Abstract
Secretory carcinoma (SC) is a newly defined and exceedingly rare pathologic subtype of lung cancer. We present a case of SC that was initially misdiagnosed as stage IIIB (cT2aN3M0) invasive mucinous adenocarcinoma harboring a programmed cell death ligand 1 tumor proportion score of 10% and an ETV6-MET fusion gene. The patient received five cycles of toripalimab and pemetrexed plus carboplatin at a local hospital without treatment-related adverse events. Radiologic evaluations prior to third- and fifth-cycle immunochemotherapy indicated that the best response was stable disease. The patient was subsequently reclassified as having stage IB (cT2aN0M0) primary pulmonary malignancy after invasive N staging at our institution, and the primary tumor and mediastinal lymph nodes were removed via a thoracoscope. The tumor exhibited typical morphologic features of SC with immunohistochemical positivity for Mammaglobin and S100 but not for NapsinA or TTF1. Tumor regression was scarcely observed in the surgically resected specimens. This patient was the fifth reported case of primary SC of the lung and the first case of SC treated with immune checkpoint inhibitor therapy that was evaluated in terms of comprehensive safety and efficacy.
Introduction
Secretory carcinoma (SC) was first defined by Skálová et al. 1 as a distinct malignancy based on its strong resemblance to SC of the breast with respect to morphologic, immunohistochemical (IHC), and molecular features. The breast and salivary glands are reportedly the most prevalent anatomic sites of primary SC.2,3 Recently, SC has been detected in the skin, 4 thymus, 5 and esophagus 6 in case reports or case series based on its pathognomonic lobulated growth pattern, diffuse expression of mammaglobin and S100 via IHC staining, and prerequisite ETV6 gene rearrangement. Since the first case of primary pulmonary SC was reported by Huang et al. 7 in 2018, only four cases (Table 1) of this rare, low-grade, primary malignancy of the lung have been described in the literature worldwide.7–10 Therefore, the clinical characteristics, optimal therapeutic approaches, and prognostic outcomes have yet to be fully elucidated.
Summary of the five reported cases of primary secretory carcinoma of the lung.

IHC, immunohistochemical.
To the best of our knowledge, this case study describes the fifth instance of primary SC of the lung. Moreover, the patient described herein was the first SC patient who received immune checkpoint inhibitor (ICI) therapy and was also the first patient to have primary SC of the lung containing an ETV6-MET gene rearrangement.
Case presentation
The reporting of this study conforms to the CARE checklist (Supplemental Material). 11
A 53-year-old Chinese male with a previous history of cigarette smoking (approximately 30 pack-years) and without a family history of malignancy or any known comorbidity, presented with a 1-year history of irritating cough and wheezing that did not respond to medications. With worsening symptoms and new chest tightness, he was admitted to a low-volume hospital on January 10, 2024. The plain and subsequent contrast-enhanced computed tomography (CT) scans of his chest revealed a hypervascular tumor measuring 2.3 × 1.5 cm located in the right intermediate bronchus, right lower paratracheal and subcarinal lymphadenectasis, partial atelectasis in the right middle and lower lobes, and a 3.1 × 2.7 cm smooth homogeneous anterior mediastinal mass at the level of the aortic arch (Figure 1). A whole-body positron emission tomography (PET)/CT scan revealed high uptake of 2- 18 F-fluoro-2-deoxy-D-glucose ( 18 F-FDG) in the primary tumor (the maximum standard uptake value (SUVmax) = 11.1), low to moderate uptake in the right supraclavicular and lower paratracheal lymph nodes (LNs, SUVmax = 6.9), and moderate uptake in the anterior mediastinal mass (SUVmax = 4.7) which was first considered to be thymomas (Figure 1). Subsequent magnetic resonance imaging (MRI) with contrast scanning ruled out brain metastasis. According to the above radiologic findings, the pulmonary lesion was staged as IIIB (cT2aN3M0, the 8th edition American Joint Committee on Cancer (AJCC) TNM staging criteria). Fiberoptic bronchoscopy (FB) revealed tumor surface vascularity and complete right intermediate bronchus obstruction. The FB biopsy samples revealed monolayers of bland epithelial cells with acinar and papillary patterns containing ciliated cells and epithelium, and were initially diagnosed as invasive mucinous adenocarcinoma. Next-generation sequencing (NGS) of a customized 733-gene panel revealed ETV6-MET gene rearrangement (mutation abundance, 28.93%; Figure 2(a)) and a low tumor mutation burden (TMB, 0.56 Muts/Mb). The tumor proportion score (TPS) and combined positive score of programmed cell death 1 (PD-1)-ligand 1 (PD-L1) were 10% and 10, respectively (Figure 2(b)).

