Abstract
Hepatocellular carcinoma (HCC) is a highly lethal disease that exhibits a rising global incidence, and most HCC patients are at an advanced stage at the time of diagnosis. Although lenvatinib has become the first-line option for HCC and its overall survival is not inferior to sorafenib, not all HCC patients could derive benefits from lenvatinib. Therefore, it was urgent to predict which group of HCC patients could derive benefits from lenvatinib and monitor its therapeutic effectiveness during treatment. This review summarized various biomarkers that could be used for prediction of lenvatinib response, including 7 biomarkers that are applied in clinical practice, 11 biomarkers that are applied in clinical practice, and 6 biomarkers that are under exploratory stage. By conducting this first comprehensive analysis of predictive biomarkers for lenvatinib in HCC patients, this review aims to help doctors to assess the response of lenvatinib treatment more accurately and, thus, develop more effective individualized treatment plans.
Plain language summary
Hepatocellular carcinoma (HCC) is the sixth most common cancer worldwide and the third leading cause of cancer-related death. Lenvatinib is a widely used treatment for advanced HCC, but not all patients benefit equally from it. This review summarizes the current biomarkers that could be used to predict treatment response to lenvatinib, and categorized the biomarkers into three groups based on their clinical relevance: (1) biomarkers which are applied in clinical practice (e.g., A-fetoprotein, systemic inflammatory indicators, etc.), (2) biomarkers which are highly relevant to clinical practice (e.g., serological biomarkers, changes in body composition), and (3) biomarkers which are under exploratory stage (e.g., non-coding RNA, Single nucleotide polymorphisms, etc.), aiming to provide theoretical support for individualized treatment and future translational research.
Background
Hepatocellular carcinoma (HCC) ranks as the sixth most prevalent cancer globally and stands as the third major cause to cancer-related fatalities, with approximately 906,000 new cases and 830,000 deaths annually.1 –3 The prognosis for HCC is unfavorable, with a mere 12.7% 3-year survival rate and a median survival of just 9 months. 4 Although radical treatments, such as surgery, radiofrequency ablation, and liver transplantation (LT), are available for early-stage HCC patients, the majority of patients are typically diagnosed at an advanced stage.5,6 In such cases, lenvatinib shows a survival benefit for patients with preserved liver function who have HCC. 5
Lenvatinib is a small molecule tyrosine kinase inhibitor (TKI) that has shown therapeutic efficacy as a first-line targeted drug in HCC. 7 According to the REFLECT trial, lenvatinib was non-inferior to sorafenib in terms of overall survival (OS) and showed superior outcomes in progression-free survival (PFS) and objective response rate (ORR) in patients with unresectable HCC (u-HCC). 8 However, some patients are unable to take long-term lenvatinib due to the advancement of tumor and the occurrence of adverse events (AEs) like diarrhea, fatigue, and decreased appetite. 9 Moreover, ORR of lenvatinib was generally less than 30% according to response evaluation criteria in solid tumors (RECIST) 1.1, which is mainly because some HCC patients are insensitive to lenvatinib. 10 Therefore, it is an urgent need to discover effective predictive markers that could identify which group of patients with HCC is most likely to derive benefits from lenvatinib.
Biomarkers have greater potential in the prediction of disease progression after HCC treatment compared to imaging assessments like RECIST and modified RECIST (mRECIST).11,12 Moreover, liquid biopsy has become a valuable strategy for obtaining tumor biomarkers, providing a less or non-invasive alternative to traditional tissue biopsies.13,14 Importantly, liquid biopsy-derived biomarkers could reduce intratumoral heterogeneity compared to tissue biopsy, improving diagnostic sensitivity and accuracy and enhancing patient prognosis.11,15 Currently, many trials have reported biomarkers that could be used to predict the efficacy and survival benefit in HCC patients treated with lenvatinib.
This review is the first to summarize various biomarkers used to predict the efficacy of lenvatinib in HCC, including 7 biomarkers that are applied in clinical practice, 11 biomarkers that are applied in clinical practice, and 6 biomarkers that are under exploratory stage. This review provides a comprehensive analysis of predictive biomarkers for lenvatinib treatment, aiming to help doctors to evaluate the response of lenvatinib treatment in HCC patients more accurately and, thus, develop more effective individualized treatment plans.
Methods
Through searching four databases (PubMed, Web of Science, Embase, and the Cochrane Library) from inception to June 2025, we selected studies about biomarkers that could predict the efficacy of lenvatinib in HCC and conducted a narrative review on this topic. The search strategy included combinations of the following terms: “hepatocellular carcinoma,” “HCC,” “lenvatinib,” “biomarkers,” “predictive markers,” and “response markers,” without restriction on publication year. Studies that did not report the source of the biomarker were excluded, as well as prognostic biomarker studies. Letters, editorials, expert opinions, case reports, and non-English language publications were excluded. Full-text reviews were conducted when abstract was insufficient for eligibility assessment. References of all included articles were also screened for relevant studies.
Biomarkers that are applied in clinical practice
Currently, several biomarkers that are already widely used in clinical practice have been identified as predictors of the therapeutic efficacy of lenvatinib in HCC, as summarized in Table 1.
Baseline characteristics biomarkers that are applied in clinical practice.
