Abstract
Ultra-low locally advanced rectal cancer presents significant challenges for sphincter-preserving surgery due to its anatomical constraints and limited response to conventional neoadjuvant therapies. This case report describes a patient diagnosed with stage IIIb (cT3N2M0) ultra-low rectal cancer who had a strong preference for sphincter preservation. Given the suboptimal efficacy of standard treatments in achieving sufficient tumor downstaging, a comprehensive 6-month neoadjuvant regimen was implemented, combining photodynamic therapy (PDT), chemotherapy, targeted therapy, and immunotherapy. PDT selectively induced tumor necrosis, disrupted the tumor vasculature, enhanced therapeutic agent penetration, and transformed the tumor microenvironment into an immune-responsive state. This multimodal approach resulted in significant tumor regression, facilitating sphincter-preserving radical resection. Postoperative pathological examination confirmed a pathological complete response, and the patient remains disease-free, with a progression-free survival exceeding 48 months. This case highlights the potential of a multimodal treatment approach, combining PDT with systemic therapies, to enhance tumor downstaging, potentiate the efficacy of immunotherapy, and improve sphincter preservation rates in ultra-low locally advanced rectal cancer. This integrated strategy offers a promising approach for optimizing clinical outcomes in these challenging cases.
Keywords
Introduction
Ultra-low rectal cancer is defined as a tumor with its lower margin located ⩽5 cm from the anal verge, accounting for approximately 70%–80% of all rectal cancer worldwide. 1 Its treatment is associated with two major challenges: First, sphincter preservation remains difficult. Due to the tumor’s close proximity to the anal sphincter, a permanent stoma is often required in conventional surgical approaches. Although techniques such as intersphincteric resection and transanal total mesorectal excision (TaTME) have been developed to improve sphincter preservation rates, postoperative bowel dysfunction continues to be a significant concern, potentially affecting patients’ quality of life.2–5 To address this, conformal sphincter-preserving operation has been introduced, which can achieve relatively satisfactory postoperative anal function. 6 Furthermore, studies have shown that approximately 7% of rectal cancer patients experience local recurrence within 5 years after surgery, with a higher risk in ultra-low rectal cancer. 7 Positive resection margins are an important predictor of recurrence. 7 Locally advanced ultra-low rectal cancer often requires individualized neoadjuvant therapy, yet clinical response rates remain relatively low with regimens primarily based on chemoradiotherapy. Multiple studies have shown that total neoadjuvant therapy can improve the pathological complete response (pCR) rate in patients with locally advanced rectal cancer.8,9 The addition of targeted therapy provides new possibilities for neoadjuvant treatment.10,11 In addition, immunotherapy has demonstrated revolutionary potential in the neoadjuvant treatment of deficient mismatch repair (dMMR)/microsatellite instability (MSI)-H rectal cancer. In 2015, Le et al. 12 reported that immunotherapy significantly increased the objective response rate in MSI colorectal cancer patients. The Voltage study explored the use of neoadjuvant immunotherapy with nivolumab following long-course chemoradiotherapy in locally advanced rectal cancer, with a pCR rate of 60% in the dMMR group. 13 However, immunotherapy has shown limited efficacy in proficient mismatch repair (pMMR)/microsatellite stable (MSS) colorectal cancer. 14 Nevertheless, pMMR/MSS colorectal cancers account for more than 90% of all colorectal cancer cases, indicating that most patients do not benefit from immunotherapy. 15 Converting these “cold tumors” that are unresponsive to immunotherapy into “hot tumors” that are sensitive to treatment has become a new focus. Photodynamic therapy (PDT) is a localized treatment method that uses a photosensitizer, specific wavelength light, and oxygen to kill tumors. It not only directly destroys tumor cells and vasculature to eliminate the tumor but also activates the patient’s immune system.16–18 Furthermore, PDT can convert “cold tumors” into “hot tumors,” enhancing the efficacy of immune checkpoint inhibitors. 19 Therefore, PDT combined with chemotherapy, targeted therapy, and immunotherapy represents a novel multimodal strategy for the neoadjuvant treatment of locally advanced rectal cancer.