Schematic diagram of the patient’s timelines of diagnosis, treatment, radiologic evaluation, abnormal tumor marker, and follow-up.
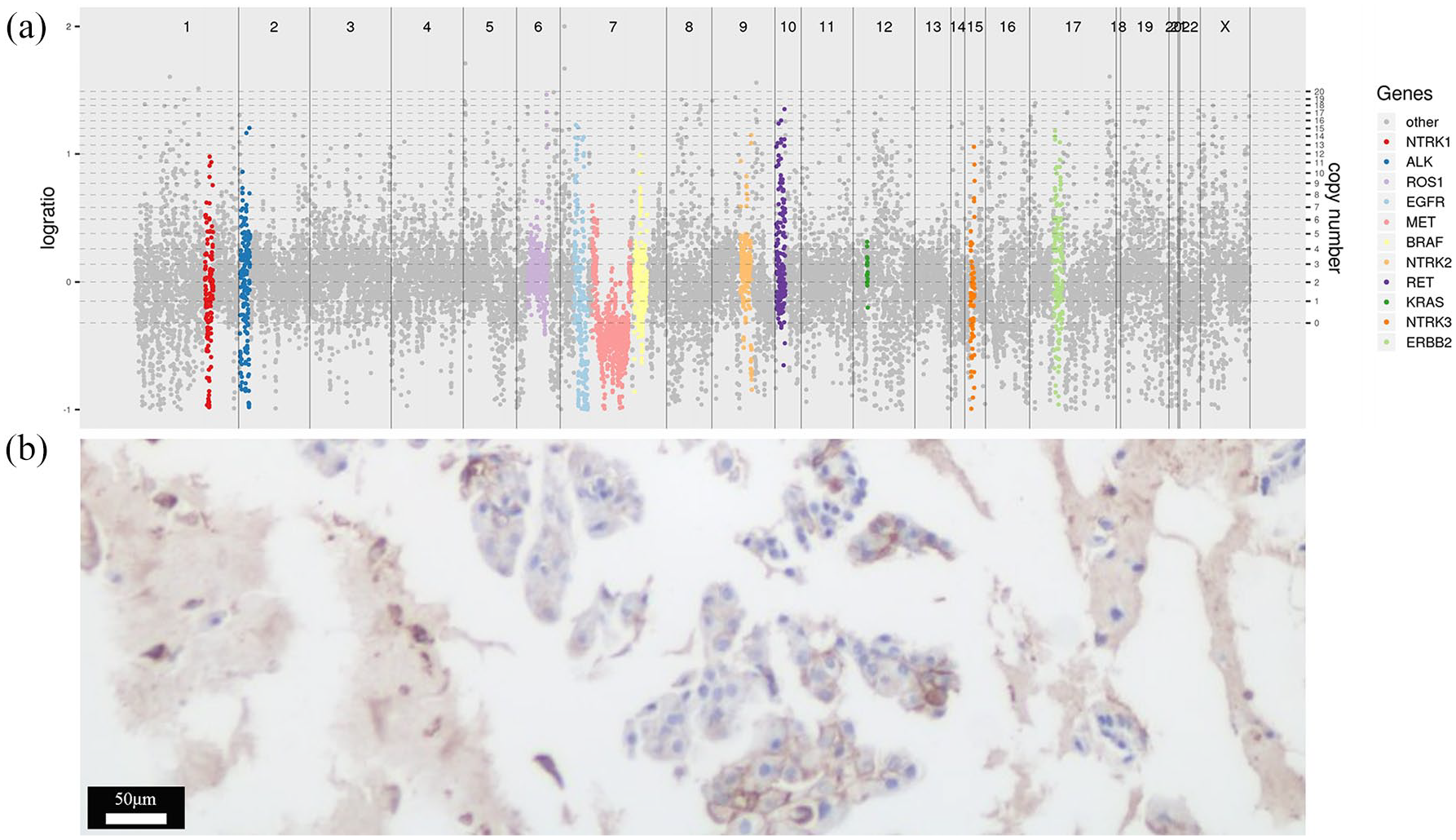
Next-generation sequencing of a customized 733-gene panel covering all exons and partial intronic regions (3D Biomedicine Science & Technology Co., Limited, Shanghai, China) (a) and an immunohistochemistry 22C3 pharmDx assay (Dako, Agilent Technologies Inc., Denmark) for PD-L1 testing (b, 200×) were performed on pretreatment biopsy samples.