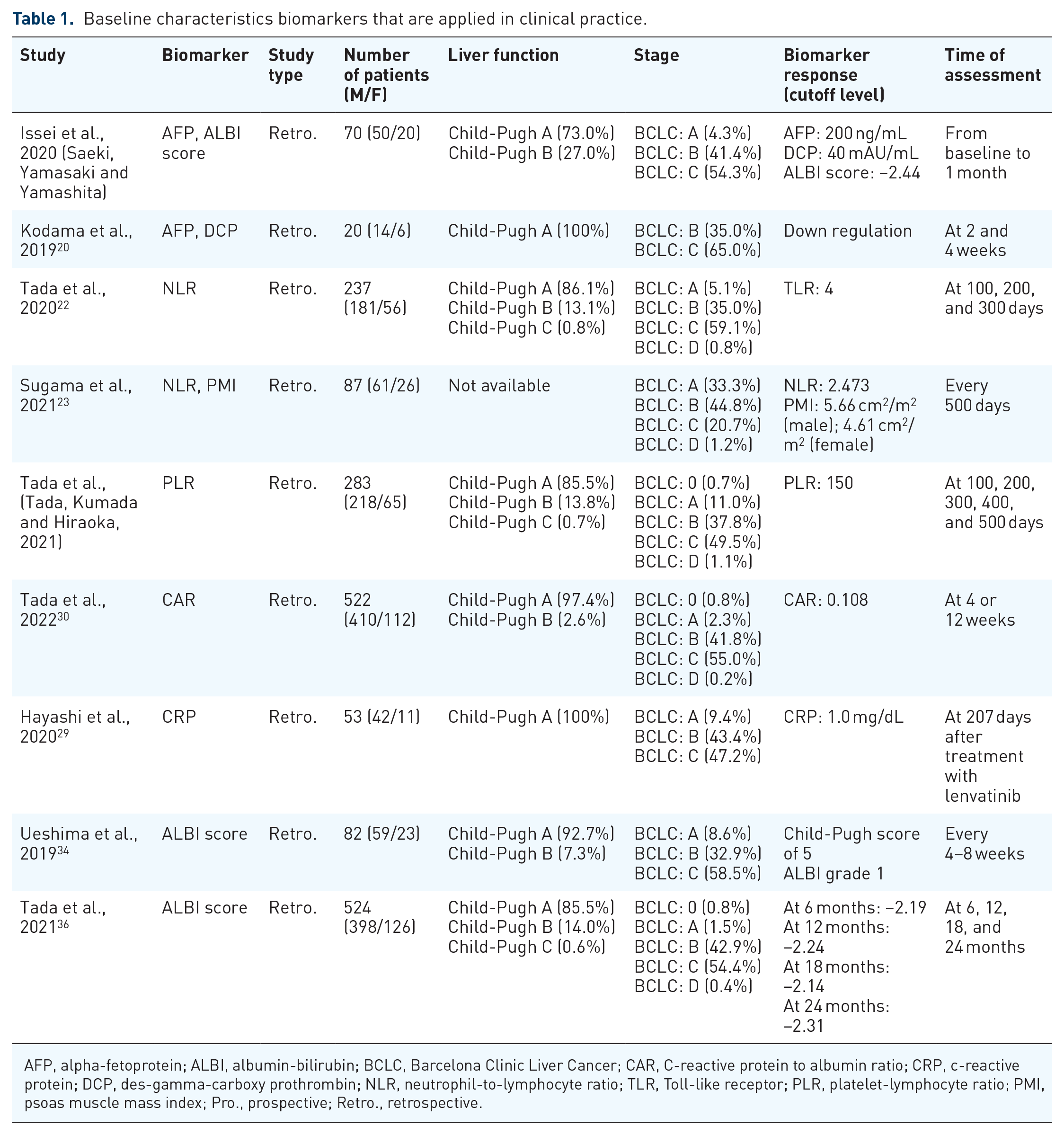
AFP, alpha-fetoprotein; ALBI, albumin-bilirubin; BCLC, Barcelona Clinic Liver Cancer; CAR, C-reactive protein to albumin ratio; CRP, c-reactive protein; DCP, des-gamma-carboxy prothrombin; NLR, neutrophil-to-lymphocyte ratio; TLR, Toll-like receptor; PLR, platelet-lymphocyte ratio; PMI, psoas muscle mass index; Pro., prospective; Retro., retrospective.
Alpha-fetoprotein
Approximately 60%–70% of patients diagnosed with primary liver cancer exhibit elevated plasma levels of alpha-fetoprotein (AFP). 16 Many studies have demonstrated that AFP is closely related to the diagnosis and prognosis of HCC patients.17,18
AFP response was defined as a decrease of 40% from the baseline (sensitivity: 1.000, specificity: 0.778, AUC: 0.396), which was a significant independent predictor of high ORR in the high AFP group (AFP ⩾ 10 ng/mL) in HCC patients treated with lenvatinib (odds ratio (OR): 51.389, 95% confidence interval (CI): 4.888–540.281, p = 0.001). 19 Similarly, it was reported that levels of AFP decreased at weeks 2 in most of lenvatinib-treated HCC patients, and the patients with sustained reduction in levels of AFP at weeks 4 was associated with a higher ORR. 20 The study also noted that levels of AFP increased when lenvatinib was reduced or discontinued, suggesting some correlation between levels of AFP and efficacy of lenvatinib. 20 Therefore, the anti-tumor effect of lenvatinib is closely related to AFP levels. However, all the above studies have the limitations of retrospective study design and sample limitation, and lack the consistency of cutoff value; more prospective studies with large sample sizes were needed to further verified.
