Abstract
Background:
Lenvatinib (LEN) is a multikinase inhibitor used for different tumors: advanced differentiated thyroid cancers progressed or resistant to radioiodine (RAIR DTC), adenoid cystic carcinoma of salivary glands (AdCC), hepatocellular carcinoma (HCC), and endometrial carcinoma (EC). In the real-world setting, patients eligible for LEN often have multiple comorbidities and/or impaired Eastern Cooperative Oncology Group performance status (ECOG PS). In HCC, patients with body weight (BW) < 60 kg require a low starting dose of LEN; in thyroid cancers (TC), BW has a clinically relevant effect on steady-state exposure to LEN. It is currently unknown whether BW can vary during LEN treatment across different histologies.
Objectives:
This study aims to investigate toxicities, duration of treatment (DoT), and to describe BW and body mass index (BMI) intrapatient variations in the real-world setting.
Design:
Retrospective observational study conducted in a tertiary cancer center.
Methods:
We collected clinical features and AEs from consecutive adult patients who started LEN for DTC, AdCC, HCC, or EC from January 2015 to April 2023. LEN first and last doses were estimated as a percentage of the standard dose approved for each indication. Differences in the BMI distribution assessed at baseline and end of treatment were assessed with Mann–Whitney test. Differences in intrapatient BMI variation were assessed with Kruskal–Wallis test. DoT and overall survival (OS) were estimated with the Kaplan–Meier method.
Results:
Over a period of 8 years and 3 months, n.161 patients were included: n.64 DTC, n.31 AdCC, n.41 HCC, and n.25 EC. Median follow-up (mFUP) was 72.8 months. Overall, median age at diagnosis was 61.8 years, with an ECOG PS 2 in 2%–9% of cases. The most frequently observed AE was fatigue. For the net of different cancer types, baseline BW/BMI, LEN doses, and mFUP, the BMI distribution was lower at last LEN administration compared to baseline (p = 0.045).
Conclusion:
Despite the higher prevalence of elderly subjects and ECOG PS 2, our series confirmed the long-term manageability and effectiveness of LEN in the real world. Patients with a baseline low BMI require special attention, as LEN treatment can be implicated in further BW decrease.
Keywords
Introduction
Lenvatinib is a multikinase inhibitor (MKI) targeting the vascular endothelial growth factor receptors (VEGFR)-1/2/3, fibroblast growth factor receptors (FGFR)-1/2/3/4, platelet-derived growth factor receptor alpha (PDGFRα), stem cell factor receptor (KIT), and ret proto-oncogene (RET). It interferes with tumor growth and cancer progression through a multifactorial mechanism of action combining anti-angiogenesis, immunomodulation, and direct antitumor activity. Currently, in Italy, lenvatinib is approved as monotherapy for the treatment of differentiated thyroid carcinoma (DTC) progressed or become resistant to radioiodine (RAIR), adenoid cystic carcinoma (AdCC) of the salivary glands, hepatocellular carcinoma (HCC), and in combination with pembrolizumab for endometrial carcinoma (EC). Full-dose lenvatinib differs for each indication. In HCC, patients (pts) with body weight (BW) < 60 kg require a low starting dose; in TC, BW has a clinically relevant effect on steady-state exposure to lenvatinib. 1 It is currently unknown whether BW can vary significantly during lenvatinib treatment across different histologies.
