Abstract
Alcohol-associated hepatitis is an extreme form of alcohol-related liver disease associated with high short-term mortality. Currently, there are no authorized therapies for the treatment of severe alcohol-associated hepatitis. Important diagnostic steps for alcohol-associated hepatitis include recognizing the presence of an alcohol use disorder, distinguishing alcohol-related liver disease from metabolic-dysfunction-associated steatotic liver disease, ruling out alternative causes of acute hepatitis, confirming the diagnosis with validated criteria or a liver biopsy, and using the model for end-stage liver disease score to predict clinical outcome and initiate therapy. Due to the lack of other effective therapy options, corticosteroids continue to be used as initial treatment for patients with severe alcohol-associated hepatitis. Patients who do not improve while on steroid treatment and are ideal candidates should be considered for curative liver transplantation as soon as possible. Avoiding unnecessary and ineffective pharmacological and interventional therapy can help to keep costs down. If a patient is not a good candidate for a transplant or is rapidly deteriorating in health due to a condition such as acute or chronic liver failure, a salvage/bridge to transplant should be pursued through enrolment in a clinical trial program. The role of healthy donor stool transplant and targeted bacteriophage therapy seems promising, pending prospective controlled trials.
Keywords
Alcohol-related liver disease: An introduction
Alcohol-related liver disease (ALD) spans the spectrum of liver steatosis, steatohepatitis, alcohol-associated hepatitis (AAH), alcohol-related cirrhosis (ALC), and the acute on chronic liver failure (ACLF) syndrome secondary to AAH. 1 The development of ALD depends not only on the dose, frequency, and duration of alcohol consumption but also on other factors such as gender, presence of metabolic traits, associated smoking, and genetic predisposition.2,3 A patient does not progress linearly through these stages and can present inadvertently at any stage, including AAH-ACLF as the index presentation. Women are at higher risk of developing liver injury due to alcohol use with rapid progression to fibrosis than men. Early studies showed that the relative risk (RR) of developing ALC in women was 17.0 compared to 7.0 in men who consumed the same amount. 4 Similarly, drinking 40 g/day showed RRs of 9.35 in women and 2.82 inmen, while drinking 80 g/day presented RRs of 23.32 in women and 7.93 in men in a recent study. 5 Dietary components and food consumption patterns also determine liver injury from alcohol use. Pre-clinical studies have shown that diets rich in saturated fats and feeding relative to the maximum blood alcohol level were found to protect against alcohol-induced liver injury, possibly due to the maintenance of the indigenous gut microbiome and its beneficial metabolites.6,7 However, subsequent studies in both animal models as well as humans have maintained that a high-fat diet with heavy alcohol use was associated with increased liver steatosis and inflammation.8,9 In humans, genome-wide association studies have shown that modifications of PNPLA3, TM6SF2, and MBOAT7 were associated with a higher risk of alcohol-induced liver injury and ALC.3,10 Additionally, metabolic syndrome (obesity, diabetes, hypertension, dyslipidemia, or cardiovascular disease) increases the risk of ALD development even with mild-to-moderate drinking. 11
