from a prospectively maintained institutional database, including demographic characteristics, tumor-related features (tumor size and Ki-67 index before NIT, tumor site, and gene mutation), and treatment details. Based on the receiver operating characteristic (ROC) curve, the optimal cutoff value for the maximum tumor diameter before NIT was determined, categorizing patients into two groups: ⩽10 and >10 cm. The duration of NIT was classified, according to the NCCN and ESMO guidelines,18,19 into three categories: <6, ⩾6 to ⩽12, and >12 months. Radiological response was evaluated in accordance with the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. 22 Pathological response was assessed using criteria proposed by Antonescu et al., 9 based on the proportion of tumor necrosis and fibrosis, and interpreted according to the Chinese Guidelines for the Diagnosis and Treatment of GIST. 10 A fibrotic area occupying more than 90% of the tumor was defined as a high pathological response. All histological slides were independently reviewed and confirmed by two experienced pathologists in a double-blind manner.
Follow-up
During NIT, patients were followed up every 2–3 months. Postoperatively, follow-up was conducted every 3–4 months for the first 3 years, every 6 months during years 3–5, and annually thereafter. Follow-up assessments included abdominal and pelvic computed tomography or magnetic resonance imaging. The primary endpoint was relapse-free survival (RFS), defined as the interval from treatment initiation to either tumor recurrence or death from any cause. Patients without recurrence by the last follow-up were censored at that point. The final follow-up date was May 24, 2025, with a median follow-up duration of 50 months (range: 8–153 months). The loss to follow-up rate in this study was 2%, and patients who were lost to follow-up were considered censored.
Development and evaluation of the nomogram
Candidate variables were first screened using univariate logistic regression (p < 0.05), followed by multivariate stepwise logistic regression to identify independent predictors of high pathological response (p < 0.05), which were used to construct the nomogram. An independent dataset was used for external validation. The concordance index (C-index) was used to assess the model’s discriminatory ability, while calibration curves and the Hosmer–Lemeshow test were used to evaluate its predictive accuracy. 23 Decision curve analysis (DCA) quantified the net benefit across a range of risk thresholds, 24 and clinical impact curves (CICs) were generated to visualize the model’s effectiveness in identifying high-response individuals. 25 Construction and evaluation of the nomogram were performed in R using the “rms,” “caret,” “pROC,” and “rmda” packages.
Prognostic value of the model
To evaluate the prognostic utility of the nomogram for patients with LA-GIST, the optimal cutoff value was determined based on the maximum Youden index in the training cohort. Patients were classified into high-response and low-response groups, and differences in RFS between the two groups were analyzed across different cohorts.
Statistical analysis
All statistical analyses were conducted using R (version 4.4.2, The R Foundation for Statistical Computing, Vienna, Austria). The sample size was estimated using the 10 events per variable principle. Continuous variables were expressed as medians with interquartile ranges and compared using the Mann–Whitney U test. Categorical variables were presented as percentages (%) and compared using the Chi-square test or Fisher’s exact test, as appropriate. Pre-NIT hematological indicators were screened using LASSO regression with 10-fold cross-validation implemented via the “glmnet” package in R to eliminate multicollinearity, and subsequently used to construct the SINI classification system. The optimal cutoff for each marker was determined by maximizing the Youden index on the ROC curve. Variables significantly associated with a high pathological response were assigned 1 point; others were assigned 0. Total scores of 0–1, 2–3, and 4–5 defined the low-, medium-, and high-risk groups, respectively. The cumulative SINI score was calculated as follows: SINI score = LYM (0/1) + NEU to ALB ratio (NAR) (0/1) + PLT to HB ratio (PHR) (0/1) + PLR (0/1) + PNI (0/1). Differences in RFS between groups were analyzed using the Kaplan–Meier method and compared using the log-rank test. Kaplan–Meier survival curves were generated using the “survminer” and “survival” packages in R. A two-sided p-value < 0.05 was considered statistically significant.
Results
Baseline characteristics
A total of 200 patients were included in this study, comprising 135 from FMUUH (training cohort) and 65 from FCH (external validation cohort; Figure S1). Except for Ki-67 (p = 0.016), no statistically significant differences in clinicopathological characteristics were observed between the two cohorts (Table 1). The median age of the study population was 57 years, with 121 men (60.5%) and 79 women (39.5%). According to the RECIST 1.1 criteria for radiological response, 109 patients (54.5%) achieved partial response (PR), while pathological evaluation indicated that 61 patients (30.5%) attained a high response.
Baseline demographic and clinical characteristics of patients.

Two patients with PD.