Systemic inflammatory indicators
Neutrophil/lymphocyte ratio
Neutrophil/lymphocyte ratio (NLR) is a systemic inflammatory index that is inexpensive and easy to measure, reflecting a potential equilibrium between inflammation caused by neutrophils and the immune function against tumors mediated by lymphocytes. 21
NLR was independently associated with PFS (hazard ratio (HR): 1.897, 95% CI: 1.268–2.837) and OS (HR: 1.874, 95% CI: 1.097–3.119) in lenvatinib-treated HCC patients, and it was shown that patients who had high levels of NLR (⩾4) had significantly worse prognosis than those with low levels of NLR (<4; p = 0.007). 22 Additionally, the use of splines in the HR analysis clarified an appropriate cutoff of approximately 3.0–4.5 for the NLR to predict OS in HCC patients. 22 Conversely, it was reported that NLR did not show significant predictive value for lenvatinib treatment, but was a significant predictor of transarterial chemoembolization (TACE) in a retrospective study. 23 Furthermore, it was reported that the combined application of NLR and psoas muscle mass index (PMI) could play a predictive role. 23 They created a formula (=0.555 × [up-to-seven criteria out: 1, in: 0] + 0.161 × NLR − 0.191 × PMI), which showed that the low-level group in the lenvatinib group had better prognosis compared with the high-level group and could predict the 1 year survival rate better than Barcelona Clinic Liver Cancer (BCLC) alone, making it a promising predictive tool. 23 In conclusion, NLR could be a prognostic biomarker for u-HCC patients treated with lenvatinib, and the formula combined with PMI and NLR could help to predict the efficacy of lenvatinib in HCC. However, the relationship between NLR and efficacy of lenvatinib needs to be further analyzed and discussed in a large number of prospective studies.
Platelet-lymphocyte ratio
Platelet-lymphocyte ratio (PLR) is an indicator of systemic inflammation, which is cheap, easy to calculate, and was reported as a prognostic biomarker in HCC.24,25 It was shown that high PLR (⩾150) was significantly correlated with shorter OS (p = 0.021) and PFS (p = 0.035) in u-HCC patients treated with lenvatinib, but not with lenvatinib treatment response. 26 Therefore, PLR could predict the survival of lenvatinib-treated u-HCC patients. However, due to the selection bias caused by the limitation of the study population, future studies need to be further verified.
C-reactive protein
C-reactive protein (CRP), a common inflammation marker, and elevated levels indicate systemic inflammation. 27 It was reported that elevated CRP levels (cutoff value: 1.0 mg/dL) were related to poor prognosis in HCC patients, while also being closely related to HCC stage. 28
CRP could be used to predict OS in lenvatinib-treated u-HCC patients in a retrospective study, indicating that high levels of CRP (>1.0 mg/dL) are significantly related to worse OS (HR: 10.9, 95% CI: 3.68–32.0, p < 0.001). 29 Therefore, it could screen patients who would benefit from lenvatinib. In addition, CRP is simple and easily accessible, and its baseline levels also correlate with time to treatment failure (TTF), relative dose intensity (RDI), and response to lenvatinib. 29 Therefore, CRP holds promise as a practical and effective biomarker to support personalized treatment strategies and optimize therapeutic decision-making in HCC.
C-reactive protein-to-albumin ratio
It was found that C-reactive protein-to-albumin ratio (CAR) is an easily measurable and inexpensive biomarker that could be used as a biomarker to reflect liver function and HCC stage. 30 It was reported that lenvatinib-treated u-HCC patients with high CAR (⩾0.108) had significantly shorter OS and PFS than those with low CAR (p < 0.001). 30 Therefore, CAR could be used as a biomarker to predict OS and PFS in u-HCC patients treated with lenvatinib. However, due to the lack of a standardized cutoff value, further studies should validate the cutoff value to improve the accuracy of CAR in predicting the response to lenvatinib treatment.
Biomarker based on comprehensive risk scores
The albumin-bilirubin score
Albumin-bilirubin (ALBI) score, an evidence-based scoring system, was designed for the evaluation of liver function in HCC patients in 2014. 31 Moreover, it was reported that ALBI was classified into three grades and was independently correlated with OS of liver diseases such as HCC and cirrhosis.32,33
The baseline ALBI score and the variation from baseline to 1 month in ALBI score are predictors of ORR in lenvatinib-treated HCC patients, especially in the low-AFP group (OR: 6.866, p = 0.039). 19 Similarly, it was reported that ALBI grade 1 was an independent predictor of ORR in HCC patients receiving lenvatinib (p < 0.05). 34 Additionally, compared with ALBI scores ⩾ −2.44, HCC patients with ALBI scores <−2.44 had a lower rate of discontinuation, resulting in a longer lenvatinib treatment duration (6.1 vs 13.3 months, p = 0.014). 19
It was reported that ALBI grading has an impact on prognosis in a post hoc analysis of lenvatinib-treated u-HCC patients. 8 Besides, ALBI grade 1 and ALBI grade 2 HCC patients had longer median OS, higher ORR, and a lower incidence of grade ⩾ 3 treatment-related AEs in the lenvatinib group. 35 Moreover, mALBI grade 2b or 3 were independently associated with OS in patients with u-HCC who received lenvatinib therapy (HR: 2.123, 95% CI: 1.267–3.555, p = 0.004). 22 Similarly, it was found that the ALBI score is a more predictive measure of OS than the Child-Pugh score in treating u-HCC patients with lenvatinib, and the mALBI classification could forecast outcomes in patients with Child-Pugh scores of 5. 36 Furthermore, it was reported that ALBI grade 3 could be used to independently predict OS in HCC patients treated with lenvatinib (adjusted HR: 6.699, p = 0.0039). 37 Additionally, mALBI grade 1 is the most favorable indication for the use of lenvatinib in a retrospective study of 375 HCC patients. 38
In conclusion, ALBI grading, including mALBI classification, could serve as prognostic indicators and predict lenvatinib treatment outcomes in HCC patients. However, ALBI score is only a static indicator and could not reflect the dynamic changes during the treatment process with lenvatinib, which could not comprehensively and accurately predict the efficacy of lenvatinib in HCC patients when used alone.