The first change in the liver that occurs in ALD is steatosis. Steatosis (alcohol-related fatty liver) can occur in 90% of patients with significant drinking, over 60 g/day over decades or in heavy drinkers who consume alcohol in the range of 120–150 g/day for 2–3 weeks.12,13 To reiterate, one standard is equivalent to 10–14 g of pure alcohol (depending on the region)—which equates to 12 ounces (350 ml) of beer/8 ounces (235 ml) of malt liquor/5 ounces (150 ml) of wine or 1.5 ounces (45 ml) of distilled spirit. 13 Steatosis typically begins as small and large droplets of fat in the hepatocytes surrounding the central veins (perivenular, zone 3), which then progresses outward to involve the lobular hepatocytes and, finally, the periportal hepatocytes with increasing severity of the disease and continued use of heavy alcohol.14,15 In those who continue to use alcohol, close to 40% progress to fibrosis and another 20% develop cirrhosis without abstinence. It is pertinent to realize that AAH can occur tangential to the natural progression of ALD. For example, patients with relatively mild fibrosis can develop AAH, as can those with established cirrhosis. AAH is an acute insult usually on a background of progressive ALD. Nonetheless, the true incidence of AAH remains unknown, although a prevalence of approximately 20% among people with alcohol use disorder can be considered reasonable based on liver biopsy-based cohort studies. Twenty to 40% of patients with ALC without abstinence develop decompensation, while 3%–10% are at risk of developing hepatocellular carcinoma (Figure 1).16,17
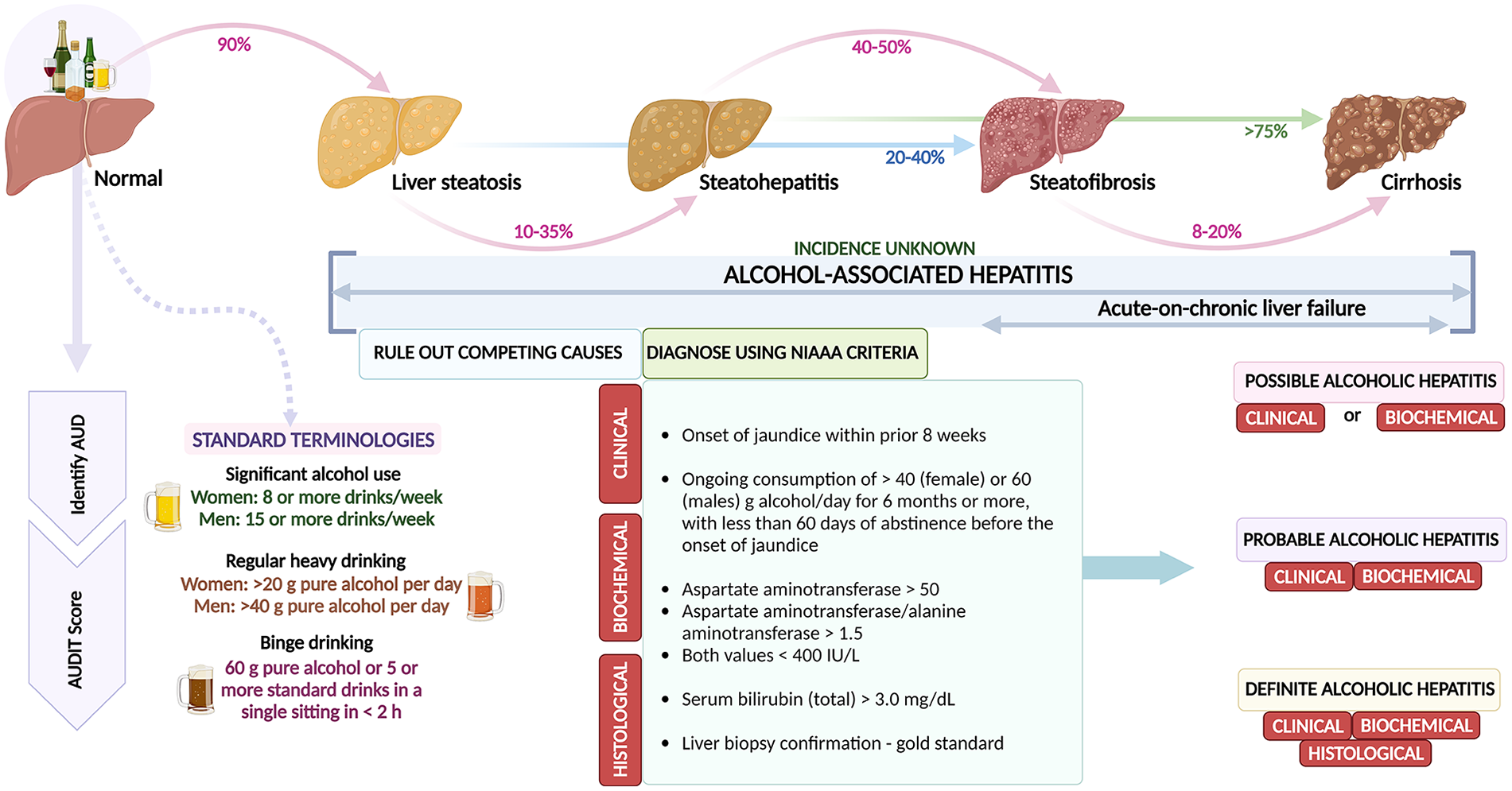
Natural history of alcohol-related liver disease and salient features of identification and diagnosis of AAH.
Diagnosis of ALD
The first step in diagnosing ALD is to assess significant alcohol use and confirm the presence or absence of alcohol use disorder (AUD). When a patient with AUD has liver steatosis on ultrasound or elevated liver enzyme levels (aspartate transaminase (AST) > alanine transaminase (ALT)), serum bilirubin < 3 mg/dl, in the absence of other competing causes, it is determined that the patient has alcohol-related fatty liver disease. Pertinent steps include identifying the age at which alcohol consumption was initiated, the total duration of consumption, the last date of consumption, the pattern of drinking, and concurrent substance abuse. Ideally, the identification of harmful alcohol use and dependence on alcohol is performed using a structured and validated screening tool such as the alcohol use disorders identification test (AUDIT) or the Cut, Annoyed, Guilty, and Eye Substance Abuse Screening Tool.18,19 AUDIT-C screening is an abbreviated version that helps identify harmful alcohol use or dependence at the bedside or in the outpatient department through three simple questions:
(a) How often have you had a drink containing alcohol in the past year?
