Abstract
Background:
Hepatocellular carcinoma (HCC) is a leading cause of cancer-related deaths in Germany. National data on the management of HCC remain scarce.
Objectives:
This study aims to provide an up-to-date overview of clinical characteristics, treatment modalities, and survival outcomes among patients with HCC in Germany.
Design:
This is a real-world retrospective study using health insurance data from BARMER.
Methods:
Patients with an HCC diagnosis between 2016 and 2020 were identified in the BARMER database. Comorbidities, anticancer therapies, and treatment pathways of those patients were evaluated using descriptive statistics and survival analysis.
Results:
A total of 2778 patients with HCC were identified. Of these, 1569 (56.5%) received any anticancer therapy. Transarterial chemoembolization (TACE; 22.3%), liver resection (LR; 20.9%), and systemic therapy (18.9%) were the most frequently used approaches. Survival varied significantly, with liver transplantation (LTx) offering the best outcomes, with a 5-year survival rate of 76%, followed by LR with 40%. Treatments with curative intent, including LTx, LR, and ablation, had a median survival of 40.4 months, compared to 9.7 months for non-curative modalities.
Conclusion:
In Germany, a substantial proportion of HCC patients remain untreated. Therapies with curative intent, particularly transplantation, provide relevant survival benefits. Improving surveillance efforts could enhance the proportion of patients eligible for these modalities and may represent a critical step toward improved outcomes for patients with HCC.
Introduction
Hepatocellular carcinoma (HCC) poses a substantial health challenge, with an increasing incidence in Western countries. 1 A shift in HCC etiologies has occurred in recent years. The successful implementation of vaccination against the hepatitis B virus (HBV) and the development of effective antiviral therapies have led to a decline in viral cases. 2 Non-viral etiologies, such as alcoholic liver disease (ALD) and metabolic dysfunction-associated steatotic liver disease (MASLD), have become increasingly prominent. This shift is evidenced by a high prevalence of metabolic disorders, including obesity and diabetes mellitus, frequently diagnosed prior to HCC diagnosis in Germany in a recent nationwide analysis. 3
Significant progress has also been made in managing HCC over the past decade. The adoption of biannual screening recommendations for patients with chronic liver disease, as recommended by the German guideline on HCC, 4 has facilitated earlier detection and reduced the proportion of patients presenting with advanced-stage disease. 5 The introduction of immune checkpoint inhibitors and sequencing therapy with molecular-targeted agents have expanded treatment options and been associated with prolonged survival for patients with advanced HCC.2,6
The management and outcomes of patients in routine clinical practice may diverge from the results of randomized clinical trials. Patient populations in clinical studies usually do not adequately reflect the heterogeneity and high comorbidity burden of patients in real-world scenarios, particularly in HCC, with a high proportion of patients suffering from chronic liver disease. 6 Health data derived from cross-sector, nationwide analyses provide valuable insights complementing clinical trial findings to inform healthcare providers. In this claims-based study, we utilize a comprehensive nationwide insurance database to provide an up-to-date overview of clinical characteristics, treatment modalities, and survival outcomes among patients with HCC in Germany.
Materials and methods
Study design and data sources
This retrospective cohort study was based on routinely collected health insurance claims data from BARMER (Berlin, Germany), providing insurance coverage for approximately 8.7 million citizens in 2022 (representing 10.4% of the German population). All diagnoses relevant to patient treatment are reported by healthcare providers for billing purposes. Diagnoses provided in medical claims data are coded according to the German Modification of the International Statistical Classification of Diseases and Related Health Problems, 10th Revision (ICD-10-GM). 7 The reporting of this study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: guidelines for reporting observational studies statement 8 (Supplemental Table 1), the ESMO-GROW checklist, 9 and the RECORD statement. 10
Patient identification
Patients with incident HCC were identified according to ICD-10-GM code C22.0 between January 1, 2016 and December 31, 2020. Patients aged 18 years and older with either one primary diagnosis of HCC (C22.0) in an inpatient setting or two confirmed diagnoses of HCC (C22.0) in an outpatient setting during consecutive quarters were included. 11 Patients with previous cancer diagnoses other than carcinoma in situ or non-melanoma skin cancer within 5 years prior to HCC diagnosis were excluded. Individuals without BARMER insurance coverage for at least 10 years before and 2 years after the date of the HCC diagnosis (unless death occurred before) and those insured by Deutsche BKK, a health insurance that integrated into BARMER in 2017, were excluded to ensure sufficient insurance records during the observation period. The index date of the first HCC diagnosis was defined as the date of the earliest inpatient or outpatient visit with a diagnosis of HCC (C22.0).