RAIR DTC are highly vascularized cancers with poor chemosensitivity. Lenvatinib is recommended as first-line (1L) therapy based on the results of the phase III placebo-controlled study SELECT, showing a benefit of lenvatinib in terms of progression-free survival (median PFS: 18.3 vs 3.6 months) and objective response rate (ORR: 64.8%). 2 Treatment-related adverse effects (TRAEs) of grade 3 or higher (⩾G3) occurred in 75.9% of patients (41.8% hypertension, 10% proteinuria, and 9.2% fatigue), leading to treatment discontinuation in 14.2% of cases. The median duration of treatment (mDoT) was 13.8 months and median overall survival (mOS) was not reached. 2 The 24 mg/day starting dose is currently recommended in all patients whenever feasible, while 10–18 mg/day can be considered for patients with Eastern Cooperative Oncology Group (ECOG) performance status ⩾ 2 and/or high risk of fistula/bleeding. 3 Compared to 24 mg/day, the 18 mg/day starting dose failed to prove non-inferiority in terms of efficacy (6-months ORR 40.3% vs 57.3%). 4 Dose interruptions of lenvatinib lasting ⩾ 10% of the total treatment duration have been correlated to worse efficacy and shorter PFS. 5 Thus, special attention has been addressed to the management of toxicities, to optimize the adherence to treatment. 6
AdCC may arise from the salivary glands, breast, lung, and skin. It can be treated by surgery followed by radiotherapy (RT), or by exclusive RT/particle therapy in unresectable cases. 7 Metastatic AdCC of the salivary glands can show either an indolent evolution, usually observed in subjects with a long disease-free interval and lung metastases, or a more aggressive behavior with liver and/or bone involvement. 8 MKIs lenvatinib and axitinib have shown activity in metastatic AdCC. 9 The indication of lenvatinib in 1L therapy is based on the results of two phase II single-arm trials conducted in the United States and Europe on 33 and 26 patients, respectively, receiving lenvatinib at a dose of 24 mg daily.10,11
HCC accounts for approximately 80% of all primary liver cancers. 12 Patients with unresectable early stage disease can receive locoregional treatments such as ablation and/or transarterial chemoembolization (TACE), but many are diagnosed with extensive/advanced disease amenable only to systemic treatment. 13 In 2018, lenvatinib was approved as a 1L treatment for advanced HCC as an alternative to sorafenib, based on the results of the phase III multicenter, randomized, open-label study REFLECT, designed as non-inferiority trial involving 954 patients. 14 Patients received lenvatinib 12 mg/day (for bodyweight ⩾ 60 kg) or 8 mg/day (for bodyweight < 60 kg). Compared with sorafenib, lenvatinib resulted to be non-inferior in terms of mOS (13.6 vs 12.3 months, HR, 0.92, 95% CI, 0.79–1.06), and significantly improved secondary endpoints such as mPFS (7.4 vs 3.7 months) and ORR (24.1 vs 9.2%). In the lenvatinib group, treatment-emergent adverse events (TEAEs) ⩾ G3 occurred in 57% of cases (23% hypertension, 8% decreased weight, and 7% increased blood bilirubin). 14
EC is the most common gynecological malignancy, in 20%–30% of cases caused by mismatch repair deficiency (dMMR) or high microsatellite instability (MSI-H). MMR status has both prognostic and therapeutic implications, as single-agent PD-1 blockade is less effective for MMR proficient (pMMR) tumors. Lenvatinib 20 mg/day in combination with pembrolizumab 200 mg every 3 weeks is currently approved for the treatment of patients with advanced EC with disease progression following prior platinum-based chemotherapy (ChT), based on the phase III multicenter, open-label, active-controlled Study 309/KEYNOTE-775 15 where 827 patients were randomized (1:1) to receive lenvatinib plus pembrolizumab or Investigator’s choice ChT. Independently of the MMR status, compared to ChT, the lenvatinib arm showed a significant benefit in ORR (33.8% vs 14.7%), mPFS (7.2 vs 3.8 months; HR, 0.56; 95% CI, 0.48–0.66; p < 0.001), and mOS (18.3 vs 11.4 months; HR, 0.65; 95% CI, 0.55–0.77; p < 0.001). 15 TEAEs ⩾ G3 occurred in 90.1% patients who received lenvatinib plus pembrolizumab (39.2% hypertension, 10.8% weight decrease, and diarrhea 8.1%) and 35.7% discontinued lenvatinib due to AEs. 15
Methods