(b) How many drinks containing alcohol did you have on a typical day drinking in the past year?
(c) How often did you have six or more drinks on one occasion in the past year?
A score of five or more suggests a positive screen, while scores above 10 indicate possible dependence. 18 Significant alcohol consumption is defined in multiple ways, depending on daily consumption, weekly beverage burden, or consumption patterns. Significant alcohol use is defined as eight or more drinks per week for women and 15 or more drinks per week for men. Regular heavy drinking is defined as >40 g of pure alcohol per day for men and >20 g per day for women. Irregular heavy drinking or binge drinking is defined as drinking at least 60 g of pure alcohol, equivalent to five standard drinks in one sitting, within 2 h.20,21
Recent alcohol use can be assessed using conventional or newer tools. Conventionally, gamma-glutamyl transpeptidase (GGT) has been used to identify recent heavy alcohol consumption. The level of GGT can rise above the normal upper limit with alcohol consumption as low as 7–14 g per day. With the half-life of GGT being 14–26 days, levels typically decrease to the normal reference range within 2–6 weeks of abstinence.22,23 Heavy alcohol consumption is not the only cause of elevated GGT levels. It may also increase due to hepatobiliary disorders, obesity, diabetes, hypertension, and hypertriglyceridemia. Levels around two to five times the upper limit of normal are notable in metabolic-dysfunction-associated steatotic liver disease (MASLD). 24 Carbohydrate-deficient transferrin (cutoff >3% fractional elevation) increases after alcohol consumption of 60–80 g per day for 2 or 3 weeks and normalizes with a mean half-life of 2–4 weeks of abstinence, providing a 2-week detection window period (specificity 88%, sensitivity 77%). False negatives are highly likely, and good detection is possible in heavy drinkers only. 25 Recently, the use of phosphatidyl ethanol (PEth) has gained wide acceptance to differentiate complete abstinence (<10 ng/ml), social drinking (20–150 ng/ml), and recent excessive alcohol use (>500 ng/ml). PEth is very sensitive (~100%), specific (90%), ideal for close monitoring, and useful for identifying relapse, but is unstable at room temperature in collection tubes, and PEth formation of PEth may continue after sampling, leading to falsely elevated concentrations. It is suggested that the PEth analysis be performed on dried blood spots (on filter paper) or after the addition of sodium metavanadate (enzyme phospholipase D) to stabilize the sample.26,27 Unfortunately, PEth is not done routinely, is not widely available, and is expensive. Detection of ethylglucoronide (EtG, >30 pg/mg) in hair (or urine) has 93% specificity and 81% sensitivity. After the complete elimination of ethanol, EtG has a half-life of 2–3 h and is hence detectable in urine for a longer time than ethanol, with a window of detection of approximately 72 h. In hair samples, the detection window is longer, lasting up to 6 months after alcohol use.28,29 However, this test is not always easy to perform due to sampling collection logistics. Interpreting the results could be complicated because extraneous exposure to alcohol-containing products, such as mouthwashes, sanitizers, over-the-counter alcohol-containing medications, or ethanol in food, can also produce false-positive test results. 30 The “rule of 3” is based on the detection of alcohol from various sites depending on the window period, hair and nail samples (window period 3 months), dry blood spots (3 weeks), or urine (3 days). 31 In patients who consume mild-to-moderate alcohol, with concurrent metabolic syndrome, who are at risk for MASLD, to identify whether the underlying liver disease is due to ethanol or the latter, a simple scoring system called the Alcoholic Liver Disease—Non-alcoholic Fatty Liver Disease Index or ANI can be utilized. 32 This index utilizes body mass index, gender, transaminase levels, and mean red blood cell volume or mean corpuscular volume to provide a composite score. The ANI score provides a percentage probability of ALD and is most accurate when the model for end-stage liver disease (MELD) score is below 20. The ANI also distinguishes MASLD and ALD, with individual cut-offs within the intermediate zone indicating Met-ALD, a new category, outside pure MASLD, which defines those with MASLD who consume greater amounts of alcohol per week (140 and 210 g/week for women and men, respectively). The ANI was also shown to outperform AST/ALT ratio or GGT.33,34
Once AUD is identified, the next step is to diagnose the stage of ALD. Noninvasive methods, such as transient elastography (TE), can be easily performed to diagnose advanced fibrosis in ALD. Cut-off values of 8.0 and 12.5 kPa are reasonable for identifying patients with F3 and F4 fibrosis, respectively. 35 However, the values can be falsely elevated in patients with ALD, especially those with recent alcohol consumption, high transaminase, or AAH. In this context, unusually higher cut-off points for cirrhosis (19.5–22.6 kPa) have been reported in patients with ALD compared to chronic viral hepatitis. 36 Especially in patients with AAH, it has been suggested to postpone the evaluation of fibrosis during alcohol withdrawal until AST (also called glutamic oxaloacetic transaminase) drops below 100 U/ml to improve diagnostic precision.35,36 Among patients with initial liver stiffness of 10–25 kPa signifying compensated advanced chronic liver disease (or cACLD), more than half of those without cACLD, showed liver stiffness of less than 10 kPa at 1- and 2-month follow-up testing. A piece of pragmatic advice is to repeat TE (Fibroscan®) after at least 1 month of abstinence to improve diagnostic accuracy. 37