This report describes a case of ultra-low locally advanced rectal cancer in which sphincter-preserving radical resection was successfully performed following treatment with PDT combined with chemotherapy, targeted therapy, and immunotherapy. Postoperative pathological examination confirmed the absence of residual tumor cells, and long-term survival has been achieved, with progression-free survival (PFS) exceeding 48 months. The successful management of this case suggests that a higher likelihood of sphincter preservation may be achieved in patients with ultra-low locally advanced rectal cancer through PDT combined with systemic therapy, thereby opening new possibilities for treatment strategies. This case report conforms to the CARE guidelines 20 (Supplemental Material 1).
Clinical data
Admission condition
Medical history
The patient, a 35-year-old female, was admitted to the hospital with a confirmed diagnosis of rectal malignancy for 1 month, following a history of intermittent hematochezia lasting over 7 years. The hematochezia was described as small amounts of bright red blood, accompanied by discomfort and intermittent episodes of pain. No symptoms of nausea, vomiting, abdominal distension, or diarrhea were reported. The patient has no significant past medical history, no family history of malignancies, no known psychosocial stressors, and no relevant genetic findings. One month before admission, a diagnosis of rectal cancer was established at an external institution, where sphincter preservation was considered unfeasible. As a result, the patient declined the recommended surgery and sought further evaluation at our hospital.
Specialized physical examination findings
Upon examination in the knee-chest position, a firm, poorly mobile mass was palpated 2 cm from the anal verge, with no obvious tenderness, and blood staining observed on glove withdrawal.
Auxiliary examinations
Magnetic resonance imaging (MRI): Irregular thickening and enhancement of the lower rectal wall with multiple enlarged perirectal lymph nodes. Colonoscopy: A cauliflower-like protruding lesion was identified 2 cm from the anal verge. Histopathology: Moderately differentiated rectal adenocarcinoma. Peripheral blood immune profiling: PD1+CD8+ positive cells: 205 per 10,000 cells, indicating a positive result. Tumor genomic profiling: MSI-H, KRAS-mutant. Tumor markers and other laboratory tests: No significant abnormalities.
Initial diagnosis
Rectal adenocarcinoma (cT3N2M0, stage IIIb), KRAS-mutant, MSI-H.
Treatment process
Based on the findings from auxiliary examinations, a diagnosis of ultra-low locally advanced rectal cancer was established. Given the patient’s strong preference for sphincter preservation, immediate surgical intervention was deemed infeasible. Consequently, a personalized precision neoadjuvant treatment plan was developed by our team.
First cycle of PDT
Following the diagnosis, the first cycle of endoscopic PDT was administered, consisting of four treatment sessions. Forty-eight hours before the procedure, the patient was transferred to the PDT ward, where an intravenous injection of hematoporphyrin (Hiporfin; Milone Biopharmaceutical Co., Ltd, Chongqing, China) at a dose of 3 mg/kg was administered. The procedure was performed using an SMA905 fiber-optic system and a semiconductor laser PDT device (PDT630II; Guilin Xingda Optoelectronic Therapeutic Equipment Co., Ltd, Guilin, Guangxi Province, China).
First treatment
A 5 cm optical fiber was introduced to the rectal lesion site for segmental irradiation. The first segment was irradiated for 15 min at a power of 1000 mW and an energy of 900 J; the second segment was irradiated for 5 min at 1000 mW and 300 J.
Second treatment
A 4 cm optical fiber was introduced for segmental irradiation. The first segment was irradiated for 15 min at 1000 mW and 900 J; the second segment for 10 min at 1000 mW and 600 J.
Third and fourth treatment
Since a large amount of grayish-white necrotic tissue was observed covering the tumor surface under endoscopy, segmental irradiation was not performed. Instead, a 4 cm optical fiber was directly introduced to the tumor site, and irradiation was administered for 10 min at 1000 mW and 600 J. During PDT, immunohistochemical analysis of tumor tissues and flow cytometric analysis of peripheral blood were performed to evaluate changes in immune cell populations before and after PDT. Following PDT, strict light avoidance was maintained for 1 month. The entire process of PDT is detailed in Supplemental Material 2.