This is an observational study, conducted at Fondazione IRCCS Istituto Nazionale Tumori (INT) in Milan, Italy, in compliance with Good Clinical Practice guidelines and in accordance with the Declaration of Helsinki. The protocol was approved by the institutional Ethical Committee (INT 256/23). All adult patients who started lenvatinib treatment for advanced RAIR DTC (subsequently abbreviated as DTC), AdCC, HCC, and EC from January 2015 to April 2023 were retrospectively retrieved through the Institutional database of delivered oral therapies. Clinical features at baseline and toxicities observed during the lenvatinib treatment were retrieved from the Institutional electronic medical records. For EC, we excluded all immuno-related AEs related to Pembrolizumab. Disease extension at diagnosis was evaluated according to the TNM Staging System (eighth Edition) for DTC, AdCC, and HCC, and the 2009 FIGO Staging system for EC. The doses of lenvatinib have been estimated in terms of percentage of the standard dose approved for each indication (24 mg/day for DTC and AdCC, 20 mg/day for EC, 8 or 12 mg/day for HCC according to the bodyweight < 60 kg or ⩾60 kg, respectively). Adverse events grading has been reported according to the Common Terminology Criteria for the Adverse Events (CTCAE) v.4. Differences in the body mass index (BMI) distribution assessed at baseline and end of treatment were assessed using Mann–Whitney test. Differences in intrapatient BMI variation were assessed using the Kruskal–Wallis test. Median duration of lenvatinib treatment (mDoT; defined as the median number of months from day 1 of lenvatinib to the last day of treatment due to any cause) in months was estimated using the Kaplan–Meier method. Duration of treatment (DoT) was compared with the log-rank test stratifying patients according to basal body weight (BW) < or ⩾60 kg. The cutoff date for data collection was September 30th, 2023.
Median follow-up was estimated with the reverse Kaplan–Meier method. Overall survival (OS) was measured from the date of first lenvatinib administration to the date of last follow-up (for censored cases) or death. The DoT was defined as the interval from the date of start to the date of end of lenvatinib administration, independently of the reason for treatment stop (event). Alive patients with ongoing treatment were censored at last follow-up. Both OS and DoT were measured as time-to-event intervals and estimated with Kaplan–Meier method.
The reporting of this study conforms to the STROBE statement for reporting observational studies (Supplemental File). 16
Results
Across a period of 8 years and 3 months, 161 consecutive patients met the criteria to be included in the study, covering different spans of years for each histology-specific group: 64 DTC (2015–2023), 31 AdCC (2019–2023), 41 HCC (2019–2023), and 25 EC (2022–2023) (Figure 1). Median follow-up (mFUP) in months was 72.8 (range 51.9–97.6) in TC, 24.8 (17.1–NR) in AdCC, 23.5 (21.1–NR) in HCC, and 16.0 (13.1–21.4) in EC.

Study population distribution by cancer histotypes.
Among non-gynecological cancers, a female prevalence was observed, especially in HCC (85.4%) and DTC (59.4%). Overall, the median age at diagnosis was 61.8 years (y), the highest in HCC (64 y) and the lowest in AdCC (53 y). The most frequent comorbidities found at diagnosis were hepatitis C virus infection (47.8% of HCC and 6.2% of DTC), venous thrombosis (12.9% of AdCC, 8% of EC, and 6.2% of DTC), and hepatitis B virus infection (17.1% of HCC, 4% of EC, and 1.6% of DTC). The stage of disease was available for 96.3% of cases: in all groups, the majority of patients had been diagnosed with early/locally advanced cancer and received locoregional treatments upfront. Except for the EC group, lenvatinib was chosen as first-line systemic therapy in more than 80% of patients treated for DTC/AdCC and in all HCC patients (Table 1).
Baseline patients characteristics.

Patients’ clinical features at lenvatinib start (ECOG PS, BMI, cardiovascular parameters, metastatic spread), medication’s starting dose expressed as a percentage of standard dose, and best responses observed across different cancer types.
BMI, body mass index; ECOG PS, Eastern Cooperative Oncology Group performance status; RT, radiotherapy; TACE, transarterial chemoembolization.
