Abstract
Background:
We have previously demonstrated that carboplatin AUC10 is a viable alternative to cisplatin-based combination chemotherapy for patients with metastatic good-risk seminoma. The International Germ Cell Cancer Collaborative Group (IGCCCG) update identified lactate dehydrogenase (LDH) as an adverse prognostic marker in those receiving cisplatin-based therapies but its relevance in patients treated with carboplatin AUC10 is unknown.
Objectives:
To update survival outcomes of patients treated with carboplatin AUC10, explore patterns of treatment relapse and determine the impact of clinical and biochemical factors on oncological outcomes.
Design:
Multi-centre retrospective cohort study of 236 patients treated with carboplatin AUC10 from January 2000 to December 2021.
Methods:
Clinical parameters including age, stage, number of cycles delivered, primary tumour location, LDH level, relapse date, post-relapse treatment and cause of death were extracted. Progression-free survival (PFS) and overall survival (OS) were estimated using Kaplan–Meier method and event-time distributions were compared using the log-rank test. Variables predictive of PFS and OS were identified using Cox proportional hazards model.
Results:
The primary tumour site was testis in 95% (n = 225) of patients. Stage distribution was II (86%, n = 204) and III (14%, n = 32). Fourteen patients (6%) relapsed with 11 receiving salvage bleomycin, etoposide and cisplatin. Updated 5-year PFS and OS were 89% and 93%, respectively, over a median follow-up of 88 months. Twenty-three patients (10%) had LDH ⩾2.5 upper limit normal, with 5-year PFS and OS of 78% and 86%, respectively. Age was not associated with risk of relapse. The addition of a fourth cycle of carboplatin was not associated with better outcomes even after adjusting for other risk factors on multivariate analysis. Approximately 50% of relapsed patients were successfully salvaged.
Conclusion:
Carboplatin AUC10 remains an effective de-escalation regimen for metastatic seminoma, with robust survival rates at prolonged follow-up. Elevated LDH indicates poorer oncological outcomes with carboplatin AUC10, though survival remained comparable to cisplatin-based chemotherapy.
Plain Language Summary
Keywords
Background
Testicular germ cell tumours (GCT) are the most common solid malignancy affecting young men between 15 and 35 years of age. 1 Testicular GCT are broadly divided into seminoma and non-seminomatous germ cell tumours (NSGCT). Seminoma is distinguished by its slower progression and higher age at presentation than its counterpart NSGCT and has an excellent response to treatment whether chemotherapy or radiotherapy making it highly curable in most cases. 2
Cisplatin is the cornerstone of treatment for metastatic GCT and has revolutionised their management. Cisplatin forms the backbone of first-line regimens like bleomycin, etoposide and cisplatin (BEP) 3 and etoposide and cisplatin (EP) for patients unlikely to tolerate for bleomycin4 –6 establishing it as standard care for metastatic good-risk seminoma. For metastatic good-risk seminoma, the International Germ Cell Cancer Collaborative Group (IGCCCG) Update reports a 5-year overall survival (OS) of 95%. 7
Despite the effectiveness of BEP and EP, their acute and long-term toxicities remain significant. Common acute toxicities of BEP include myelosuppression, nausea, fatigue and febrile neutropenia, with pulmonary toxicity primarily linked to bleomycin. Bleomycin-induced pneumonitis, a rare but serious complication, affecting 1%–3% of patients, typically presents shortly after therapy. 8 The risk is higher in those with poor kidney function, older age, stage IV disease and higher cumulative bleomycin doses. 9 Long-term toxicities include peripheral neuropathy, ototoxicity, secondary malignancies, cardiovascular, renal and pulmonary toxicity and hypogonadism which can impair fertility, contribute to metabolic syndrome and lower overall quality of life.10,11
De-escalation therapy was introduced in metastatic good-risk seminoma to minimise toxicity while preserving cure rates. Carboplatin AUC7 monotherapy was investigated as an alternative regimen to BEP/EP but was found to have significantly inferior outcome, with relapse rate of 18%. 12 Subsequently, using higher dose carboplatin AUC10, our group demonstrated a 3-year progression-free survival (PFS) of 93.2% in a study of 61 patients. 13 In an expanded retrospective analysis of 216 patients treated with carboplatin AUC10, we observed 3-year PFS of 96.5%, and the 5-year disease-specific survival of 98.3%, with a third experiencing grade >3 haematological toxicity. 14 In light of these outcomes, we now offer carboplatin AUC10 as a standard of care (SOC) for IGCCCG metastatic good-risk seminoma.