The bold values indicate statistically significant p-values (p < 0.05).
BMI, body mass index; IM, imatinib; IQR, interquartile range; PD, progressive disease; PR, partial response; SD, stable disease.
Prognostic value of high pathological response
In the overall cohort, the 5-year RFS was 92.1% in patients with a high response and 58.8% in those without, with a statistically significant difference between the two groups (p < 0.001; Figure S2(A)). Similarly, in both the training and validation cohorts, patients with a high response exhibited significantly improved 5-year RFS compared to those without (p < 0.05; Figure S2(B) and (C)). Univariate analysis demonstrated that the preoperative imatinib (pre-IM) duration, number of lesions, Ki-67 index, tumor size, radiological response, and pathological efficacy were all significantly associated with 5-year RFS (p < 0.05). In multivariate analysis, pre-IM duration, number of lesions, Ki-67 index, radiological response, and pathological efficacy were identified as independent prognostic factors for 5-year RFS (Table S1).
Construction of the SINI and its prognostic value
LASSO regression analysis identified five key inflammation- and nutrition-related indices (Figure 1(c) and (d)). Compared with patients without a high pathological response, those with a high response had significantly higher LYM and PNI values, while NAR, PHR, and PLR were significantly lower (Figure S3). ROC analysis determined the optimal cutoff values for LYM, NAR, PHR, PLR, and PNI as 1.740 × 10⁹/L, 0.124, 2.332, 118.144, and 49.325, respectively (Table S2). Based on these thresholds, patients were stratified into high-risk and low-risk groups. A scoring system was then used to construct the SINI, which was further divided into three subgroups.
In the overall cohort, the 5-year RFS for the low-, medium-, and high-SINI groups were 46.0%, 75.3%, and 94.8%, respectively, with statistically significant differences among the three groups (p < 0.05; Figure 2(a)). Similar results were observed in the training cohort (p < 0.05; Figure 2(b)). In the validation cohort, 5-year RFS increased with higher SINI scores, although the difference between the medium-SINI group (72.6%) and the high-SINI group (91.7%) was not statistically significant (p = 0.266; Figure 2(c)).
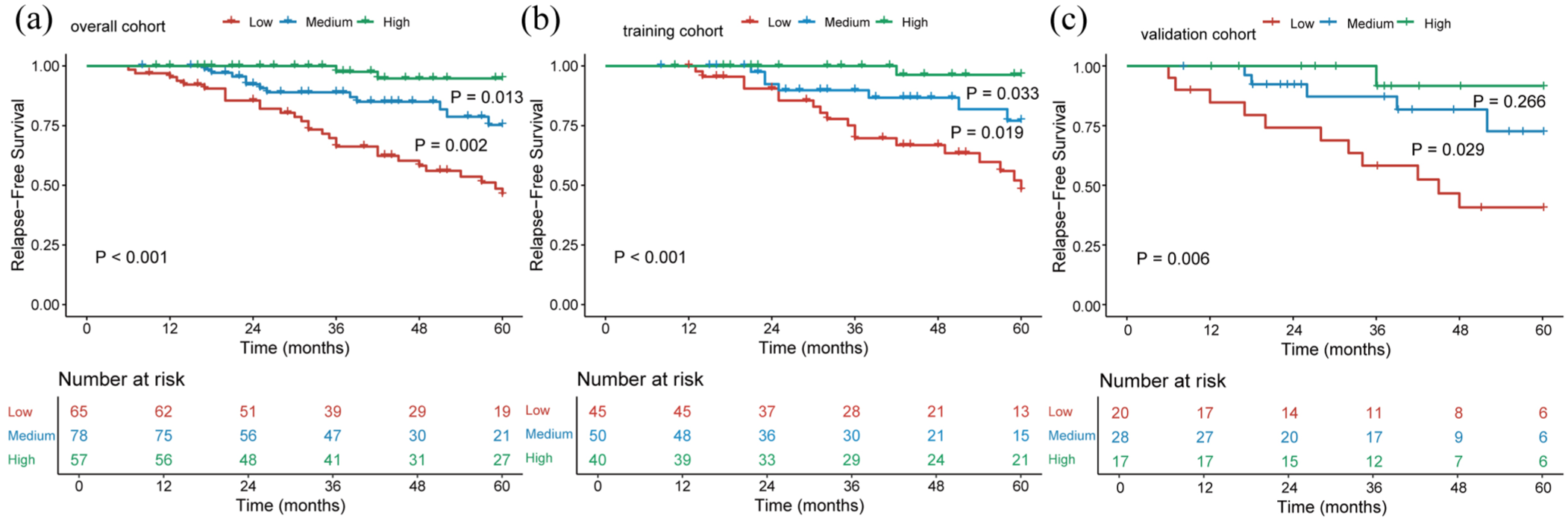
Kaplan–Meier analyses of RFS based on SINI score in the overall cohort (a), the training cohort (b), and the validation cohort (c).