AFP-GGT-Hangzhou (AGH) scoring system
The AGH scoring system included three factors: preoperative AFP levels above 200 µg/L, gamma-glutamyl transferase (GGT exceeding 96 U/L, and surpassing the Hangzhou criteria. 39 It was shown that these three factors were independently related to poor disease-free survival (DFS) in patients with HCC undergoing LT in multivariate Cox analysis (Li, Chen and Xing, 2023). Moreover, HCC patients were divided into high-, intermediate-, low-risk cohorts using the AGH scoring system, and the differences in OS and PFS among the three groups were statistically significant (p < 0.05), while patients with high-risk AGH score could be more effective with lenvatinib adjuvant therapy after LT. 39 Therefore, the AGH scoring system could be a simple and effective method to screen patients who derive benefits from lenvatinib after LT, and its integration of biomarkers and clinical criteria is worthy of further exploration and promotion. However, due to the small sample size of patients and the lack of mention of the application effect in other populations, its universality needs to be verified in a large number of studies.
Biomarkers that are highly relevant to clinical practice
There is growing evidence that several biomarkers, though not yet widely used in clinical practice, are highly clinically relevant and possess strong predictive and translational potential. Relevant studies are summarized in Table 2.
Baseline characteristics biomarkers that are highly relevant to clinical practice.

Ang2, angiopoietin 2; BCLC, Barcelona Clinic Liver Cancer; cfDNA, cell-free DNA; ctDNA, circulating tumor DNA; Cu/Zn: Copper/zinc; CXI, cachexia index; ECW/TBW, extracellular water to total body water ratio; FGF, fibroblast growth factor; LEP, lenvatinib prognostic index; M2BPGi, Mac-2 binding protein glycosylation isomer; Pro., prospective; Retro., retrospective; SMA, skeletal muscle attenuation; SMI, skeletal muscle index; TACE, transarterial chemoembolization; VAF, variant allele frequency; VEGF, vascular endothelial growth factor; MTA, molecular-targeted agent; PNI, prognostic nutritional index; RFA, radiofrequency ablation.
Biomarkers in serology
The predictive effect of biomarkers in serology on lenvatinib treatment for HCC has received extensive attention.
Angiopoietin 2
Angiopoietin 2 (Ang2), a ligand of the tyrosine kinase receptor Tie2, which exhibits widespread expression in embryonic tissues and promotes angiogenesis and tumor growth.40 –42 It was reported that levels of Ang2 were increased in 131 HCC patients compared with healthy controls in a retrospective study (p < 0.0001). 43 Besides, Llovet et al. 44 showed that elevated levels of Ang2 were related to worse OS in HCC patients in a randomized controlled trial.
There was a significant correlation between lenvatinib response and baseline serum levels of Ang2 (p = 0.017) and all HCC patients (9/9) receiving lenvatinib who had low baseline levels of Ang2 achieved an objective response (cutoff level: 3108 pg/mL). 45 Similarly, it was shown that levels of Ang2 were significantly lower in the objective response group compared with the lenvatinib non-objective response group with HCC in a prospective cohort study (p = 0.0002). 46 Besides, it was reported that baseline levels of Ang2 were significantly negatively associated with treatment response to lenvatinib in a retrospective study (p = 0.002). 47 Additionally, it was identified that Ang2 as an independent biomarker associated with poor prognosis in HCC patients and high levels of Ang2 correlates with a shorter OS in 279 HCC patients treated with lenvatinib in a randomized controlled trial (HR: 1.436; p < 0.0001). 48 Moreover, it was reported that increased levels of Ang2 could be associated with worse liver function reserve (cutoff level: 3108 pg/mL), predicting poor PFS and OS in lenvatinib-treated HCC patients in a retrospective study. 49 Furthermore, high baseline Ang2 was found to be linked with a shorter median OS in the subgroup with hepatitis B virus etiological of lenvatinib-treated HCC patients, but not in the hepatitis C virus etiological subgroup. 48 Therefore, Ang2 could be used as a predictive marker to predict the efficacy of lenvatinib treatment and could determine whether u-HCC patients are suitable for systemic therapy. However, the expression of Ang2 is affected by many factors, and the use of Ang2 alone could not accurately predict the efficacy of HCC patients. Therefore, the combination of other predictors can improve the accuracy of predicting the treatment effect of lenvatinib.
Fibroblast growth factors
Fibroblast growth factor 19 (FGF19) plays a necessary role in glycogen synthesis, gluconeogenesis, and protein synthesis.50,51 The objective response group had a significantly higher levels of FGF19 at 4 and 8 weeks compared with the non-objective response group in lenvatinib-treated HCC patients in a retrospective study (cutoff value: FGF19/baseline: 1.51). 46 Besides, it was reported that there was a significant negative relationship between baseline FGF19 and the response of lenvatinib in HCC patients in another retrospective study (cutoff value: 194 pg/mL, p < 0.001). 45 Besides, it was reported that the combination of increased FGF19 levels and decreased Ang2 levels is not only an indicator of favorable response to lenvatinib therapy (p = 0.0012) but is also associated with longer PFS in HCC patients (p = 0.0240; cutoff value: FGF19/baseline: 1.51, Ang2/baseline: 0.67). 46 Similarly, it was found that the combination of low baseline levels of Ang2 and FGF19 could predict favorable efficacy of lenvatinib in u-HCC patients in a retrospective study (cutoff value: Ang2: 3108 pg/mL, FGF19: 194 pg/mL). 45 However, due to inconsistent cutoff values, small sample sizes, and retrospective design, further studies with larger samples are needed.