At the start of lenvatinib, the lowest median age was found in EC (64 y) and the highest in AdCC (71.4 y). DTC and AdCC groups showed the highest age ranges (24.7–88.5 y and 43.8–87 y, respectively). The most prevalent baseline ECOG PS was 0 in HCC (90.2%) and 1 in DTC (59.4%), AdCC (74.2%), and EC (80%). About 2%–9% of all treated patients have a PS of 2. Baseline BMI was available for at least 2/3 of cases. A prevalence of BMI > 25 was observed in DTC (62.7%). All groups showed a trend of BMI decrease across lenvatinib treatment, with the highest delta found in AdCC (−3.9), followed by DTC (−1.49), EC (−1.0), and HCC (–0.78). Except for DTC, in all other groups, most patients did not have baseline hypertension. At least 2 hypertensive medications were reported at baseline by 12.9% of AdCC, 12.5% of DTC, and 9.7% of HCC patients. Anticoagulant/antiaggregant therapy was ongoing for 29% AdCC, 11% DTC, 8% EC, and 7.3% HCC patients. A high-burden disease (⩾3 metastatic sites) was found in 35.9% of DTC, 22.6% of AdCC, 20% of EC, and 7.3% of HCC; liver metastases were present in 22.6% AdCC, 12.5% DTC, and 8% EC; and brain metastases in 9.7% AdCC, 7.8% DTC, and 4% EC. Compared to histology-specific standard doses, the median starting dose of lenvatinib (defined as dose intensity in the first 21 days) was full in HCC, 58% in DTC, and AdCC, 50% in EC. Dose reductions during the treatment period shifted the median end-of-treatment doses versus baseline in HCC (67% vs 100%) and AdCC (42% vs 58%) groups. The most prevalent best response observed across all groups was stable disease. Disease control during lenvatinib treatment was met in 90% DTC, 76% EC, 74% AdCC, and 65.8% HCC (Table 2).
Overview table: patients and tumor characteristics; lenvatinib start and stop dose; and best response to lenvatinib.
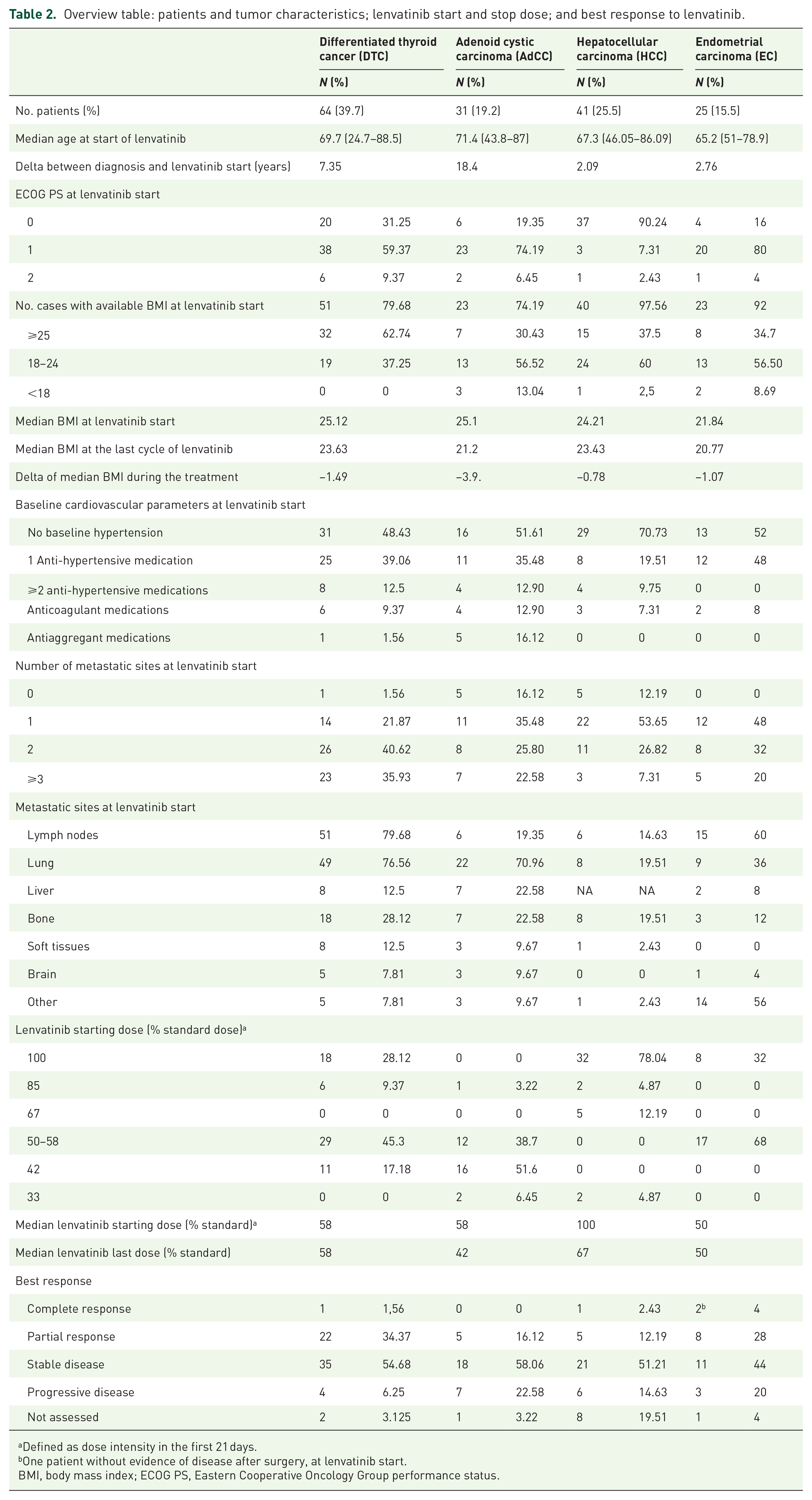
Defined as dose intensity in the first 21 days.
One patient without evidence of disease after surgery, at lenvatinib start.
BMI, body mass index; ECOG PS, Eastern Cooperative Oncology Group performance status.