The recent IGCCCG update demonstrated that patients previously classified as having a good prognosis (nodal disease with or without pulmonary metastases) could be further stratified based on lactate dehydrogenase (LDH) levels. Specifically, patients were divided into two subgroups: those with LDH <2.5 times the upper limit of normal (ULN) and those with LDH ⩾2.5×ULN. In the elevated LDH subgroup, prognosis was poorer, with a PFS of 80% following first-line cisplatin-based combination chemotherapy compared to 92% in the <2.5×ULN group but the relevance of LDH in patients treated with carboplatin AUC10 is unknown.
Here, we provide an updated survival analysis for our patients treated with carboplatin AUC10. In particular we wished to ascertain if the presence of an elevated LDH (⩾2.5×ULN) was associated with poorer survival outcomes and how the survival outcomes compared to those treated with combination cisplatin-based chemotherapy. Further to that, we aim to analyse patient and treatment-related parameters which may influence response to carboplatin AUC10, as well as treatment outcomes following relapse after carboplatin AUC10.
Methodology
Methods and patients
Retrospective data was collected from all patients with metastatic good-risk seminoma who received carboplatin AUC10 between January 2000 and December 2021, with the data cutoff set for 31 December 2023. This cohort included patients from the original pilot study (n = 20), 15 the CARPET trial (n = 48) 16 and all patients treated with carboplatin AUC10 thereafter.
Data were gathered from St Bartholomew’s Hospital (SBH) and Mount Vernon Cancer Centre (MVCC). Electronic healthcare systems were utilised to collect all the data. The collected dataset includes age, primary tumour location, disease stage according to eight edition American Joint Committee on Cancer TNM system, tumour marker (LDH, alpha-fetoprotein (AFP), beta-human chorionic gonadotropin (β-HCG)), number of cycles delivered, incidence of relapse, types of salvage chemotherapy, survival status and the documented cause of death. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement 17 (Supplemental Material 1).
Eligibility
All patients had a histologically confirmed diagnosis of metastatic good-risk seminoma as defined by the IGCCCG criteria. Inclusion criteria required patients to be male, aged 18–80 years, with a performance status of 0–2. Exclusion criteria included prior chemotherapy or radiotherapy, and elevated AFP levels.
Treatment regimen
Following a specialist multidisciplinary team review of imaging and pathology, patients were offered carboplatin AUC10 as an alternative to BEP/EP. Carboplatin was administered every 21 days and delivered as a 1-h outpatient infusion. Carboplatin was dosed at AUC10 based on Calvert formula (total dose in mg = target AUC (glomerular filtration rate ml/min + 25)). Filgrastim was not routinely given as primary prophylaxis but was added if neutropenia caused a delay in the next cycle or if febrile neutropenia occurred.
Follow-up
Patients were followed up every 2 months in the first year, every 3 months in the second year, every 4 months in the third year, every 6 months in years 4 and 5 and annually from year 6 onwards. A CT scan was performed at the end of both year 1 and year 2. Follow-up blood tests at each visit include a full blood count, renal function, liver function tests and serum tumour markers (AFP, HCG, LDH). Patients with residual disease larger than 3 cm following a period of observation were advised to proceed with retroperitoneal lymph node dissection (RPLND), in accordance with current practices. If a complete response was achieved, a final scan was performed at the end of year 2 (see follow-up protocol in Supplemental Material 2).
Statistical analysis
Statistical analysis was conducted using STATA MP version 18. Descriptive analysis was performed to examine the demographic characteristics. Two survival outcomes namely OS and PFS were reported. PFS was defined as the time from the first day of treatment to either disease progression or death from any cause, whichever occurred first while OS was defined as the time from the first day of treatment until death from any cause. 5-year OS and PFS were estimated using the Kaplan–Meier approach. We also performed univariable (UVA) and multivariable analyses (MVA) using Cox regression to examine association between the variables which were age at diagnosis, number of cycles, stage and LDH and the risk of relapse. In the UVA, each variable was examined separately for its association with the risk of relapse. In the MVA, the association between the number of cycles and LDH level with risk of relapse was assessed after adjusting for other variables in the model. Age was dichotomised at 40 years, a threshold supported by prior evidence showing worse outcomes in metastatic GCTs in patients aged ⩾40. 18
Results
Data were collected from 236 male patients treated at SBH (n = 187) and MVCC (n = 49) between January 2000 and December 2021, with a cutoff date of 31 December 2023. The median follow-up was 88 months. The median age at diagnosis was 39 years (range 22–76). The majority (95%) had a testicular primary, while other primary sites included retroperitoneal (3%) and other extragonadal (2%) locations. The baseline characteristics are shown in Table 1.