Identification of predictive factors for high response
In the training cohort, univariate logistic regression analysis identified three variables significantly associated with a high pathological response: pre-IM duration, tumor size, and SINI score. Multivariate logistic regression analysis further confirmed that pre-IM duration, tumor size, and SINI score were independent predictors of high pathological response following NIT in patients with LA-GIST (Table 2).
Logistic univariate and multivariate analysis of predictors for high response.

The bold values indicate statistically significant p-values (p < 0.05).
BMI, body mass index; CI, confidence interval; IM, imatinib; OR, odds ratio; SINI, systemic inflammation and nutrition index.
Development and validation of the predictive nomogram
A nomogram for predicting high pathological response was constructed based on the results of multivariate logistic regression analysis (Figure 3(a)). An interactive web application for individualized patient prediction, based on the developed nomogram, is available at https://gist.shinyapps.io/my_shiny_app/ (accessed on August 12, 2025). The nomogram demonstrated good discriminatory ability, with area under the curve (AUC) values of 0.798 (95% confidence interval (CI), 0.716–0.879) in the training cohort and 0.849 (95% CI, 0.740–0.958) in the validation cohort (Figure 3(b) and (c)). Compared to individual predictors, the nomogram achieved superior performance in both cohorts, with higher AUC values than those for pre-IM duration (0.607, 95% CI, 0.517–0.698; 0.560, 95% CI, 0.434–0.686), maximum tumor diameter (0.675, 95% CI, 0.598–0.753; 0.675, 95% CI, 0.554–0.796), and SINI score (0.722, 95% CI, 0.635–0.809; 0.805, 95% CI, 0.686–0.924). Additionally, calibration curves (Figure 3(d) and (e)) demonstrated strong concordance between predicted and observed probabilities in both cohorts. The Hosmer–Lemeshow goodness-of-fit test indicated no significant deviation from ideal calibration (both p > 0.5).

Nomogram, ROC curves, and calibration curves for predicting a high response in patients with LA-GIST after NIT. (a) Three variables were included in the nomogram. (b, c) ROC curves of the nomogram and individual variables in the training and external validation cohorts are shown. (d, e) Calibration curves of the nomogram in the training and external validation cohorts are presented.
Clinical utility of the model
DCA (Figure S4(A) and (B)) demonstrated that the nomogram yielded a higher net clinical benefit across a broad range of threshold probabilities (0.01–0.76), with particularly pronounced advantages observed in the external validation cohort. Furthermore, the CIC (Figure S4(C) and (D)) showed strong alignment between the number of individuals predicted to be at high response and the actual number of events, indicating excellent model calibration. These results underscore the nomogram’s potential value in guiding clinical decision-making.
To evaluate its prognostic utility, individual response scores were calculated using the nomogram. The optimal cutoff value was determined through ROC curve analysis, based on which patients were stratified into high-response and low-response groups. Across the overall cohort, as well as in both the training and validation cohorts, the high-response group exhibited significantly superior 5-year RFS compared to the low-response group (all p < 0.01; Figure 4(a)–(c)), suggesting a clear survival benefit among LA-GIST patients with a higher probability of achieving a high pathological response. A Sankey diagram (Figure 4(d)) illustrated the relationships among pre-IM duration, tumor size, SINI score, pathological response, nomogram-predicted response group, and recurrence status in the overall cohort.

Kaplan–Meier survival curves for patients with LA-GIST in the overall cohort (a), training cohort (b), and validation cohort (c). The Sankey diagram (d) illustrates the relationships among tumor size, mitotic index, SINI score, treatment response, RFS, and nomogram-based response groups.