The most frequently observed AEs across all groups were fatigue, hypertension, and diarrhea. Fatigue was predominantly of low–moderate grade. High-grade hypertension (⩾G3) was observed in 22.5% AdCC, 15.6% DTC, 8% EC, and 2% HCC. Diarrhea was reported by 36.6% HCC, 23.4% DTC, 12% EC, and 6.4% AdCC. Hemorrhagic events were observed, especially in DTC (29.7%) and AdCC (16%), venous thrombosis in 17.2% DTC, 8% EC, and 6.4% AdCC. Palmar-plantar erythrodysestesia syndrome was observed in 28% EC, 14% DTC, and 9.7% HCC. Osteonecrosis of irradiated sites was observed in AdCC (19.4%). At least one dose reduction during lenvatinib treatment was required by 64% DTC, 45.2% AdCC, 44% EC, and 41.5% HCC. The therapy was temporarily withdrawn in 89% DTC, 67.7% AdCC, 56% HCC, and 36% EC. After 1 year of lenvatinib, 68.5% DTC, 35.5% AdCC, 32% EC, and 14.6% HCC patients were still on treatment, and late AEs ⩾ G3 were annotated for available cases (Table 3). Across all cohorts, the definitive stop of lenvatinib was due to progression more frequently than to toxicities. At the data cutoff date, the majority of DTC and AdCC experienced an OS event, due to PD in more than 80% cases.
Lenvatinib treatment characteristics: main adverse events, dose reductions/withdrawals, and treatment duration.

Further four patients ongoing with pembrolizuamb monotherapy.
In the whole population, lenvatinib treatment correlated with BMI decrease (p = 0.045). Intrapatient BMI decrease was higher in patients with cancers originating from the head and neck district (HND) (TC + AdCC, p = 0.039). mDoT did not differ stratifying according to baseline BW in all subgroups (Table 4).
BW, intrapatient BMI variation, and duration of treatment during LEN therapy across different histotypes.

BMI, body mass index; BW, body weight; DoT, duration of treatment; DTC, differentiated thyroid carcinoma; EC, endometrial carcinoma; HCC, hepatocellular carcinoma; LEN, lenvatinib; mDoT, median duration of treatment; mOS, median overall survival.
Median follow-up, duration of treatment, and mOS for each cohort are reported in Table 5.
Duration of treatment and overall survival.