The patient was intravenously treated with toripalimab (240 mg, a humanized PD-1 inhibitor), pemetrexed (500 mg/m2) plus carboplatin (area under the curve = 5) on day 1 once every 3 weeks, which was approved by China’s National Medical Products Administration according to the CHOICE-01 trial, 12 for five cycles (Figure 1). The follow-up contrast-enhanced CT scans of the chest prior to the third and fifth cycles of treatment revealed that the primary pulmonary lesion had slightly reduced in size, and the lymphadenectasis and anterior mediastinal mass remained stable. Therefore, the best response was interpreted as stable disease (SD) via the immune-related response evaluation criteria in solid tumors. 13 On May 31, 2024, he was transferred to our high-volume center for the primary purpose of receiving additional radiotherapy. Ultrasound-guided right supraclavicular LN fine-needle aspiration cytologies and endobronchial ultrasound-guided transbronchial needle aspiration stations 4R and 7 LN biopsies were negative for malignancy. Therefore, the pulmonary malignant tumor was restaged as IB (cT2aN0M0), and video-assisted thoracoscopic surgery (VATS) was planned (Figure 1). Surgical resection included two steps. First, the patient was placed in a semisupine position, and total thymectomy and complete excision of the anterior mediastinal tumor via VATS were performed through a single, 4-cm-long incision in the anterior axillary line of the 3rd intercostal space. Second, the patient was placed in the lateral decubitus position and underwent VATS right middle and lower bilobectomy plus systematic mediastinal LN dissection (stations 2R, 3A, 4R, 7, 8, and 9) via a single, 4-cm-long incision located in the 5th intercostal midline.
A well-defined solid homogeneous mass measuring 2.5 × 1.7 × 0.8 cm was found in the right intermediate bronchus. Hematoxylin and eosin (HE)-stained slides revealed that the tumor was well circumscribed and consisted of mucin-filled cuboidal to columnar tumor cells arranged in variable growth patterns, accompanied by fibrotic foci and inflammatory cells (Figure 3(a)). IHC staining was selectively performed, and the interpretation results were as follows: negative for GCDFP15, pan-TRK, CK5/6, P40, ALK(D5F3), BRAF-V600E, cMET, Her2, TTF1 (Figure 3(b)), and NapsinA (Figure 3(c)); strongly positive for Mammaglobin (Figure 3(d)), S100 (Figure 3(e)), SOX10, AE1/AE3, and CK7; and moderately positive for Vimentin. All 23 LNs from the 9 stations examined were negative. This patient was diagnosed with primary pulmonary SC, assessable for a pathologic nonresponse according to the immune-related pathologic response criteria (irPRC), 14 and staged as IA3 (ypT1cN0M0). No further adjuvant therapy was given to this primary carcinoma of the lung. In addition, HE stained sections of the anterior mediastinal tumor revealed that the tumor cells were uniform in appearance with polygonal shapes, finely granular nuclear chromatin, inconspicuous nucleoli, moderate to abundant eosinophilic cytoplasm, and >2 mitoses per 2 mm2. Strong positive CD56, ChrA, and Syno staining was observed on the tumor cell membrane. The combination of features supported a diagnosis of atypical carcinoid. In view of a Ki-67 proliferation index of 30%, this patient received adjuvant radiation therapy of 50 Gy/25 fractions to the tumor bed located in the left anterior mediastinum. As of May 24, 2025, the follow-up time was 11.5 months since the day of surgery without disease recurrence.