Discussion
The efficacy of NIT in LA-GIST has been well supported by previous studies. In the phase III BFR14 trial, 60% of 25 LA-GIST patients achieved a PR after a median NIT duration of 7.3 months. 26 A pooled analysis conducted by the EORTC-STBSG, involving 161 patients, reported an objective response rate of 80.1%, a disease control rate of 98.7%, and a 5-year overall survival rate of 87%. 27 For patients with LA-GIST, NIT can reduce tumor volume and clinical stage, facilitate organ-preserving surgeries by minimizing the extent of resection, and lower the risk of intra-operative rupture and peritoneal dissemination. Collectively, these benefits contribute to a higher rate of complete resection and improved survival outcomes.8,28 As a result, NIT has been endorsed as an optional treatment strategy for LA-GIST in international clinical guidelines.3,10,29 However, previous studies have shown that only 21.1% of LA-GIST patients achieve a high pathological response following NIT, 11 slightly lower than the rate observed in our study (30.5%). In addition, tumor progression during NIT has been documented, causing some patients to miss the optimal window for curative surgery, 30 which underscores the risk of response heterogeneity. In our study, pathological response was identified as an independent prognostic factor for RFS (p = 0.013). Using LASSO regression to select features from pre-NIT hematological parameters, we developed a novel composite indicator—the SINI—which was significantly associated with 5-year RFS. Furthermore, by integrating SINI with clinicopathological features, we established a preoperative predictive model for high response. This model effectively discriminated between high- and low-response groups (p < 0.001), enabling better selection of NIT candidates and optimization of surgical timing. As such, it offers a reliable tool for guiding individualized precision treatment.
Systemic inflammatory responses and nutritional-metabolic imbalances play pivotal roles in tumor progression, and their biological effects can be dynamically monitored via peripheral blood inflammatory markers.31,32 NEU facilitate extracellular matrix remodeling and angiogenesis by secreting matrix metalloproteinases (MMP8/9), vascular endothelial growth factor, and pro-inflammatory cytokines such as interleukin-8 (IL-8), thereby promoting tumor invasion and metastasis.33,34 LYM, as key effectors of antitumor immunity, when decreased in number, signify impaired immune surveillance and are strongly associated with poor prognosis.35,36 PLT regulate the tumor microenvironment’s transition from inflammation to malignancy by releasing growth factors and inflammatory mediators, with their activation closely linked to both tissue repair and the exacerbation of chronic inflammation.37,38 ALB, a core marker of nutritional status, when reduced, reflects compromised immune function and correlates with diminished anticancer treatment efficacy and adverse outcomes in gastrointestinal tumors.39,40 Additionally, hypoxia induced by anemia-driven low HB promotes tumor progression through reactive oxygen species accumulation. 41 Recent studies have validated that composite inflammatory and nutritional indices—including PLR, NLR, PNI, NAR, and PHR—dynamically reflect systemic inflammation intensity and nutritional status, effectively predicting long-term prognosis and treatment response in patients with solid tumors.42 –45 However, investigations into pre-NIT hematologic markers in LA-GIST remain scarce, and their associations with pathological response and prognostic value are yet to be systematically clarified. This study found that LYM, NAR, PHR, PLR, and PNI were all significantly associated with high pathological response following NIT (all p < 0.05). Accordingly, we developed a composite scoring system—SINI—whose three-tier stratification (low, medium, high) effectively distinguished patients’ long-term prognosis, with 5-year RFS of 46.0%, 75.3%, and 94.8%, respectively, demonstrating significant stepwise differences (all p < 0.05). Furthermore, SINI independently predicted high pathological response (p = 0.001). These findings suggest that SINI integrates features of inflammation activation, immune exhaustion, and nutritional imbalance, providing a comprehensive reflection of tumor microenvironment heterogeneity in LA-GIST patients. Patients with low SINI scores should undergo intensified imaging surveillance during NIT to facilitate early detection of primary resistance, whereas those with high SINI scores might benefit from extended NIT to achieve enhanced tumor downstaging.
Tumor size is a critical indicator of tumor burden and aggressiveness in GIST and has been integrated into established risk stratification systems. 46 In this study, it was identified for the first time as an independent predictor of pathological response in patients with LA-GIST receiving NIT (p = 0.012). Larger tumors, characterized by high proliferative activity and aberrant angiogenesis, often give rise to hypoxic and necrotic microenvironments,47,48 which may diminish treatment efficacy through two principal mechanisms: (1) greater tumor volume is associated with increased intratumoral heterogeneity and enrichment of resistant clonal subpopulations; and (2) hypoxic zones and impaired drug diffusion directly compromise IM efficacy. The optimal duration of NIT remains controversial. Our findings showed that treatment response plateaued between 6 and 12 months, consistent with previous studies suggesting a “window of maximal response.”10,49 Although extended treatment may enhance tumor shrinkage and improve surgical resectability, therapeutic benefits are limited beyond the plateau phase and may increase the risk of resistance or delay definitive surgery. These results support determining the duration of NIT based on individualized dynamic assessment, in order to maximize therapeutic efficacy while minimizing the risk of overtreatment.