FGF21 could be a biomarker for assessing the functional status of hepatocellular injury, which is significantly correlated with prognosis in HCC patients. 52 It has been shown that a higher baseline level of FGF21 is correlated with shorter OS in lenvatinib-treated HCC patients (HR: 2.475, 95% CI: 1.565–3.922, p = 0.0001). 48 However, it was reported that FGF21 levels were higher in lenvatinib-treated HCC patients with an objective response than in those with non-objective response in a retrospective study, but there was no statistical significance (p = 0.147). 45
FGF23 is produced and released into the blood primarily by bone cells and acts as a hormone in the kidney, stimulating the excretion of phosphate and inhibiting the synthesis of 1,25(OH)2D3.53 –55 It was found that changes in the increased levels of FGF23 were correlated with the favorable efficacy of lenvatinib treatment in HCC patients (p = 0.0022). 48 Furthermore, high baseline levels of FGF23 are one of the factors that constitute the signature of cytokines and angiogenic factors (CAFs), which are correlated with low RDI and shorter PFS in 41 lenvatinib-treated HCC patients. 56
In conclusion, FGFs could be important candidate biomarkers to predict the response to treatment with lenvatinib. However, more large-sample prospective studies are needed to confirm them as predictors of lenvatinib efficacy in HCC patients. Furthermore, whether the combination of FGFs with other biomarkers offers better predictive value is worth further investigation in future research.
Vascular endothelial growth factor
Lenvatinib, an inhibitor of tyrosine kinase receptors, effectively inhibits the phosphorylation of vascular endothelial growth factor (VEGF) receptors (VEGFR1, VEGFR2, and VEGFR3), which leads to an increase in VEGF levels compared to baseline after treatment with lenvatinib. 57 A significant correlation has been reported between higher baseline VEGF and shorter OS in 279 lenvatinib-treated HCC patients (HR: 1.181, p = 0.0037). 48 Additionally, low serum levels of VEGF (cutoff value: 514.9 pg/mL) were significantly correlated with worsening hepatic functional reserve in HCC patients at 8 weeks from the initiation of lenvatinib treatment (p = 0.0259). 49 However, VEGF levels were not significantly different between the objective response group and the non-objective response group in HCC patients at 4 weeks of lenvatinib treatment compared with baseline in a prospective cohort study (p = 0.540). 46 Therefore, VEGF has great potential in predicting HCC survival with lenvatinib. However, whether serum VEGF can predict the efficacy of lenvatinib in HCC still needs to be confirmed by further prospective studies.
Combination of Ang2 and VEGF levels
Ang2 and VEGF were significantly correlated with survival after lenvatinib treatment, so the combination of Ang2 and VEGF levels was further investigated in HCC.48,49 It was shown that most HCC patients (8/9) with low baseline levels of VEGF and high levels of Ang2 showed worse Child-Pugh scores at 8 weeks from the initiation of lenvatinib treatment (cutoff value: Ang2: 3108 pg/mL, VEGF: 514.9 pg/mL), whereas patients with high baseline VEGF and low levels of Ang2 (0/9) did not showed worse Child-Pugh scores. 49 Therefore, the combination of Ang2 and VEGF could serve as a potential predictive biomarker of survival in u-HCC patients receiving lenvatinib. Previous study reported that the combination of Ang2 and VEGF could predict liver function after lenvatinib treatment; considering that their combination was also associated with survival after lenvatinib in HCC patients, whether it could be used as a biomarker for predicting the effectiveness of lenvatinib needs to be verified in future prospective studies.
Other serum growth factors
It was reported that patients with increased growth factors were significantly associated with shorter post-progression survival (PPS) in lenvatinib-treated HCC patients (p = 0.032). 47 Additionally, it was reported that lenvatinib-treated HCC patients who displayed favorable 9-CAFs characteristics are significantly associated with longer PFS (HR: 0.42, 95% CI: 0.18–0.96, p = 0.040). 56 Therefore, serum growth factors could serve as predictors to predict lenvatinib treatment response; more studies are needed due to the retrospective design and small sample size of current research.
Cell-free DNA
Circulating cell-free DNA (cfDNA) is a fragment of DNA released into the bloodstream by apoptosis, necrosis, or living cells, including circulating tumor DNA (ctDNA). 58 Moreover, previous studies reported that cfDNA could be used to reflect the progression of HCC, and the higher the concentration of cfDNA, the larger the tumor size and the higher tumor grade.59 –61
It was reported that in patients with HCC who received lenvatinib, cfDNA levels generally increased after therapy, and the magnitude of this increase was significantly greater in those achieving an objective tumor response (OTR) compared to non-OTR group (p = 0.048). 62 In another study involving 24 HCC patients, monitoring the changes in the mean variant allele frequency (VAFmean) of single-nucleotide variants in ctDNA revealed that a decline in VAFmean at week 4 could accurately predict partial response, with high sensitivity (1.0) and moderate specificity (0.67). 63
Although cfDNA elevation and ctDNA VAF reduction may appear contradictory, they in fact reflect distinct biological processes in response to treatment: an increase in total cfDNA likely indicates apoptosis or necrosis of tumor cells induced by therapy, while a decrease in ctDNA VAF reflects a reduction in the proportion of tumor-derived DNA. These two biomarkers thus provide complementary insights into therapeutic efficacy. Therefore, cfDNA holds promise as a noninvasive and dynamically monitorable predictive biomarker in clinical practice. However, its predictive reliability may be limited when used alone due to its susceptibility to non-tumor-related effects such as liver injury or inflammation. It is recommended to comprehensively evaluate cfDNA in combination with other biological indicators to improve the predictive performance.
