Abstract

Introduction
In 2016, the late Valerie Guild, Founder and then President of AIM at Melanoma, was attending a melanoma conference when she noticed that most of the attendees were male oncologists who held leadership roles at various academic institutions. For those who attend these meetings, there are many benefits, including the opportunity for increased visibility as a leader in one’s chosen field and the opportunity to meet other like-minded individuals and learn about their ongoing work. These discussions can often lead to future collaborations, thereby helping to advance research and one’s own career.
Val recognized that she had an opportunity to do something important. A 2014 American Medical Association (AMA) survey showed that half (48%) of hematology/oncology fellowship slots were filled by women. 1 Despite the rising proportion of women oncologists, their distinct lack of prominent visibility at many key conferences was evidence that they needed additional support to reach leadership roles in academia. Seeing the growing number of young women oncologists with an interest in melanoma and AIM at Melanoma’s track record for bringing together experts in the field, Val captured the opportunity to create a forum to advance the careers of women in melanoma, with a parallel goal of improving patient outcomes and the patient experience.
The inaugural 2-day meeting for Women in Melanoma Initiative (WIM) took place in August 2017 in Leesburg, VA, USA. Since that summer, AIM at Melanoma has brought together approximately 30 women, mostly medical oncologists, with a focus on melanoma and skin cancers. This group has also included dermatologists, surgical oncologists, basic scientists, and industry leaders. The purpose of the initiative is to:
Support the academic careers of junior to mid-career women with a research interest in melanoma.
Foster mentoring relationships and collaborations among women physicians and scientists focused on melanoma.
Support the improvement of melanoma patient outcomes through a focus on strengthening the physician–patient relationship.
Provide insights into best practices in communicating with patients with melanoma to improve outcomes.
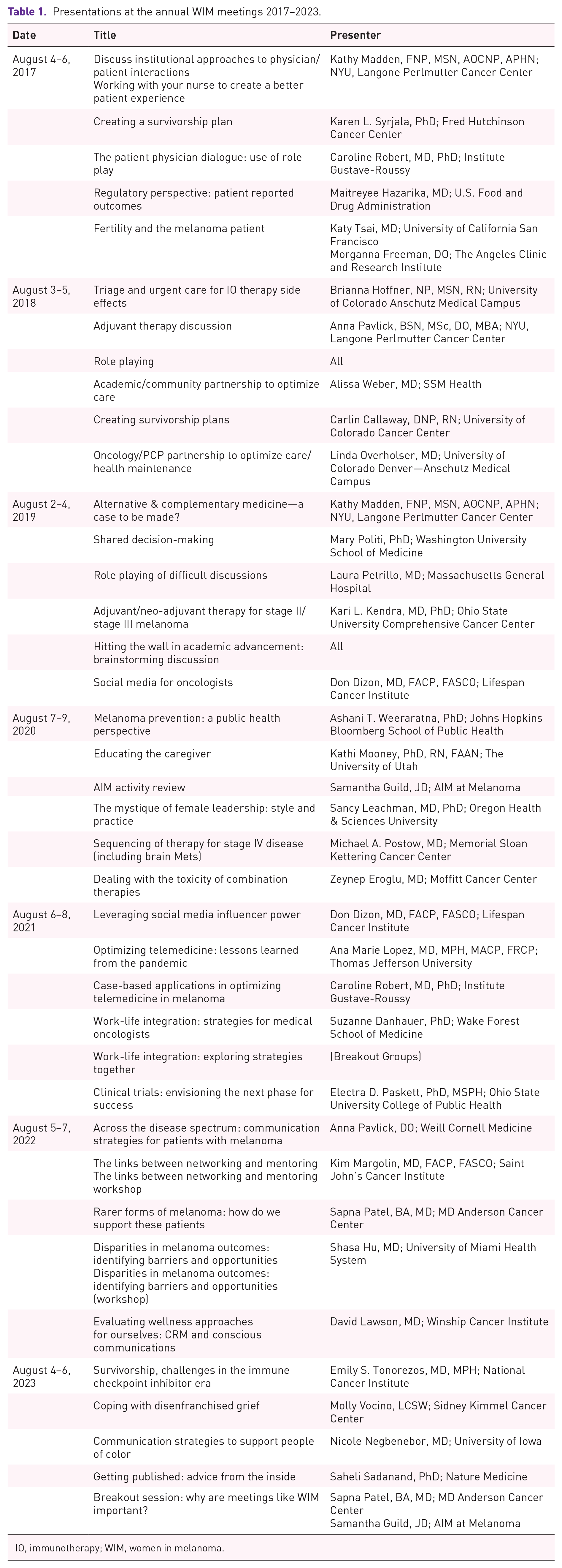
Over the course of the past 7 years, a broad range of topics has been covered. These are listed in Table 1. These annual “touch points” have proven invaluable in fostering productive discussion among all who participate. Importantly, the scope of the meetings has been impacted by the scientific and therapeutic landscape each year. For example, in 2017, as we started to see patients with metastatic disease having prolonged survival due to the widespread utilization of immunotherapy (IO), there were individual sessions on creating a survivorship plan, and on fertility and the melanoma patient. Sessions in 2018 (soon after the first regulatory approval for adjuvant IO) focused on discussions on adjuvant therapy, in addition to triage and urgent care for IO side effects. Here we summarize the proceedings of the seventh Annual WIM Meeting, grouped by the meeting agenda topics.