Treatments
HCC treatment procedures given in the inpatient setting, including liver transplantation (LTx), liver resection (LR), thermal ablation (TA), transarterial chemoembolization (TACE), transarterial radioembolization (TARE), and systemic therapies, were coded according to the respective operation and procedure classification system (OPS codes), version 2020. 12 Prescriptions of systemic therapies were considered by using their respective ATC codes. Dates of drug approvals were extracted from the European Medicines Agency database. To assess the main treatment for every patient, six groups were operationalized according to the hierarchical order of decreasing treatment efficacy as suggested by Vitale et al. 13 : LTx, LR, TA, TACE/TARE, and systemic therapy. Cases without a documented HCC treatment were grouped into “No therapy.” In addition, treatment was operationalized into two groups: “curative therapy,” including treatment modalities with curative intent such as LTx, LR, and TA cases, and “non-curative therapy,” including TACE, TARE, and systemic therapy. The treatment patterns of patients who underwent LTx or LR were visualized using Sankey diagrams. Information on hospitalization (days in hospital, number of hospital visits) was analyzed from the date of treatment until 1 year after treatment.
Comorbidities
The comorbidities of patients with incident HCC were identified using the Charlson Comorbidity Index (CCI), using the respective ICD-10-GM codes. 14 Categories including diagnoses of malignant neoplasms were not considered because they could be related to the index disease (HCC). Comorbidities were identified by one documented primary or secondary diagnosis in an inpatient setting or two diagnoses in an outpatient setting during two consecutive quarters, 12 months before or 3 months after the HCC diagnosis.
Risk factors
Risk factors prior to the HCC diagnosis were identified by retrieving either one primary or secondary diagnosis in an inpatient setting or two diagnoses in an outpatient setting during consecutive quarters within 10 years before the index date using ICD-10-GM codes. MASLD, which was used according to the new nomenclature for steatotic liver disease, 15 is not yet available in the ICD-10-GM system. Hence, it was defined based on a combination of several ICD-10-GM codes (K76.0 or K75.8, and any of the following: I10, I11, I12, I13, I15, or E78.0-5).
Statistics
Baseline characteristics were determined using descriptive statistics: numbers and percentages for categorical variables, median with interquartile range (IQR), or mean with standard deviation (SD) for continuous variables. Survival was estimated using the Kaplan–Meier method. To control for age, sex, comorbidity, and risk factors, a multivariate Cox Proportional Hazards regression analysis was additionally performed. Variables were selected using the backward selection method. We examined patients’ outcomes from a 3-month landmark to allow for a comprehensive assessment of long-term survival outcomes. Patients with no documented date of death were censored on December 31, 2022. A significance level of α = 0.05 was prespecified for all statistical tests. Data processing and statistical analyses were conducted using SAS software (version 9.4; SAS Institute Inc., Cary, NC, USA) and R software (version 4.2.1; R Foundation, Vienna, Austria).
Results
Between January 1, 2016 and December 31, 2020, 4574 patients with an incident HCC diagnosis were identified. In total, 1796 patients were excluded due to insufficient insurance coverage, prior history of malignancy, or age <18 years (see Figure 1). In all, 2778 patients were included in the analysis.

Study flowchart of patient identification.
Treatment patterns and clinical characteristics
For 1569 patients (56.5%), at least one HCC treatment was documented: LR was performed in 580 patients (20.9%), LTx in 65 (2.3%), TA in 190 (6.8%), TACE in 620 (22.3%), TARE in 156 (5.6%), and systemic therapy in 525 patients (18.9%). No anticancer treatment was documented for 1209 patients (43.5%) in the BARMER database. Using the hierarchical treatment framework proposed by Vitale et al., 13 we categorized patients according to the most effective HCC therapy received. According to this framework, 2.3% of patients received LTx, 20.6% LR, 4.9% TA, 16.5% TACE/TARE, and 12.2% systemic therapy. Among the 12.2% of patients receiving systemic therapy, sorafenib was the mainstay of treatment (Supplemental Table 2). During the study period, we detected a progressive evolution of agents corresponding to their respective approval, notably the increasing use of lenvatinib after 2018. Frequent agents approved for second-line therapy included cabozantinib and ramucirumab. Regorafenib and immune checkpoint inhibitors were used infrequently.