Diagnosis and prognosis of AAH
At the outset, one must be aware that AAH is a clinical syndrome, and the histopathological counterpart is called alcoholic steatohepatitis (ASH). The latter can occur even in the absence of the clinical syndrome. AAH is defined as an abrupt development of jaundice in patients with recent, very high, recent alcohol consumption with a slight increase in transaminase levels and the exclusion of other causes of liver disease. Mortality from AAH at 28 days is estimated to be between 25% and 40%. 38 A diagnosis is made according to the criteria laid down by the National Institute of Alcohol Abuse and Alcoholism (NIAAA) which include clinical, biochemical, and histological parameters. Clinical criteria include heavy alcohol use (>2 drinks in women and >3 drinks in men) for >5 years; active alcohol use until at least 6 weeks before the presentation and recent (<1 month) onset or worsening of jaundice. Biochemical criteria encompass serum bilirubin >3 mg/dl, AST > 50 IU/L and <500 IU/L, AST > ALT with AST: ALT ratio ⩾ 1.5:1. If both criteria are met, then, in the absence of a liver biopsy, a diagnosis of probable AAH is reached while, in its presence and classical histopathological features, definite AAH is confirmed. If only one of the two criteria is met and a liver biopsy is not available, a diagnosis of possible AAH is made (Figure 1).39,40
Various blood biomarkers were studied as candidates for diagnosis of AAH. The AshTest is a unique panel consisting of 10 serum biomarkers designed to diagnose ASH in patients with alcohol consumption over 50 g/day. With a threshold of 0.50, it achieved a sensitivity of 80% and a specificity of 84%. Additionally, the AshTest showed a strong correlation with the histological grading of AAH, particularly demonstrating high predictive values in severe cases. 41 Plasma levels of cytokeratin-18 fragments were found to be reliable non-invasive markers of AAH. Two-thirds of liver biopsies could be avoided using the proposed cut-off points for cytokeratin 18-M65 fragment (2000 IU/L, positive predictive value 91%; 641 IU/L negative predictive value 88%).42,43 The trimethylamine (TMA) and pentane (TAP) score combining pentane and TMA levels in the breath (TAP score ⩾36) had excellent prediction accuracy in diagnosing AAH, which requires further validation. 44
The trigger for ordering a liver biopsy (the gold standard to confirm AAH) depends on the need to exclude competing causes. Classical histological features of AAH include lobular neutrophil-rich inflammation, ballooning of hepatocytes, satellitosis (neutrophils surrounding ballooned hepatocytes), numerous Mallory Denk bodies (which can persist up to 6 months after abstinence), glycogenated nuclei, megamitochondria and peculiar fibrosis that starts in zone 3, perivenular region and extends in a pericellular/perisinusoidal pattern, giving rise to the classic chicken wire fibrosis. 45 Mallory-Denk bodies are clumps of intrahepatocyte eosinophilic material notable near the perinuclear region, which are assortments of ubiquitinated keratin, protein p62, and heat shock proteins. 46 Other possible findings include mixed inflammatory infiltrate, apoptotic hepatocytes called councilman or acidophil bodies, cholestasis with canalicular bile plugs, alcoholic foamy degeneration (microvesicular steatosis in almost all hepatocytes without steatohepatitis or steatofibrosis, very high GGT without transaminase elevation). 45 The characteristics of the liver biopsy that can effectively distinguish AAH from steatohepatitis of MASLD include marked cholestasis (indicates acute decompensation associated with strongly positive hepatocyte keratin-7 immunostaining) and sclerosing hyaline necrosis (central vein obliteration) in the former. In AAH, neutrophilic inflammation is widespread, Mallory-Denk bodies are much higher, and megamitochondria are more plentiful. 47
AAH can be mild or severe and occur at any stage of ALD. Nonetheless, it is important to note that the mortality of non-severe AAH is up to 3%–7% in the short-medium term (up to 6 months) and 13%–20% at 1 year. Recently, the MELD score was found to be the best prognostic indicator of survival for AAH compared to all other conventional prognostic scores. Severe AAH is diagnosed when the MELD score is above 20 (with approximately 20% mortality at 30 days), Maddrey’s discriminant function (MDF) score is greater than or equal to 32, or the presence of overt hepatic encephalopathy. 48 A MELD score >30.5 has an excellent performance in predicting mortality with a sensitivity of 100% and a specificity of 94%. 49 A recent multinational study showed that the new MELD 3.0 demonstrated better performance compared with MELD-Na and MDF in predicting 30-day and 90-day mortality and was also the best predictor of renal replacement therapy requirements during admission for AAH. 50 The MDF score predicted a mortality rate of approximately 20%–50% within the first month of diagnosis of severe AAH. Other less frequently used scoring systems include the Age-Bilirubin-INR-Creatinine (severe AAH, ABIC > 9) score and the Glasgow Alcoholic Hepatitis Scale (GAHS, >8), and the lesser known Beclere model (determines prognosis in ALC).51,52 The other well-utilized score is the Lille model, which provides insights into prognosis in the context of the response to treatment (corticosteroids). Patients above the ideal cut-off point of 0.45 showed a marked decrease in survival compared with those below the cutoff, which identified approximately 75% of the observed deaths. 