Baseline characteristics.
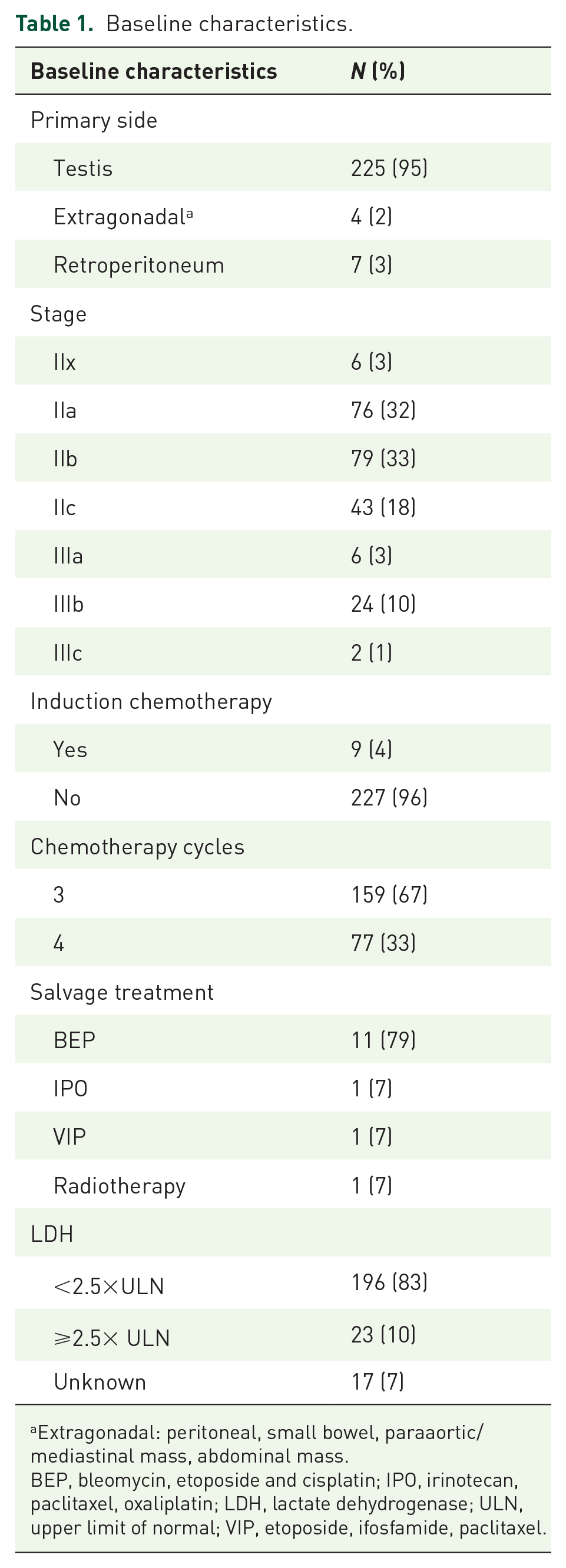
Extragonadal: peritoneal, small bowel, paraaortic/mediastinal mass, abdominal mass.
BEP, bleomycin, etoposide and cisplatin; IPO, irinotecan, paclitaxel, oxaliplatin; LDH, lactate dehydrogenase; ULN, upper limit of normal; VIP, etoposide, ifosfamide, paclitaxel.
There were 204 (86%) patients with stage II and 32 (14%) patients with stage III disease. Among those with stage II disease, 76 (32%) were classified as stage IIa, 79 (33%) as stage IIb and 43 (18%) as stage IIc. 159 (67%) patients received three cycles of carboplatin AUC10 and 77 (33%) patients received four cycles.
At the time of analysis, 14 (6%) patients had experienced relapse. Among these 14 patients, 7 remains progression-free. BEP was the first-line salvage chemotherapy regimen for 11 (79%) patients. All relapses occurred within 2 years, except for one case, where the patient relapsed 88 months later (patient 8). Of the patients who relapsed, three patients were cured by BEP alone. The overall salvage rate after carboplatin AUC10 was 50%. The outcomes of patients who relapsed are summarised in Table 2.