Cachexia index
Cancer cachexia negatively impacts quality of life and survival in many patients with advanced cancer. 64 A new cachexia index (CXI) has been developed and calculated using (L3 skeletal muscle index) × (serum albumin)/NLR, which was originally proposed for non-small cell lung cancer. 65 It was shown that low CXI (<53) was independently related to shorter PFS (HR: 1.84, 95% CI: 1.09–3.09, p = 0.02) and OS (HR: 2.07, 95% CI: 1.17–3.65, p = 0.01) in lenvatinib-treated HCC patients. 66 Furthermore, disease control rate (DCR) and anorexia rates were also significantly lower in HCC patients with low CXI (<53) compared to patients with high CXI (⩾53) during the first 2 months of lenvatinib treatment. 66 Therefore, CXI could be a potential biomarker to predict the efficacy of lenvatinib in the treatment of patients with HCC. The weight and optimal cutoff value of each component of CXI need to be further explored in large-sample prospective studies.
Lenvatinib prognostic index
Lenvatinib prognostic index (LEP) is a new prognostic score that incorporates five factors, including lymphocytes, bilirubin, albumin, BCLC stage, and previous TACE (yes/no). 67 The LEP index categorizes HCC patients receiving lenvatinib treatment into low-, intermediate-, and high-risk cohorts, with significant differences observed in OS among them (29.8, 17.0, and 8.9 months; p < 0.0001). 67 Moreover, it was shown that lenvatinib has a positive predictive effect in the low-risk HCC group (p < 0.0001). 67 Therefore, LEP could be a potential indicator for predicting the efficacy of lenvatinib in the treatment of HCC patients. However, further investigation is needed because of retrospective design of current research, and a small number of low-risk patients were included.
Biomarkers based on changes in body composition
Changes in body composition play a crucial role in predicting outcomes in HCC treatment. 68 It was reported that there was a significant association between changes in body composition and TTF along with prognosis after 2 weeks in lenvatinib-treated HCC patients. 68 Besides, lenvatinib treatment led to a reduction in mean skeletal muscle attenuation within 2 weeks (p = 0.004), which was proved to be a reliable predictor of TTF in lenvatinib-treated HCC patients. 68 Similarly, decreased skeletal muscle index (SMI) is related to the worse TTF and OS in lenvatinib-treated HCC patients (TTF, p = 0.010; OS, p = 0.021). 69 Moreover, it was revealed that SMI of arm before lenvatinib treatment could reflect longer PFS in HCC patients (HR: 2.12, 95% CI: 1.23–3.64, p = 0.0069). 70 Furthermore, extracellular water to total body water ratio ⩽ 0.400 before lenvatinib treatment was significantly related to longer PPS (HR: 3.08, 95% CI: 1.32–7.18, p = 0.0093), OS (HR: 4.72, 95% CI: 12.03–11.00, p < 0.001), and PFS (HR: 2.66, 95% CI: 1.33–5.34, p = 0.0057) in HCC patients. 70 Therefore, assessment of body composition before and after lenvatinib treatment could be helpful to predict the efficacy of lenvatinib in HCC patients. However, changes in body composition are a complex process, and further large-scale studies and clinical trials are still needed to validate and confirm the effectiveness and practicality of using changes in body composition to predict lenvatinib efficacy.
Copper/zinc and metallothionein
Copper (Cu) and zinc (Zn) are basic nutritional metals related to metabolism in human cells. 71 Cu has a close relationship with cell proliferation and tumor angiogenesis. 31 Impaired zinc and/or copper function could occur at different stages of liver disease and could correlate with the severity of liver disease.72 –77 Additionally, metallothionein (MT) is a protein of low molecular weight containing large amounts of cysteine, which could be highly induced by metal ions like Cu and Zn. 78
A higher survival rate was shown in HCC patients receiving lenvatinib with Cu/Zn <0.999 compared to Cu/Zn ⩾ 0.999 (p < 0.001). 71 They also observed that lenvatinib could induce MT in HCC patients, and there was a significant positive relationship between MT levels and Cu/Zn in HCC patients. 71 Furthermore, it was reported that elevated levels of MT could be related to decreased survival in lenvatinib-treated HCC patients, and this needs to be further investigated. 71 Therefore, Cu/Zn and/or MT could be useful in assessing lenvatinib treatment resistance and survival outcomes of HCC patient. However, serum MT was not correlated with serum Cu or Zn levels measured separately, so the relationship between MT and other heavy metals in HCC and alterations in MT expression during Zn supplementation could be further explored in future studies.