53 A modified version of the Lille Model found that patients could be further classified into complete responders (Lille score ⩽ 0.16, 28-day survival 91%), partial responders (Lille score 0.16–0.56, 79%), and null responders (Lille ⩾ 0.56, 53%) based on clinical outcomes. 54 Similarly, a combination of scores was found to predict the prognosis of AAH patients better. For example, the MELD plus Lille model, predicted 6-month mortality of complete responders with MELD scores of 15–45 (Lille score, 0.16) at 8.5%–49.7%, compared with 16.4%–75.2% for non-responders (Lille score, 0.45). 55 Other indicators of prognosis in AAH include the presence of acute kidney injury (AKI, 90-day mortality 65% vs 7% in those without); early change in serum total bilirubin (bilirubin level 7 days lower than that on the first day of steroid therapy, 6-month survival 83% in responders); proportional decrease in bilirubin (25% reduction in serum bilirubin after 6–9 days of steroid treatment, 56-day mortality 58% in non-responders) and severe protein-energy malnutrition (PEM score < 60%) correlated with 45% death at 180 days follow-up. 56 It is also pertinent to note that acute variceal bleeding at the time of diagnosis of severe AAH did not affect clinical outcomes in the short and long term. 57 Finally, the alcoholic hepatitis histologic score (AHHS) consisting of four independent histologic characteristics (fibrosis stage, presence of bilirubinostasis, severity of neutrophilic infiltration, and presence of megamitochondria) was combined to provide a composite score that classified AAH patients into low (0–3, survival 97%), intermediate (4–5, 81%) and high-risk (6–9, 49%) groups. 58 However, a recent study found that AHHS was not predictive of short-term survival in a cohort of patients with severe AAH. 59
AAH and ACLF
The syndromic entity ACLF was first defined and described in the context of acute liver injury due to severe AAH superimposed on cirrhosis by Ohnishi and colleagues in the Japanese medical literature in 1995. 60 Cirrhosis can be classified into compensated, early decompensated, and late (multiple complicating events) decompensated stages. At each stage, an acute—direct or indirect—precipitant liver injury can lead to ACLF, characterized by liver failure with or without extrahepatic organ failure. The higher the number of organ failures, the higher the mortality. In large European studies, excessive alcohol consumption (defined as more than 14 units per week in women and 21 units per week in men) in the past 3 months was recognized as the second most frequent precipitating event leading to ACLF after bacterial sepsis.61,62 The European Association for the Study of Liver-Chronic Liver Failure Consortium (EASL-CLIF) classifies ACLF into three grades—ACLF grade 1: patients with single kidney failure; patients with non-renal organ failure plus renal dysfunction (creatinine 1.5–1.9 mg/dl) or overt hepatic encephalopathy; ACLF grade 2: patients with two organ failures, and ACLF grade 3: patients with three or more organ failures. The 28-day mortality of ACLF-1 is 21%, while it is 87% in ACLF-3. 63 Critically ill patients with AAH-related ACLF have worse mortality than patients with non-AAH ACLF. Thus, in ACLF related to AAH, the prognosis depends not only on the severity of AAH but also on the etiology as well as the severity of ACLF. 64 In the absence of approved therapies, treating patients with AAH and AAH-ACLF remains a challenge even after decades since the original publication that shed light on short-term benefits of corticosteroid treatment of AAH. 65
Present options and future therapies for AAH
Treatment for severe AAH must begin with alcohol abstinence, the most important predictor of long-term survival. 66 Evaluation and interventions for malnutrition should be mandatory during the treatment of all patients with severe AAH with avoidance of restrictive hospital diets. Mid-arm muscle circumference or the 24-h creatinine excretion can be utilized as an indirect measure of body muscle mass assessment (1 g excreted creatinine related to 18.5 kg of muscle mass) in patients without ascites. 67 In patients with AAH with ascites, the creatinine-height index appears to be reliable for assessing muscle mass-related nutritional status. The optimal nutrition goals (using estimated euvolemic weight) for recovery include 1.5 g of protein/kg body weight and 30–40 kcal/kg body weight per day, initiated as soon as altered per oral intake is noted. 68 In the context of anorexia or altered mental status that results in poor oral intake, a feeding tube should be considered for early enteral feeding. Nonetheless, it is important to note that intensive enteral nutrition was difficult to implement in patients with AAH and did not increase survival. However, the authors of the study noted that low daily energy intake was associated with greater mortality, and hence adequate nutritional intake should be a main goal for treatment. Patients consuming <21.5 kcal/kg/day experience higher short-term mortality and adverse clinical events. 69 Nutritional deficiencies must be looked for and corrected, especially vitamin B12, thiamine, and folic acid. Furthermore, various nutritional societies also suggest zinc supplementation (50 mg elemental, once a day with a meal) that helps attenuate liver injury and improve hepatic encephalopathy. 70