Systemic therapy
Following PDT, the CAPOX chemotherapy regimen was administered, consisting of oxaliplatin (130 mg/m2, intravenous infusion on Day 1) and capecitabine (1000 mg/m2, orally twice daily, from Days 1 to 14). Genetic testing revealed a KRAS mutation and a PD-1+ cell count of 205 per 10,000 cells. Therefore, the patient received bevacizumab (5 mg/kg, intravenous infusion on Day 1) and tislelizumab (200 mg, intravenous infusion on Day 1). All treatments were repeated every 3 weeks.
Second cycle of PDT
To ensure complete tumor cell elimination, a second cycle of endoscopic PDT was performed during the eighth treatment cycle. Endoscopic examination revealed post-PDT scar tissue extending from 2 to 5 cm from the anal verge.
First treatment
A 5 cm optical fiber was introduced to the rectal lesion site for two-segment irradiation. The first segment was irradiated for 10 min at 800 mW with an energy of 480 J; the second segment for 5 min at 800 mW with an energy of 240 J.
Second treatment
A 4 cm optical fiber was introduced to the rectal lesion site for continuous irradiation. The lesion was irradiated for 10 min at 800 mW with an energy of 600 J.
Third and fourth treatment
A 4 cm optical fiber was introduced to the rectal lesion site, and irradiation was performed for 5 min at 800 mW with an energy of 240 J.
Surgical treatment
One month after the completion of two cycles of PDT and eight cycles of systemic therapy, the patient underwent sphincter-preserving radical resection for rectal cancer at an external institution. Postoperatively, immunotherapy was maintained for 1 year. Since then, regular postoperative follow-up has been conducted.
Evaluation of therapeutic effect
MRI evaluation
During the treatment process, the patient underwent three MRI examinations, which revealed a significant reduction in the irregular thickening of the lower rectal wall compared to the initial diagnosis, along with a marked decrease in the extent of enhancement. No significantly enlarged perirectal lymph nodes were observed. According to RECIST 1.1 criteria, the treatment response was evaluated as a partial response (PR; Figure 1).

MRI changes before and after treatment. (a–c) MRI images of the primary lesion were obtained before treatment, and at 3 and 6 months post-treatment, respectively. (d–f) MRI images of the metastatic lymph nodes before treatment, and at 3 and 6 months after treatment.
Endoscopic evaluation