Primary secretory carcinoma of the lung. (a) Well-circumscribed tumor displaying mucin-filled cuboidal to columnar tumor cells arranged in variable growth patterns (20×, HE); ▲, scarce plasma cells infiltrated among the tumor cells (100×, HE); *, mucin pools (200×, HE). Negative staining for TTF1 (b, 200×) and NapsinA (c, 200×). Diffuse staining for Mammaglobin (d, 200×) and S100 (e, 200×).
Discussion
In this case report, we first described the safety and efficacy of ICIs plus chemotherapy in a patient with SC worldwide. This patient maintained good tolerability throughout five cycles of toripalimab, pemetrexed, and carboplatin therapy, with no adverse events reported. However, his primary tumor showed almost no radiologic or pathologic response to the immunochemotherapy. Notably, this was also the first ETV6-MET gene rearrangement identified in primary SC of the lung, which may provide implications for future targeted therapy.
The most common age group among patients with primary SC of the lung is older adults, with a mean age at diagnosis of 55.4 years (ranging from 51 to 62 years); however, there were no trends in terms of sex and lobe prevalence.7–10 Irritating cough (3/5, 60%) and wheezing (2/5, 40%) were the most common discomfort presentations (Table 1) because the primary lesion usually originates from the bronchial glands and forms an endobronchial mass, which can cause obstructive symptoms and pneumonia. CT findings have revealed that primary pulmonary SC usually appears as a central, hypervascular, and well-circumscribed tumor with enlarged LNs in the peribronchial or hilar zones.7,9 Consistent with our case, all three patients who received pretreatment PET/CT scans showed radionuclide uptake in primary lesions.7–9 Notably, although these enlarged LNs were considered metastases in the present case due to the 18 F-FDG avidity, all biopsies were negative. Therefore, pretreatment biopsy is recommended to obtain a definite pathologic diagnosis because of the lack of any specific diagnostic imaging features, and invasive LN evaluation methods should be performed for accurate staging.
As previously described in primary pulmonary SC cases, the microscopic features observed from the HE sections of the small biopsy samples may support an initial diagnosis of (mucinous) adenocarcinoma.7,9 However, negative staining for TTF1, NapsinA, p40, or p63 should raise awareness to refine the pathologic subtypes in this situation. S100 protein and mammaglobin immunopositivity tend to indicate salivary gland neoplasms. The detection of a chromosomal rearrangement between the ETV6 gene on chromosome 12 and NTRK3 on chromosome 15 (ETV6-NTRK3 rearrangement), which can be confirmed via reverse-transcriptase polymerase chain reaction, fluorescent in situ hybridization, and pan-TRK IHC staining, is usually regarded as the gold standard for diagnosing SC.1,15,16 Furthermore, this specific rearrangement may be a divergent oncogene and a potential therapeutic target.15,17 Drilon et al. reported a patient diagnosed with SC harboring an ETV6-NTRK3 rearrangement. After receiving pan-Trk inhibitor entrectinib therapy, the patient achieved a dramatic partial response that lasted for 7 months. 17 Similarly, a patient with stage IV lung atypical carcinoid harboring the ETV6-NTRK3 gene fusion received repotrectinib (a next-generation TRK inhibitor) treatment and achieved a partial response of more than 10 months. 18 However, although Baněčková et al. 15 recently reported that ETV6-NTRK3 rearrangement was identified in 176 (81.9%) of 215 patients, less frequent fusion mutations, including ETV6-unknown partners (8.4%), ETV6-RET (5.6%), and VIM-RET (0.5%), were also observed in the molecular profiles. Recently, two other rearrangement patterns, that is, CTNNA1-ALK 19 and ETV6-MAML3, 20 were identified in minority cases. Although the first ETV6-MET translocation was detected in SC via NGS in 2018, 21 it was not detected in primary pulmonary SC until the current case report. Notably, multikinase MET inhibitors and c-Met-selective inhibitors have demonstrated promising outcomes in a wide range of malignancies with nuclear basket protein gene-MET fusion. 22 In summary, owing to varied mutation patterns and a lack of specific drug therapy in SC, we recommend the use of NGS for detection to avoid missing these uncommon rearrangements, which could guide targeted cancer therapies and aid in differential diagnosis.17,18