The landmark STOPAH trial showed that while pentoxifylline did not improve survival in patients with severe AAH, prednisolone use was associated with a reduction in 28-day mortality that did not reach significance and with no improvement in outcomes at 90 days or 1 year. 71 Thus, the only treatment recommended to improve short-term mortality in severe AAH is corticosteroids. Oral prednisolone (not prednisone as it requires conversion within the liver to its active form) 40 mg for a total of 28 days (without tapering) is the first-line treatment for severe AAH. In patients unable to tolerate oral steroids, it is rational to initiate intravenous methylprednisolone 40 mg daily until a switchover is possible. Anecdotal studies have shown that oral methylprednisolone (32 mg once daily) within 1 week of admission for 28 days or intravenous methylprednisolone (500 mg daily for 3 days followed by prednisolone 40 mg daily for 25 days along with prophylactic antimicrobials could lead to clinical and biochemical response in index AAH patients and prednisolone non-responders, respectively.72,73 A quarter of patients with severe AAH harbor bacterial infections and once thought an absolute contraindication to steroid therapy, this notion has been challenged and closely monitored steroid use in controlled infections may be attempted. However, infections developing after steroid therapy (most commonly pneumonia; 46% survival) portend a worse prognosis than infection at diagnosis (most commonly, spontaneous bacterial peritonitis; 78% survival). 74 Therefore, it is very important to differentiate an infection on admission from an infection that occurs after the initiation of steroid therapy in patients with AAH. It is also imperative to note that incident AKI (that is renal injury after admission to the hospital) is associated with 90-day mortality independent of liver function. Prednisolone therapy was associated with reduced incident kidney injury. 75 Similarly baseline neutrophil-to-lymphocyte ratio (NLR) predicted response to corticosteroids and was also associated with infection and AKI in AAH patients. An NLR score of 5–8 at baseline identifies those most likely to benefit from corticosteroids. 76 A large multinational study showed that corticosteroids improved 30-day survival only among patients with severe AAH, especially those with MELD scores between 25 and 39. 77 According to the Lille score (checked after 7 days of treatment), steroid therapy is beneficial for patients who are complete responders or partial responders but not null responders. Complete responders must complete their 28-day course of steroids.53,54 Partial responders (Lille score 0.16–0.56) have two options: —complete steroid monotherapy or initiate additional therapy to improve steroid response. 54 Two additional therapies that have shown some promise in improving clinical outcomes in the short term are N-acetyl cysteine (NAC) and metadoxine. N-acetyl cysteine is infused on day 1 at a dose of 150, 50, and 100 mg/kg/body weight in 250, 500, and 1000 ml of 5% glucose solution over 30 min, 4 h, and 16 h, respectively, and on days 2 through 5 at 100 mg/kg/body weight/day in 1000 ml of 5% glucose solution).78,79 The survival benefit of NAC + prednisolone was demonstrated in a systematic review and network meta-analysis study. 80 A small randomized trial showed that adding metadoxine, 500 mg three times daily for 30 days to steroid treatment for severe AAH improved 3-month survival. 81
In patients with severe AAH who do not respond to steroids, liver transplantation is the only treatment option that has shown long-term survival benefit. Initial studies were stringent on patient selection (first presentation of liver disease, commitment to lifelong sobriety, and absence of characteristics predictive of relapses, such as legal consequences of drinking, other substance abuse, or significant mental health disorders) and were associated with excellent short-term survival. 82 However, recent studies from the United States and France have shown that shedding stringent criteria increased transplant rates for severe AAH with high MELD scores, although associated with more frequent relapses, resulting in improved short- and long-term survival.83,84 The study by the American Consortium for Early Liver Transplantation for Alcoholic Hepatitis (ACCELERATE-AH) found that the 1- and 3-year survival was 94% and 84%, with sustained alcohol use after transplantation in 10% and 17%, respectively. In this study, mortality without transplantation was 70% at 6 months. 85 The QuickTrans prospective controlled study confirmed the important survival benefit related to early liver transplantation for severe AAH providing objective data on survival and alcohol relapse to tailor the management of patients with severe AAH. 86 Thus, the current 6-month abstinence rule for transplant patients with ALD does not affect relapse rates or survival and should not be utilized. An algorithmic approach toward diagnosis and management of AAH is shown in Figure 2.