Presentations at the annual WIM meetings 2017–2023.
IO, immunotherapy; WIM, women in melanoma.
Survivorship
The National Cancer Institute (NCI) Office of Cancer Survivorship aims to support research that examines and addresses the long- and short-term effects of cancer and its treatment among cancer survivors and their families. In the current era, the definition of a survivor has evolved: historically, a survivor was thought of as an individual with a history of cancer who is now cancer free. More recently, the NCI adopted a definition stemming from cancer survivor advocates. An individual is considered a cancer survivor from the time of diagnosis through the balance of life, encompassing those living with cancer and those free of cancer. This conceptualization of survivorship allows inclusion of individuals receiving continuous treatment as well as those who have a diagnosis that is likely incurable. It is important to acknowledge, however, that the term survivor does not resonate with every individual patient, and thus, individual preference is key when considering which terms or labels to use. 2
The incidence of melanoma has continued to linearly increase over the past several decades, with an incidence of 97,610 in 2023. Further, IO has led to improved outcomes, with almost 50% of patients with metastatic disease alive at 7.5 years. 3 This progress has resulted in a significant decline in melanoma deaths: of the >600,000 individuals living with advanced cancer in the USA, 40,000 are living with advanced melanoma. 4 In addition, the number of patients with earlier stage melanoma receiving IO has increased significantly, as IO is FDA approved in the adjuvant setting for resected stage IIB and higher.5–8 Thus, the number of patients with melanoma who receive treatment has continued to steadily increase. It is important in this context to emphasize not only the significant number of individuals who are survivors of melanoma, but also the number of patients exposed to IO and its long-term sequelae, which are, to date, little understood.
Given the growing number of individuals living with a history of advanced melanoma, including patients who have completed treatment for metastatic disease, comprehensive survivorship care is essential. The National Academy of Medicine recommends that quality care for cancer survivors should include prevention and surveillance for recurrence, surveillance, and management of physical and psychosocial effects, and the promotion of a healthy lifestyle. 9 However, in the current era of highly effective therapies for melanoma and with a growing number of survivors, there are no evidence-based guidelines to inform ongoing care. The duration, frequency, and mode of imaging surveillance for survivors of melanoma varies based on shared decision making with the patient, according to the risk of recurrence as well as patient preference. Providing anticipatory guidance regarding the immediate and long-term adverse effects of IO in the adjuvant and metastatic settings is an essential element of quality survivorship care.
Furthermore, as data emerge regarding long-term or delayed adverse effects of treatment, oncologists should remain vigilant in monitoring for new or changing comorbidities. For example, there is a need for a unified effort to follow long-term survivors after immune checkpoint blockade for cardiovascular health and/or diagnoses. There also needs to be consensus surrounding evidence-based monitoring, and which clinicians (e.g., oncologists, primary care providers, or advanced practice providers) will follow patients 5 or 10 years or even later into their survivorship. Ultimately, the implementation of quality metrics for survivorship care in melanoma will serve patients. To this end, the NIH has many opportunities for survivorship research, including areas such as improving the prognostic understanding and communication between clinicians, patients, and caregivers. 10 Further, it is essential for non-oncology providers to understand long-term prognosis to inform the need for preventative or surveillance care, and to inform their language in speaking to survivors.