Mac-2 binding protein glycosylation isomer
Wisteria sinensis agglutinin positive Mac-2 binding protein glycosylation isomer (M2BPGi) could serve as a serum biomarker to predict the stage of liver fibrosis and was found to be associated with nutritional status for HCC patients (p < 0.001).79,80 Pretreatment levels of M2BPGi could be used as predictive markers for PFS (HR: 0.52, p = 0.0358) and also predict the tolerance and response to lenvatinib in HCC patients. 81 Moreover, low serum levels of M2BPGi (<1.5) were correlated with longer cumulative PFS (p = 0.0003), higher RDI (p < 0.0001), ORR (p = 0.0004), DCR (p < 0.0001), and longer duration of lenvatinib treatment (p < 0.0001) compared with high levels of M2BPGi in HCC patients. 81 Therefore, M2BPGi could be used as a potential biomarker in HCC patients treated with lenvatinib, and HCC patients with low levels of M2BPGi could benefit from lenvatinib treatment. However, further research is needed due to the retrospective design, small sample size of current researches, and lack of external validation.
Biomarkers that are under exploratory stage
Circular RNA
Circular RNAs (circRNAs) are formed by a 5′-3′-phosphodiester bond, regulating biological processes and transcription, altering epigenetic modifications, and acting as competing endogenous RNAs.82 –84 Also, circRNAs were reported to be used as a prognostic biomarker to predict the prognosis of HCC and the therapeutic effectiveness of targeted therapy. 85
The expression of circKCNN2 significantly downregulated in HCC tissues compared to normal tissues (p < 0.001), and elevated levels of circKCNN2 significantly predicted longer OS (p = 0.013) and recurrence-free survival (p = 0.011) in lenvatinib-treated HCC. 86 However, the presence of high levels of circKCNN2 in HCC cells counteracts the anti-tumor effects of lenvatinib (possibly because both of them inhibit FGFR4 expression) and confers resistance to lenvatinib. 86 Moreover, it was reported that HCC cells that express circKCNN2 ectopically or have low intrinsic levels of circKCNN2 are more sensitive to lenvatinib and have a better treatment effect with lenvatinib in HCC patients. 86 Therefore, circKCNN2 could be used as a potential biomarker for the efficacy of lenvatinib in HCC patients, but it requires further translational research before conducting clinical trials.
MicroRNA
MicroRNAs (MiRNAs) are a group of non-coding, single-stranded RNA molecules with a length of around 22 nucleotides encoded by endogenous genes. MiRNAs are closely associated with the proliferation, invasion, and metastasis of HCC, and some miRNAs were already used as potential targets to develop novel anticancer drugs.87,88
It was reported that miR-3154 was increased in HCC and liver cancer stem cells compared with adjacent normal tissue, and upregulation of miR-3154 was associated with short OS and DFS in HCC patients (p < 0.05). 89 Furthermore, miR-3154 could inhibit HNF4α (a tumor suppressor), promote HCC progression, and determine the response of HCC cells after lenvatinib treatment (as knockdown of miR-3154 makes HCC cells more sensitive to treatment with lenvatinib). 89 Additionally, the combination of low expression of miR-3154 and high expression of HNF4α was related to better prognosis and predicted better DFS in lenvatinib-treated HCC patients. 89 Therefore, miR-3154 had the potential to serve as a new biomarker for the individualized treatment of lenvatinib in HCC and improve the accuracy of prediction when combined with HNF4α. However, it should be noted that this result is based on laboratory studies, and the existing evidence may have false positives and biological heterogeneity. Therefore, its predictive potential urgently requires further validation through rigorous preclinical studies, large-scale clinical trials, and standardized detection methods to confirm its effectiveness.
Single-nucleotide polymorphisms
Previous studies have reported that single-nucleotide polymorphisms (SNPs) were related to the risk of liver cancer, which is important for understanding the genetic risk and pathogenesis of HCC.72,90 –95 It was reported that the TC/CC genotypes of NOS3 rs2070744 correspond to poor outcomes in u-HCC patients treated with lenvatinib, but the results showed no statistical significance. 96 Furthermore, the combined SNP pattern of FGFR4 rs351855 and NOS3 rs2070744 genotype could be useful as predictors of treatment response and prognostic factors in lenvatinib-treated HCC patients (PFS: (HR = 2.56, p = 0.006); OS: (HR = 3.36, p = 0.013)). 96 Additionally, ABCG2 421C > A C/A or A/A was significantly related to the occurrence of anorexia (OR: 9.009, 95% CI: 1.750–46.383, p = 0.009) and could affect exposure to lenvatinib in HCC patients in a prospective study. 97 Moreover, a retrospective study found that a reduction in VAFmean of single-nucleotide after 4 weeks of lenvatinib treatment was associated with partial response in 24 HCC patients (specificity: 0.67, sensitivity: 1.0). 63 Therefore, SNPs could be a potential biomarker in predicting the efficacy of lenvatinib in HCC. However, the combination and interaction of genetic variations have a more significant impact on lenvatinib efficacy compared with individual SNPs.
Mucin 15
Mucin 15 (MUC15) is a member of the transmembrane mucin family, which acts primarily to hydrate, lubricate, and protect epithelial surfaces. 98 It was reported that downregulation of MUC15 is associated with poor late differentiation and metastasis of HCC. 99 It was reported that MUC15 expression is significantly lower in lenvatinib-resistant HCC patients and recurrent HCC, compared to primary lesions. 100 Besides, lenvatinib efficacy was upregulated in HCC cells that overexpress MUC15, which could predict the benefit of lenvatinib in HCC patients. 100 Therefore, MUC15 could serve as a potential biomarker for lenvatinib-treated HCC patients, which deserve further research in biomarker-driven basic and clinical trials.