Algorithm for diagnosis and management of severe AAH, present options.
Various therapies that are without benefit and therefore should not be used for the treatment of severe AAH include NAC monotherapy, pentoxifylline, ursodeoxycholic acid, vitamin E supplementation, silymarin (milk thistle extract), anabolic steroids (oxandrolone), S-adenosyl methionine supplementation, probiotics/prebiotics, antioxidant cocktail, granulocyte-colony stimulating factor, tumor necrosis factor-alpha antagonists (infliximab/etanercept), glucagon/growth hormone therapy. Liver assist devices, such as extracorporeal cellular therapy, plasmapheresis, and the molecular adsorbent recirculation system, were also disadvantageous in reducing mortality at 3 and 6 months in severe AAH. They may be used only within clinical research protocols.87,88 A summary of beneficial and non-beneficial therapies in severe AAH is shown in Figure 3.

Summary of beneficial and non-beneficial therapies for alcohol-associated hepatitis.
Newer options for treating severe AAH include fecal microbiota transplantation (FMT) and bacteriophage therapy. Bench-to-bedside studies have shown that alcohol use is associated with intestinal dysbiosis, deviation from the normal gut microbiome composition and function, or a change in the host’s indigenous or autochthonous microbial taxa. 89 Gut dysbiosis is central to the causation and progression of severe AAH. Specific bacteria were associated with discrete clinical events in patients with severe AAH. For example, reduction in Akkermansia muciniphila was associated with mucin degradation and gut barrier dysfunction, Dehalobacteriaceae and Turibacteraceae were associated with MELD > 25, reduction in Bifidobacterium/Lactobacillus was related to continued drinking, and Enterococcus/Acinetobacter/Mitsuokella was associated with the development of AKI in severe AAH. 90 Beneficial modulation of dysbiotic taxa by healthy donor stool transplant has been shown to improve clinical outcomes in the short and long term.91,92 Healthy donor FMT was associated with reduced infections, control of portal hypertensive complications, fewer episodes of hepatic encephalopathy, and improved transplant-free survival.92,93 These clinical results were also associated with favorable changes in pathogenic bacterial taxa, such as a reduction in the relative abundance of deleterious Proteobacteria and an increase in beneficial short-chain fatty acid-producing Actinobacteria.90,91 Similarly, healthy donor FMT was also associated with improved clinical outcomes when compared to pentoxifylline therapy in severe AAH. 94 Response to corticosteroid therapy was demonstrably poor in patients with AAH-ACLF with higher grades of ACLF. 95 The first study on FMT in AAH-related ACLF demonstrated an overall survival rate of 66% at 548 days, and, interestingly, the survival rate in higher grades of ACLF (2 + 3) at the end of 548 days was 58.3%, demonstrating proof of concept on the benefits of FMT in this sickest group of AAH patients. 96 A summary of preliminary evidence of healthy donor FMT is shown in Figure 4. Another proof-of-concept study found that cytolysin-producing Enterococcus fecalis was overexpressed and associated with the development and severity of AAH. With bacteriophage therapy targeting E. fecalis, the authors found that the development and severity of AAH were reduced in an animal model. 97 More well-designed, prospective, controlled, and clinically translational studies are excitingly expected on the utility of targeted bacteriophage therapy and FMT in severe AAH.