Summary of patients who relapsed, detailing first-line salvage treatments at relapse and patient outcomes.
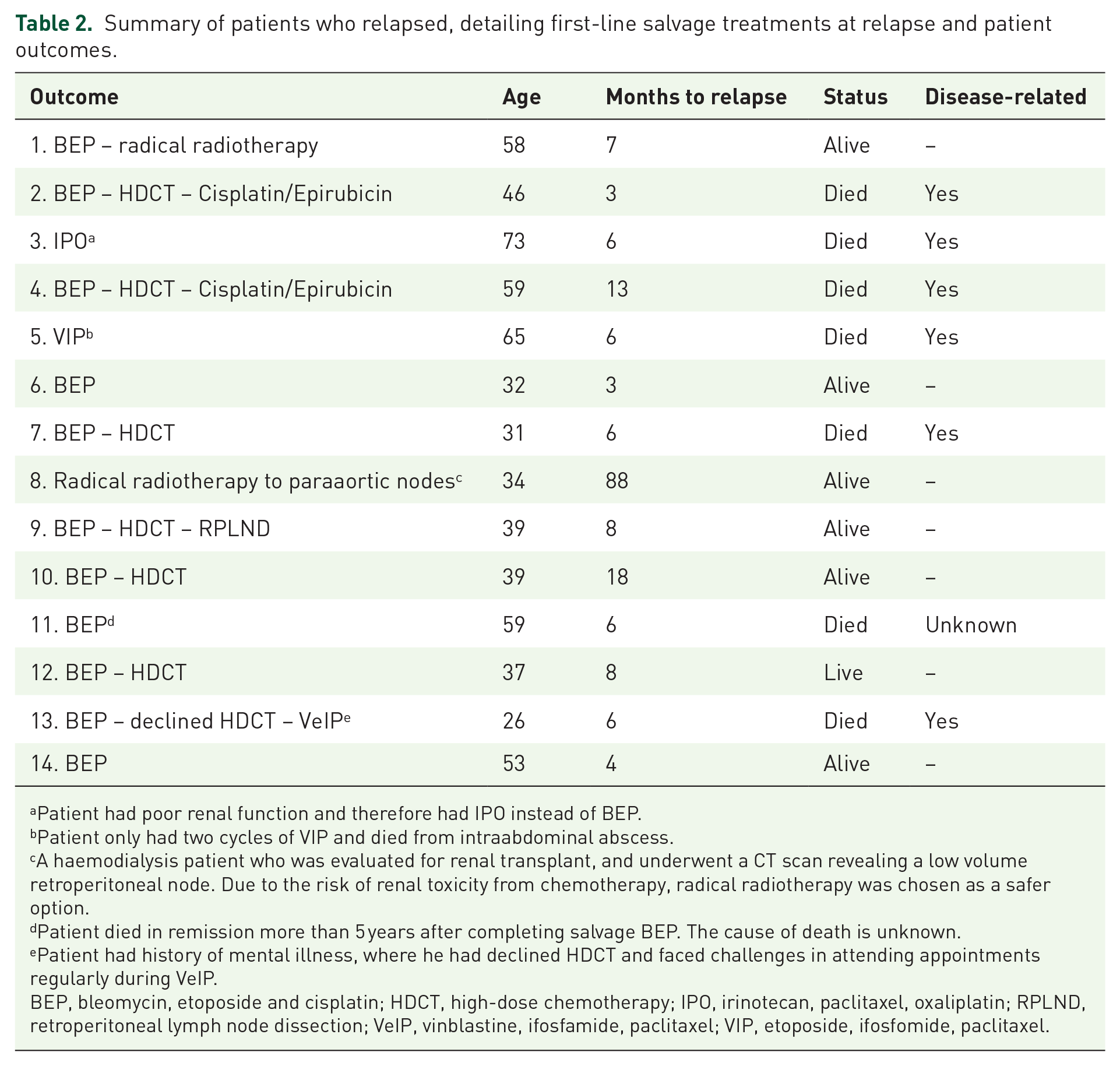
Patient had poor renal function and therefore had IPO instead of BEP.
Patient only had two cycles of VIP and died from intraabdominal abscess.