Grief along the cancer spectrum
It is a sad truth that half of all patients seen by an oncologist will die of their disease. 11 A life-changing diagnosis, a treatment plan including treatment toxicities, recurrence risk, and potential progression of cancer must be mentally processed and summarized by the clinical team in a short amount of time. These important pieces of information must then be presented to the patient and their loved ones with proficiency and empathy: not a small task. As one member of the WIM meeting attendees put it, “do they {administrators} know that we talk about life and death in those 15-minute appointments?.” Further, the eventual death of a patient impacts the physician and the entire team caring for the patient. Physician grief and even feelings of guilt or shame at the perceived failure of treatment are common. 12 Physicians take care of their patients first and foremost, and so managing their own grief around a patient’s outcome may come late or never at all. It is an interesting paradox that oncologists have high rates of depression but surprisingly high rates of job satisfaction. 13
Dr Kenneth J. Doka defined disenfranchised grief as grief that is not usually openly acknowledged, mourned publicly, or socially accepted. Grief is complex, non-linear, and does not follow a predicted time course. Each physician also brings their own personal experience to grief. Indeed, the grief response can be accentuated the more we identify with patients.
The prolonged exposure to the hardships endured by patients, their families, and coworkers can be a form of trauma to healthcare workers. Both grief and trauma that are not acknowledged can become internalized and even manifest as physical signs. 14 Post-traumatic symptoms include anxiety, sleep disturbance, dissociation, and re-experiencing the traumatic event. First, physicians must recognize the impact of prolonged exposure to grief and trauma: “What has changed within me as a result of doing this work?” Practicing vulnerability can help physicians in acknowledging grief. This is further complicated in the patient-doctor relationship, as physicians are expected to “have it together,” and showing vulnerability may be misinterpreted as incompetence. Disconnection can result from unprocessed grief and trauma and can lead to increased anxiety and fatigue, and decreased camaraderie and job satisfaction.
As a result of medicine’s long history of propagating disenfranchised grief, many oncologists will resort to the maladaptive coping strategies they have learned and witnessed. 15 These mechanisms may include compartmentalization, depersonalization, dissociation, desensitization and numbing, minimizing, deliberate avoidance, and cynicism. While these coping mechanisms may be useful in the short term, they are not appropriate for the type of chronic grief that medical oncologists face over the many years of their career. Over time, leaning on these strategies can result in “compassion fatigue,” which has been linked to burnout resulting in oncologists leaving patient care. 16 Burnout is one of the more common signs of physician distress, and the rate of emotional exhaustion and/or depersonalization related to burnout among oncologists is estimated to be 45%.17,18
While advances in the management of melanoma have improved overall survival, melanoma oncologists continue to experience complex and sustained grief. We frequently deal with locoregional or metastatic recurrences in patients whom we hoped had been cured with adjuvant therapy. While patients are surviving longer, there is still suffering and death in half of our patients with advanced disease. Over the last decade, there has been a greater recognition that acknowledging death, dying, suffering, and loss in a more open and supportive way is highly beneficial. Implementing adaptive coping strategies such as setting boundaries, seeking out social support, meaning making, practicing intentionality, and paying attention to one’s own physical and mental wellness have been shown to boost resilience and promote growth, stability, healing, and satisfaction. Another strategy is to reconnect to the “big why” and remind oneself of their purpose and reasons for choosing oncology. It is apparent, however, that though an oncologist can individually implement these coping strategies, they only work if the institution and system in which they operate are equally committed. It is an institution’s responsibility to create a safe and supportive space for grieving as well as acknowledge the uniqueness and challenges of caring for cancer patients.
Supporting persons of color
While melanoma occurs most often among white people, individuals across all races and ethnicities can develop a melanoma. A lack of awareness of this risk among clinicians as well as among patients has compounded existing health care disparities in surveillance, diagnosis, and outcomes. Recent public health initiatives have begun to close this gap by increasing education through efforts in transparency, community outreach, and ad campaigns. Nicole Negbenebor, MD, Director of the Skin of Color Clinic at the University of Iowa, notes that melanoma has a prevalence of 5% in Hispanics, 4% in Asians, and 2% in Black individuals.19,20 One of the major confounders in melanoma detection is that individuals of color often have less common patterns of presentation, and thus are more frequently diagnosed with higher stage melanoma.21–23 Further, these individuals have less ultraviolet-related melanoma, and higher rates of acral lentiginous and mucosal melanoma, 24 which are known to be associated with more advanced disease at diagnosis. For example, Asians and Pacific Islanders have the highest proportion of mucosal melanomas, particularly anorectal melanomas. 24 Acral melanomas are the most common subtype in Black patients and are more common in Hispanics compared to Caucasian patients.20,24 These individuals often face delays in diagnosis: one retrospective study noted that 34% of acral-lentiginous melanomas were misdiagnosed, and half of misdiagnosed cases were amelanotic melanomas. 25 And while the biggest progress in melanoma treatment has been against cutaneous melanoma, people of color are more likely to be diagnosed with these rarer subtypes where treatment successes are more modest.