Of the 1569 patients treated, 49.3% received a modality with curative intent (LTx, LR, and TA) and 50.7% had non-curative treatment (TACE, TARE, systemic therapy). Clinical characteristics for the whole cohort and patients stratified by therapy subgroups are shown in Table 1. The mean age in the total cohort was 71.9 years (SD ±9.7). In the LTx group, patients were about 10 years younger compared to the other groups. Patients without any documented HCC treatment were significantly older than treated patients, with a mean age of 73.9 years (SD ±10.2). The burden of comorbidities was high in every treatment group, with a median CCI of 4.0. A higher proportion of patients with impaired liver function could be noted in the LTx group compared to the other groups (Supplemental Table 3). Patients receiving no treatment had the highest proportion of CCI ⩾7, and high rates of congestive heart failure and dementia. Diabetes mellitus (51.9%) and obesity (55.5%) were the most common HCC risk factors in all patients. Diabetes, obesity, and MASLD were equally distributed among all groups. The proportion of patients with fibrosis/cirrhosis ranged from 28% in the LR group to 91% in the LTx group. In patients receiving LR, alcohol abuse (26.9%) and ALD (14.7%) were lower as compared to the whole cohort. The proportion of viral hepatitis was highest in patients treated with LTx (33.9%) and TA (25.7%), whereas in the other groups, it was 14%–15%. In Table 2, characteristics are presented separately for patients receiving curative and non-curative modalities. Patients receiving non-curative approaches were significantly older (p = 0.0002), had a higher proportion of men (p = 0.0001), and a significantly higher comorbidity burden (p < 0.0001) compared to patients treated with curative intent. Distribution of metabolic risk factors such as diabetes (p = 0.0989), obesity (p = 0.1653), and MASLD (p = 0.5488) was similar among both groups. The proportion of patients with alcohol abuse (p = 0.0006), ALD (p = 0.0155), and fibrosis/cirrhosis (p = 0.0045) was higher in the non-curative group, while patients with viral hepatitis (p = 0.0146) were more frequent among curatively treated individuals.
Characteristics of HCC patients according to main treatment groups (all patients, n = 2778).

Several risk factors in one patient are possible.
ALD, alcoholic liver disease; CCI, Charlson Comorbidity Index; HCC, hepatocellular carcinoma; IQR, interquartile range; LR, liver resection; LTx, liver transplantation; MASLD, metabolic dysfunction-associated steatotic liver disease; SD, standard deviation; TA, thermal ablation; TACE/TARE, transarterial chemoembolization/radioembolization.
Characteristics of HCC patients according to curative versus non-curative treatment (treated patients, n = 1569).

Several risk factors in one patient are possible.
ALD, alcoholic liver disease; CCI, Charlson Comorbidity Index; HCC, hepatocellular carcinoma; IQR, interquartile range; MASLD, metabolic dysfunction-associated steatotic liver disease; SD, standard deviation.
Survival outcomes and treatment sequences