A haemodialysis patient who was evaluated for renal transplant, and underwent a CT scan revealing a low volume retroperitoneal node. Due to the risk of renal toxicity from chemotherapy, radical radiotherapy was chosen as a safer option.
Patient died in remission more than 5 years after completing salvage BEP. The cause of death is unknown.
Patient had history of mental illness, where he had declined HDCT and faced challenges in attending appointments regularly during VeIP.
BEP, bleomycin, etoposide and cisplatin; HDCT, high-dose chemotherapy; IPO, irinotecan, paclitaxel, oxaliplatin; RPLND, retroperitoneal lymph node dissection; VeIP, vinblastine, ifosfamide, paclitaxel; VIP, etoposide, ifosfomide, paclitaxel.
At the time of analysis, 16 patients (7%) had died. Among those who died, 6 (38%) deaths were related to testicular cancer, while 7 (44%) deaths were due to non-testicular cancer-related causes, including non-Hodgkin lymphoma (n = 1), chronic disseminated intravascular coagulation (n = 1), hepatitis C/intravenous drug use (n = 1), vascular disease (n = 3) and colorectal cancer (n = 1).
LDH levels were <2.5×ULN in 196 (83%) patients, while 23 (10%) had LDH levels ⩾2.5×ULN. LDH data were missing for 17 of 236 patients (7%). LDH ⩾2.5×ULN was associated with advanced disease, observed in 47% of stage III patients versus 4% in stage II. LDH ⩾2.5×ULN was more frequent in patients receiving four cycles (24%) compared to three cycles (4%; Table 3). Both disease stage and number of cycles delivered showed statistically significant associations with LDH level (number of cycles delivered: p < 0.01; disease stage: p < 0.01).
LDH level based on stage and number of cycles.
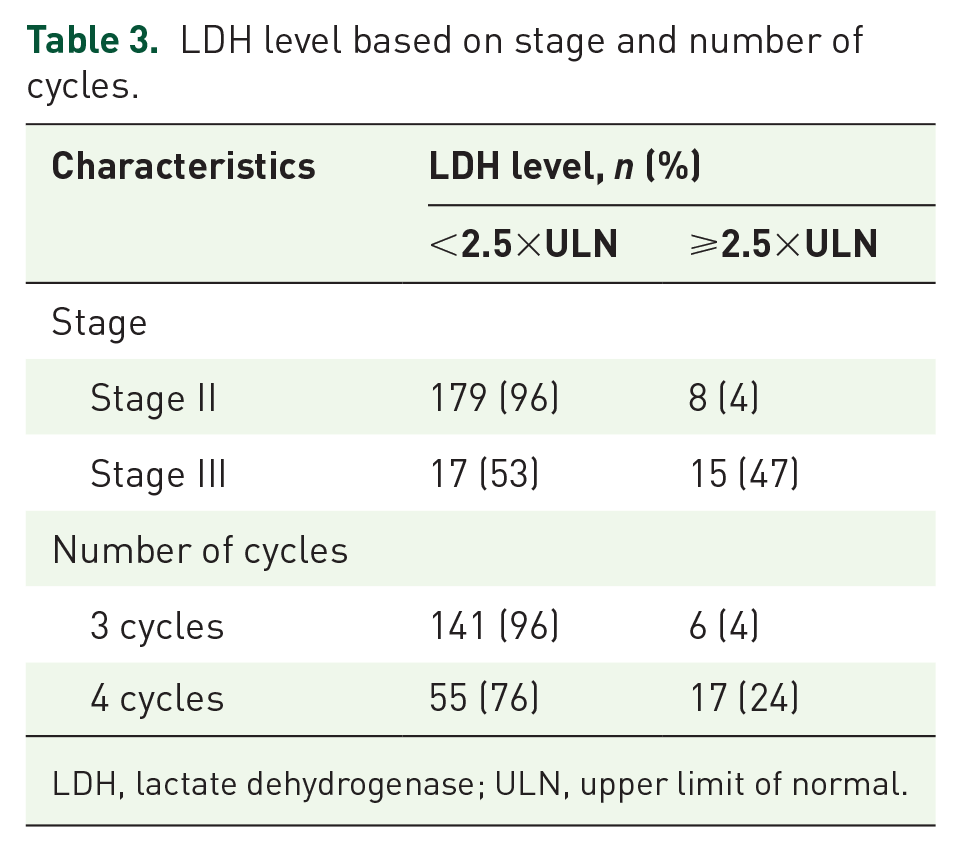
LDH, lactate dehydrogenase; ULN, upper limit of normal.