A nomogram, as a user-friendly and intuitive clinical prediction tool, has demonstrated robust accuracy and discriminative capacity in estimating the probability of outcome events, and is thus widely applied in clinical practice. 50 Multivariate regression analysis identified pre-IM duration, tumor size, and SINI as independent predictors of high pathological response. Based on these variables, we developed a predictive model capable of quantitative scoring and precise estimation of the probability of achieving a high pathological response. This model exhibited good predictive performance in both the training and external validation cohorts, with C-indices of 0.798 and 0.849, respectively. Calibration curves demonstrated excellent concordance between predicted probabilities and actual observations. Moreover, DCA and CIC analyses further confirmed that the model provides substantial clinical net benefit within a reasonable range of threshold probabilities. When stratified using the nomogram-derived probabilities, LA-GIST patients were classified into high-response and low-response groups, with a significant difference in RFS observed between them. Patients in the high-response group exhibited notably superior prognoses. Collectively, these findings suggest that the model’s variable selection is appropriate and that it holds considerable clinical utility.
This study has several limitations. First, although the most commonly used pathological response assessment method for GIST was employed in this study, the relevant criteria have not yet been standardized internationally, underscoring the need for a unified scoring system. Second, although the study incorporated data from two high-volume centers, its retrospective design may introduce selection bias, highlighting the necessity for validation through large-scale, prospective, multicenter studies. Third, the absence of gene mutation data for some patients may limit a comprehensive evaluation of the relationship between specific gene mutations and treatment response. Finally, this study did not assess post-treatment tumor molecular characteristics or the tumor microenvironment, leaving the biological mechanisms underlying SINI’s predictive value unexplored. Future prospective studies integrating multi-omics approaches are warranted to elucidate the causal pathways through which inflammation-nutrition indices influence therapeutic response.
Conclusion
This study demonstrated that a high response is an independent prognostic factor for 5-year RFS in patients with LA-GIST. We developed a novel prognostic index, SINI, based on pre-NIT hematological parameters, which was significantly associated with 5-year RFS and served as an independent predictor of high response. The SINI-based predictive model showed promising accuracy in estimating the likelihood of achieving a high response following NIT and provides a quantitative tool to facilitate individualized treatment decision-making.
Supplemental Material
sj-doc-2-tam-10.1177_17588359251378286 – Supplemental material for The predictive value of the systemic inflammation-nutrition index for treatment response and prognosis in patients with locally advanced gastrointestinal stromal tumors receiving neoadjuvant imatinib therapy: a dual-center retrospective cohort study
Supplemental material, sj-doc-2-tam-10.1177_17588359251378286 for The predictive value of the systemic inflammation-nutrition index for treatment response and prognosis in patients with locally advanced gastrointestinal stromal tumors receiving neoadjuvant imatinib therapy: a dual-center retrospective cohort study by Zhiming Cai, Jinhu Chen, Zhenrong Yang, Lv Lin, Tao Lin, Xincheng Su, Shichai Hong, Weibin Song, Xinyu Chen and Yongjian Zhou in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-doc-3-tam-10.1177_17588359251378286 – Supplemental material for The predictive value of the systemic inflammation-nutrition index for treatment response and prognosis in patients with locally advanced gastrointestinal stromal tumors receiving neoadjuvant imatinib therapy: a dual-center retrospective cohort study
Supplemental material, sj-doc-3-tam-10.1177_17588359251378286 for The predictive value of the systemic inflammation-nutrition index for treatment response and prognosis in patients with locally advanced gastrointestinal stromal tumors receiving neoadjuvant imatinib therapy: a dual-center retrospective cohort study by Zhiming Cai, Jinhu Chen, Zhenrong Yang, Lv Lin, Tao Lin, Xincheng Su, Shichai Hong, Weibin Song, Xinyu Chen and Yongjian Zhou in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-1-tam-10.1177_17588359251378286 – Supplemental material for The predictive value of the systemic inflammation-nutrition index for treatment response and prognosis in patients with locally advanced gastrointestinal stromal tumors receiving neoadjuvant imatinib therapy: a dual-center retrospective cohort study
Supplemental material, sj-docx-1-tam-10.1177_17588359251378286 for The predictive value of the systemic inflammation-nutrition index for treatment response and prognosis in patients with locally advanced gastrointestinal stromal tumors receiving neoadjuvant imatinib therapy: a dual-center retrospective cohort study by Zhiming Cai, Jinhu Chen, Zhenrong Yang, Lv Lin, Tao Lin, Xincheng Su, Shichai Hong, Weibin Song, Xinyu Chen and Yongjian Zhou in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