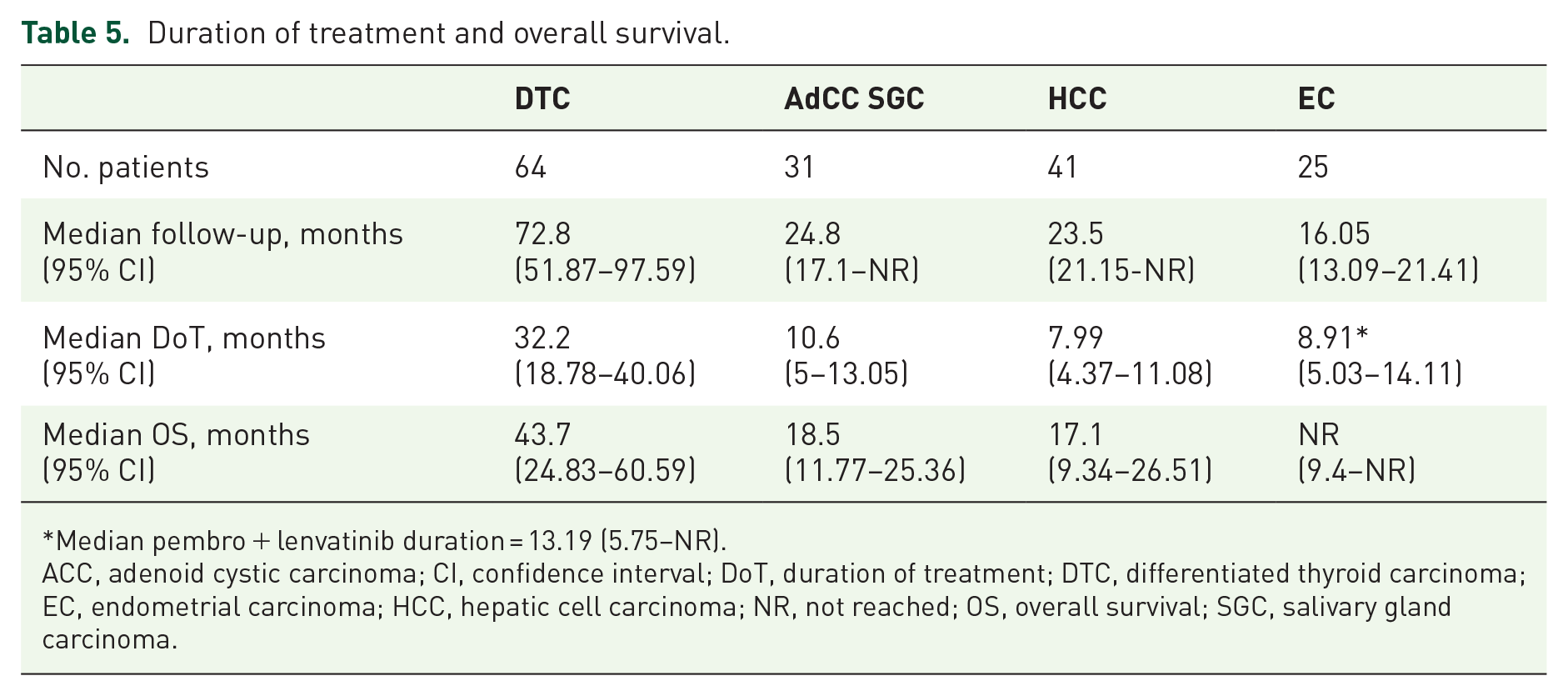
Median pembro + lenvatinib duration = 13.19 (5.75–NR).
ACC, adenoid cystic carcinoma; CI, confidence interval; DoT, duration of treatment; DTC, differentiated thyroid carcinoma; EC, endometrial carcinoma; HCC, hepatic cell carcinoma; NR, not reached; OS, overall survival; SGC, salivary gland carcinoma.
Discussion
Few data are available regarding direct comparisons on management and outcomes of patients affected by different types of cancers treated with lenvatinib in clinical trials17,18; moreover, to the best of our knowledge, no data are currently available in the literature about direct comparisons in the real-world setting. In the following sections, we will highlight how the results of unselected patients can be compared with those reported in the lenvatinib arms of the referral clinical studies for each cancer type.