Survival analysis
Among the 236 patients recorded, only 219 were included in the survival analysis due to 17 missing LDH data points. The 5-year PFS was 89% (95% confidence interval (CI): 84%–93%; Figure 1). The 5-year OS was 93% (95% CI: 89%–96%; Figure 2). The 5-year PFS for LDH ⩾2.5×ULN was 78% (95% CI: 55%–90%, p = 0.0334; Figure 3), while the 5-year OS for LDH ⩾2.5×ULN was 86% (95% CI: 65%–96%, p = 0.0451; Figure 4).
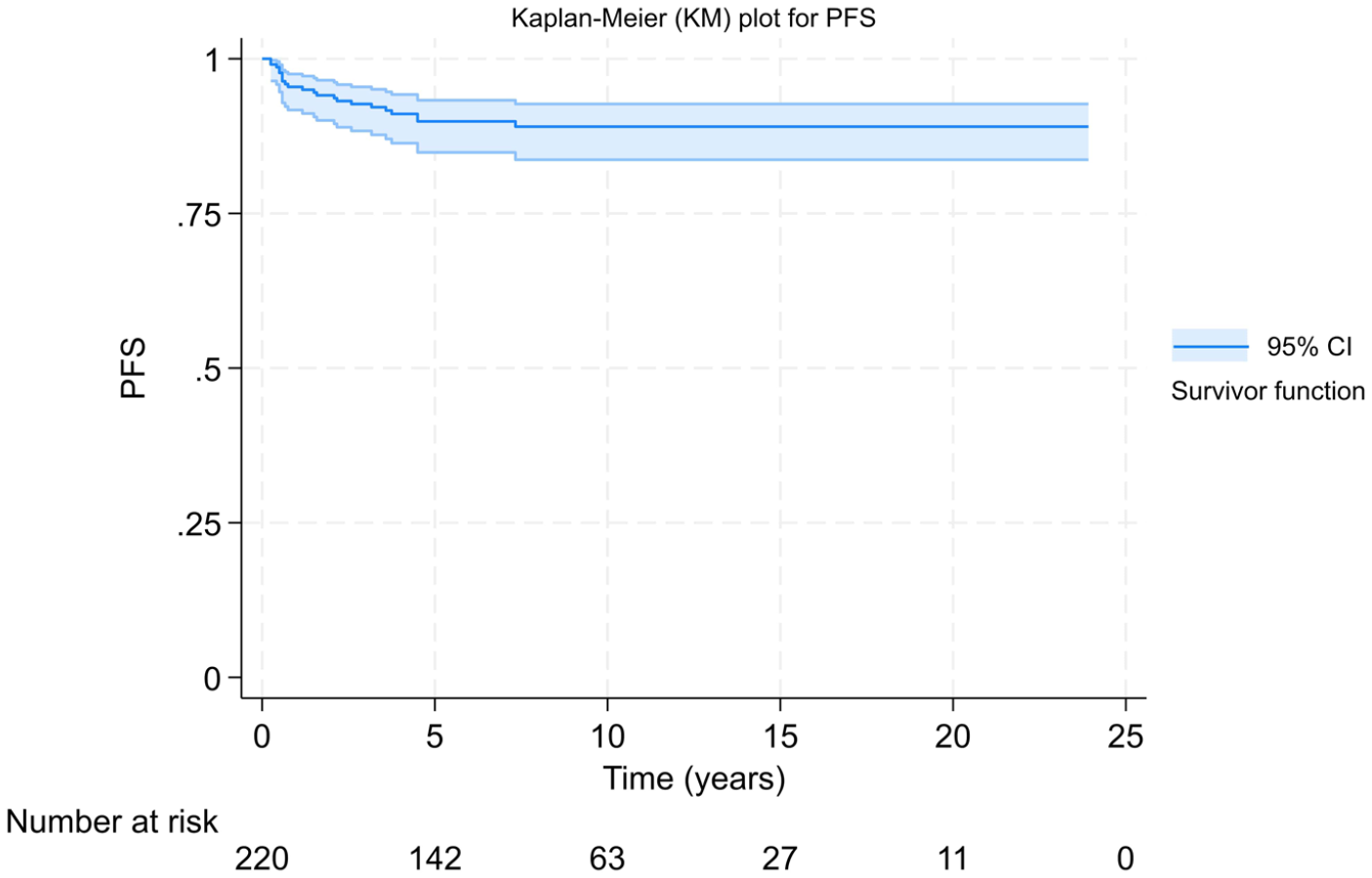
Kaplan–Meier curve showing PFS.