Potential causes for disparities in diagnosis and treatment include access to care, patient awareness for skin or symptom changes of concern, and physician awareness and/or bias related to including melanoma in the differential. Methods to address awareness include improvements in outreach at the community level via drop-in screenings and coordination with community and religious leaders to improve health literacy. Rectifying the myth that melanoma does not develop in individuals with darker pigmentation is critical to emphasizing attention to skin exams and concerning findings among communities of color. Community-organized health screenings will decrease the barrier to medical evaluation and help overcome socioeconomic factors related to insurance or delays in accessing care. These events may also provide additional layers of cultural, linguistic, or religion-centered support and provide more culturally humble care, 26 a term emphasized by the AMA (rather than culturally competent care, the terminology previously used). Having a broader appreciation and respect for the complexity of cultural factors influencing medical decisions allows providers to tailor care and improve efforts to understand and develop practices related to modesty, relatable education materials, and enhanced shared decision making. Recently, the CDC launched an ad campaign to increase the awareness of melanoma across ethnicities by including pictures from individuals of Black, Hispanic, Native American, and Asian descent. Given the diversity of populations impacted by melanoma, incorporating interpreters will assist in addressing language barriers to care.
On a provider-patient level, methods to improve confidence in the healthcare system and relationships with medical personnel will aid in establishing care and securing follow-up. Additionally, interventions within medical education can help decrease barriers, including addressing training gaps on the early diagnosis of melanoma. Medical images incorporating a wider representation of melanomas from individuals of diverse racial and ethnic backgrounds will help decrease the delay in recognition by health care providers. Providers and health systems are also encouraged to increase access to specialized care including multidisciplinary skin cancer teams as well as to clinical trials by decreasing barriers and actively recruiting underrepresented minorities to participate. Skin-specific concerns and beauty standards across racial backgrounds may have a pivotal impact on diagnosis and treatment, particularly as current therapies may induce vitiligo. Indeed, skin color is an innate part of self-identification, and treatments (such as IO) that impact this can be perceived as detrimental. Understanding an individual’s priorities and concerns paves a path toward more empowered care and informed decision making. Ultimately, ongoing acknowledgements of differences in melanoma presentation and care can ultimately culminate in more equitable access to care and outcomes.
Insights into publishing high-impact manuscripts
Publication is essential to further academic advancement, disseminate research results, and advance patient care. Understanding the steps involved in publication from the perspective of Saheli Sadanand, Ph.D. Deputy Editor for Nature Medicine provided the attendees with an “inside perspective” on the high-impact medical publishing world.
Pre-submission inquiries provide an early answer on an article’s suitability for a journal and are best utilized while still finalizing a manuscript. The pre-submission inquiry should include a cover letter, title, abstract, and drafts of the main figures if available. An inquiry can be submitted through the system or by direct email to the relevant editor. Cover letters should be a brief description of the article which puts it in the context of broader melanoma research and can include timing factors such as competition, an upcoming conference presentation or inclusion in a registrational packet to the FDA. Cover letters should include an acknowledgment that everyone signed off on the work and that it is not under consideration elsewhere.
Upon submission, the editorial team evaluates the paper’s fit within the scope of the journal. The paper is also evaluated for broadness of interest, novelty, relevance for management and/or fundamental understanding of disease, adherence to clinical reporting guidelines, and availability of data (including trial protocols). Once a paper is determined to be appropriate for review, two to four reviewers are selected with a balance of new and experienced reviewers with relevant expertise and a focus on diversity. A statistical reviewer is included for all clinical trial papers. The editors will honor three to four excluded referee requests but cannot honor long lists of exclusions.
Following completion of the reviews, the primary handling editor brings the manuscript and reviewer reports for discussion with the editorial team. Considerations include enthusiasm of the reviewers, the significance of the contribution, major flaws, and whether experiments need to be redone. The decision to send a revise or reject letter is then made. Revise decisions will highlight what reviewer comments to prioritize, note what is being overruled, and note aspects that can be improved in presentation or transparency of the data. Reject letters will indicate decision rationale and provide transfer options. The Deputy Editor sees their role as a champion for the manuscript and finding it the best possible “home.”