During PDT, the tumor mucosa appeared whitened, followed by gradual necrosis and sloughing. One week after PDT, extensive necrotic tissue was detected on endoscopy. Three months post-treatment, colonoscopy revealed localized mucosal healing changes at 2 cm from the anal verge, with disrupted vascular patterns observed under NBI imaging (Figure 2). Pre- and post-treatment tumor tissue biopsies were performed, and post-treatment biopsy results demonstrated inflammatory infiltrates and necrosis, with no residual tumor cells detected (Figure 3(a) and (b)).
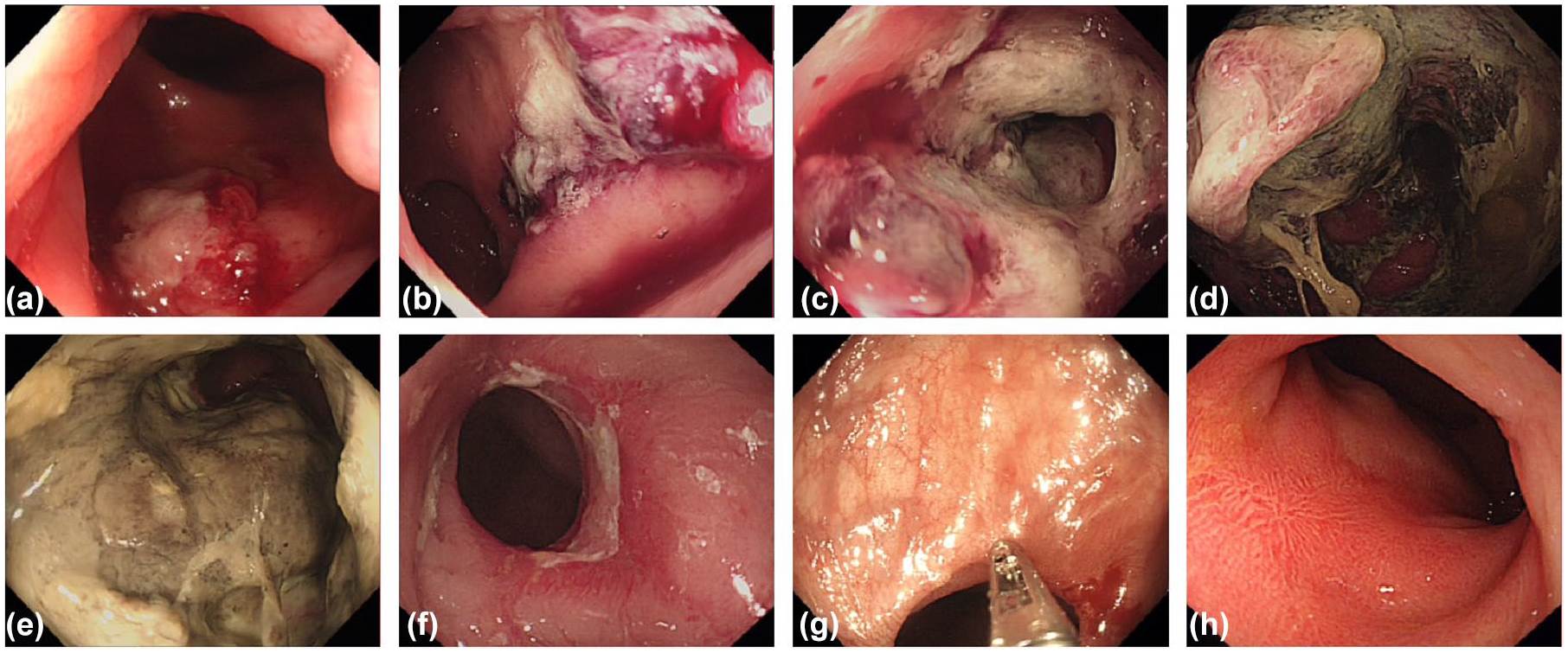
Endoscopic findings before and after PDT. (a–d) Endoscopic images of the rectal lesion during the first, second, third, and fourth PDT of the first cycle, showing progressive whitening of the tumor mucosa. (e–h) Endoscopic images at 1 week, 1 month, 3 months, and 6 months after completion of comprehensive treatment, demonstrating detachment of necrotic tissue and emergence of regenerated mucosa.

HE staining and immunohistochemical images before and after treatment. (a, b) HE staining results before treatment and at 3 months post-treatment. (c–j) Changes in CD8+ T cells, CD20+ B cells, CD68+macrophages, and CD163+ macrophages in tumor tissues before and after PDT of the first cycle.
Changes in immune markers before and after PDT
Immunohistochemical analysis
Tumor tissues collected before and after the first cycle of PDT were subjected to immunohistochemical analysis. The results showed a significant increase in CD8+ T cells and CD20+ B cells, while CD68+ and CD163+ macrophages decreased after treatment (Figure 3(c)–(j)).
Peripheral blood analysis
Peripheral blood samples collected before and after the first cycle of PDT were analyzed using flow cytometry. The results demonstrated that following PDT, an increase in the percentage of natural killer (NK) cells (from 9.4% to 20.2%) and CD8+ T cells (from 9.7% to 20.1%) was observed, while CD4+ T cells decreased (from 48.5% to 33.5%) and Tregs declined (from 9.4% to 3.7%; Figure 4).

Flow cytometry analysis of peripheral blood before and after PDT. The changes in NK cells, CD8+ T cells, CD4+ T cells, and Treg cells before and after PDT of the first cycle.
Postoperative pathological examination
One month after the completion of neoadjuvant therapy, the patient underwent sphincter-preserving radical resection for rectal cancer at an external institution. Histopathological assessment confirmed the absence of residual tumor cells, indicating that pCR had been achieved following neoadjuvant therapy. In addition, gradual recovery of bowel function was observed postoperatively, achieving successful sphincter preservation.