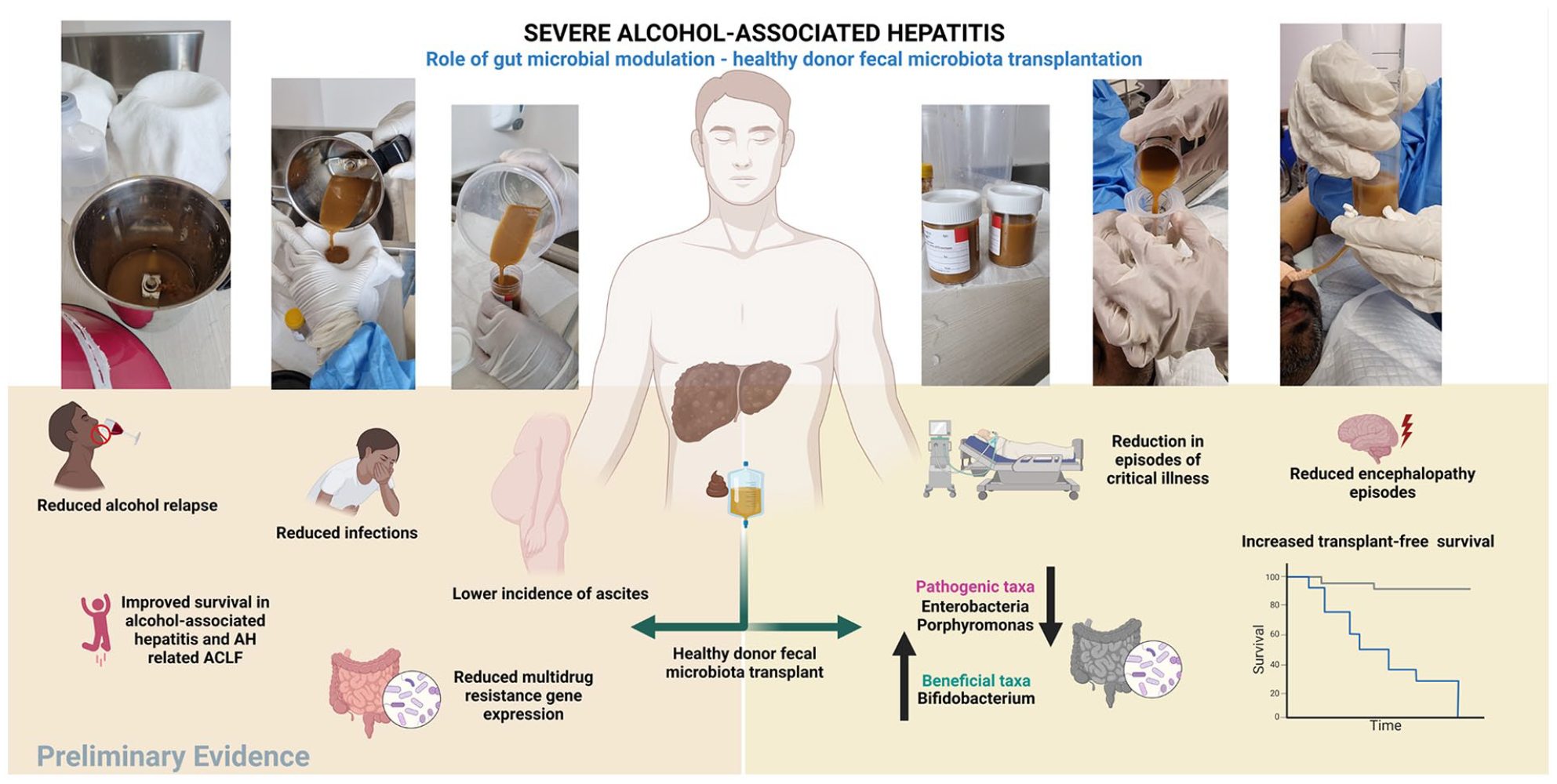
Role of and benefits of healthy donor stool transplantation in alcohol-associated liver disease and severe AAH.
Summary and conclusions
Severe AAH is a clinical syndrome associated with high mortality without approved treatments. Pertinent steps in the diagnosis of AAH include the identification of AUD in the patient, the differentiation of ALD from MASLD, excluding other competing causes of acute liver disease, using the NIAAA criteria or liver biopsy to confirm the diagnosis, and finally, to forecast clinical outcome and decide the initiation of therapy based on MELD scores. Corticosteroids remain the first-line treatment for severe AAH in the absence of contraindications. Early referral to liver transplantation must be entertained for patients who do not respond to steroid therapy. Pharmacological and interventional therapies that are not beneficial should be avoided to reduce financial burden. In case of poor transplant candidacy or rapid clinical deterioration such as ACLF, the patient should be enrolled in a clinical trial program (based on availability) as a salvage/bridge option (liver assist devices, FMT) to transplant.
Footnotes
Acknowledgements
None.
Credit author contributions
Cyriac Abby Philips: conceptualization, methodology, data curation, writing (review and editing), validation.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Not applicable.
Informed consent
Not applicable.