Kaplan–Meier curve showing OS.

Kaplan–Meier curve showing PFS stratified by LDH levels.

Kaplan–Meier curve showing OS stratified by LDH levels.
Univariate and multivariate analyses
In the UVA, Stage 3 disease (hazard ratio (HR) = 4.10, 95% CI: 1.752–9.569, p = 0.001), receiving 4 treatment cycles (HR = 2.84, 95% CI: 1.211–6.674, p = 0.016) and having an LDH level ⩾2.5×ULN (HR = 2.80, 95% CI: (1.032–7.596), p = 0.043) were each significantly associated with an increased risk of relapse. In the MVA, receiving 4 treatment cycles (HR = 2.019, 95% CI: (0.796–5.118), p = 0.139) and LDH level ⩾2.5×ULN (HR = 1.044, 95% CI: (0.318–3.427), p = 0.944) were no longer statistically significant associated with risk of relapse after adjustment. Age at diagnosis showed no significant association with risk of relapse. These results are presented in Table 4.
Results of univariate and multivariate analyses for risk of relapse.

CI, confidence interval; HR, hazard ratio; LDH, lactate dehydrogenase; ULN, upper limit of normal.
Discussion
There is growing international interest in de-escalation strategies for metastatic good-risk seminoma to reduce long-term toxicities while maintaining oncological outcomes. The SEMITEP trial used two cycles of EP followed by PET/CT to guide further treatment with either one cycle of carboplatin or two additional cycles of EP, achieving a 3-year PFS of 90% across both groups. 19 The SAKK 01/18 trial combined a single cycle of EP with involved-node radiotherapy, reporting a 3-year PFS of 93.7% with minimal toxicity. 20 Surgical approaches such as RPLND are also promising; the SEMS and COTRIMS trials reported 2-year recurrence-free survival rates of 80%–90%, with 100% OS at last follow-up.21,22 None of these strategies has yet been adopted as an internationally recognised SOC. Treatment strategies will continue to evolve towards more individualised approaches.
De-escalation strategy with monotherapy carboplatin AUC10 has become the institutional SOC in our centre for patients with metastatic good-risk seminoma as an alternative to cisplatin-based chemotherapy. The IGCCCG Update recently reported updated survival outcomes in the cisplatin-based chemotherapy cohort, with 5-year PFS and OS rates of 89% and 95%, respectively. 7 These outcomes are comparable to those in our carboplatin AUC10 cohort, which had a 5-year PFS and OS of 89% and 93%, respectively. Besides being reasonably close efficacy to cisplatin-based chemotherapy, carboplatin AUC10 has also been associated with a better health-related quality of life than combination chemotherapy. 23
The IGCCCG Update identified elevated LDH as an adverse prognostic factor in the cisplatin-based chemotherapy cohort, with patients exhibiting LDH ⩾2.5×ULN demonstrating reduced survival outcomes. Specifically, the 3-year PFS and OS rates were 80% and 92%, respectively. 7 These findings are comparable with our analysis, which demonstrated 5-year PFS and OS rates of 78% and 86%, respectively. Both UVA and MVA in our study confirmed that serum LDH remains a robust prognostic factor, although statistical significance was not achieved in the multivariate analysis. This lack of significance is likely attributable to the small number of events.
A retrospective analysis of a first-line cisplatin-based regimen by Fléchon et al. 24 showed that the highest risk of relapse occurred within the first 18 months, with 85% of relapses happening during this period and a median time to relapse of 6 months. In our cohort, 13 patient (93%) relapses occurred within the first 2 years. Additionally, in our cohort our relapse rate is 6% which is the same as reported in the IGCCCG Update. These findings suggest that carboplatin AUC10 exhibits relapse characteristics comparable to BEP/EP, with most patients relapsing within the first 2 years of treatment.