Adverse reactions
Throughout the entire treatment process, chemotherapy-related adverse effects, including nausea, vomiting, and transient liver function impairment, were observed, all of which were resolved with antiemetic and hepatoprotective treatment. No grade 3–4 hematologic toxicities (leukopenia or thrombocytopenia) or other intolerable adverse events were recorded (Figure 5). The patient experienced a 4 kg weight reduction during treatment. No hypertension, bleeding, or other common adverse effects associated with targeted therapy were detected, and no significant immune-related adverse events occurred. In addition, no photosensitivity reactions, severe pain, bleeding, perforation, or infection were observed.

Changes in laboratory parameters during treatment. (a–c) Changes in WBC count, Hb, and PLT count during each treatment cycle. (d–f) Changes in ALT, AST, and ALB during each treatment cycle.
Follow-up results
The patient remains under active surveillance, having achieved PFS exceeding 48 months since initial treatment. The entire treatment process is illustrated in Figure 6.

The treatment process of the patient is reported in this study.
Discussion
For patients with ultra-low locally advanced rectal cancer, sphincter preservation and the maintenance of normal bowel function remain significant clinical challenges. The rates of sphincter preservation can be improved with neoadjuvant therapy, and the introduction of immunotherapy has further increased pCR rates in rectal cancer neoadjuvant treatment. However, within the complex tumor microenvironment (TME), immune cells have been shown to contribute to immune evasion by reducing effective homing to tumor tissues and promoting the expansion of immunosuppressive cell populations, thereby inhibiting the host immune response. 21
PDT has been widely adopted in the clinical treatment of tumors, particularly for superficial or luminal malignancies. Several studies have demonstrated that PDT based on novel nano-photosensitizers exhibits potent antitumor effects in melanoma.22–24 Li et al. 25 reported that PDT induces pyroptosis via the PKM2/caspase-8/caspase-3/GSDME axis in esophageal squamous cell carcinoma. Our team reported a case of advanced esophageal cancer achieving long-term survival after PDT combined with stent implantation and systemic therapy. 26 In addition, studies have demonstrated that the combination of PDT and immunotherapy can effectively inhibit the progression of gastric cancer. 19 A systematic review that included 137 patients with advanced colorectal cancer revealed that nearly all patients underwent PDT as a palliative treatment, with a CR rate of 40%, a PR rate of 43.2%, and symptom improvement observed in 51.9% of patients. 27 Gu et al. 28 found that patients in the PDT group, including those receiving PDT alone and those receiving PDT combined with systemic treatment, had longer overall survival and higher survival rates compared to the non-PDT group (38.2% vs 83.3%, p = 0.006). Multiple studies have demonstrated that PDT combined with systemic treatment exerts significant antitumor effects against various malignancies, including rectal cancer.
PDT exerts its antitumor effects through three primary mechanisms: direct tumor cell destruction, tumor vasculature damage, and remodeling of the immune microenvironment, ultimately leading to the activation of systemic antitumor immunity.29,30 As a highly complex system, the TME is regulated by immune cells, which play critical roles in tumor progression and immune evasion. PDT induces changes in immune cell populations within the TME. Following PDT, tumor-associated antigens are captured and processed by antigen-presenting cells, including dendritic cells (DCs), macrophages, neutrophils, and lymphatic endothelial cells, and presented via MHC class I and II molecules to T-cell receptors, activating naïve CD8+ and CD4+ T cells in draining lymph nodes and stimulating antitumor immunity. 29 Tong et al. 31 demonstrated that the combination of pheophorbide A-mediated PDT and αPD-L1 therapy enhances treatment efficacy by increasing intratumoral PD-L1 expression and promoting T-cell infiltration. Yu et al. 32 reported that PDT using Ce6 as a photosensitizer effectively induces tumor-associated macrophages to polarize toward the M1. Ji et al. 33 further demonstrated that PDT recruits M1 macrophages, thereby inhibiting the growth of cutaneous squamous cell carcinoma. Our team’s study further revealed that PDT enhances intratumoral immune cell infiltration and promotes the conversion of tumors to a “hot” immune phenotype. In addition, PDT induces the clonal expansion of tumor-specific T cells while suppressing the clonality of Tregs, thereby enhancing antitumor immunity. 19 PDT remodels the TME through three primary mechanisms. First, PDT induces immunogenic cell death and promotes the release of damage-associated molecular patterns, including calreticulin, high mobility group box 1 protein, adenosine triphosphate, and heat shock proteins, thereby activating DCs and enhancing antigen presentation.34,35 Second, PDT stimulates the release of cytokines such as tumor necrosis factor-α and interleukin-6, which regulate the recruitment and enhance the activity of immune cells. 36 Third, PDT significantly modulates the expression of immune-related genes in tumor cells, such as SLC11A1, thereby strengthening the cytotoxic function of immune cells. 37