Median follow-up time was 8.7 months (range 0–83 months). In the total cohort, the median overall survival (mOS) was 8.7 months (95% confidence interval (CI), 7.7–9.7 months). The estimated 1- and 5-year survival rates were 44% and 16%, respectively. Stratification into subgroups according to the main treatment revealed significant differences in OS, as presented in Figure 2(a). Patients treated with LTx had the longest survival: mOS was not achieved, the 5-year survival rate was 76% (Supplemental Figure 1). The unadjusted hazard ratio (unadj. HR) for LTx versus LR was 0.33 (95% CI, 0.19–0.55), and the adjusted hazard ratio (adj. HR) was 0.29 (95% CI, 0.17–0.48, adjusted for CCI, risk factors fibrosis/cirrhosis, viral hepatitis, and other treatments after backward variable selection, see Supplemental Table 4). mOS was 40.4 months (95% CI, 36.1–49.8) for LR, 27.0 months (95% CI, 21.0–37.3) for TA, 14.5 months (95% CI, 12.1–16.9) for TACE/TARE, and 5.6 months (95% CI, 4.6–6.9) for systemic therapy (Figure 2(a)). For patients receiving curative modalities, mOS was 40.4 versus 9.7 months for patients treated with non-curative modalities (Figure 2(b)). Patients with curative intent had a 68% higher survival probability than patients with non-curative treatments (unadj. HR 0.31, 95% CI, 0.27–0.35; adj. HR 0.32, 95% CI, 0.28–0.36, adjusted for CCI and viral hepatitis after backward selection, see Supplemental Table 5). Since the best oncological outcomes were achieved with LTx and LR, we aimed to further characterize the role of therapeutic sequencing in these patients. The treatment pathways of LTx patients are illustrated in Figure 3(a). Most patients received TACE or TARE as a bridging therapy before transplantation (n = 53, 81.5%); other approaches were less frequently performed: TA in eight patients (12.3%), LR in six (9.2%) and systemic therapy in three (4.6%). Only six patients (9.2%) did not undergo bridging before LTx. The mean time from HCC diagnosis to LTx was 9.2 months (SD ±5.9) and ranged from 0 to 23.5 months. In the year following LTx, the mean hospital stay was 52 days (SD ±47, range 2–191 days) with a mean of 4.0 hospital visits (SD ±3.2, range 1–15). Treatment sequencing for LR differed substantially from LTx. Most resected patients (n = 541, 93.3%) had LR as their initial treatment. From those, n = 190 (32.8%) had no other treatment, n = 210 (36.2%) died during the observation period, and n = 180 (31.0%) had additional treatments like TACE/TARE, TA, or systemic therapy. Eight patients had LTx after resection (see Figure 3(b)). Until 1 year after LR, the mean hospital stay was 52 days (SD ±29, range 1–262 days) with a mean of 2.6 hospital visits (SD ±2, range 1–13).

Overall survival according to main treatment groups (a) and to curative/non-curative treatment (b) n = 1373, n = 196 were excluded due to 3-month landmark).
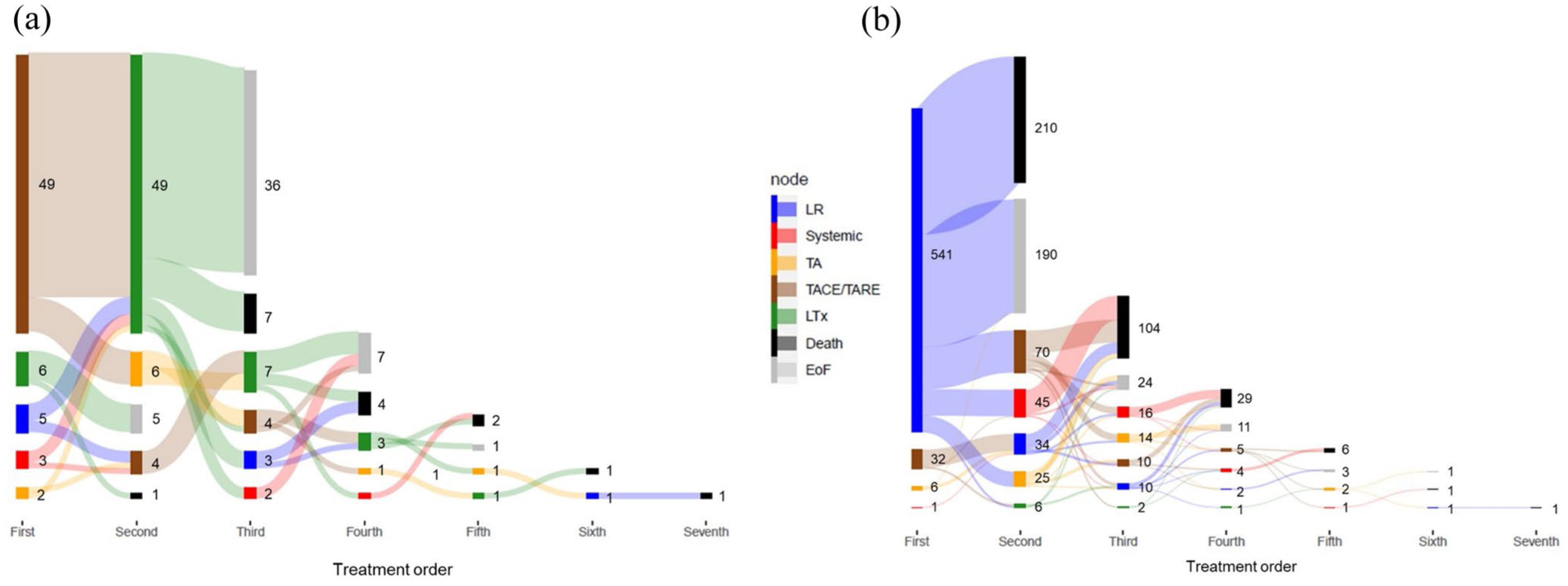
Sankey diagram showing the number and sequence of treatments for HCC patients with LTx (a) and with LR (b) within 2 years after diagnosis (n = 65).