Cisplatin-based chemotherapy is typically preferred for bulky disease in good-risk (stage IIc and above) due to its high cure rates25,26 as recommended by SOC given concerns about relapse and the suboptimal outcomes with radiotherapy. 27 Domont et al. 28 reported that the 5-year relapse-free survival decreased from 83% in non-bulky cases to 44% in bulky cases treated with radiotherapy. The Swedish Norwegian Testicular Cancer Study Group observed that, despite using standard regimens such as BEP/EP, relapse rates in bulky disease (14.9%) were higher compared to Clinical Stage IIa/b cases. 29 In our cohort, despite having 32% of patients with bulky disease, favourable relapse outcomes were achieved even with this de-escalated regimen, similar to the cisplatin-based chemotherapy.
Among the 14 patients in our cohort who experienced relapse, 50% achieved long-term remission with salvage therapy, consistent with published outcomes in relapsed seminoma, including those previously treated with first-line BEP.30 –32 Among the 11 patients who received salvage therapy, 3 (27%) were successfully treated with BEP alone. Whether a more intensive regimen is necessary in this setting remains unclear and warrants further investigation, as the optimal salvage approach for relapsed GCTs has yet to be defined. Both conventional-dose chemotherapy and high-dose chemotherapy continue to be widely used in clinical practice.
A limitation of our cohort is that some patients received three or four cycles of carboplatin AUC10. Previously, four cycles of carboplatin AUC10 were administered for bulky metastatic good-risk seminoma. However, following evidence from the CARPET trial, 16 which demonstrated that haematological toxicities mainly occurred in the fourth cycle, standard practice shifted towards adoption of a 3-cycle regimen. By retrospectively analysing outcomes across the entire cohort of patients treated with carboplatin AUC10, MVA showed no strong evidence of a meaningful difference in outcomes between patients receiving three versus four cycles. These findings reassure that three cycles of carboplatin AUC10 are sufficient for effective management for metastatic good-risk seminoma, especially as this approach helps minimise haematological toxicity for patients. The inclusion and analysis of treatment cycles in this context represents a novel aspect of our study.
Our study is limited by its retrospective design. Although the initial patient cohort was larger, cases with incomplete data had to be excluded. Additionally, our study has a lower proportion of patients with elevated LDH levels (10%) compared to the 21% reported in the IGCCCG Update cohort, which may limit the generalisability of our findings to broader populations.
Conclusion
To our knowledge, our study represents the largest series of seminoma treated with carboplatin AUC10 and first-ever study that looked into the impact of LDH on carboplatin AUC10 cohort. Our study supports that oncological outcomes with carboplatin AUC10 are comparable to those achieved with cisplatin-based chemotherapy. While we observed a trend towards poorer outcomes in patients with LDH ⩾2.5×ULN, this was not statistically significant in the MVA. Importantly, our results indicate that three cycles of carboplatin AUC10 are sufficient for good-risk seminoma. This is also the first study demonstrating treatment outcomes after relapse on carboplatin AUC10, with significant patients requiring high-dose chemotherapy.
Carboplatin AUC10 offers a promising alternative to cisplatin-based chemotherapy, particularly for patients seeking a regimen with lower toxicity. Further investigation of this de-escalation monotherapy approach through a larger-scale prospective study is warranted.
Supplemental Material
sj-doc-1-tam-10.1177_17588359251369040 – Supplemental material for Treatment de-escalation for metastatic good-risk seminoma with carboplatin AUC10: predictive factors and patterns of relapse
Supplemental material, sj-doc-1-tam-10.1177_17588359251369040 for Treatment de-escalation for metastatic good-risk seminoma with carboplatin AUC10: predictive factors and patterns of relapse by Nasreen Abdul Aziz, Didjier Danger Masangwi, Sushila Ganguli, Anand Sharma, Kenrick Ng, Prabhakar Rajan and Jonathan Shamash in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-doc-2-tam-10.1177_17588359251369040 – Supplemental material for Treatment de-escalation for metastatic good-risk seminoma with carboplatin AUC10: predictive factors and patterns of relapse
Supplemental material, sj-doc-2-tam-10.1177_17588359251369040 for Treatment de-escalation for metastatic good-risk seminoma with carboplatin AUC10: predictive factors and patterns of relapse by Nasreen Abdul Aziz, Didjier Danger Masangwi, Sushila Ganguli, Anand Sharma, Kenrick Ng, Prabhakar Rajan and Jonathan Shamash in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We would like to thank the clinical teams at St Bartholomew’s Hospital and Mount Vernon Cancer Centre for their support in facilitating access to patient records and data used in this study. Most importantly, we sincerely thank the patients whose data made this research possible.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