ST6GAL1
ST6GAL1, a secreted protein derived from tumors, is positively regulated by FGF19 in hepatoma cells, while FGF19 could be a potential tumor biomarker for lenvatinib-susceptible HCC. 101 Additionally, lenvatinib was significantly related to better survival than sorafenib in HCC patients with high serum levels of ST6GAL1 (p < 0.05). 101 Therefore, serum ST6GAL1 could be used to identify patients with FGF19-driven invasive HCC who benefit from lenvatinib treatment and could be a novel and potential predictor. However, as the current evidence mainly comes from laboratory studies and has not been verified in clinical cohorts, more preclinical and translational studies are needed to further evaluate its application potential in the future.
Ongoing trials of biomarkers for lenvatinib in HCC
After consulting the website (http://apps.who.int/trialsearch), Table 3 provides a summary of the ongoing trials of biomarkers that predicted the effect of lenvatinib treatment in HCC patients, which could guide clinical practice in future.
Overview of ongoing trials of biomarkers for lenvatinib in HCC.

AFP, alpha-fetoprotein; ANG2, Angiopoietin 2; DCR, disease control rate; DLT, dual liver transplantation; FGF, fibroblast growth factor; HBsAg, hepatitis B surface antigen; HCC, hepatocellular carcinoma; IV, intravenous; LPI, liver perfusion imaging; LT, liver transplant; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; PVTT, portal vein tumor thrombosis; RBC, red blood cell; RFS, recurrence-free survival; SBRT, stereotactic body radiotherapy; TACE, transarterial chemoembolization; TTP, time to progression; VEGF, vascular endothelial growth factor; CEA, carcinoembryonic antigen.
Conclusion
Lenvatinib shows great promise as a treatment option for HCC. Numerous predictive biomarkers are currently being identified and studied, which serve as crucial tools to provide HCC patients with a more effective and individualized treatment plan (Figure 1). Based on clinical relevance of biomarkers, we classified the predictive biomarkers for lenvatinib in HCC into three groups: (1) biomarkers that are applied in clinical practice, (2) biomarkers that are highly relevant to clinical practice, and (3) biomarkers that are under exploratory stage. Although some biomarkers are still in the exploratory stage, this review includes them with the aim of providing promising directions for future research. As future trials continue to shed light on predictive biomarkers for the efficacy of lenvatinib in HCC, these biomarkers promise to help doctors to evaluate the response of lenvatinib treatment more accurately and, thus, improve prognosis of HCC patients.

Biomarkers for prediction of lenvatinib treatment in hepatocellular carcinoma.
It is noteworthy that the superior adaptability of these predictive biomarkers in lenvatinib treatment is closely associated with its unique target inhibition characteristics. Therefore, it is essential to further investigate the differences in biomarker responsiveness between lenvatinib and other TKIs based on their molecular mechanisms of action. Lenvatinib possesses a unique multi-target inhibition profile (VEGFR1–3, FGFR1–4, PDGFR-α/β, RET, KIT) which distinguishes it from other TKIs. 102 For instance, FGF19/21 is more likely to predict the efficacy of lenvatinib due to its potent FGFR inhibition, whereas sorafenib—primarily targeting VEGFR2/PDGFR—shows greater sensitivity to VEGF-A but weaker FGFR suppression. 103 Similarly, Ang-2 synergistically promotes angiogenesis with VEGF, and the potent VEGFR inhibition of lenvatinib may make it more sensitive to Ang-2 levels, whereas other TKIs may have different effects on vascular remodeling. 104 These mechanistic differences underscore the need for biomarker selection tailored to specific drugs.
Except for mechanism-driven biomarkers, general indicators like AFP and liver function metrics (e.g., ALBI score) exhibit predictive value across TKIs. AFP regulates angiogenesis, and high AFP levels may be associated with VEGF signaling pathway activation, which have shown significant predictive value in the non-lenvatinib TKI treatment of liver cancer. 105 Moreover, the liver function evaluation index ALBI score was confirmed as an important predictor of TKIs (such as lenvatinib and sorafenib), reflecting the effect of hepatic metabolic function on drug effectiveness. 106 Furthermore, factors such as tumor heterogeneity, dynamic changes during tumor growth, the degree of affinity between biomarkers and drugs, and the sensitivity of detection methods may also influence the predictive performance of different biomarkers for various TKIs. These findings collectively suggest that although some markers have broad predictive value, the predictive efficacy of biomarkers is often modulated by the mechanism of action of specific TKIs, and the most appropriate predictor should be selected in combination with specific drugs in clinical application.
Although this review provides a comprehensive summary of candidate biomarkers for predicting lenvatinib response in HCC, the majority of studies are retrospective currently, which limited by small sample sizes and inconsistent cutoff values, which hinder their direct translation into clinical practice. Moreover, most studies lack detailed descriptions of pre-analytical technical variables, such as sample collection timing, assay platform consistency, and data normalization procedures, which further impedes comparability across studies.
In our opinion, not all biomarkers could be recommended for future clinical trials. Biomarkers that are applied in clinical practice and biomarkers that are highly relevant to clinical practice in our review are promising for clinical applications and should be prioritized for validation in future large-scale, prospective, multicenter clinical trials. Moreover, although certain markers, such as circKCNN2, miR-3154, SNPs (e.g., NOS3 rs2070744), MUC15, and M2BPGi, have so far been proposed in preclinical experiments, they should require further translational researches before conducting clinical trials.
In conclusion, whether to advance a specific biomarker to subsequent clinical trials should not only depend on technical feasibility but also take into account its clinical convenience and patient acceptability. In particular, as there are limitations in single indicator, it is suggested to carry out clinical trials in future that focus on comprehensive scoring of multiple indicators in HCC patients.