Discussion
This claims-based study provides a comprehensive, up-to-date overview of clinical characteristics, treatment modalities, and survival outcomes in individuals with HCC in Germany. Utilizing a large cohort of 2778 patients from the BARMER insurance database—representing approximately 10% of the German population—we address the scarcity of national data on the HCC treatment landscape.
Only 56.5% of HCC patients received any form of anticancer therapy, mirroring observations from studies in the United States, Taiwan, and Sweden.16,17 The untreated cohort was older and had a higher prevalence of comorbidities, such as congestive heart failure and dementia. However, the significant proportion of untreated patients may not be fully attributed to poor performance status or a high comorbidity burden. A subset of these patients may have been diagnosed at a very advanced stage, which often limits therapeutic options, particularly in the presence of reduced liver function due to underlying cirrhosis. 18 In addition, only 27.8% of patients in this study were eligible for treatments with curative intent. This underscores the importance of early detection measures, such as biannual sonography for patients with liver cirrhosis, as recommended by most Western HCC guidelines. 4 However, screening in Germany seems to require optimization, as evidenced by the superior outcomes reported from Japan, where the majority of patients are diagnosed at early stages (BCLC 0 or A). 19 This is associated with an improved survival of 80 months for all patients with HCC in Japan, exceeding outcomes reported from other regions. 19 By contrast, in Western countries, only a minority of patients are estimated to receive adequate surveillance, 20 contributing to a higher proportion of advanced-stage diagnoses. 21 The high proportion of patients in this study who did not receive anticancer therapy or had non-curative treatment options may indicate existing barriers to HCC surveillance. Enhanced screening efforts could facilitate timely diagnosis and expand access to potentially curative treatments, ultimately improving patient outcomes.
Among treated patients, 49.3% received interventions with curative intent such as LR, TA, or LTx. Palliative therapies, including locoregional therapies (TACE/TARE) or systemic therapy, were applied in 50.7%, which is comparable to other studies.22,23 mOS significantly differed by treatment type. Patients receiving curative procedures had an mOS of 40.4 months, compared to 9.7 months with non-curative approaches. This disparity persisted after adjusting for other explanatory factors, underscoring the substantial impact of treatment modality on patient outcomes. Our analysis also highlighted differences in the allocation of curative treatments. Patients with alcohol abuse and alcohol-related liver disease were less likely to receive curative therapies. This may be due to lower screening rates leading to a more advanced disease at diagnosis, since patients with ALD showed lower adherence to HCC surveillance programs in Italian and French cohorts.24,25 Patients with viral hepatitis were slightly more likely to receive curative treatments, while those with diabetes, obesity, and MASLD showed similar treatment rates across groups. These findings emphasize the need for improved screening efforts, particularly in vulnerable subgroups with chronic liver disease.
Using the hierarchical framework proposed by Vitale et al., 13 we categorized patients according to the most effective therapy received. LR was the most common treatment (20.6%), followed by locoregional therapies such as TACE or TARE (16.5%), and systemic therapy (12.2%). TA (4.9%) and LTx (2.3%) were less frequent. The best survival outcomes were observed in LTx patients, with a 5-year survival rate of 76%, followed by LR at 40%. This finding may not only be attributed to the beneficial effects on oncological outcomes in patients undergoing LTx but also to the causal treatment of the underlying liver disease. The majority of LTx patients underwent bridging treatments, most commonly TACE or combinations of other approaches, highlighting the critical role of multimodal therapeutic sequencing as a bridge to transplantation. Our data underscore the importance of referring suitable HCC patients to liver transplant centers for evaluation and appropriate bridging therapies.