Differentiated thyroid cancers
In the SELECT study, 2 261 patients received lenvatinib, and 52.1% were female. At lenvatinib start, as compared to SELECT, our real-world patients presented higher median age (69.7 vs 64 y) and ECOG PS 2 (9.4% vs 5%). In comparison with the post hoc analysis of SELECT, 19 this series presented higher prevalence of nodal (vs 79.7% vs 52.9%) and brain metastases (7.8% vs 3.4%), and lower prevalence of lung/liver/bone metastases (76.6 vs 86.5%, 12.5 vs 16.8%, 28% vs 39.8%, respectively). The ORR in the real world was lower than the one reported in SELECT (36% vs 64.8%), probably due to lower median starting dose leading to higher SD rates (54.7% vs 29.9%) and lower PR rates (34.4% vs 63.2%). This confirms the previous evidence of the ORR advantage given by starting lenvatinib at full dose. 4 A lower median starting dose can also be related to the lower incidence of hypertension ⩾G3 (15.6% vs 41.8%), diarrhea (23.4% vs 59.4%), and palmar-plantar erythrodysesthesia (14% vs 31.8%) observed in the real world. Conversely, a higher rate of all-grade fatigue has been reported here (78% vs 59%). SELECT study reported a lower rate of discontinuation of lenvatinib because of AEs (14% vs 26.5%). However, the two groups are not comparable on this point, due to the significantly different median follow-ups (72.8 months in real-world vs 17.1 months in SELECT). Despite the higher prevalence of elderly subjects, ECOG PS 2, and rate of dose reductions, our series confirmed the long-term manageability and effectiveness of lenvatinib in the real world (mDoT of 32.2 vs 13.8 months in SELECT, mOS of 43.7 months vs NE in SELECT).
Adenoid cystic carcinoma of the salivary glands
In the American phase II clinical study of lenvatinib in AdCC recurrent/metastatic patients, 10 the ORR was 15.6%, mPFS 17.5 months, with a treatment duration ranging from 13.7 to 22.6 months; the most common G3 or G4 TRAEs were hypertension (28.1%) and oral pain (9.4%); 18 of 32 patients discontinued lenvatinib for drug-related issues. In the Italian study, 11 the ORR was 11.5% and mPFS 9.1 months; 50% of cases presented TRAEs ⩾ G3; 24 of 26 patients required a dose reduction of lenvatinib. Although our real-world experience cannot be directly comparable with the results of phase II clinical trials, our data on response are in line with the ORR cited in the literature. The treatment duration of 10.6 months is comparable to the 9-month PFS of the Italian study, while the median OS is shorter (18.5 months in the current study vs 27 months in the trial). However, the survival is still in line with the evidence on the use of lenvatinib provided by a multicenter British study which included 23 patients treated with lenvatinib, whose median OS was 12 months. 20 The frequency of G3+ arterial hypertension of the current work (22.5%) is comparable to the one reported in the cited phase II studies (21%–28%).10,11
Hepatocellular carcinoma
In the phase III REFLECT study, 14 median OS in the lenvatinib arm was 13.6 months, with a median duration of treatment of 5.7 months; the most common any-grade TRAEs were hypertension (42%) and diarrhea (39%). In our real-world experience, OS was higher (17.1 m vs 13.6) with the same relevant TRAEs: hypertension (39.03%) and diarrhea (36.6%). However, we recorded a higher rate of any-grade fatigue (39%) than in the regulatory trial (30%). The duration of treatment in our study was 7.99 months, about 2 months longer than in the REFLECT trial (5.7 months). Moreover, 31.7% of our patients had a drug interruption versus 40% in the regulatory trial with similar dose reduction rates (41.5 vs 37%). The ORR in the REFLECT trial was 24.1% and 14.6% in our population; this difference could be due to 19.51% of our patients not being evaluated for radiological response due to clinical progression of disease. Our data are in line with other real-life experiences.21 –23
Endometrial carcinoma
In the Study-309-KEYNOTE-775 phase III clinical trial 15 of pembrolizumab plus lenvatinib for recurrent EC, the ORR was 30.3%, with a median duration of treatment of lenvatinib of 211.5 days (7 months) and median OS of 17.4 months (range 14.2–19.9); the most frequent TRAE was hypertension (64%) with 66.5% of lenvatinib dose reduction for AE of any grade, and 69.2% of drug interruption. In our experience, the ORR is 32% with a median treatment duration of 267.3 days (8.91 months), in line with regulatory trial data. Median OS is not reached in our population (range 9.4–NR) due to the recent availability of the treatment by the Italian National Health Service. Median age of population is similar to the literature (65.2 vs 64 y.o.); however, we also included patients with PS ECOG 2 (4%) and who received more than 2 previous lines of chemotherapy (19%), who were not included in the KEYNOTE-775 trial. Within the first cycle of therapy (21 days from the start of lenvatinib), most of our patients, being frailer than the population included in the clinical trial, needed dose reductions for TRAEs. Thus, the starting dose of lenvatinib was reported as the dose maintained during the first 21 days of treatment. In our real-world experience, only 48% of patients required a dose reduction (vs 70.9%). A lower median starting dose led to a lower incidence of hypertension ⩾G3 (8% vs 37.9%) and a lower drug discontinuation due to TRAEs (40% vs 69.2%).