This report presents a case of a patient with ultra-low locally advanced rectal cancer who achieved successful sphincter-preserving surgery following a combination of PDT, chemotherapy, targeted therapy, and immunotherapy. Postoperative pathology confirmed pCR, and the patient achieved long-term disease stability. Peripheral blood samples collected before and after PDT were analyzed by flow cytometry, revealing a significant increase in NK cells and CD8+ T cells, alongside a marked reduction in Tregs, indicating antitumor immune activation. In addition, immunohistochemical analysis of pre- and post-PDT tumor tissues showed a significant increase in CD8+ T cells and CD20+ B cells, accompanied by a decrease in CD68+ and CD163+ macrophages. These findings suggest that PDT may enhance T-cell-mediated cytotoxicity and B-cell-driven humoral immunity, thereby improving antigen presentation and antibody-mediated antitumor immunity. Furthermore, PDT attenuates tumor immune evasion and promotes antitumor immune responses (Figure 7).

The effects of PDT on the immune microenvironment. PDT promotes the release of TAAs, activating T cells to mediate tumor cell killing, while also enhancing immune cell infiltration and remodeling the tumor immune microenvironment, thereby converting “cold tumors” into “hot tumors.”
While PDT plays a pivotal role in enhancing the tumor immune microenvironment, its combination with chemotherapy, targeted therapy, and immunotherapy likely synergizes to achieve the observed clinical outcomes. These findings offer translational evidence to support sphincter preservation strategies and prolonged survival in locally advanced rectal cancer. However, as a single-case report, these findings have inherent limitations in generalizability. The mechanisms underlying the interactions between PDT and chemotherapy, targeted therapy, and immunotherapy are complex. Our team is currently conducting a single-center clinical trial to assess the efficacy, safety, and clinical applicability of this combined therapeutic approach in gastrointestinal tumors (ChiCTR2200064280, ChiCTR2300076208). In the future, we aim to initiate large-scale, multicenter randomized controlled trials to further validate the impact of PDT in combination with systemic treatments on sphincter preservation rates and long-term survival in patients with low-lying rectal cancer.
Conclusion
This case report highlights the potential of a multimodal, individualized neoadjuvant treatment strategy—combining PDT, chemotherapy, targeted therapy, and immunotherapy—to downstage ultra-low locally advanced rectal cancer, increase sphincter preservation rates, and potentially improve long-term survival.
Supplemental Material
sj-pdf-1-tam-10.1177_17588359251345927 – Supplemental material for Successful sphincter preservation and long-term survival in a patient with ultra-low locally advanced rectal cancer treated with photodynamic therapy combined with multimodal treatment: a case report and literature review
Supplemental material, sj-pdf-1-tam-10.1177_17588359251345927 for Successful sphincter preservation and long-term survival in a patient with ultra-low locally advanced rectal cancer treated with photodynamic therapy combined with multimodal treatment: a case report and literature review by Dan Zou, Baohong Gu, Yifan Li, Ying Yan, Huanhuan Ma, Kai Wang, Weiyi Chen, Bofang Wang and Hao Chen in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-2-tam-10.1177_17588359251345927 – Supplemental material for Successful sphincter preservation and long-term survival in a patient with ultra-low locally advanced rectal cancer treated with photodynamic therapy combined with multimodal treatment: a case report and literature review
Supplemental material, sj-pdf-2-tam-10.1177_17588359251345927 for Successful sphincter preservation and long-term survival in a patient with ultra-low locally advanced rectal cancer treated with photodynamic therapy combined with multimodal treatment: a case report and literature review by Dan Zou, Baohong Gu, Yifan Li, Ying Yan, Huanhuan Ma, Kai Wang, Weiyi Chen, Bofang Wang and Hao Chen in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