Systemic therapy was administered to 12.2% of patients as the main treatment, and we detected moderate outcomes with a median survival of 5.6 months, confirming previous research. 2 In the observation period, the armamentarium of agents evolved rapidly. Following its approval in 2018, lenvatinib became increasingly used. However, sorafenib remained the mainstay of pharmacotherapy. This predominance could reflect the broader clinical experience of treating physicians across sectors in Germany with sorafenib, compared to the newer agent lenvatinib. Cabozantinib and ramucirumab were frequently administered drugs approved as second-line therapies. Regorafenib, another agent approved in the second-line, was rarely detected, probably due to its withdrawal from the German market by the manufacturer, after which it was only available as an import medication. 26
This study’s limitations include its reliance on claims data, which lack clinical metrics such as liver function scores, BCLC stage, and ECOG performance status. Detailed information on tumor stage or performance status is not relevant to reimbursement in Germany, and therefore unavailable in this dataset. The study compensates for this by classifying liver disease severity through ICD codes and providing a detailed comorbidity analysis. In claims data, diagnoses and treatments could be overreported due to billing purposes, but the characteristics and treatment proportions were comparable to those of other HCC studies,16,17 supporting the reliability of the analysis. Despite these limitations, our study offers a comprehensive and current overview of HCC treatment patterns and survival outcomes in a large, German real-world cohort.
Recent drug approvals, with the introduction of immune checkpoint inhibitors, have advanced the pharmacotherapy of HCC.2,27 This promises to prolong the median survival of patients treated with systemic therapy in the future with respect to the outcomes reported in this study.28 –32 Furthermore, combinatorial approaches such as TACE plus immune checkpoint inhibitors have recently demonstrated efficacy in large phase III trials and could lead to improvements for patients with intermediate-stage disease.33,34 However, these approaches remain largely palliative. The key to survival improvement remains increasing the rate of patients treated with curative intention. HCC screening is an effective measure to improve early detection of disease and enhance the rate of patients eligible for curative therapies. 35 In Germany, patients at high risk for HCC should undergo surveillance with abdominal sonography every 6 months, in line with national guidelines. 4 Measurement of AFP is recommended in combination with ultrasound, but not as a standalone parameter. At-risk conditions defined in the German HCC guideline are liver cirrhosis Child-Pugh A or B, liver cirrhosis Child-Pugh C on a transplant waiting list, chronic HBV infection, high-grade liver fibrosis, and metabolic dysfunction-associated steatohepatitis. In our opinion, early detection of HCC in Germany relies on two key factors: (1) detection of chronic liver disease as the basis for initiating surveillance and (2) regular surveillance for all at-risk patients. The recent implementation of routine screening for chronic HBV and HCV as part of the preventive German medical examination “Check-Up 35+” has enhanced the identification of patients at risk and could expand the rate of early HCC detection in the viral subgroup. 36 Patient empowerment through educational activities and awareness campaigns could increase screening uptake across all HCC etiologies. Future research should explore barriers to screening, particularly in patients with alcoholic and metabolic liver diseases. Emerging liquid biomarkers for HCC screening show promise and may offer advantages over standard sonography by reducing the time and expertise required.36,37 However, these methods necessitate high accuracy, validation in randomized studies, and cost-effectiveness assessments before they can be integrated into clinical practice.
Conclusion
In summary, this nationwide claims-based analysis reveals a relevant proportion of untreated HCC patients in Germany. Curative therapies, particularly transplantation, provide substantial survival benefits. Improving surveillance efforts might be crucial to increasing the number of patients eligible for curative treatments and may significantly enhance outcomes for patients with HCC in Germany.
Supplemental Material
sj-pdf-1-tam-10.1177_17588359251363202 – Supplemental material for Treatment gaps and survival outcomes of hepatocellular carcinoma: insights from a nationwide, claims-based study
Supplemental material, sj-pdf-1-tam-10.1177_17588359251363202 for Treatment gaps and survival outcomes of hepatocellular carcinoma: insights from a nationwide, claims-based study by Simone Schrodi, Karin Berger-Thürmel, Selina Becht, Michael von Bergwelt-Baildon, Peter Buggisch, Wolf-Peter Hofmann, Ursula Marschall, Christian M. Lange, Florian P. Reiter, Julia Mayerle, Bernhard Mörtl, Sami Orabi, Alexander Philipp, Jörg Trojan, Tobias Weiglein, Enrico N. de Toni and Najib Ben Khaled in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
This study was partly funded by Roche Pharma AG. Najib Ben Khaled was supported by the Bavarian Cancer Research Center, FöFoLe of LMU Munich funding program (1122), and the German Cancer Consortium.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