Multi-malignancies considerations
In a real-world setting, independent of different cancer types, baseline BW and BMI, lenvatinib starting doses, and mFUP duration, the BMI distribution was lower at last lenvatinib administration compared to baseline, especially for patients affected by cancers arising in the head and neck district (TC and ACC). In AdCC and HCC, a wider delta between first and last lenvatinib doses was observed, compared to DTC and EC, opening the question about the duration of tolerability of standard doses in the real-life setting, especially for patients affected by AdCC and HCC. These findings, if confirmed by multicenter studies, could inform future clinical guidelines about the importance of including a comprehensive nutritional assessment at baseline and during the whole treatment, especially for patients with a baseline low BMI who could experience further unintended weight loss.
Conclusion
In our real-world experience, patients treated with lenvatinib, net of different cancer types, show toxicities comparable to selected patients included in clinical trials. Despite the higher prevalence of elderly subjects, ECOG PS 2, and rate of dose reductions, our series confirmed the long-term manageability and effectiveness of lenvatinib in the real-world practice. Moreover, further studies should investigate the impact of nutrition on the tolerability of lenvatinib and possible strategies to overcome weight loss during treatment.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251359905 – Supplemental material for Lenvatinib in advanced thyroid, salivary gland, endometrial, and hepatocellular carcinomas: a real-world study on treatment duration and toxicity management
Supplemental material, sj-docx-1-tam-10.1177_17588359251359905 for Lenvatinib in advanced thyroid, salivary gland, endometrial, and hepatocellular carcinomas: a real-world study on treatment duration and toxicity management by Elena Colombo, Monika Ducceschi, Stefano Cavalieri, Maggie Polignano, Marta Bini, Paolo Ambrosini, Matteo De Monte, Giulia Montelatici, Alessandra Russo, Valentina Bellia, Sharrie Bhoori, Cristiana Bergamini, Salvatore Alfieri, Imperia Nuzzolese, Arianna Ottini, Vincenzo Mazzaferro, Francesco Raspagliesi, Lisa Licitra and Mara Mantiero in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251359905 – Supplemental material for Lenvatinib in advanced thyroid, salivary gland, endometrial, and hepatocellular carcinomas: a real-world study on treatment duration and toxicity management
Supplemental material, sj-docx-2-tam-10.1177_17588359251359905 for Lenvatinib in advanced thyroid, salivary gland, endometrial, and hepatocellular carcinomas: a real-world study on treatment duration and toxicity management by Elena Colombo, Monika Ducceschi, Stefano Cavalieri, Maggie Polignano, Marta Bini, Paolo Ambrosini, Matteo De Monte, Giulia Montelatici, Alessandra Russo, Valentina Bellia, Sharrie Bhoori, Cristiana Bergamini, Salvatore Alfieri, Imperia Nuzzolese, Arianna Ottini, Vincenzo Mazzaferro, Francesco Raspagliesi, Lisa Licitra and Mara Mantiero in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