To date, all four reported patients with primary pulmonary SC have undergone surgical resection after the exclusion of distant metastasis, with two patients (50%) having one hilar LN metastasis.7–10 Consequently, mediastinal LN dissection or systematic LN sampling is the standard of care for this rare malignancy to obtain more accurate staging. Whether patients with SC could benefit from chemotherapy remains controversial. Our patient was observed to have no radiologic or pathologic response to platinum-based chemotherapy, which was similar to the case report of primary SC of the breast by Arce et al. 2 Recently, Wang et al. 10 reported that a pulmonary SC lesion shrank dramatically after one cycle of pemetrexed plus carboplatin chemotherapy; however, the pathologic features of response in resected lung tumors and LNs were not described in their report. Although encouraging progress in lung cancer immunotherapies has been made to improve outcomes, the efficacy and safety of immunotherapy in patients with SC have yet to be reported, which may be primarily attributable to the high proportion of genetic rearrangement mutations inherent in this rare malignancy. 15 Because this otherwise healthy, middle-aged, male patient with no comorbidities and a normal body mass index was initially diagnosed with stage T2aN3M0 invasive mucinous adenocarcinoma with PD-Ll TPS = 10% and TMB-low, five cycles of first-line immunochemotherapy were administered at the first-visit hospital. Two consecutive CT examinations revealed no response, and no treatment-related adverse events (TRAEs) occurred. He was restaged at T2aN0M0 through invasive N staging in our hospital, and radical resection plus mediastinal LN dissection was subsequently performed. Microscopic examination of the resected pulmonary tumor specimen revealed (a) scarce lymphocytes and plasma cells infiltrated among the tumor cells with rare granuloma formation; (b) almost no tumor cell death; and (c) no neovascularization or new, proliferative fibrosis. 14 Owing to >90% residual viable tumor remaining in posttreatment specimen, this patient was evaluated as having a pathologic nonresponse according to the irPRC. 14 Consistent with the findings of previous studies,23,24 the low TMB and concurrent PD-L1 plus tumor-infiltrating lymphocyte expression in the present case may underlie the above clinical observations. Therefore, we recommend that comprehensive molecular profiling and immune phenotyping should be carried out at the onset of immunotherapy in this rare entity to identify potential beneficiaries.
Conclusion
In summary, this is the first case report regarding the efficacy and safety of immunochemotherapy in SC, and the ETV6-MET fusion gene was also found for the first time in primary SC of the lung. According to our report and literature review, radical surgical resection is the preferred treatment option for this rare, low-grade, primary malignancy. In addition, targeted therapy based on comprehensive molecular profiling has the potential to be a promising therapeutic alternative. This case report indicates that although no TRAEs occurred during the preoperative immunochemotherapy phase, primary SC of the lung might have a limited response to platinum-based chemotherapy and PD-1 inhibitors. Taken together, matched basket trials evaluating targeted therapies are warranted, and future basic studies regarding molecular profiles and immune phenotypes should develop more tailored and efficient therapeutic strategies for SC.
Supplemental Material
sj-pdf-1-tam-10.1177_17588359251357520 – Supplemental material for First case report of immune checkpoint inhibitor therapy to treat secretory carcinoma
Supplemental material, sj-pdf-1-tam-10.1177_17588359251357520 for First case report of immune checkpoint inhibitor therapy to treat secretory carcinoma by Xiangyang Yu, Xiaoxing Ye, Mengqi Zhang, Wenting Huang, Calvin S. H. Ng, Zhentao Yu and Kai Ma in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