When authors receive revise letters, they should read the comments of the reviewers and editorial team carefully, noting anything the editor prioritized or overruled. If an author feels that addressing a specific reviewer concern is either irrelevant or not feasible, they should reach out to the handling editor. All concerns should be addressed before re-submission as only one round of major revisions is generally allowed. If a concern cannot be addressed, the reasons for this should be included in the response letter.
If an editor rejects a paper in pre-review, this indicates a fundamental issue with the article fitting in the journal’s scope. If a paper has been rejected on review, appeal should be considered carefully and only done if new information to rebut/address grounds for rejection is provided. A point-by-point response and revision plan should be submitted with an appeal.
Three top items to prioritize in a paper submission are the abstract, cover letter, and data availability. Authors should ensure that all sections are included in the abstract. The cover letter, on the other hand, affords the author the opportunity to “pitch” the novelty and significance of their work in a way that is usually more understated in the manuscript. Finally, authors should ensure that all data are available, including the study protocol and coding for the database. A request for an expedited review is unlikely to be honored when sections or data are missing.
Benefits of WIM
Over the last 7 years, WIM has served as a complement to other research forums by focusing on the humanity of medicine. It provides a space to consider and reflect on the experience of the patient and the caregiver, in addition to the experience of physicians and other practitioners in the melanoma field. It strongly emphasizes the art and practice of oncology. It is a unique forum for patient centered content to be presented to physicians, including discussion of patient support services. The meeting has fostered collaborations, friendships, and camaraderie that carries far beyond the 36 h of the weekend. The success of WIM is evidenced by the continued growth in the careers of women and their achievements and contributions to the melanoma field.
Several tangible benefits emerge each year from the WIM conference. As the group matures, the experience and expertise of the collective group creates a network of referrals and curbsides for complicated patient cases. WIM has an expanding rolodex of experts not only in cutaneous melanoma, but also in uveal melanoma, non-melanoma skin cancer, immune toxicity, genetics, clinical trials, tumor infiltrating lymphocytes (TIL) therapy, and beyond, which serves as a built-in sounding board for patients and providers. It also creates a top-tier list of speakers for continuing medical education (CME) conferences, Grand Rounds, Visiting Seminars, professional committees, medical podcasts, and other academic and professional exposures, which the group has frequently tapped into to invite one another to appropriate forums. Several manuscripts have emerged including, but not limited to, those listed in Table 2, as well as a number of collaborative abstract submissions and clinical trials.
Publications originating from WIM collaborations.

As noted by one WIM attendee, this conference stands apart as a patient-centered meeting where physicians are the audience. As a result, it tends to lend itself to the emergence of specific patient needs, such as role-play for communication, real-world registries for rare melanomas, development of a pregnancy database, and projects related to male and female fertility. Professional development as a result of the WIM conference has also materialized in the form of sponsorship, references for promotion, recruitment for jobs, and transition into leadership positions.
At a time when physician burnout is very high due to workload demands, the COVID pandemic, and systemic issues in healthcare, the WIM conference mitigates some of this damage by allowing us to emphasize the joy in medicine. Attendees participated in an activity to describe some of the intangible gains from attending the WIM weekend. These meetings energize, inspire, and nourish participants who report a sense of camaraderie, feeling valued, connectivity/community, collegiality, and a celebration of intentional and compassionate peer physicians. Figure 1 demonstrates other popular themes that were verbalized by WIM attendees this year.

Intangible advantages of WIM expressed by attendees.
Discussion
The seventh Annual WIM Conference provided opportunity for discussion around cancer survivorship, grief including disenfranchised grief that may be experienced by providers, melanoma and skin cancer in persons of color, insights into publishing, and a collective exercise describing the benefits of meetings such as WIM. Networking and open communication around support and wellness are common each year, and so-called “life hacks” are often freely shared over meals. The diverse presentations, guest speakers, and dovetailed conversations make WIM’s compact weekend agenda dense with information and resources.
Each year, WIM attendees regale the meeting as a highly anticipated event of camaraderie, resources, and support for ourselves and our patients. Many of us happily plan our summer travel around it. The male speakers have described their experience with equal affection and resonance. Other disease streams have taken note of WIM and have remarked on the desire to do something similar. Too often, competing interests or capacity to host such gatherings, particularly in large medical specialties, is limiting.
Indeed, what Valerie Guild did 7 years ago is akin to capturing lightning in a bottle with the timing and uniqueness of this event, and it remains one of the most important melanoma meetings of the year to those who are fortunate to attend.
